Abstract
αB-crystallin is regarded as a biomarker for triple-negative and/or basal-like breast cancer. In normal breast cells, over-expression of αB-crystallin leads to neoplastic-like changes, which likely relate to enhanced expression of phosphorylated ERK1/2 (pERK1/2). In this study, we investigated whether αB-crystallin expression is correlated to pERK1/2 expression in breast cancer. In a balanced tissue microarray the expression of αB-crystallin and pERK1/2 kinase were determined immunohistochemically, together with the triple-negativity and basal-like markers CK5/6 and SMA and the signaling molecules pAKT, pmTOR, EGFR, and IGF-1R. αB-crystallin expression significantly correlated with triple negativity and basal-like markers CK5/6 and SMA (Pearson Chi-square test p=0.004, p=0.001, and p<0.001, respectively). A significant correlation was also observed with pERK1/2 expression (p=0.002). siRNA-mediated knockdown of αB-crystallin in the triple-negative breast cell line MDA-MB468 did not show an effect on pERK1/2 expression levels, indicating that lowering the level of αB-crystallin does not reduce pERK1/2 expression. Our results confirm that αB-crystallin can be used as a biomarker for triple-negative and/or basal-like breast cancer. The expression of αB-crystallin correlates with pERK1/2 expression in breast cancer tissue suggesting that therapies targeting αB-crystallin might be considered for treatment of triple-negative or basal-like breast cancer.
Introduction
Breast cancer consists of a heterogeneous group of tumors with varying histological appearances and prognoses (1). Using gene expression profiling, breast cancer can be categorized in several subsets, of which the most well-known are normal breast- like, luminal A and B, epidermal growth factor receptor 2 (HER2)-enriched, and basal-like breast tumors (1, 2). Because of the clinically aggressive nature, basal-like breast cancers have the worst prognosis (2).
Triple-negative breast cancers are defined by the absence of immunohistochemical staining for estrogen receptor (ER), progesterone receptor (PR), and HER2. This tumor type accounts for 15% of all breast cancer cases (3). Around 75% of the triple-negative cases have the basal-like gene expression profile showing, among other markers, the expression of high-molecular weight basal cytokeratins (CKs) and smooth muscle actin (SMA) (4–7).
Expression of αB-crystallin, a molecular chaperone able to interfere with several cellular processes resulting in cell maintenance under stress conditions (8–10), is enhanced in triple-negative and basal breast cancer, and is a marker of poor prognosis (5, 11–14). αB-crystallin may directly contribute to malignant progression as overexpression of wild type αB-crystallin in MCF-10A immortalized mammary epithelial cells leads to the formation of abnormal mammary acini and induction of neoplastic changes (13). Constitutive overexpression of αB-crystallin in MCF10A cells was found to upregulate expression of extracellular signal-regulated protein kinase 1/2 (ERK1/2) and phospho-ERK1/2 (pERK1/2), as well as AKT and phospho-AKT (pAKT). Only inhibition of the ERK1/2 pathway could oppose the abnormal acini formation induced upon αB-crystallin overexpression. In cell lines and tumor models, AKT and/or ERK1/2 are implicated in cancer progression and metastasis formation (15).
However, it is not known whether αB-crystallin expression in breast tumors is correlated to the enhanced activity of these kinases. Therefore, in this study we determined whether endogenous αB-crystallin expression levels in breast tumors are correlated, first, with the expression of phosphorylated, and thus activated, forms of ERK1/2, AKT and mTOR in breast cancer tissue and, second, with the expression of epidermal growth factor receptor (EGFR) and insulin-like growth factor 1 receptor (IGF-1R), which can activate these kinases (16, 17). Furthermore, the direct effect of αB-crystallin depletion on pERK1/2 expression was studied in vitro, using siRNA-mediated knockdown of αB-crystallin.
Materials and Methods
Patients
The study population was selected from a cohort of breast cancer patients that were surgically treated between January 1991 and December 1996 at the University Medical Centre St. Radboud in Nijmegen, the Netherlands. Selection criteria were: no axillary lymph node invasion, no adjuvant systemic treatment, and at least 5 years of follow-up or an earlier recurrence during follow-up (18, 19). In order to increase the power of our analysis, we designed a tissue microarray (TMA) with a high percentage of triple-negative cancer tissues, and we obtained approximately equally sized groups of breast cancer subtypes. This ultimately led to a cohort of 122 patients. As approved by the institutional review board and according to national law, coded tumor tissues were used.
Immunohistochemistry on breast cancer TMAs
Immunohistochemical analyses were performed on 4-μm thick formalin-fixed, paraffin-embedded human TMA, containing sections of human breast tumors. The specifications for each IHC assay are listed in Table I. Tissue sections were deparaffinized in xylol and rehydrated through a graded ethanol into water series. All sections were pretreated with a 10 mM citrate buffer (pH 6.0) in a microwave oven for 3 minutes at 800 Watt, followed by 10 minutes at 180 Watt. After cooling for 1.5 hours at room temperature (RT), slides were washed in distilled water. To block endogenous peroxidases activity, slides were treated with 3% hydrogen peroxide in distilled water for 10 minutes at RT, followed by washing in distilled water and Tris buffered saline (TBS). The primary antibodies were diluted in 1% BSA-TBS and incubated overnight in a humidified chamber at 4°C. The following day, the sections were washed with TBS and incubated with PowerVision (ImmunoLogic) for 30 minutes at RT. Subsequently, the sections were washed with TBS and the antibody bound was visualized by incubating for 5 minutes at RT with PowerVision DAB (3,3′-diaminobenzide) (ImmunoLogic). After rinsing in distilled water, the sections were counterstained with Mayer's hematoxylin solution, dehydrated through graded ethanol, cleared in xylol and enclosed with Permount (Fisher Scientific). Control tissues were used as a positive controls for each IHC assay as listed in Table I. As negative controls we substituted the primary antibody with 1% BSA-TBS.
Antibodies used for Immunohistochemical Staining
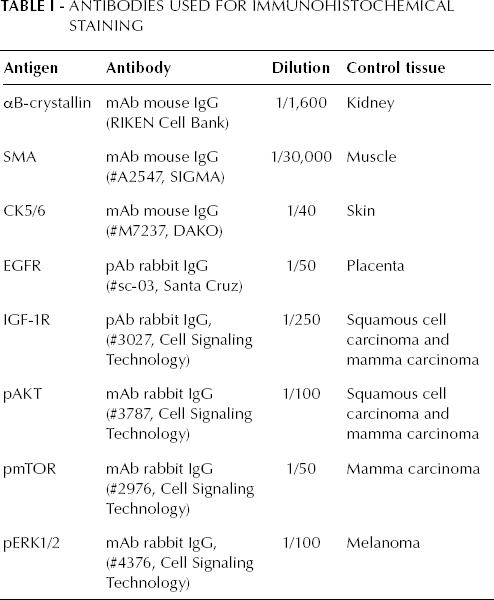
Scoring of IHC results
Scoring of the stainings was performed by eye according to a scoring system where a score ‘0’ is a negative staining and '1' is a positive staining. Cutoff points were determined for each staining. For pAKT, pERK1/2, pmTOR, EGFR, and IGF-1R any observable staining was scored as positive; conversely, CK5/6 and SMA were scored positive when >5% of the cells expressed the protein, and αB-crystallin when >15% of the cells expressed it.
Statistical analysis
Analysis of results was performed with SPSS 16.0 using the Pearson Chi-square test.
Cell culture of triple-negative cell lines
MDA-MB231, MDA-MB436 and MDA-MB468 cells were seeded into T25 flasks and maintained in DMEM + Glutamax™(Invitrogen), supplemented with 10% fetal calf serum (Gibco-BRL). At 80% confluency, the cells were harvested, trypsinized and washed 2 times with PBS; 40 μL of 2% sodium dodecyl sulfate (SDS, Gibco) was then added. Samples were heated twice for 5 minutes at 100°C and sonicated for 10 minutes (1 minute cycles, 30 seconds on, 30 seconds off, Bioruptor®, Diagenode). The samples were then stored at -20°C.
αB-crystallin knockdown in triple-negative breast cancer cells
Approximately 7.75∗10 5 MDA-MB468 cells were seeded into T25 flasks (with 2 flasks per condition) and maintained in DMEM + Glutamax™ (Invitrogen) supplemented with 10% fetal calf serum (Gibco-BRL). After 24 hours, confluency was 40% and cells were transfected with 1.25 ng siRNA/flask using Lipofectamine™ 2000 Reagent according to the manufacturer's protocol (Invitrogen). One mock transfection and 5 different siRNA's were used: mock transfection using MQ; 2 negative control siRNAs: si-EGFP (CGAGAAGCGCGAUCACAUGdTdT) and si-LUC (CGUACGCGGAAUACUUCGAdTdT); and 3 αB-crystallin specific siRNAs: si-αB1 (GCACCCAGCUGGUUUGACAdTdT), si-αB2 (CCCUGAGUCCCUUCUACCUdTdT) and si-αB3 (CCGGAUCCCAGCUGAUGU AdTdT). The medium was refreshed 5 hours after transfection. Forty-eight hours after transfection, cells were harvested, and proteins were isolated as described above. The samples were then stored at -20°C.
Western blotting
The protein concentrations of the lysates were determined with BCA Protein Assay Kit (Thermo Scientific) according to the manufacturer's protocol. Protein samples were separated by electrophoresis on a 12.5% SDS-polyacrylamide gel and transferred to a nitrocellulose membrane (Protran). The membranes were blocked with 5% non-fat dry milk (Elk, Campina) in 1x PBS for 1 hour, and then washed 3 times for 10 minutes with 1x PBS + 0.0025% v/v Nonidet P-40. The membranes were then incubated with the following antibodies: monoclonal mouse-anti-human-αB-crystallin antibody (undiluted, RIKEN), mouse-anti-human-gamma-tubulin as reference (1:3000, Sigma-Aldrich), mouse-anti-human- ERK1/2 (1:200, Cell Signaling) and rabbit-anti-human-phospho-ERK1/2 (1:200, Cell Signaling) diluted in 0.025% w/v Nonidet P-40 and completed with 2% Elk in 1x PBS. After washing, blots were incubated for 1 hour with a 6,000-fold diluted goat-anti-mouse IRDye® 800CW (LI-COR) and 6,000-fold diluted goat-anti-rabbit IRDye® 680CW (LI-COR). Proteins were visualized with the Odyssey scanner (LI-COR). Analysis was performed using Odyssey 2.1 software.
Results
αB-crystallin expression is correlated with triple-negative and basal-like markers
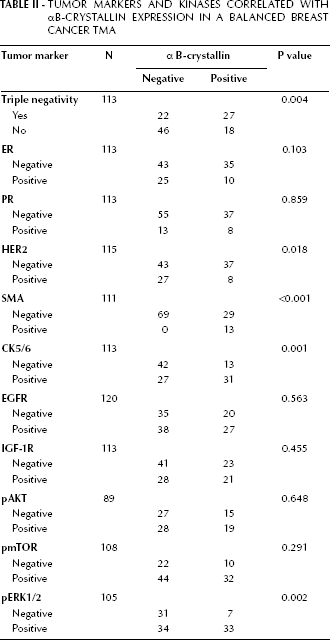
In this study, we used a balanced breast cancer TMA in which 69% of the sections were negative for ER expression, 81% were negative for PR expression, 70% were negative for HER2 expression and 43% were triple-negative. Endogenous αB-crystallin expression was assessed by immunohistochemistry and 39% of the sections scored positive. αB-crystallin expression appeared to correlate with triple negativity (Tab. II, p=0.004), and not, or less well, with the lack of expression of one of the 3 receptors per se (ER, p=0.103; PR, p=0.859; and HER2, p=0.018). Moreover, a correlation of αB-crystallin expression was found also with the basal-like markers SMA (p<0.001), and CK5/6 (p=0.001) (Tab. II). These results suggest that αB-crystallin is a good biomarker for triple-negative tumors and/or basal-like tumors.
Tumor Markers and Kinases Correlated with αB-Crystallin Expression in a Balanced Breast Cancer TMA
αB-crystallin expression is correlated with pERK1/2 expression
As constitutive overexpression of αB-crystallin in the immortalized mammary epithelial cell line MCF-10A was found to upregulate expression of pERK1/2 and pAKT (13), we determined whether a correlation exists between αB-crystallin and pAKT and pERK1/2 in the TMA. Besides pERK1/2 and pAKT, we also analyzed pmTOR, which is an AKT substrate, as well as IGF-1R and EGFR, which are both able to activate the pERK1/2 and pAKT pathways (16, 17) (Tab. II). Remarkably, only pERK1/2 expression showed a significant correlation with αB-crystallin expression (p=0.002), whereas pAKT (p=0.648), pmTOR (p=0.291), IGF-1R (p=0.455) and EGFR (p=0.563) did not show any significant correlation.
αB-crystallin depletion does not affect pERK1/2
The results described above suggest that αB-crystallin may directly or indirectly affect pERK1/2 expression. To further investigate the effects of a reduction of αB-crystallin expression on pERK1/2, the triple-negative breast cell lines MDA-MB231, MDA-MB436, and MDA-MB468 were selected for αB-crystallin knockdown experiments. All 3 cell lines showed expression of pERK1/2 and ERK1/2, but αB-crystallin was only detected on Western blot in MDA-MB468 cells (Fig. 1). Therefore, αB-crystallin was depleted in MDA-MB468 cells, by transfection with 3 different αB-crystallin-specific siRNAs. As controls, mock-transfected cells and cells transfected with unrelated siRNAs (si-LUC and si-EGFP) were included. Although αB-crystallin levels were successfully reduced (Fig. 2a), the results showed that reduction of αB-crystallin in the triple-negative breast cell line MDA-MB468 did not detectably affect the total pERK1/2 levels (Fig. 2b).
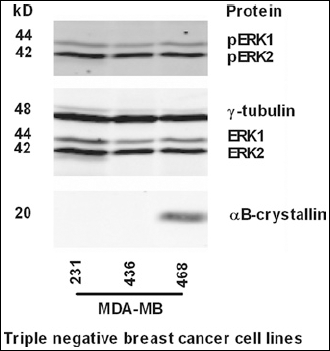
αB-crystallin expression in 3 cell lines derived from triple-negative breast tumors. Western blot analysis of αB-crystallin, ERK1/2 and pERK1/2 expression in the 3 triple-negative breast cancer cell lysates: MDA-MB231, MDA-MB436, and MDA-MB468. α-γ-tubulin was used as loading control.
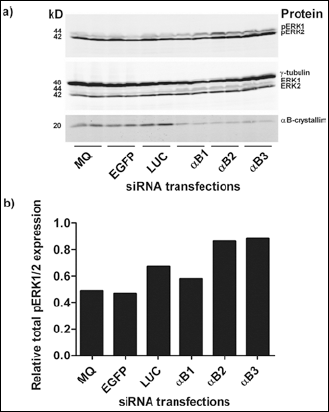
pERK1/2 and ERK1/2 expression upon αB-crystallin depletion. (a) siRNA-mediated αB-crystallin knockdown was performed in MDA-MB468 and analyzed by western blotting using an antibody against αB-crystallin. pERK1, pERK2, ERK1 and ERK2 expressions were detected by using anti-pERK1/2 and anti-ERK1/2 antibodies. α-γ-tubulin was used as a loading control. (b) Quantification of total pERK1/2 expression levels, relative to γ-tubulin expression. The bars in the graph represent the mean of the 2 adjacent samples on Western blots.
Discussion
In this study we describe how in human breast cancer tissues αB-crystallin is related to triple negativity and basal-like tumor markers CK5/6 and SMA. The correlation of αB-crystallin with basal-like breast cancers is in line with other studies that also reported that αB-crystallin is related to poor prognosis in breast cancer (5, 11–14). EGFR, which is also often used as a basal-like tumor marker (6), did not correlate with αB-crystallin in our study, which is in contrast with the findings described by Tsang and coworkers (12). This discrepancy, however, might be explained by the lack of a well-documented standard assay for this marker protein (20).
Patients with triple-negative breast cancers have a poor prognosis (2), partly because specific treatments are not available for these patients. Anti-breast cancer treatments targeting the estrogen, progesterone or HER2 receptors are ineffective in triple-negative breast cancers since they do not express these receptors. Other strategies in cancer treatment involve the use of small molecule kinase inhibitors, which target the intracellular downstream signaling pathways (21). Assessing which kinases are specifically enhanced in triple-negative breast cancers might therefore reveal new targets for treatments using kinase inhibitors in these tumors. For triple-negative tumors, an increased activation of the AKT and ERK1/2 pathways has been described (22), suggesting that targeting these pathways might be beneficial in the treatment of this type of breast cancer. Disruption of the RAS/RAF/MEK/ERK1/2 pathway could be an alternative strategy in particular breast cancer subtypes (23, 24). Alternatively, proteins or mechanisms involved in the upregulation of these kinases might be a potential target for alternative treatment options. Reportedly, αB-crystallin could have an effect on the aggressiveness of tumor cells via upregulation of the AKT or ERK1/2 signaling pathways (13). The correlation between αB-crystallin and pERK1/2 expression, observed in the present study, suggests that αB-crystallin expression specifically influences the ERK1/2 pathway and not the AKT pathway, and is in agreement with the observations of Moyano and coworkers (13). However, in the triple-negative cell line MDA-MB468, knockdown of αB-crystallin did not detectably change pERK1/2 expression. Moyano and coworkers reported an upregulation of pERK2 upon αB-crystallin overexpression in the MCF-10A breast cell line (13) (pERK1 is not expressed in this cell line [23]), and thus a downregulation of at least pERK2 was expected upon siRNA treatment. The reasons why αB-crystallin knockdown did not show an effect on ERK1/2 phosphorylation are not clear. Since αB-crystallin is a heat shock protein, of which the expression is specifically enhanced during stress conditions, it is possible that αB-crystallin affects pERK1/2 expression only when the protein is overexpressed. The correlation of αB-crystallin with triple-negative tumors and ERK1/2 suggests that therapies targeting αB-crystallin might be considered for treatment of triple-negative or basal-like breast cancers. In conclusion, our data suggest that not an αB-crystallin downregulation but rather preventing αB-crystallin upregulation might be an option for such an alternative treatment.
