Abstract
Background and aims
The aim of the present study was to evaluate serum Dickkopf-1 (Dkk-1) as a marker for early detection of hepatocellular carcinoma (HCC), as well as for prognostic prediction of early HCC after hepatic resection.
Methods
One-hundred and four cases of matched fresh tissue specimens of early HCC and adjacent non-tumorous liver tissue (ANLT) were obtained for RT-PCR, qRT-PCR, western blot and immunohistochemistry assays. Sera were collected from patients with early HCC (n=184), benign liver tumors (n=29), cirrhosis (n=174), non-cirrhotic hepatitis B (n=193), and from healthy individuals (n=202). The levels of Dkk-1 and alpha fetoprotein (AFP) were measured.
Results
The Dkk-1 mRNA and protein levels were both upregulated in early HCC. Serum levels of Dkk-1 in patients with early HCC were significantly higher than in the other 4 groups (p<0.001). Dkk-1 had a better sensitivity and accuracy than AFP (p<0.05). More importantly, 73.1% of the patients negative for AFP could be diagnosed with early HCC using Dkk-1. A combination of Dkk-1 and AFP further improved the diagnostic efficacy. Patients with a high serum Dkk-1 level had poorer overall and relapse-free survivals than those with a low Dkk-1 level (p=0.028 and p=0.045, respectively). These results were shown in a testing cohort and confirmed in a validation cohort of patients. Univariable and multivariable Cox regression analyses showed serum Dkk-1 level to be an independent prognostic factor for overall survival.
Conclusions
Our data show that Dkk-1 is a diagnostic and prognostic serologic marker for early HCC.
Introduction
Hepatocellular carcinoma (HCC) ranks as the 5th most common malignancy and the second leading cause of cancer death worldwide, resulting in more than 695,900 deaths each year (1, 2). Despite the great improvements in treatment, the long-term survival of patients with HCC remains unsatisfactory as most patients are diagnosed late, with advanced and metastatic disease (3, 4). However, for HCC detected at a very early and asymptomatic stage by screening, the literature reports 5-year survival rates of 89%-93% after liver resection, and of 71% after percutaneous treatment (5, 6), thus suggesting the importance of detecting and treating HCC at an early stage.
In the past decades, the widespread use of screening has resulted in the detection of small and asymptomatic HCC (7, 8). The screening programs most commonly adopted use ultrasound (US) and serum tumor markers. Computed tomography (CT) and magnetic resonance imaging (MRI) are mainly used to investigate abnormalities identified with ultrasound. Due to their high costs they are rarely used for primary screening (9). The sensitivity and specificity of US for HCC ≥1 cm are, respectively, of 70% and 48%. However, to detect lesions <1 cm, the US has a sensitivity of only 34% (10, 11). Furthermore, the distinction of early HCC from dysplastic nodules in cirrhotic patients by medical imaging without the aid of tumor markers has been shown to be extremely difficult (12). Serum tumor markers have also been used for the diagnosis of HCC. Alpha-fetoprotein (AFP) is the most commonly used “positive control” for novel serum biomarkers screening and identification (13). AFP has a specificity ranging from 76% to 96%, which increases with a higher cutoff value. However, the sensitivity ranges only between 20% and 40% (14–16), as elevated levels of AFP may also be found in patients with chronic liver disease, including hepatitis B, hepatitis C and cirrhosis (17). Other tumor markers, such as Des-γ-carboxy-prothrombin (DCP), Glypican-3 (GPC3), AFP lectin-3 fraction (AFP-L3), and Golgi protein73 (GP73) have significant limitations in diagnosing early HCC (12, 13, 18–20). Recently, the National Comprehensive Cancer Network (NCCN) clinical practice guidelines for HCC (version 1, 2011) indicated that AFP, as well as DCP and AFP-L3, were not considered optimal in detecting early-stage or very early-stage HCC. Therefore, there is an urgent need for novel serum biomarkers with higher sensitivity and specificity for early HCC diagnosis.
We have previously shown that Dickkopf-1 (Dkk-1) was significantly elevated in HCC tissues (21). Dkk-1 is a secreted glycoprotein that plays important roles in Wnt signaling (22, 23), and aberrant Dkk-1 expression has been found in many types of human cancers (24–26). However, data on the clinical significance of Dkk-1 expression in serum of cancer patients are limited (27) and its role in the diagnosis and prognosis of early HCC is still not completely clear.
In the present study, we evaluated the use of Dkk-1 as a possible diagnostic and prognostic serum marker for early HCC.
Materials and Methods
Collection of specimens
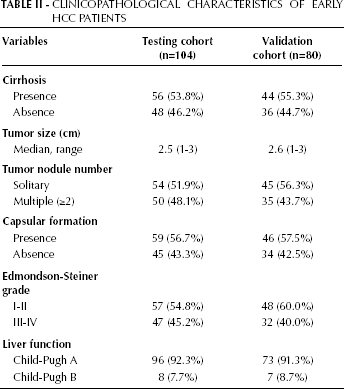
The Guidelines for Tumor Marker Prognostic Studies were used for this analysis (28). During the study period, lasted from June 2001 to October 2008, we collected sera for the testing cohort from consecutive patients operated for early HCC (n=104) or benign liver tumors (n=19) at the Xiangya Hospital of Central South University. For the early HCC patients, matched fresh tissue specimens of early HCC and adjacent non-tumorous liver tissue (ANLT) were also obtained. Specimens from 10 patients with traumatic liver rupture or hemangioma who underwent hepatic resection were also included as normal liver tissue. In this cohort, 43 early HCC patients had a recurrence. During the same time, in our clinics we also collected sera from consecutive patients who were diagnosed with cirrhosis (n=102), and non-cirrhotic chronic hepatitis B (n=109). Sera from healthy individuals who attended our clinic for minor surgical conditions, such as lipoma, sebaceous cyst or hernia, were collected as controls (n=112). We classified tumors with BCLC stage 0+A as early-stage HCC (29). The benign liver tumors included focal nodular hyperplasia (n=7) and hepatic cavernous hemangioma (n=12). The diagnosis in all patients with early HCC or benign liver tumors was confirmed histopathologically. Patients with chronic liver disease, including cirrhosis and non-cirrhotic chronic hepatitis B, were diagnosed using the Guidelines from the American Association for the Study of Liver Diseases (AAS-LD). Healthy individuals were chosen from people with no history or blood-test results suggestive of liver disease or hepatitis (individuals with negative virus profile or those who only had positive anti-HBs). Patients who had a history of other solid tumors were excluded from the study.
At the end of the first part of the study, sera from a second set of patients were similarly collected from November 2008 to March 2012 for the validation cohort. This cohort consisted of patients who were operated for early HCC (n=80) or benign liver tumors (n=10), patients with cirrhosis (n=72), non-cirrhotic chronic hepatitis B patients (n=84), and healthy individuals (n=90). In this cohort, 33 early HCC patients had a recurrence.
Demographic and clinical information were collected prospectively from each patient (Tab. I and II). Prior informed consent was obtained from all the individuals involved in this study for collection of serum specimens, in accordance with the Guidelines of the Xiangya Hospital, Central South University. The study protocol was approved by the ethics committee of the Xiangya Hospital.
Demographic Characteristics of Patients in the Testing and Validation Cohort
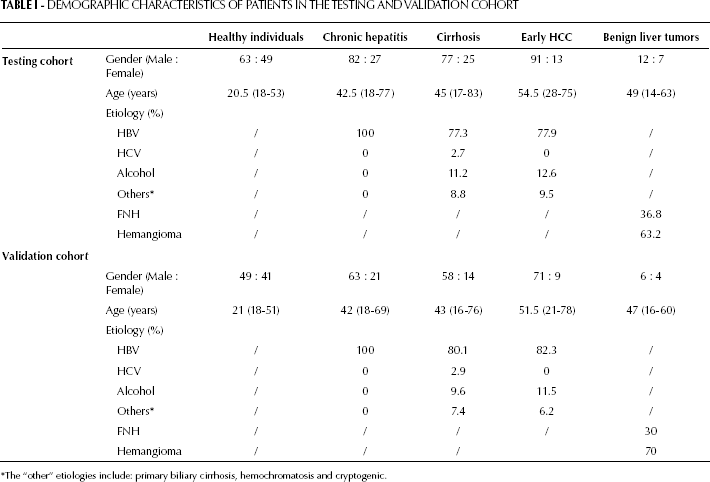
The “other” etiologies include: primary biliary cirrhosis, hemochromatosis and cryptogenic.
Clinicopathological Characteristics of Early HCC Patients
Serum Dkk-1, AFP or Combined Dkk-1 and AFP in the Differentiation of Early HCC Patients from Non-HCC Patients and Cirrhotic Patients
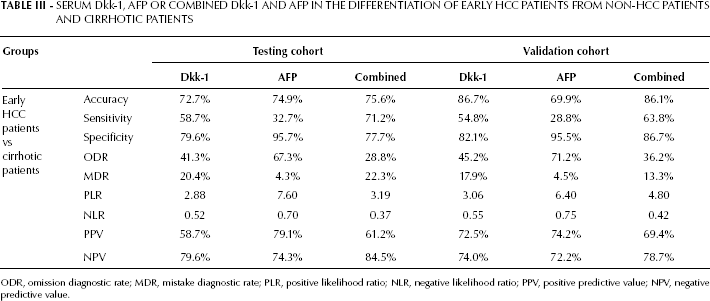
ODR, omission diagnostic rate; MDR, mistake diagnostic rate; PLR, positive likelihood ratio; NLR, negative likelihood ratio; PPV, positive predictive value; NPV, negative predictive value.
Semi-quantitative RT-PCR
Total RNA was isolated by using Trizol reagent (Invitrogen, CA, USA) according to the manufacturer's instructions. The Dkk-1 primers we used are the following: forward, 5′-AGCACCTTGGATGGGTATTC-3′; reverse, 5′-CCAGGTTTTTGGAAGGTGTT-3′. GAPDH expression was determined as control using the following primers: forward, 5′-GGTGAAGGTCGGAGTCAACG-3′; reverse, 5′-CAAAGTTGTCATGGATGACC-3′. Total RNA (2.0 μg) was reverse transcribed and amplified by initial polymerase activation for 5 minutes at 94°C, 32 cycles at 94°C for 30 seconds, 53°C for 30 seconds, 72°C for 30 seconds, followed by extension at 72°C for 4 minutes. The PCR product (5 μL) was then electrophoresed on 2.0% agarose gel, and the intensity of the bands was quantified using the Eagle Eye II laser densitometry program (Strategene, La Jolla, USA). The level of Dkk-1 mRNA was expressed as the relative intensity of the PCR product bands of the target sequences to that of GAPDH. All PCR experiments were done in triplicates.
qRT-PCR
qRT-PCR was performed on ABI Prism 7300 Real-Time PCR System (Applied Biosystems, Foster, CA). The primers for Dkk-1 were the following: forward, 5′-GACCCAGGCTTGCAAAGTGAC-3′; reverse, 5′-CGCTACCATCGCGACAAAGA-3′. GAPDH expression was determined as control using the following primers: forward, 5′-GCACCGTCAAGGCTGAGAAC-3′; reverse, 5′- TGGTGAAGACGCCAGTGGA-3′. qRT-PCR amplification was conducted in a 25 μL reaction using the SYBR® Premix Ex Taq™ (Takara, Dalian, China). All amplification reactions were performed in triplicates. PCR product quality was monitored using post-PCR melting curve analysis. The results were analyzed using the 2-ΔΔCT method. For analyzing the expression of Dkk-1 in HCC tissues, ΔΔCt = (CtHCC-CtGAPDH) - (CtANLT-CtGAPDH).
Western blotting
The total protein content was extracted from fresh tissue specimens and 100 μg of total protein were separated by SDS-PAGE and then transferred onto a PVDF membrane (Millipore, Bedford, MA, USA). The blotted membranes were incubated first with rabbit anti-human Dkk-1 polyclonal antibody (Santa Cruz, CA, USA) and then horseradish peroxidase-linked goat anti-rabbit antibody (Santa Cruz, CA, USA). The beta-actin protein was also determined as a loading control by using the specific antibody (Santa Cruz, CA, USA). All experiments were repeated in triplicates and the levels of Dkk-1 protein were quantified by densitometry (Beckman, South Pasadena, Canada).
Immunohistochemistry
The formalin-fixed paraffin-embedded sections were stained for Dkk-1 (Santa Cruz, CA, USA, 1:100) using the streptavidin-peroxidase system (Zhongshan Goldenbridge Biotechnology, Beijing, China). Antigen retrieval was performed by microwave treatment with EDTA buffer (1 mM, pH 8.0).
Measurement of serum expressions of Dkk-1 and AFP
The expression of serum Dkk-1 was measured by ELISA. In brief, pre-coated 96-well microplates (Greiner Bio-One, Frickenhausen, Germany) with a mouse monoclonal antibody specific to Dkk-1 (R & D Systems, Minneapolis, USA) were placed at 4°C overnight. After washing with PBS, they were blocked with 10% bovine serum albumin (BSA) and 0.5% Tween 20 in PBS for 2 hours at room temperature. Then, 2-fold diluted sera were added to the wells and incubated for 2 hours at room temperature. After washing away any unbound substances, a rabbit polyclonal antibody specific to Dkk-1 (Santa Cruz, CA, USA) was added to the wells as a capture antibody and the wells were incubated for 2 hours at room temperature with horseradish peroxides-linked goat anti-rabbit antibody (KPL, Gaithersburg, USA). Subsequently, a substrate solution was added to the wells. After incubating at room temperature for 30 minutes, the reaction was stopped with 0.2 M H2SO4. Finally, the optical density was measured at a wavelength of 450 nm and corrected for the absorbance at 650 nm. The concentrations of Dkk-1 were determined by interpolation by a standard curve, which was established by using serial dilutions of recombinant human Dkk-1 (R & D Systems, Minneapolis, USA). All measurements were done in duplicates. Using the same specimens, serum AFP was measured with a commercially available ELISA (R & D Systems) according to the manufacturer's instructions. When the concentration of AFP was less than 0.31 ng/mL (the lowest limit of the standard curve), the value was set as equal to 0. All measurements were done in duplicates.
Additionally, intra and inter-assay controls were performed to check for the quality of the data in the present study. We randomly selected 2 serum samples and tested the Dkk-1 and AFP levels as described above. The calculated intra-assay coefficients of variation (CV) for these 2 samples were 2.3% and 2.7% for Dkk-1 and 1.9% and 2.4% for AFP. These results confirmed a good intra-assay repeatability of these measurements (CV <5%). To assess the inter-assay CV, the Dkk-1 and AFP levels were measured in 2 different dilutions of the Dkk-1 standard samples (6.25 ng/mL and 1.56 ng/mL) and of the AFP standard samples (12.5 ng/mL and 100 ng/mL). Measurement of each of these samples was repeated 6 times in 6 different 96-well microplates. The inter-assay CVs were 11.2% and 12.0% for Dkk-1 and 12.1% and 12.9% for AFP. These results indicated that the inter-assay repeatability was not satisfactory (CV >10%). Therefore, a standard curve for each 96-well microplate was needed.
In this study, we adopted the STARD statement for reporting studies of diagnostic accuracy (28). To avoid any subjective bias of the researchers, the serum samples were randomly allocated and the researchers were double-blinded throughout the entire serological test. The serum collectors did not take part in the measurement of the markers. Every serum specimen had an equal opportunity to be distributed into any well of the ELISA plate using the random number table. The inspector did not know the origin and the group the specimens belonged to. We uncovered the blinded data and analyzed the data only after all the serum specimens had been tested.
Follow-up
Patients with HCC were regularly followed up at intervals of once every 1 to 2 months in the first year after surgery. Subsequent follow-up was at an interval of once every 3 months. At each follow-up visit, an US was performed and serum AFP was measured. Patients who were suspected to have tumor recurrence were further investigated with CT or MRI. The follow-up period was defined as the interval between the date of the operation and the date that the patient died or the date of the last visit. This first part of the study, involving the testing cohort, was censored on October 31, 2010 and deaths from causes other than HCC were included. The second part of the study, involving the validation cohort, was censored on March 31, 2012. The relapse-free survival was defined as the interval between the operation and the date that tumor recurrence or metastasis were diagnosed. Overall survival was defined as the interval between the operation and the date of death of the patient. Thirty-nine patients died in the testing cohort and 24 patients died in the validation cohort. To determine the factors influencing survival after hepatic resection, 10 variables were considered in patients with early HCC in the testing cohort: gender, age, hepatitis B status, preoperative AFP level, cirrhosis, number of tumor nodes, capsular formation, Edmondson-Steiner grade, vascular invasion and Dkk-1 expression level.
Statistical analysis
SPSS 13.0 for Windows (SPSS Software, Chicago, USA) and GraphPad Prism 5 Demo (Graph Pad Software, San Diego, CA) were used for statistical analysis. Fisher's exact test was used for categorical data, whereas the independent t test was used for continuous data. Dkk-1 and AFP values are reported as mean ± standard deviation (SD). The area under the receiver operating curve (AUROC) was calculated and compared using the Z test. Survival curves were constructed using the Kaplan-Meier method and evaluated using the log-rank test. The Cox proportional-hazards regression model was used to identify factors that were independently associated with overall survival. All tests were two-tailed and a p<0.05 was considered statistically significant.
Results
Dkk-1 was overexpressed in early HCC tissues
To examine the expression of Dkk-1 in early HCC, semi-quantitative RT-PCR was used to measure mRNA expression in 104 cases of early HCC tissues and their correspondent ANLTs. The expression levels of Dkk-1 mRNA in early HCC tissues were higher than those in ANLTs. qRT-PCR results showed that the average expression level of Dkk-1 mRNA in early HCC tissues was 3.43 times higher than that in ANLTs (Fig. 1A). Additionally, by using western blotting and immunohistochemistry (IHC), the Dkk-1 protein levels in the same early HCC tissues were also detected (here reported as mean ± SD). Consistent with the mRNA levels, Dkk-1 protein levels in early HCC tissues were significantly higher than those in ANLTs (1.35±0.42 vs 0.72±0.27, p<0.01; Fig. 1B). The Dkk-1 expression in ANLTs was slightly higher than that in normal liver tissue but did not reach a statistically significant difference (p>0.05; Fig. 1A and B). Interestingly, we found that the Dkk-1 tissue levels were not different among early HCC patients who were either positive or negative for serum AFP (Fig. 1C). This suggests that Dkk-1 expression level in early HCC might not correlate with AFP. Besides, the IHC results showed that a positive Dkk-1 expression (Fig. 1D) was detected in 86.6% of the early HCC tissues (90 of 104), indicating that Dkk-1 is frequently upregulated in early HCC.
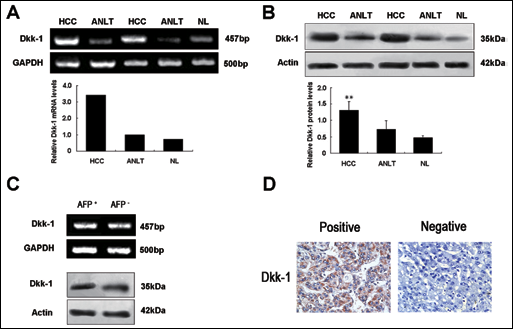
Detection of Dkk-1 mRNA and protein expression in HCC. (A) Representative results showing the expression patterns of Dkk-1 mRNA in tissue specimens determined by semi-quantitative RT-PCR (upper panel) and qRT-PCR (lower panel). (B) Representative Western blot results showing that Dkk-1 protein levels in HCC tissues were significantly higher than those in ANLTs. (C) Representative results showing the mRNA and protein expression patterns of Dkk-1 in early HCC tissues with AFP either positively or negatively expressed. (D) Representative immunohistochemistry results of Dkk-1 level in early HCC tissues. Abbreviations: Dkk-1, dickkopf-1; HCC, hepatocellular carcinoma tissue; ANLT, adjacent non-tumorous liver tissue; NL, normal liver tissue; **, p<0.01.
Dkk-1 as a serum marker for diagnosis and prognosis of early HCC
Considering that Dkk-1 is upregulated in early HCC tissues and that it is a secreted glycoprotein, we speculated that Dkk-1 may be detected in serum and may constitute a serum marker for the diagnosis and prognosis of early HCC, especially in cases in which serum AFP is negative. Therefore, we designed 2 cohorts (a testing cohort and a validation cohort) to carry out the serological study of Dkk-1 in early HCC.
Part I of the serological study on the testing cohort
Dkk-1 levels in Serum of early HCC
The Dkk-1 levels in sera from patients with early HCC (n=104), cirrhosis (n=102), non-cirrhotic chronic hepatitis B (n=109), benign liver tumors (n=19) and from healthy individuals (n=112) were detected by ELISA (Fig. 2A). The mean serum level of Dkk-1 in patients with early HCC was 5.81±2.76 ng/mL, and was significantly higher than that in patients with cirrhosis (4.28±1.98 ng/mL, p<0.001), non-cirrhotic chronic hepatitis B (3.60±2.21 ng/mL, p<0.001), benign liver tumors (3.23±1.15 ng/mL, p<0.001) and healthy individuals (2.02±1.63 ng/mL, p<0.001). Although the serum levels of Dkk-1 in cirrhosis, non-cirrhotic chronic hepatitis B and benign liver tumors were higher than in healthy individuals (p<0.05), no significant differences were found among these 3 groups (p>0.05). More importantly, serum Dkk-1 level in the group of early HCC patients decreased after surgery (3.97±2.44 ng/mL, p<0.001) and increased again when HCC recurred (n=43, 5.54±2.52 ng/mL, Fig. 2A). Additionally, serum Dkk-1 levels linearly correlated with the Dkk-1 protein levels in the corresponding HCC samples (Fig. 3, r=0.898, p<0.001).
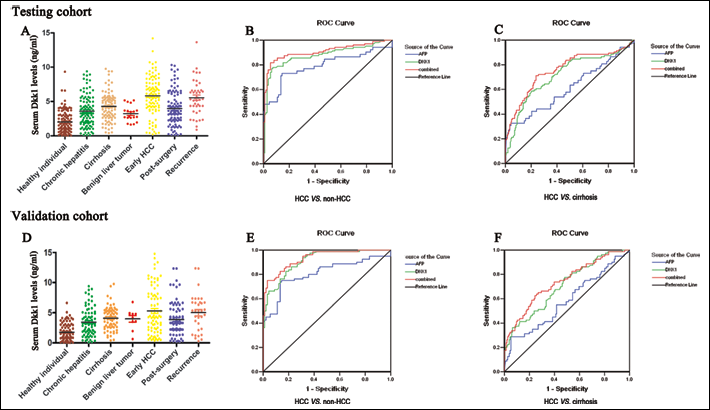
Dkk-1 serum levels in the testing and validation cohorts and their applications in the early diagnosis of hepatocellular carcinoma. (A, D) Dkk-1 levels were determined in the sera of the different groups of patients using ELISA. Data were analyzed with the independent t test. (B, E) ROC curves indicating that the serum levels of Dkk-1, AFP, or the combination of the two, could differentiate early HCC from non-HCC patients. (C, F) ROC curves showing that the serum levels of Dkk-1, AFP, or the combination of the two, could differentiate early HCC from chronic liver disease patients. ROC, receptor operation curve; AUROC, area under the ROC.
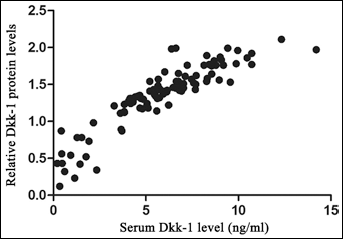
Relationship between serum Dkk-1 levels and Dkk-1 protein levels in the corresponding HCC samples. The serum Dkk-1 levels were linearly correlated with the Dkk-1 protein levels in the corresponding HCC samples (r=0.898, p<0.001).
The efficacies of serum Dkk-1 and AFP in early HCC diagnosis
The diagnostic efficacies of serum Dkk-1 and AFP in early HCC were compared using ROC curve analysis. For Dkk-1 the optimal cutoff level was set at 4.14 ng/mL (identified by the Youden index), whereas for AFP it was set at 20 ng/mL. The results of the test to discriminate patients with early HCC from non-HCC patients (including healthy individuals, patients with chronic liver disease or benign liver tumor) showed that Dkk-1 had a better sensitivity (76.0% vs 50.0%, p<0.05), specificity (93.8% vs 89.3%, p<0.05), accuracy (85.2% vs 70.4%, p<0.05), positive predictive value (91.9% vs 81.3%, p<0.05) and negative predictive value (80.8% vs 65.8%, p<0.05) (Fig. 2B). The AUROC of Dkk-1 was significantly larger than that of AFP (0.877 vs 0.793, p<0.05; Fig. 2B). Moreover, 67.3% (35/52) of the early HCC patients with a negative serum AFP could be positively diagnosed using Dkk-1 (Tab. IV).
Serum Dkk-1 in the Diagnosis of Early HCC in Patients WHO were Serum AFP-Negative
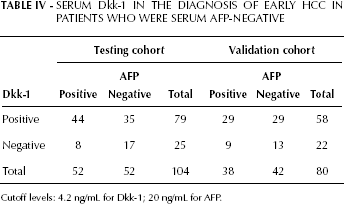
Cutoff levels: 4.2 ng/mL for Dkk-1; 20 ng/mL for AFP.
Interestingly, serum Dkk-1 and AFP levels were not correlated (Spearman rank correlation coefficient: r=0.141; p>0.05), suggesting that a combination of Dkk-1 and AFP may be helpful to further improve the diagnostic efficacy of early HCC. To verify this hypothesis, 3 different ROC curves were constructed as to assess the value that could combine these 2 tests and could thus discriminate early HCC from non-HCC patients. Our results showed that the AUROC, sensitivity, accuracy, and negative predictive value of the combined test were, respectively, 0.908, 86.5%, 85.6% and 87.2%. These parameters were significantly better than those found for AFP (p<0.001), although they were similar to those found for Dkk-1 (p>0.05; Fig. 2B).
As most patients with HCC have underlying cirrhosis, the efficacy of Dkk-1 in the differentiation between patients with early HCC and cirrhosis was investigated. Based on the ROC curves (Fig. 2C), the optimal cutoff values of Dkk-1 and AFP were respectively of 5.53 ng/mL and 502.43 ng/mL. The sensitivity of Dkk-1 was significantly better than that of AFP (58.7% vs 32.7%; p<0.05; Tab. III). Moreover, the sensitivity of the combined test was better than those of Dkk-1 or AFP alone (71.2% vs 58.7% vs. 32.7%; p<0.05; Tab. III). Additionally, the AUROC of Dkk-1 was significantly larger than that of AFP (0.717 vs 0.612; p<0.05; Fig. 2C).
Dkk-1 as a serum marker for prediction of long-term outcomes of patients with early HCC after hepatic resection
The relationship between the levels of serum Dkk-1, the clinicopathological characteristics and the postoperative survivals of patients with early HCC was investigated. The serum Dkk-1 levels were significantly higher in patients with early HCCs without capsules (p=0.039) and with vascular invasion (p=0.018). However, there was no significant association between the serum Dkk-1 levels and the other clinicopathological variables (p>0.05).
Using a cutoff value of 4.14 ng/mL for Dkk-1, patients with early HCC (n=104) were divided into a high expression group (n=79) and a low expression group (n=25). The patients with a high serum Dkk-1 level had a poorer overall survival (median survival: 32.1 months vs 41.2 months, p=0.028; Fig. 4A) and relapse-free survival (median relapse-free survival: 30.3 months vs 39.1 months, p=0.045; Fig. 4B) than those with a low expression level.
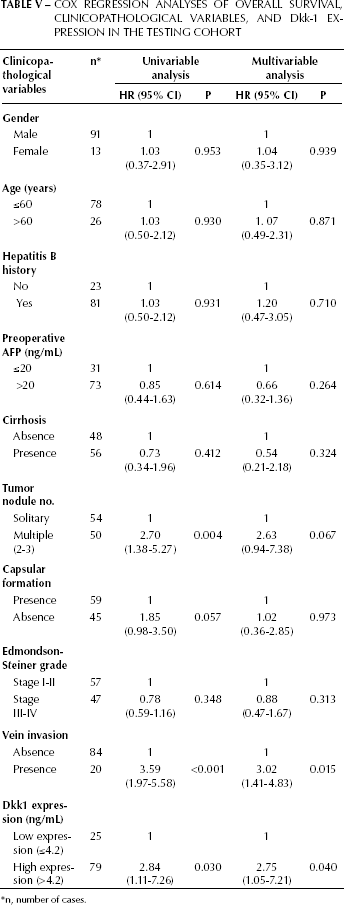
Univariable Cox regression analysis indicated that serum Dkk-1 level (hazard ratio [HR] 2.84; p=0.030), multiple tumor nodules (HR 2.70; p=0.004) and vascular invasion (HR 3.59; p<0.001) were prognostic factors for overall survival (Tab. III). Multivariable Cox regression analysis showed that serum Dkk-1 level (HR 2.75; p=0.040), and vascular invasion (HR 3.02; p=0.015) were independent prognostic factors for overall survival (Tab. V). The other clinicopathological variables, including serum AFP level, did not add any independent prognostic information.
Cox Regression Analyses of Overall Survival, Clinicopathological Variables, and Dkk-1 Expression in the Testing Cohort
n, number of cases.
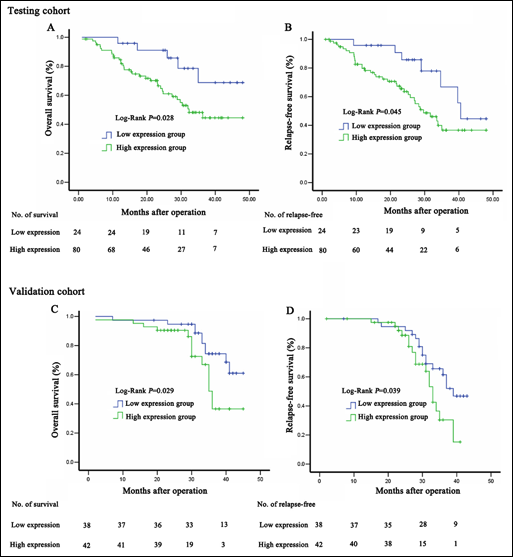
Survival analysis of the serum Dkk-1 levels with overall and relapse-free survival of patients with HCC after hepatic resection.
Part II of the serological study on the validation cohort
Dkk-1 levels in the serum of early HCC
The validation cohort consisted of patients with early HCC (n=80), cirrhosis (n=72), non-cirrhotic chronic hepatitis B (n=84), benign liver tumors (n=10), and healthy individuals (n=90). Similarly to the testing cohort, we found that serum levels of Dkk-1 in patients with early HCC (5.30±3.86 ng/mL) were significantly higher than those in patients with cirrhosis (4.11±1.90 ng/mL, p<0.001), non-cirrhotic chronic hepatitis B (3.45±2.21 ng/mL, p<0.001), benign liver tumors (3.99±1.70 ng/mL, p=0.001), and healthy individuals (1.78±1.41 ng/mL, p<0.001). Additionally, in the group of early HCC patients, serum Dkk-1 levels were significantly lower after surgery (3.87±2.74 ng/mL) when compared to the levels before surgery (p<0.001) or during HCC recurrence (n=33, 5.02±3.09 ng/mL, p=0.001; Fig. 2D).
The efficacies of serum Dkk-1 and AFP in early HCC diagnosis
To identify whether in the validation cohort the diagnosis efficacies of Dkk-1 and AFP were as good as in the testing cohort, we used the ROC curve analysis maintaining for both markers the cutoff level previously specified (4.14 ng/mL for Dkk-1 and 20 ng/mL for AFP). We obtained similar results in the validation cohort (Fig. 2E). For Dkk-1 and AFP, sensitivity was respectively 73.8% and 48.8% (p<0.05), specificity was respectively 95.6% and 88.9% (p<0.05), accuracy was respectively 80.6% and 70.0% (p<0.05), the positive predictive value was respectively 92.7% and 79.6% (p<0.05), and the negative predictive value was respectively 74.8% and 66.1% (p<0.05). Similarly, 69.0% (29/42) of early HCC patients with a negative level of AFP could be diagnosed using Dkk-1 (Tab. IV). When the combined test was used to differentiate patients with early HCC from non-HCC patients, the AUROC was 0.933, sensitivity was 78.8%, accuracy was 82.9%, positive predictive value was 84.0% and negative predictive value was 82.1%. These parameters were significantly better than those for AFP alone (p<0.05), although they were similar to those for Dkk-1 alone (p>0.05; Fig. 2E and Tab. III).
The ROC curve analysis was also used to differentiate between early HCC and cirrhosis at an optimal cutoff value of 5.53 ng/mL for Dkk1 and 502.43 ng/mL for AFP (Fig. 2F). The sensitivity, accuracy and AUROC of Dkk-1 were also significantly better than AFP (respectively 54.8% vs 28.8%; p<0.001; 86.7% vs 69.9%; p<0.001; 0.706 vs 0.581; Tab. III and Fig. 2F). When used to differentiate early HCC from cirrhosis, the combined test had a better sensitivity (63.8% vs 28.8%), accuracy (86.1% vs 69.9%), negative predictive value (78.7% vs 72.2%) and a larger AUROC (0.738 vs 0.581) than those for AFP (p<0.05). (Fig. 2F and Tab. III).
Dkk-1 as a serum marker for prediction of long-term outcomes for patients with early HCC after hepatic resection
The value of Dkk-1 as prognostic serum marker also needed to be validated. In the validation cohort 65 of the 80 patients had at least 2 years of follow-up. Similarly, patients with a high serum Dkk-1 level (>4.14 ng/mL) had a poorer overall survival (median survival: 35.6 months vs 40.3 months, p=0.029) and relapse-free survival (median survival: 33.0 months vs 39.0 months, p=0.039) than those with a low expression level (<4.14 ng/mL) (Fig. 4C and 4D). Multivariable Cox regression analysis also showed that serum Dkk-1 level (HR 2.95; p=0.036) and vascular invasion (HR 3.14; p=0.012) were independent prognostic factors for overall survival (Tab. VI).
Cox Regression Analysis of Overall Survival, Clinicopathological Variables, and Dkk-1 Expression in the Validation Cohort

Number of cases.
Discussion
As patients with early HCC are usually asymptomatic, the majority of HCC patients are incurable due to the fact that they are usually diagnosed only at a late stage with advanced liver disease (3, 4). The rationale behind screening for HCC is to diagnose these tumors early and at a stage when they are still potentially curable. Unfortunately, until now, patients are rarely diagnosed with early HCC, mainly because of the limitations of the presently available screening tests (5).
Screening using AFP and US to detect subclinical HCC has been implemented in many countries (30). Although some studies showed encouraging results, the efficacy of these screening programs in patients with high risk of developing HCC remains controversial. Bolondi et al found firstly that the annual cost to screen 2,000 chronic hepatitis B and hepatitis C carriers in order to detect one HCC case was of USA $1,167, and secondly that to detect one HCC that was treatable the cost was of USA $1,667 (31). This cost-effectiveness rate is unacceptably high, especially for developing countries, and it needs to be improved. Since the majority of HCC patients (>85%) have underlying cirrhosis, there is a great difficulty to distinguish between early HCC and dysplastic nodules using the existing diagnostic methods. Since for this clinical setting surveillance is recommended, high specific and sensitive serologic biomarkers are needed. Also, as US depends on the experience of the sonographer and the quality of the equipment, it is a rather subjective technique and the result is often non-reproducible (13). Conversely, testing of serum markers is objective, reproducible and inexpensive, suggesting that it is more suitable for early HCC screening. Many serum diagnostic markers of HCC have been identified, and AFP is the most widely used (32). However, the sensitivity of AFP is poor (approximately 20%-30%) at a cutoff value of 100 ng/mL (33). The sensitivity of AFP in our study was of only 28.8%-50% in 2 independent cohorts at a cutoff value of 20 ng/mL. AFP-L3, an isoform of AFP, displays a sensitivity which ranges from 45% for lesions <2 cm to >90% for lesions >5 cm, and a specificity of more than 95% in HCC (34, 35). The major limitation of AFP-L3 is its use in the early diagnosis of HCC. Yamashiki reported that only 35% of small-lesions HCC patients were detected by AFP-L3 (36). DCP, also known as PIVKA-II (protein induced by vitamin K absence or antagonist-II), has been proved to be a diagnostic serum marker for HCC, with a sensitivity of 48%-62% and a specificity of 81%-98%, which are comparable to AFP (37, 38). Unfortunately, only 15%-30% of early HCC present significant concentrations of serum DCP as to be detectable, and this limits its implementation in a screening program (39).
Overexpression of Dkk-1 has been found in the tissues of many types of human cancers (24–26). Yamabuki et al found that serum Dkk-1 levels were significantly higher in patients with lung and esophageal cancer than in healthy controls (27). The proportion of Dkk-1-positive patients was 70.0% in non-small cell lung cancers, and 63.0% in esophageal squamous cell cancer, whereas only 4.8% of healthy volunteers were falsely positive, suggesting the effectiveness of Dkk-1 as a diagnostic biomarker for lung and esophageal cancer (27). Yu et al found that high Dkk-1 expression in cancer tissues was an independent predictor of poor overall and relapse-free survival for HCC patients (26). Sato et al found that serum Dkk-1 was elevated in HCC patients (24). Tung et al found that the serum level of Dkk-1 was upregulated in HCC and both serum and tissue Dkk-1 levels had prognostic significance in HCC. These results indicated that serum Dkk-1 was a potential tumor marker with prognostic value for HCC (40). However, these studies did not illustrate the diagnostic efficacy of Dkk-1 in HCC, especially in early HCC. A recent large-scale and multicenter study indicated that serum Dkk-1 could be a protein biomarker for the diagnosis of HCC (41). However, this result needs more research to be confirmed. Moreover, the prognostic value of Dkk-1 was not fully illustrated in their research. In the present study, we found that Dkk-1 was highly elevated in serum of patients with early HCC. Furthermore, serum Dkk-1 had a better accuracy, sensitivity, and specificity than AFP in distinguishing between patients with early HCC from non-HCC patients and cirrhosis. Our study suggests that Dkk-1 may be useful for the detection of early HCC. Additionally, around 67.3%-69.0% of patients with early HCC but with a negative AFP could be diagnosed by Dkk-1. More importantly, the combined use of Dkk-1 and AFP significantly increased the sensitivity in the detection of early HCC, without any obvious reduction in its specificity. These data indicated that the diagnostic performance of serum Dkk-1 in early HCC was better than the other biomarkers, and that the combined testing of Dkk-1 and AFP was the best way to diagnose early HCC, especially for those patients who were negative for AFP. Additionally, in the present paper a significant difference was shown between healthy individuals and the patients' groups with risk of HCC development (chronic hepatitis and cirrhosis). These findings, which are different from the results of Shen et al (41), demonstrate that further follow-up studies are needed to estimate the diagnostic value of Dkk-1 for chronic liver disease, and to validate its use as diagnostic marker in clinical practice.
Another major concern is whether serum markers for HCC can predict the long-term outcomes of patients after hepatic resection of HCC (42). We found that the serum level of Dkk-1 was significantly correlated with the absence of tumor capsule and vascular invasion of early HCC, and an elevated level of serum Dkk-1 was associated with worse overall and relapse-free survival in patients with early HCC, after hepatic resection. Our study also showed that serum Dkk-1, but not AFP, was an independent prognostic factor for early HCC after hepatic resection for long-term survivals.
In conclusion, our study showed that the serum levels of Dkk-1 were highly elevated in patients with early HCC. Moreover, serum levels of Dkk-1 decreased after surgery for early HCC patients but increased again when these HCC patients had a recurrence. Serum Dkk-1 alone or in combination with AFP significantly improved the diagnostic efficacy of early HCC with a sensitivity, specificity and accuracy higher than AFP alone. Furthermore, a high serum Dkk-1 level was an independent poor predictor for the long-term survival of early HCC patients after hepatic resection. These results have been shown in a testing cohort and have then been confirmed in a validation cohort of patients. Our findings suggested that Dkk-1 may be a good diagnostic and prognostic serologic marker for early HCC.
