Abstract
BACKGROUND:
MicroRNAs (miRNA) are promising biomarkers for cancer diagnosis and prognosis; miR-100 expression is decreased in cervical cancer tissues.
OBJECTIVE:
To determine whether miR-100 is a useful biomarker for early cervical cancer diagnosis.
METHODS:
Total RNA was extracted from the sera of 34 healthy controls (HC), 64 cervical intraepithelial neoplasia patients (CIN), and 46 cervical cancer patients (CC). miR-100 expression levels were measured with quantitative real-time PCR. Correlations between clinicopathological factors and miR-100 expression levels were also assessed. The cut-off value for miR-100 was calculated from the Receiver Operating Characteristic (ROC) curve.
RESULTS:
Relative expression levels of miR-100 in serum were 1.84
CONCLUSIONS:
Serum miR-100 may be a useful diagnostic biomarker for CC, and for predicting lymph node metastasis and disease free survival in CC patients.
Introduction
Cervical cancer (CC) is a major cause of mortality in women; approximately 311,000 women worldwide die of this disease every year [1]. Human papillomavirus (HPV) infection is the leading cause of CC [2]. CC is histologically divided into two types: squamous cell carcinoma (SCC), which arises from cervical intraepithelial neoplasia (CIN), and adenocarcinoma, which arises from intraepithelial adenocarcinoma [3]. Based on the relative risk of cervical cancer, CIN1 and CIN2-3 are regarded as low-grade and high-grade CIN, respectively [4]. SCC antigen in blood is a well-known tumor marker for CC, and a tumor-associated antigen of SCC [5]. However, this marker rarely shows an increase in early stage cancers [6].
MicroRNAs (miRNAs) have been drawing attention as novel regulatory molecules for genomic information [7]. MiRNAs are short chain RNAs, approximately 20–25 bases, that do not directly code for proteins but rather regulate various physiological activities in living organisms by suppressing the expression of target mRNAs [7]. Many miRNAs are involved in the downregulation of target mRNAs in various biological processes [8]. MiRNAs bind mainly to the 3’-untranslated region (UTR) of target mRNAs and restrain their translation [8]. In addition, miRNAs play roles in both promoting and suppressing the tumors because they strongly influence the expression of tumor oncogenes and suppressor genes. A single miRNA can have many targets; it is possible that miRNAs regulate about one-third of human genes [9]. The function of miRNA expression in cancer cells is not completely clear. Nevertheless, previous studies have reported that various diseases, including cancers, are accompanied by changes in miRNA expression patterns [10]. Thus, miRNAs are regarded as promising biomarkers in cancer diagnosis and treatment.
Altered miRNA expression in CC lesions is partially related to chromosomal alternations. In a previous study, decreased miR-100 and miR-125 expression in CIN and CC samples was found to be associated with a chromosomal loss [11]. Li et al. have shown that miR-100 expression in CC tissues, which is associated with cell proliferation, apoptosis, and the cell cycle, was reduced [10]. Moreover, miR-100 was found to be hypo-expressed in advanced cervical small cell carcinoma tissue samples [12]. MiRNAs embedded in small exosome granules were found to stably exist in extracellular body fluids, such as saliva, serum, and urine [13]. Moldovan et al. described a quantitative real-time PCR (qRT-PCR)-based method to analyze circulating miRNAs in exosomes from serum [14]. The miR-9, miR-192, and miR-205 expression levels were elevated in serum and tissue samples of CC patients [15]. Serum miRNAs may this serve as biomarkers for diagnosis or prognosis of patients with CC.
We searched the literature for miRNAs that were downregulated in cervical cancer tissues and identified three candidate miRNAs: miR-34a, miR-100, and miR-125 [10, 11]. We measured the expression of each miRNA in the sera of 10 cervical cancer patients using RT-PCR, and miR-100, whose expression was the most downregulated, was chosen for further study. In this study, miR-100 expression levels in serum and cervical tissues were measured to determine whether miR-100 could be useful in the diagnosis and prognosis of CC.
Materials and methods
Clinical samples
The research protocol was approved by the Institutional Review Board of Tokyo Medical University (approval no. 930). This study was conducted according to the Declaration of Helsinki. From April 2010 to April 2012, 144 patients took part in this study at the Tokyo Medical University Hospital; of these individuals, 46 were patients with CC, 64 were patients, with CIN, and 34 were healthy controls (HC). Among HCs, patients with a history of uterine malignancy were excluded. Patients with CC were staged using the International Federation of Gynecology and Obstetrics (FIGO) staging system for CC. Patient backgrounds were obtained through interviews. Informed consent was obtained from all individuals participating in the study. A 10-mL volume of blood samples was collected from individuals before surgery, chemotherapy, and radiation therapy at Tokyo Medical University Hospital. Cervical tissues were collected from the patients who had had a biopsy or hysterectomy. CIN and CC were diagnosed by histopathological examinations.
Laser capture microdissection
Cervical tissue blocks were cut into 5-
Serum preparation and total RNA extraction
Blood samples were centrifuged at 3000 rpm for 5 min and separated into blood cells and serum. Serum was stored at
Characteristics of the 144 study participants
Characteristics of the 144 study participants
CC: cervical cancer; CIN1: cervical intraepithelial neoplasia grade 1; CIN2: cervical intraepithelial neoplasia grade 2; CIN3: cervical intraepithelial neoplasia grade 3; HC: healthy controls; SCC: squamous cell carcinoma.
MiRNAs were quantified using TaqMan MicroRNA Assays (Applied Biosystems, Foster City, CA, USA) with modifications and miRNA-specific stem-loop primers [17] (hsa-mir-100; Applied Biosystems). First, each miRNA was specifically converted into cDNA, according to the manufacturer’s protocol, using TaqMan miRNA RT-Kit with a stem-loop RT-primer and the Applied Biosystems 9800 Fast Thermal Cycler. Second, qRT-PCR was performed for miR-100, using RT-primers with Universal Master Mix, on the Applied Biosystems StepOnePlus
The miR-100 expression profile was used for creating a Receiver Operating Characteristic (ROC) curve, a graphical plot of the true positive rate versus the false positive rate. The cut-off value of miR-100 expression was calculated by ROC analysis.
Statistical analysis
The “EZR” Software was used to perform statistical analysis of the causal relation between the patient background and miR-100 expression pattern [18]. The expression levels of miR-100 were presented as mean
Results
Characteristics of the study subjects
Table 1 summarizes the characteristics of the study participants. Out of the 46 patients with CC, four had stage IA, 21 had stage IB, four had stage IIA, 10 had stage IIB, three had stage III, and four had stage IV (Table 2). Of the 64 patients with CIN, 14 were CIN1, 18 were CIN2, and 32 were CIN3.
Relationship between serum miR-100 expression and clinicopathological factors
Relationship between serum miR-100 expression and clinicopathological factors
We examined whether there was a difference in miR-100 expression levels in the sera of patients with CC or CIN, and HC. MiR-100 concentrations were measured in 46 patients with CC, patients with 64 CIN subjects, and 34 HC. The miR-100 expression levels were significantly downregulated in sera of patients with CC, but not CIN, when compared with those of HC (
Serum miR-100 expression levels in patients with cervical cancer (CC), patients with cervical intraepithelial neoplasia (CIN), and healthy controls (HC), as determined with qRT-PCR. This graph illustrates that the miR-100 expression level was significantly downregulated in sera of patients with CC in comparison to that in HC, but not in the sera of patients with CIN.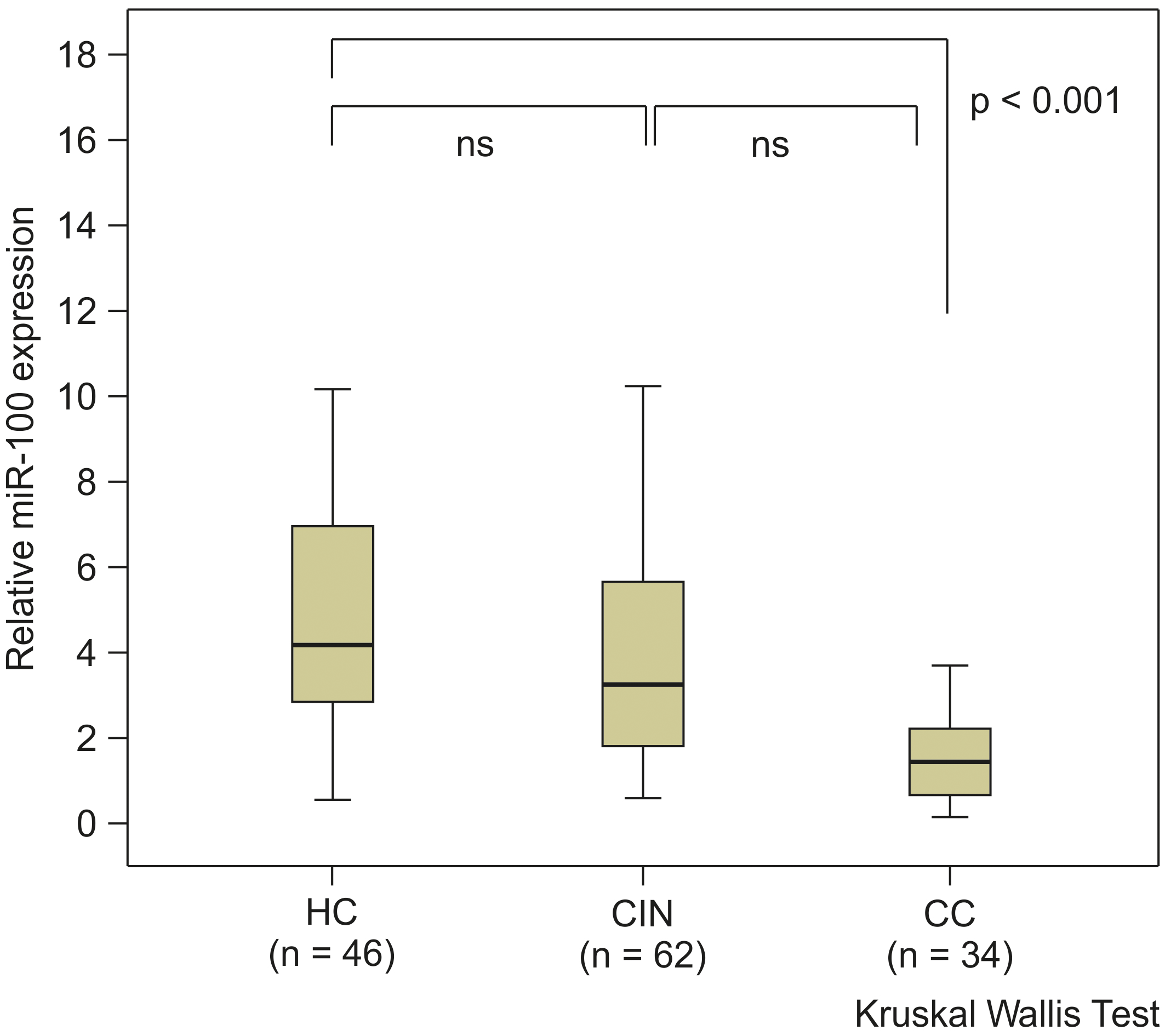
Graph indicating the receiver operating characteristic (ROC) curve. The ROC curve shows that serum miR-100 expression levels could distinguish patients with cervical cancer (CC) from healthy controls (HC), with an AUC of 0.879. With the cut-off at 2.451, miR-100 had 91.2% FPF and 80.4% TPF for patients with CC compared to HC.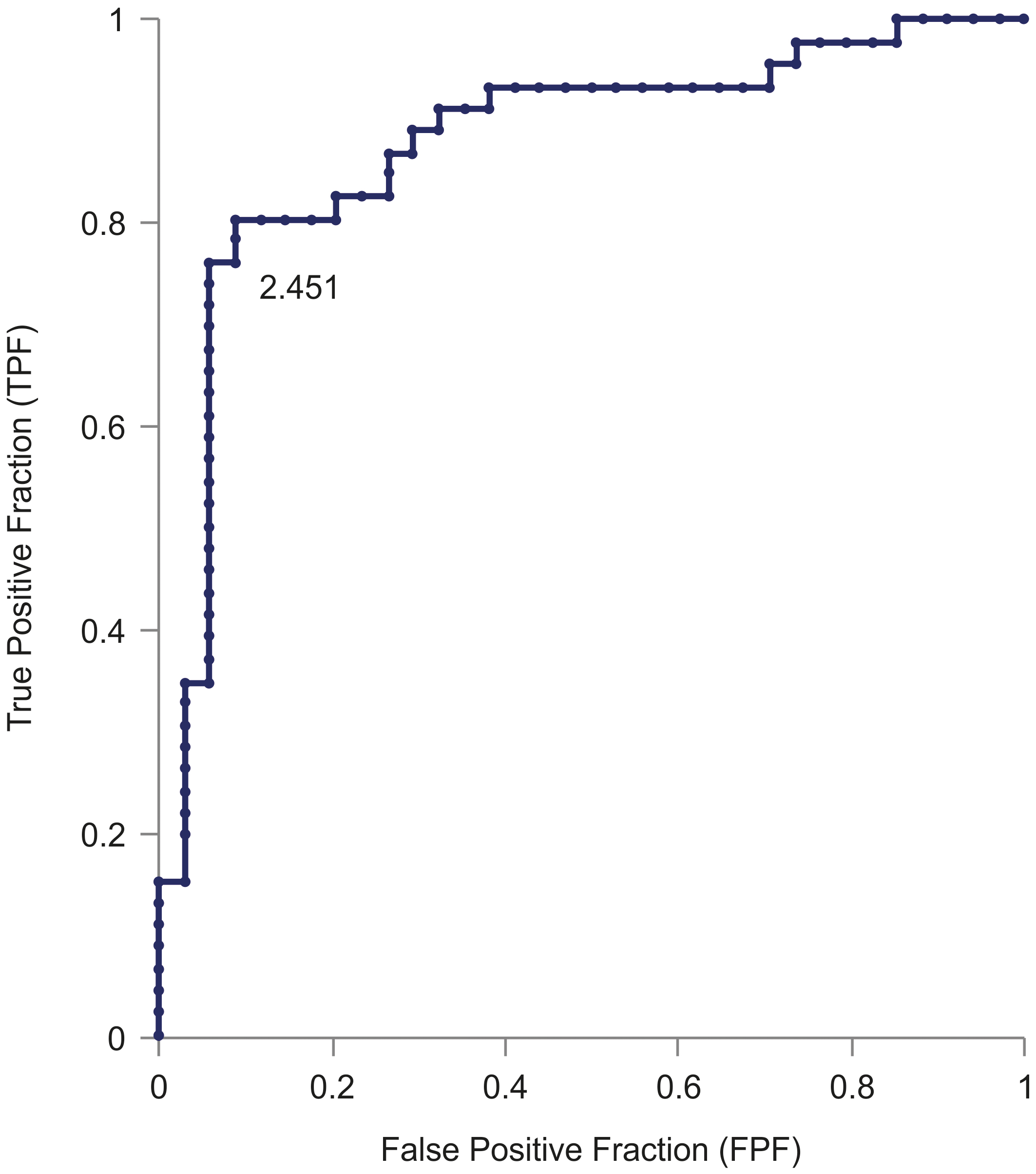
The ROC curve showed that serum miR-100 levels were able to distinguish between patients with and HC; the area of ROC was 0.879 (95% confidence interval, 0.7976–0.9609; Fig. 2). The cut-off value was calculated from the ROC curve to be 2.451 (relative expression level), at which the sensitivity of miR-100 to CC was 91.2% and specificity was 80.4% in comparison to the healthy subjects.
The expression levels of serum miR-100 in each group were as follows: HC, 4.1225; CIN1 group, 4.0055; CIN2 group, 3.1205; CIN3 group, 2.9905; CC stage IA group, 1.00575; stage IB group, 1.466; stage IIA group, 0.747; stage IIB group, 1.12; stage III group, 1.829; and stage IV group, 1.418 (Fig. 3). The median serum miR-100 expression levels decreased as the CC stage progressed. No significant difference was found in the histological type between squamous cell carcinoma and adenocarcinoma (Table 2). However, there was a significant difference in lymph node metastasis (
Serum miR-100 expression levels in patients with cervical cancer (CC) or cervical intraepithelial neoplasia (CIN) at each stage. This chart illustrates that the median expression level of serum miR-100 decreased as the CC stage progressed (healthy group: 4.1225, CIN1 group: 4.0055, CIN2 group: 3.1205, CIN3 group: 2.9905, cervical cancer stage IA group: 1.0058, stage IB group: 1.466, stage IIA group: 0.747, stage IIB group: 1.12, stage III group: 1.829, and stage IV group: 1.418).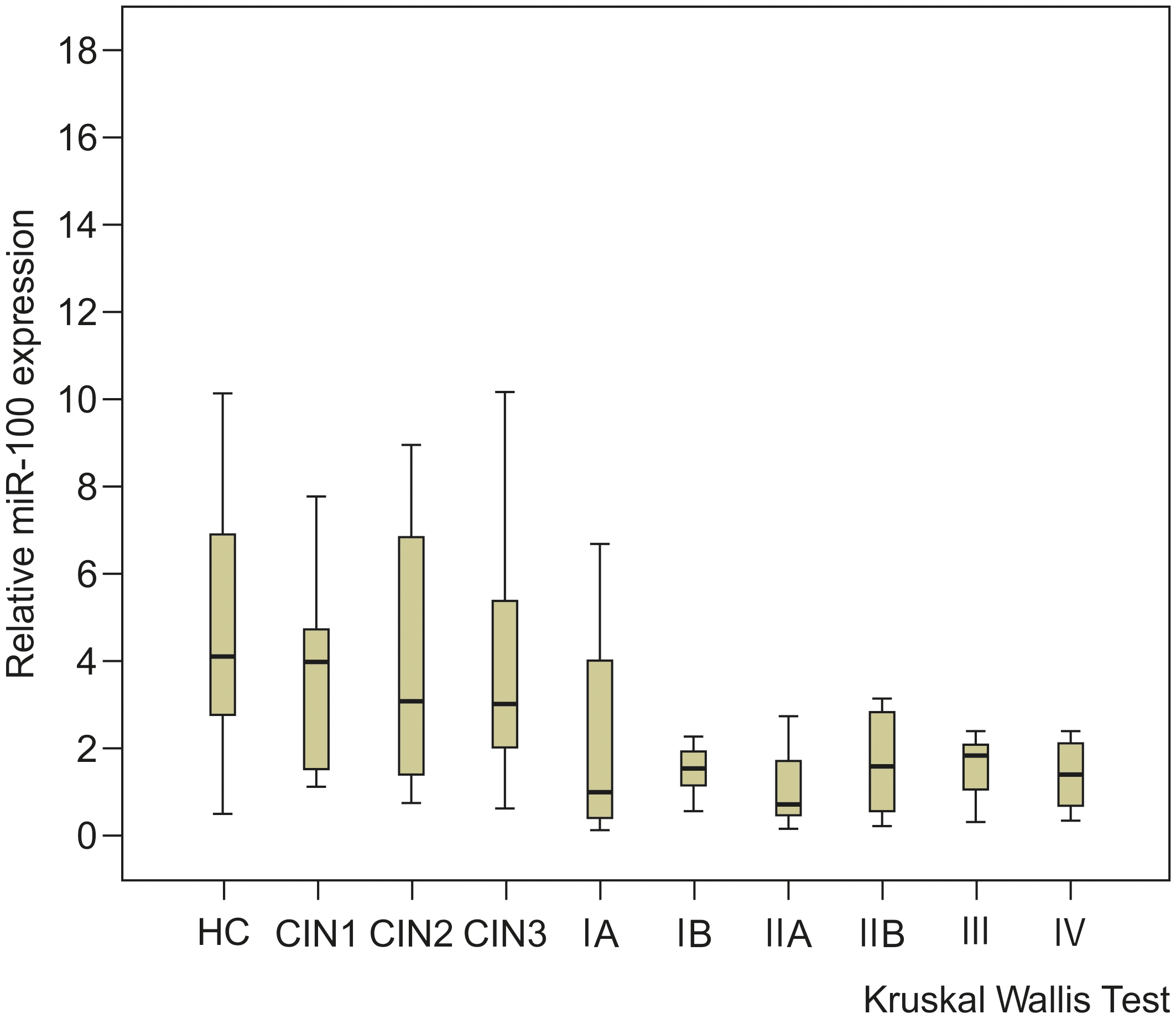
Graph indicating the expression level of miR-100 in normal cervical epithelium (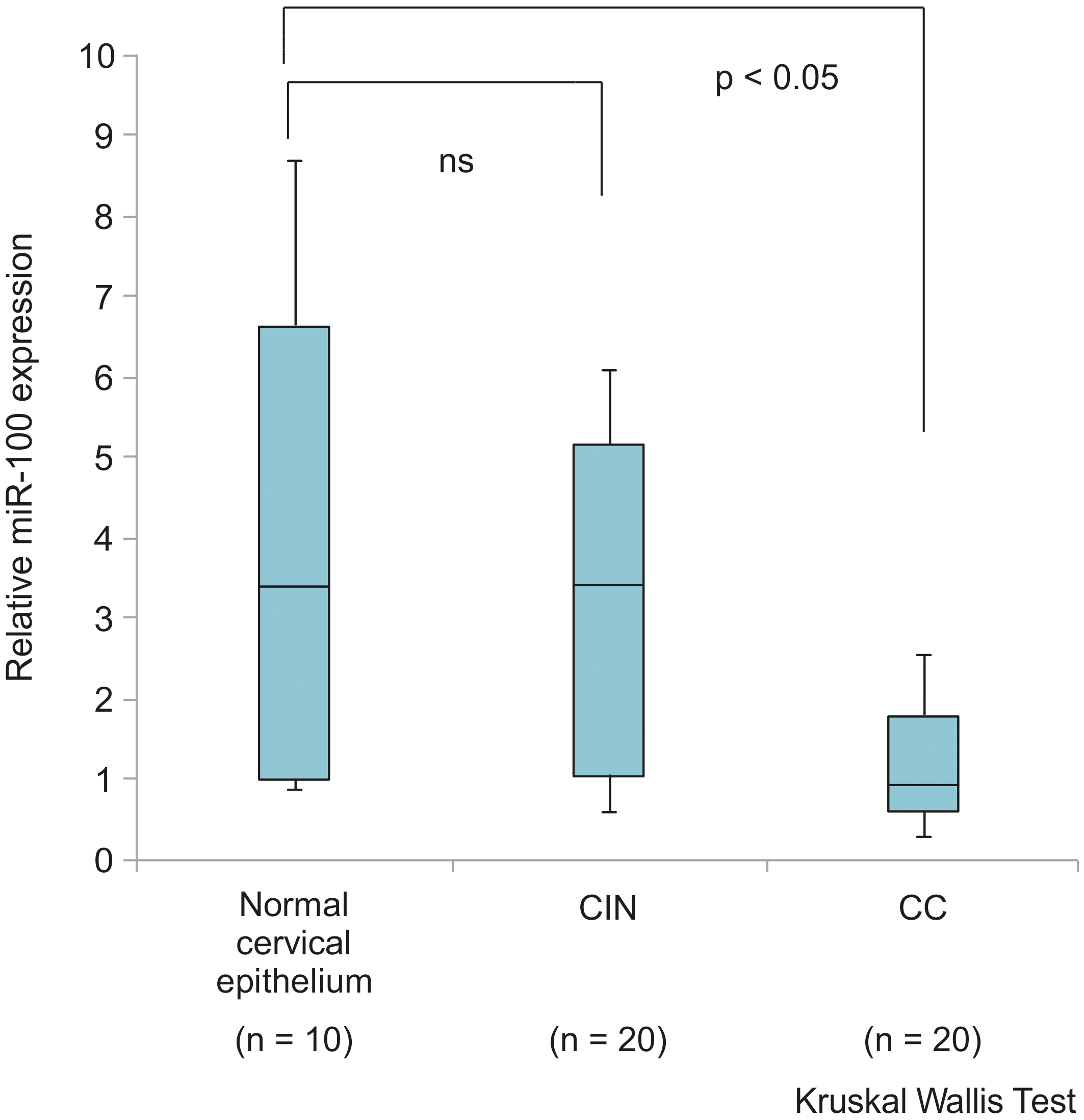
Expression of miR-100 in cervical intraepithelial neoplasia (CIN) (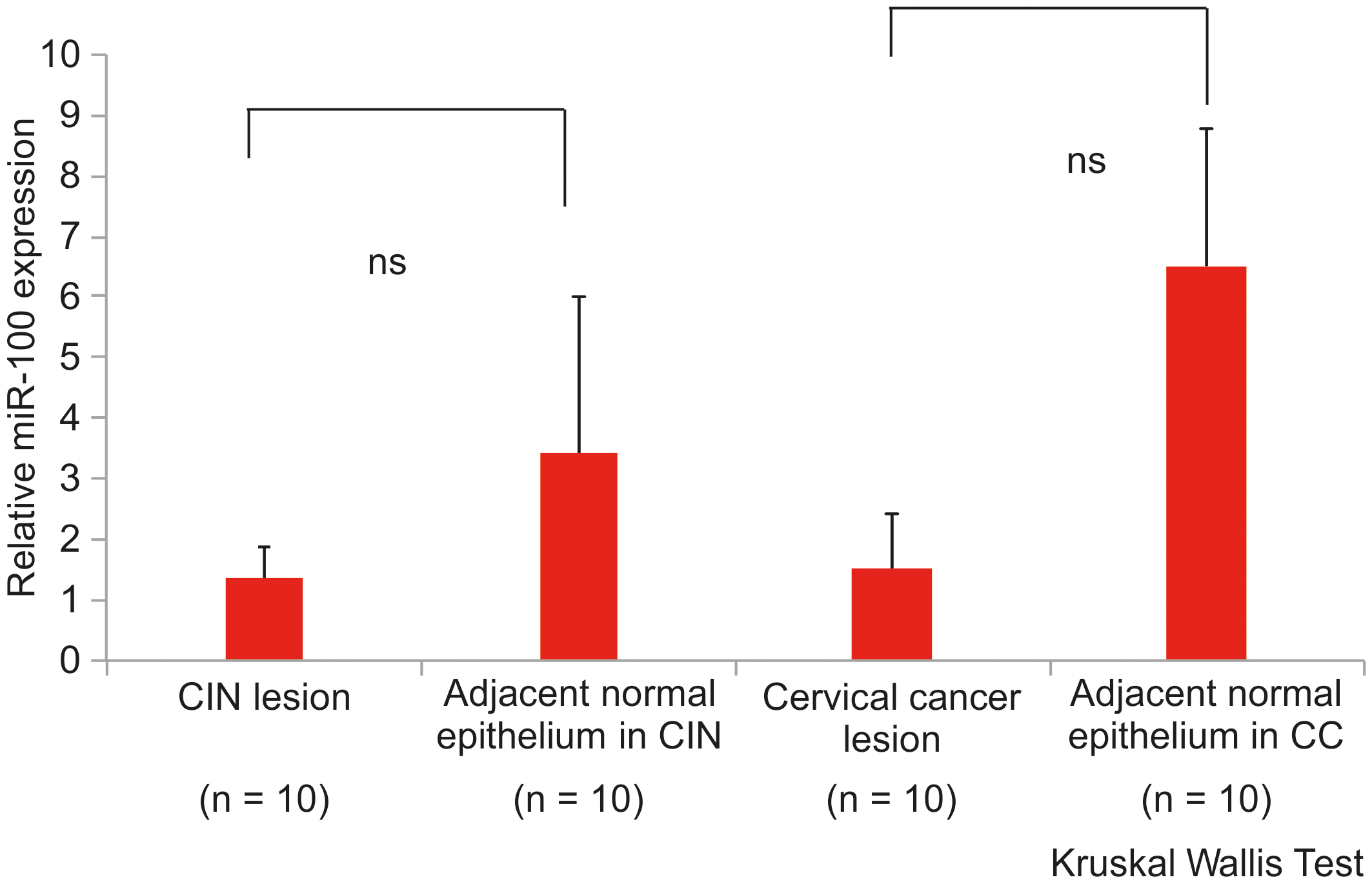
Kaplan-Meier plots of overall survival and disease-free survival curves according to the status of serum miR-100 expression. Individuals with an miR-100 expression level under 2.451 (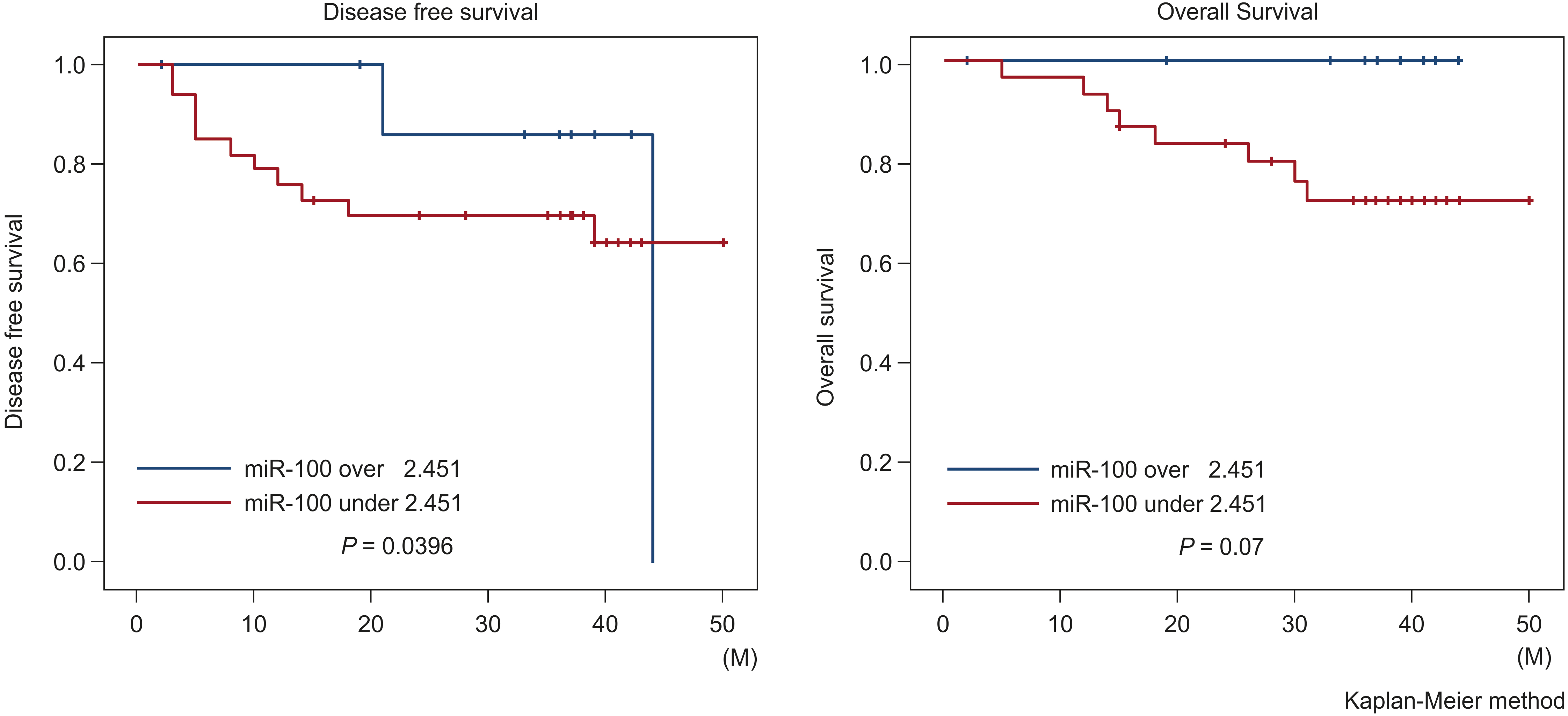
The miR-100 expression levels were significantly higher in the tissues of HC than in those of patients with CC (
Survival and serum miR-100
Kaplan-Meier curve indicated that disease-free survival (DFS) in patients with miR-100 expression level under 2.451 was significantly worse than that of patients with miR-100 level over 2.451 (
Discussion
Some biomarkers can permit early detection of cancers, and contribute to improving the survival rate. Early cancer biomarkers are thus highly important for diagnosis and prognosis of cancers. SCC antigen is a tumor marker for cervical SCC; however, the SCC antigen level is not often upregulated in the early stages of CC [19]. Many clinicians have been looking for new effective biomarkers for early CC detection.
Altered microRNA expression is observed in CC and precancerous lesions. A systematic review showed that miR-21 and miR-29a are the most frequently decreased miRNAs during CC progression [20]. In a meta-analysis, miR-100 rs1834306 was not found to be associated with CC development; however, this meta-analysis included only two studies related to miR-100 [21]. Li et al. showed that reduced miR-100 expression is associated with CC development by abolishing the inhibition of the target gene polo-like kinase 1 (PLK1), which likely accrues relatively late in carcinogenesis [22]. The miR-100 gene is present on Chromosome 11q24.1 (
The discovery of circulating miRNAs has brought great scientific progress in recent years. Circulating exosomal miRNAs have been realized to be useful biomarkers for cancer patients, because of their non-invasive approach and high stability [32]. Circulating miRNA profiles are believed to be unique to each cancer type, and reflect the primary tumor focus [33]. Several studies have revealed the usefulness of serum miRNAs as potential biomarkers that may aid in detection of cancer. It has been reported that the expression levels of serum miR-100 are correlated with different cancers, such as colorectal and lung cancers [34]. Serum miRNAs may be useful biomarkers in cancer diagnosis, and decrease the frequency of invasive biopsies. In this study, we first used qRT-PCR to examine the serum miR-100 expression in HC, and in patients with CIN and CC. The serum miR-100 expression was highest in HC, intermediate in patients with CIN, and lowest in patients with CC. Furthermore, serum miR-100 expression level was higher in patients with low-grade CIN than those with high-grade CIN. As the CC stage progressed, the miR-100 expression level tended to decrease. Furthermore, reduced serum miR-100 levels were related to positive lymph node metastasis and time to relapse. These results indicate that serum miR-100 may be useful in predicting CC progression and prognosis.
As far as we know, this is the first study to probe miR-100 expression levels and its clinical significance, in patients with CC. However, stage IIA had lower expression than stage IIB, III and IV as shown in Fig. 3. One possible cause is the small population at each stage of cervical cancer. Therefore, it is necessary in the future to expand the study with more cases and analyze the expression level of serum miR-100 at each stage of cervical cancer. There were three positive lymph node metastases cases in stage IIA, which may have contributed to the low serum miR-100 levels in this experimental group. Using LCM (not just a follow-up test), this study showed that miR-100 expression level is decreased in CC tissues. In addition, the expression pattern of miR-100 tended to be increased in the surrounding noncancerous lesions in comparison with CC and CIN lesions. These results suggest that miR-100 may have a role in the progression of CIN to CC. Furthermore, the reduced expression of miR-100 in CC tissues may lead to decreased expression of serum miR-100 in CC. Mahdieh et al. showed that the expression level of serum miR-192 was upregulated in CC tissues [16]. This result suggests that circulating miRNAs in blood reflect cancer origin-specific expression signatures.
Limitations of this study include low sample size and selection bias of healthy individuals. Blood samples and cervical tissues were collected only once, and it is unclear whether treatment for CC or CIN affects miR-100 expression levels.
In conclusion, we found that miR-100 in serum and tissue was downregulated in parallel in patients with CC. Serum miR-100 may be a useful biomarker in the diagnosis of CC, especially in the prediction of lymph node metastasis and prognosis. Further studies are needed to completely investigate the function of miR-100 in CC. Serum miR-100 may possibly not only be a predictive marker for CC, but also a marker for determining the efficacy of treatment for cervical cancer by observing the temporal changes in serum miR-100 expression levels after treatment.
Footnotes
Acknowledgments
This work was supported by the Grants-in-Aids (Grant no. 24592533) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) and the Third-Term Comprehensive Control Research for Cancer and Research on Biological Markers for New Drug Development (Grant no. 201118007B) conducted by the Ministry of Health, Labour and Welfare of Japan (MHLW). We would like to thank Editage (www.editage.com) for English language editing.
Author contributions
Conception: Zenta Yamanaka, Kazuyoshi Kato.
Interpretation or analysis of data: Akina Yamanaka, Toru Sasaki.
Preparation of the manuscript: Zenta Yamanaka.
Revision for important intellectual content: Zenta Yamanaka, Hirotaka Nishi.
Supervision: Hirotaka Nishi.
