Abstract
Aim
To explore the significance of circulating tumor cells (CTCs) detection in the course of preoperative chemotherapy (PC) and their effect on the outcomes.
Methods
Fifty-five patients with stage II/III invasive breast cancer were enrolled into a preoperative clinical trial. Patients were given PC with sequential single-agent doxorubicin and paclitaxel vs paclitaxel followed by doxorubicin. Blood samples (8 mL) were collected from patients before PC, after each phase, and at 6 months intervals during follow-up. Peripheral blood mononuclear cells were isolated and enriched for epithelial cells. Quantitative RT-PCR was used to determine the presence of cytokeratin 19 (CK19) mRNA. Samples were considered positive when the PCR curve crossed the standard threshold curve.
Results
After the first phase of chemotherapy, there was a 59% overall reduction in the median tumor volume. The percentage of volume reduction did not differ between patients who presented with detectable CTCs at baseline and those who did not (p=0.89). After the second phase of chemotherapy, there was a further decrease in the median tumor volume to 93% from baseline. There was no correlation between the lack of response and the presence of CTCs either after the first (p=0.36) or second (p=0.5391) phases of PC. The presence of CTCs was a predictor of local or distant relapse (p=0.0411). The detection of CTCs did not affect overall survival (p=0.2569).
Conclusion
CTCs can be used as predictors of relapse after definitive treatment of locally advanced breast cancer; however, CTCs detection in peripheral blood during the course of PC does not implicate a particular pattern of response to treatment.
Keywords
Introduction
Circulating tumor cells (CTCs) can be detected in the peripheral blood of 10%-40% of patients with breast cancer, depending on the clinical setting and the method of detection (1-5). However, the clinical significance and utility of CTCs remain unclear. Studies of CTCs in patients with metastatic breast cancer suggest that the presence and quantity of CTCs can predict disease progression and survival; however little is known about the relevance of CTCs in patients with earlier stages of the disease in the preoperative setting (6-9).
Preoperative chemotherapy (PC) is a generally accepted standard-of-care treatment for locally advanced breast cancer and has become an acceptable alternative for operable breast cancer (10, 11). Predicting patients' responses to a PC regimen through the detection of CTCs in peripheral blood seems to be a very exciting application that could allow changing the line of treatment without the need for repeated biopsies.
The aim of this study was to determine the ability of CTCs to predict the response to PC or failure after treatment in patients with stage II/III breast cancer.
Materials and Methods
Patient cohort
From June 2000 to May 2004, 62 patients with stage II/III invasive breast cancer were enrolled into a phase II randomized trial of PC with sequential single-agent doxorubicin (4 cycles at 60 mg/m2 every 2 weeks) and paclitaxel (9 cycles at 80 mg/m2 weekly). Patients were randomized to receive doxorubicin followed by paclitaxel (group 1) or paclitaxel followed by doxorubicin (group 2). The study was approved by the institutional review board of the Massachusetts General Hospital and the Dana-Farber Cancer Institute. Written consent was obtained from each patient (Clinicaltrials/gov NCT00096291).
Per protocol, MRI and ultrasound measurements of breast tumor size were performed before and after each treatment phase. The MRI-measured tumor volume was used as a surrogate for clinical response because it has been shown to be reliably and accurately correlated with the histologically measured tumor size (12, 13) and the final histopathologic response (14, 15). In addition, blood samples were collected from all patients for detection of CTCs as a pre-planned correlative analysis.
Circulating tumor cell collection and measurement
Several methods have been developed for the detection of CTCs, among these the most widely used include the reverse transcription-polymerase chain reaction (RT-PCR), which seems to be more sensitive, and the immunomagnetic/fluorescent approach, which seems to be more specific (2).
In the current study we used a mixture of both approaches through an initial immunoseparation step followed by the RT-PCR step. In a trial to achieve a reasonable balance between sensitivity and specificity, we used 2 types of markers for the detection of CTCs. The first was cytokeratin 19 (CK19), which is considered a marker of high sensitivity but low specificity for breast cancer cells (1-5), the second marker was BS106, also known as Small Breast Epithelial Mucin (SBEM), which exhibits a high specificity for breast cancer cells (16). It has been previously shown that a panel of markers, rather than a single marker gene, decreases the likelihood of false positive results (17).
Blood samples were collected from 55 patients prior to starting chemotherapy, after the first chemotherapy drug (doxorubicin or paclitaxel), after the second chemotherapy drug or prior to surgery, and every 6 months during follow-up. The mononuclear cell fraction from 8 ml of blood was isolated from CPT tubes using gradient centrifugation, it was washed, counted, and frozen down in 10% DMSO using a rate-controlled freezer. Cells were stored at −140°C. Circulating tumor cells were enriched by immunomagnetic selection using BerEP4 antibody (anti-EpCAM) attached to Dynal magnetic beads (Dynabeads®). RNA was purified using Qiagen RNeasy kits. RNA was amplified by RT-PCR specific for CK19, BS106 and beta-2-microglobulin (B2M). Amplification was detected in real time using Beacon probes. Blood samples were considered positive if the PCR curve crossed the threshold before 45 cycles.
Statistical analysis
The study of CTCs measurements, their changes and correlations with tumor response and clinical outcomes was conceived as a secondary pre-planned correlative analysis of clinical trial data; therefore the sample size of the clinical trial (originally planned as n=100, but accomplished as n=62, the protocol was stopped when new types of chemotherapy became available) was motivated not by this analysis but rather by the trial primary aim. Nevertheless, the sample size of the present secondary study (n=55, the number of patients with available blood samples) is large enough to detect moderate quantitative changes in CTCs levels over time and moderate correlation of CTCs with quantitative tumor response endpoints, such as measurements of tumor volume. For example, detection of a correlation corresponding to a Kendall's tau correlation coefficient of 0.3 (a moderate effect) requires, with power of 80% and type 1 error of 5%, a sample size of just n=39 (18). Similarly, a paired Wilcoxon test requires n=42 subjects to detect a change, assuming power of 80% and type 1 error of 5%, as to detect the probability of 75% for a negative sum of 2 CTCs changes (18).
Results
Patients, Treatment and Tumor Characteristics
Fifty-five patients had samples collected (29 in group 1 and 26 in group 2) and processed for CK19, BS106 and B2M. Clinical characteristics of these patients are summarized in Table I. No significant differences in clinical variables were found between the study groups, either in our cohort or in the parent study.
PATIENTS, TREATMENT AND TUMOR CHARACTERISTICS ACCORDING TO THE TREATMENT ARM
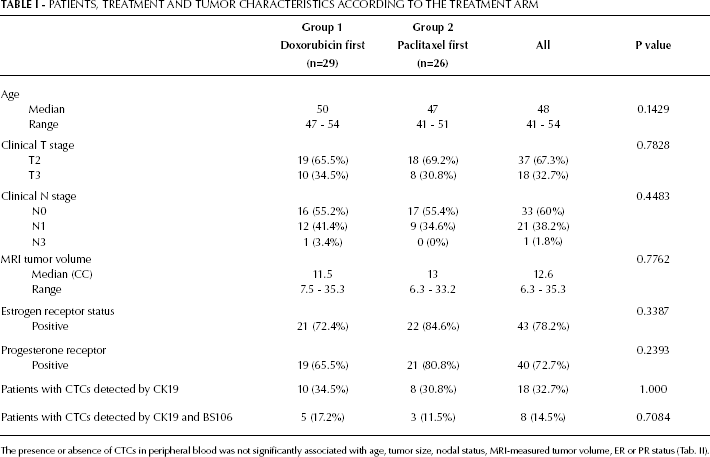
The presence or absence of CTCs in peripheral blood was not significantly associated with age, tumor size, nodal status, MRI-measured tumor volume, ER or PR status (Tab. II).
PATIENT AND TUMOR CHARACTERISTICS ACCORDING TO BASELINE CTCs
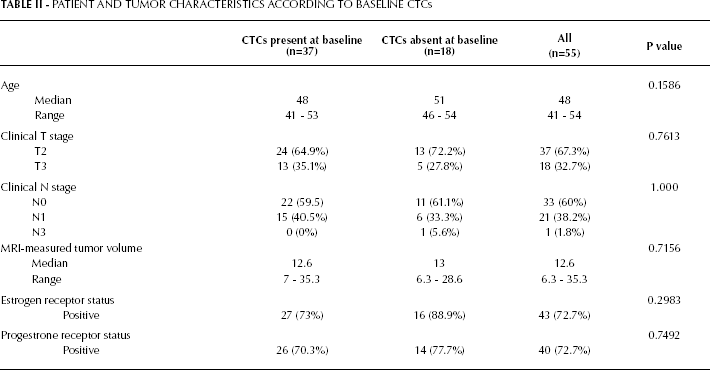
At baseline, 18 (32.7%) out of the 55 patients had CTCs detected in their peripheral blood. There were no differences in the rate of baseline CTCs detection between treatment groups (p=1.000). Using a pre-established cutoff for CTCs detection, 1 (3.3%) out of 30 healthy donors had a positive blood sample.
Results after the First Phase of Chemotherapy
After the first phase of chemotherapy the percentage of patients with CTCs detected by CK19 decreased from 32.7% to 26% in the entire cohort; however this was not statistically significant (p=0.4692). In group 1, CTCs were detected in the peripheral blood of 23% (n=6) of patients, with only 8% (n=2) of those patients having CTCs detected at baseline and 15% (n=4) with newly detected CTCs after the end of the first phase of chemotherapy. In group 2, CTCs were detected in 30% (n=6) of the cases after the end of the first phase of chemotherapy, with 15% (n=3) of the patients being CTCs-positive at baseline and 15% (n=3) with newly detected CTCs.
There was no statistically significant difference between treatment groups with regard to the total number of patients with detected CTCs after the first phase of treatment (p=0.7379).
There was an overall reduction of the MRI-measured tumor volume as calculated from the MRI-measured tumor dimensions. The median tumor volume decreased from 12.6 cc (range 6.7-34.8 cc) to 4.1 cc (range 1.9-9.6 cc), corresponding to a 67% reduction in tumor volume. The percentage of volume reduction did not differ between patients who had detectable CTCs at baseline compared to those without (p=0.89).
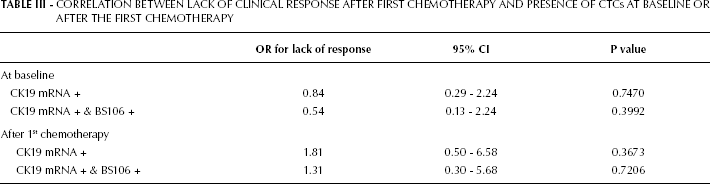
No difference in the magnitude of clinical response was found when patients were further stratified according to treatment arm (p=0.2391). The odds ratio (OR) for lack of clinical response and the presence of CTCs after the first phase of chemotherapy was 1.81 (95% CI 0.50-6.58, p=0.36), however this result was not statistically significant (Tab. III).
CORRELATION BETWEEN LACK OF CLINICAL RESPONSE AFTER FIRST CHEMOTHERAPY AND PRESENCE OF CTCs AT BASELINE OR AFTER THE FIRST CHEMOTHERAPY
Results after the Second Phase of Chemotherapy
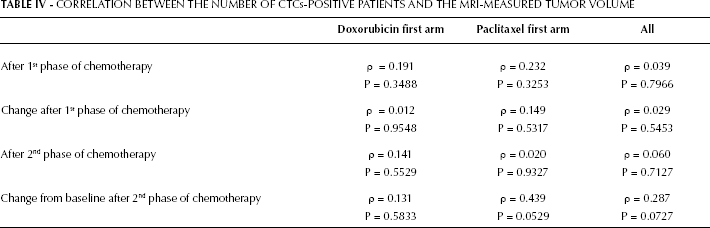
The percentage of patients with detectable CTCs in their peripheral blood declined to 12.2% (n=5) after the second phase of chemotherapy and, when compared to the number of patients with detectable CTCs at baseline, this reduction was statistically significant (p=0.0129). The distribution of these patients between the treatment arms did not differ significantly (p=0.18). The median tumor volume was 0.9 cc (range 0-4.6 cc), corresponding to a 93% volume reduction from the baseline MRI-measured tumor volume. There was no association between lack of response to chemotherapy and detection of CTCs in peripheral blood after the second phase of chemotherapy (OR=1.72, p=0.54) (Tab. IV). These results did not change when the data were reanalyzed using both the CK19 mRNA and the BS106 values.
CORRELATION BETWEEN THE NUMBER OF CTCs-POSITIVE PATIENTS AND THE MRI-MEASURED TUMOR VOLUME
CTCs detection after surgery
The median follow-up was 64.3 months (range 16-90 months). Of the 17 patients who developed either local or distant relapsed disease, 14 had detectable CTCs in their peripheral blood during follow-up and prior to the development of the relapsed disease. In a Cox proportional-hazard model with time-dependent covariates, the presence of CTCs was a predictor of imminent relapse, HR 3.28 (95% CI 0.98-10.99, p=0.04). When the model was restricted to patients who had no previously detectable CTCs and developed CTCs only during follow-up, the presence of CTCs was significantly associated with relapse, HR 1.50 (95% CI 1.05-2.15, p=0.04). No association could be found between the presence of CTCs and a worse overall survival, HR 3.46 (95% CI 0.31-38.71, p=0.25).
The table presents Spearman's rho (ρ) corrections coefficients and p values for testing whether ρ=0 for correlations between CTCs detected by CK19 and MRI-measured tumor volume. There was no statistically significant correlation.
Discussion
The prognostic significance and the clinical implications obtained from detecting CTCs in the peripheral blood of breast cancer patients have been questioned in many studies, most of which have been performed in the metastatic (6-9) and adjuvant settings (19, 20). Few studies explored the prognostic significance of CTCs detection in the preoperative setting (21-23).
The results of the current study suggest that the detection of CTCs in the peripheral blood of women with locally advanced breast cancer before the initiation of the PC is not a useful tool to predict which patients are likely to respond to chemotherapy. We were not able to demonstrate a correlation between the lack of clinical response and the presence of CTCs after the first and the second phase of chemotherapy (p=0.36 and p=0.53, respectively). It should be noted that the number of patients with detectable CTCs dropped from 32.7% to 26% after the first phase of chemotherapy and to 12.2% after the second phase. These results did not differ when we used the data obtained for both markers (CK19 mRNA and BS106) in the analysis.
Several authors have reached similar results using different methods for CTCs detection. Van der Auwera and colleagues compared 3 CTCs detection methods (Cell Search System, Adna Test and a multimarker quantitative RT-PCR). They found a significant concordance between the number of CTCs detected by the Cell Search System and the multimarker RT-PCR (24). In the “GEPARQuattro” trial, which included 213 non-metastatic breast cancer patients, the prevalence of CTCs-positive patients dropped from 22% to 11% after PC and there was no association with the primary tumor response (23). In a phase II trial on 118 patients, Pierga (21) and colleagues evaluated the prognostic significance of the detection of CTCs in the preoperative setting. The authors found that neither the detection of CTCs before or after PC, nor changes in CTCs count during the preoperative treatment was predictive of pathologic response. However, CTCs detection before and/or after PC was significantly associated with early metastatic relapse (p=0.013, median follow-up: 18 months). Pachman et al (22) evaluated a group of 30 patients receiving PC; the patients received 1 of 3 regimens: dose dense epirubicin, epirubicin/cyclophosphamide or epirubicin/cyclophosphamide/trastuzumab. The authors were able to observe a correlation between the decrease in the number of CTCs during the first cycles of treatment and the tumor response to chemotherapy. However the limited sample size of their study and the inconsistency of this finding among the 3 treatment arms (the trastuzumab arm did not show this relation between CTCs count and response to treatment) make the results of this study in need of more validation before drawing solid conclusions about the importance of CTCs quantification.
In the current study, the detection of CTCs in the postoperative setting increased the hazard of relapse, HR 1.50 (95% CI 1.05-2.15, p=0.04). This finding, however, was not translated into an overall survival difference in favor of those who did not have CTCs, HR 3.46 (95% CI 0.31-38.71, p=0.25). It is possible that such a difference exists (note that HR>1) but with a number of only 8 death events the power is too low to detect it. In another study of 41 patients undergoing surgery for breast cancer (with a mean follow-up of 8.6 months), Biggers (25) and colleagues were unable to show a difference in overall survival between patients with detectable CTCs and patients with no detectable CTCs. In a larger study of 167 patients conducted in the adjuvant setting, Xenidis (20) and associates were able to show a strong correlation between the detection of CTCs using CK19 mRNA and recurrence. The difference in recurrence rate between patients with persistently positive and those with persistently negative CK19 mRNA was highly significant (p=0.00026). The still ongoing German SUCCESS trial which enrolled non-metastatic lymph node-positive and high risk lymph node-negative patients before and after Taxane-based adjuvant therapy demonstrated that at least 1 CTC was detectable in the peripheral blood in 10% of the patients and that the patients with persistently negative CTCs had better prognosis (26). This strong correlation between CTCs detection in the post-operative setting and the development of local or distant relapse could be useful in designing a clinical trial to test the hypothesis that further systemic therapy might be helpful for patients with detectable CTCs after receiving neoadjuvant chemotherapy.
The results of the current study did not show a correlation between the detection of CTCs and other important tumor variables, such as nodal status and tumor size (Tab. II). These results mirror what was observed by Stathopoulou (27) and colleagues in their study of 142 patients with operable breast cancer patients. This dissociation between the tumor burden and the detection of CTCs may well be due to different biological phenotypes being responsible for locoregional vs hematogenous spread.
There were certain limitations to our study. First, due to the small number of patients enrolled (n=55) into the study, strong conclusions could not be drawn. Therefore, studies with a larger series of patients are needed, especially to address the question of the relationship between CTCs detection and response to PC. Second, quantitative analysis of the CTCs data in our study was not used as the clinically predictive thresholds for the non-metastatic patients are not exactly known and they are likely to lie within the range of 0-10 cell/ml (28, 29). Moreover, it has been shown that during the time of epithelial-mesenchymal transition, downregulation of the EpCAM expression might occur. This could have contributed to the false negative tests in the current study, especially since the entire study was conducted using an EpCAM-based assay. Nevertheless, the finding of a single CTC in 7.5 ml of peripheral blood was shown to be associated with an increased risk of metastases (30, 31). Third, the use of MRI to quantify the clinical response to PC might have led to either an underestimation of the actual tumor size, due to loss of tumor vasculature, or an overestimation (32, 33) of the size, due to the non-specific enhancement caused by the proliferative disease. Thus, a more robust method for quantifying tumor response in the preoperative setting needs to be established.
CONCLUSION
The results of this study have shown that the detection of CTCs in peripheral blood in the post-operative setting is correlated with local and/or distant relapse. We believe that from this point CTCs can be used as a prognostic indicator of a higher risk for relapse, a finding that could be used to develop further adjuvant therapies for patients in the high-risk group with detectable CTCs. However, the CTCs detection in peripheral blood in the course of PC does not correlate with a particular pattern of response to PC.
