Abstract
Background
Detection of circulating cancer cells by reverse transcription polymerase chain reaction (RT-PCR) has been studied as a prognostic marker in patients with colorectal cancer (CRC) but so far with conflicting results regarding specificity and prognostic value. In this study cytokeratin-20 (CK20) was evaluated by real-time RT-PCR as a marker for circulating CRC cell detection and the influence of surgical tumor resection on the presence of circulating CRC cells was analyzed.
Methods
RNA was isolated from the mononuclear cell fraction of blood samples taken from 95 CRC patients before and after tumor resection and from 23 healthy volunteers and assayed by real-time RT-PCR for CK20 expression.
Results
Among 23 healthy volunteers one was positive for CK20. Among 95 CRC patients, 25 were positive for CK20 before and 23 after surgery. Sixteen patients positive before surgery became negative after surgery, while 14 patients negative before surgery became positive after surgery. An increase in the proportion of CK20-positive samples with increasing stage of disease was observed for preoperative but not postoperative blood samples.
Conclusions
Its association with clinical stage indicates that CK20 might have prognostic value as a marker for detection of circulating CRC cells. Surgical tumor resection can both reduce and induce the presence of circulating CRC cells.
Keywords
Introduction
The majority of patients with newly diagnosed colorectal cancer (CRC) are surgically treated with curative intent. However, approximately 20%-40% of these patients ultimately develop local recurrence or distant metastases following curative surgical resection (1). Accurate prognostic markers that would identify this group of patients could improve the management of CRC patients.
One such potential prognostic marker is the presence of CRC cells in the circulation. To date, the only standardized and FDA-approved method for the detection of circulating tumor cells in patients with CRC is the method based on immunomagnetic separation called CellSearch (2). Other methods to detect circulating CRC cells have been studied; reverse transcription polymerase chain reaction (RT-PCR) is the most widely investigated, mostly due to its low threshold for detection (1 CRC cell in 1 mL blood) (3-7).
In most studies epithelial markers such as carcinoembryonic antigen (CEA) and cytokeratins (CK) 19 and 20 were analyzed for the detection of circulating CRC cells by RT-PCR (4). The expression of CK20 was shown to be restricted to gastric and intestinal epithelium, urothelium and Merkel cells, and cancers originating from these tissues (8, 9). One study showed expression of CK20 in 95% of primary and metastatic CRC samples (8). Since epithelial cells expressing CK20 are not normally present in the circulation, the expression profile of CK20 makes it a suitable, specific marker for the detection of circulating CRC cells.
However, in several studies a proportion of control subjects (healthy volunteers or patients with inflammatory diseases) were positive for CK20 expression in the circulation (5, 6, 10-17). These false-positive findings could be explained by the expression of CK20 in normal or activated leukocytes (11). The association of CK20 expression with the stage of the disease detected by RT-PCR has been studied as a surrogate indicator of prognostic value of CK20 in several studies. Some studies demonstrated this association, while others did not (3, 6, 7, 12, 13, 18-25).
The prognostic value of CK20, analyzed as a single marker or in combination with other RT-PCR markers, has also been studied directly in follow-up studies. Several studies showed that RT-PCR detection of circulating CRC is associated with shorter overall and progression-free survival (5, 19, 22, 26-28). However, some other follow-up studies did not confirm the prognostic value of RT-PCR detection of circulating CRC cells (13, 20, 29, 30).
A study in which blood samples obtained from CRC patients before and after tumor resection were analyzed suggests that clearance of CRC cells from the circulation detected by RT-PCR after tumor resection is a marker of favorable prognosis (12). This was corroborated in a study showing that the presence of circulating CRC cells in preoperative blood samples is not predictive of recurrence but the presence of circulating CRC cells in blood samples obtained after curative tumor resection is a strong predictor of CRC recurrence (10).
More recently, real-time RT-PCR has been employed for the detection of circulating CRC cells (7, 16, 22, 23, 25, 27, 28, 31). Real-time RT-PCR, as a quantitative method, has some potential advantages compared with standard RT-PCR. Real-time RT-PCR allows to precisely define cutoff values of marker expression in order to discriminate between low-level expression in control samples and higher-level expression in patients’ samples and thus increase the specificity (22, 31). Real-time RT-PCR also allows to quantitatively compare samples from the same patient obtained at different time points.
Due to conflicting results regarding specificity and prognostic value, the clinical value of RT-PCR detection of circulating CRC cells is disputable. Therefore, in this study we evaluated CK20 by real-time RT-PCR as a marker of circulating CRC cells in our group of patients. We also analyzed the influence of surgical tumor resection on the presence of circulating CRC cells.
Materials and Methods
Patients
We analyzed blood samples from 95 patients (38 women and 57 men, aged 39-86 years). All blood samples from patients and healthy volunteers were taken from the peripheral venous circulation. All patients had histopathologically confirmed primary CRC and were treated at the Department of Surgery and the Department of Oncology and Nuclear Medicine, University Hospital Center “Sestre milosrdnice”, Zagreb, Croatia. Clinical stage was determined according to the American Joint Committee on Cancer (AJCC) guidelines (32). Blood samples from 23 healthy volunteers (18 women and 5 men, aged 27-61 years) were analyzed as controls. The study was carried out in compliance with the World Medical Association Declaration of Helsinki and informed consent was obtained from all patients and healthy controls before their inclusion in the study (33).
Specimen characteristics
Two blood samples were collected from each patient, the first before tumor resection on the day of surgery and the second 5-7 days after tumor resection. Ten milliliters of peripheral blood from each patient and healthy volunteer was collected in tubes containing ethylenediami-netetraacetic acid as anticoagulant. Blood samples were processed within 2 hours of collection.
Assay methods
The mononuclear cell fraction of blood was isolated on Ficoll gradient (1.077 g/cm3; Axis-Shield PoC AS, Oslo, Norway) as described by Böyum (34). Three milliliters of Ficoll were overlaid with 5 mL blood diluted with 5 mL phosphate-buffered saline (PBS). After centrifugation for 15 minutes at room temperature at 500 g, the layer between the plasma and the Ficoll was transferred into another tube and washed twice with PBS by centrifugation for 15 minutes at 4°C at 800 g. The cell pellet was used immediately for RNA isolation.
Total cellular RNA was isolated from the mononuclear cell fraction of blood samples and from the HT29 cell line using the TriPure Isolation Reagent (Roche, Indianapolis, IN, USA) following the manufacturer's instructions. The RNA pellet was dissolved in deionized water and stored at −70°C. For reverse transcription, 4 mL of RNA was mixed with other components from the High Capacity cDNA Reverse Transcription Kit with RNase Inhibitor (Applied Biosystems, Foster City, CA, USA) in a total volume of 20 mL, according to the manufacturer's instructions. The mixture was incubated at 25°C for 10 minutes, then at 37°C for 120 minutes, and finally at 85°C for 5 seconds, and was stored at −20°C until used for real-time RT-PCR.
Real-time RT-PCR was performed in 96-well plates in the Applied Biosystems 7500 Real-Time RT-PCR System (Applied Biosystems). Four milliliters cDNA was added to the reaction mixture containing final concentrations of 1x TaqMan Universal PCR Master Mix (Applied Biosystems) and 1x TaqMan Gene Expression Assay, Assay ID: Hs00300643_m1 (Applied Biosystems) for CK20 or 1x TaqMan Endogenous Control, Human GAPD (GAPDH), FAM™/MGB (Applied Biosystems) for GAPDH in a total volume of 25 ⚟L The PCR reaction conditions were: 2 minutes at 52°C, 10 minutes at 95°C, and 45 cycles each consisting of 15 seconds at 95°C and 1 minute at 60°C. Nontemplate control was included in each experiment and all samples were analyzed in duplicate.
Quantification of CK20 expression was done by the ΔΔCT method using the Sequence Detection Software v 1.3.1 (Applied Biosystems) (35). GAPDH was analyzed as endogenous control and cDNA from HT29 cells was analyzed as the calibrator sample. The human colon adenocarcinoma grade II cell line HT29 was purchased from American Type Culture Collection (Manassas, VA, USA).
To confirm that the efficiencies of the target (CK20) and endogenous control (GAPDH) were approximately equal, which is a requirement for ⚟⚟CT quantification, a validation experiment was performed using 10-fold serial dilutions of RNA from the HT29 cell line. Serial dilutions of RNA were reversely transcribed and analyzed by real-time PCR for the expression of CK20 and GAPDH as described. For the validation experiment all samples were analyzed in triplicate.
Statistical analysis methods
The cutoff value for CK20 relative expression was determined as the mean plus 2 standard deviations for healthy controls. All samples with CK20 relative expression higher than the cutoff were considered positive. The statistical significance of the association between the distribution of values of CK20 and other nominal categorical variables (sex, tumor location, histopathological diagnosis, and presence of metastases) was determined by the chi-square (χ 2 ) test or, when ≥20% of cells had an expected value <5, by Fisher's exact test. The statistical significance of the association between the distribution of values of CK20 and stage of the disease (ordinal categorical variable) was determined by the Cochran-Armitage test for trend. The statistical significance of the association between the distribution of values of CK20 and age (numerical variable) was determined by the Mann-Whitney U test. Liddell's exact test for matched pairs was used to analyze the statistical significance of the association between the distribution of CK20 values in preoperative and postoperative blood samples.
The analysis was performed using MedCalc (version 10.4.0.0; MedCalc Software bvba; Mariakerke, Belgium) and a p value of ≤5 was considered statistically significant.
Results
Analysis of blood samples from patients and healthy volunteers
The demographic and clinical characteristics of the patients analyzed in this study are presented in Table I. None of the patients received any systemic therapy (e.g. neoadjuvant chemoradiotherapy) before blood samples were taken for analysis. All patients underwent surgery of the primary CRC after blood samples had been taken. Among 13 patients with distant metastases (stage IV), 3 were operated for metastases at the same time (1 with liver, 1 with small intestine and 1 with urinary bladder metastases). All other patients with distant metastases either did not receive surgery at the metastatic site because the metastases were unresectable or did receive surgery at the metastatic site in a subsequent procedure after the blood samples for the analysis were obtained, so it could not have influenced our results.
Patient Characteristics
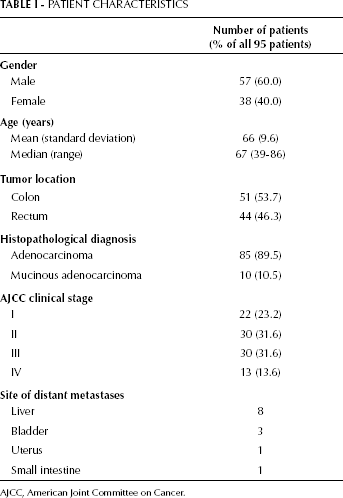
AJCC, American Joint Committee on Cancer.
The expression of CK20 in CRC patients’ and healthy volunteers’ blood samples was analyzed and quantified as described (Fig. 1). The cutoff value for CK20 relative expression of 31.25 × 10−6 was established as described above. Among 23 healthy volunteers, 1 (4.3%) was positive for CK20. Among 95 CRC patients, 25 (26.3%) were positive for CK20 before surgery (preoperative blood sample) and 23 (24.2%) were positive for CK20 after surgery (postoperative blood sample). Out of 95 patients, 56 were negative for CK20 both before and after surgery, 9 were positive both before and after surgery, 14 were negative before surgery and positive after surgery, and 16 were positive before surgery and negative after surgery (Tab. II).
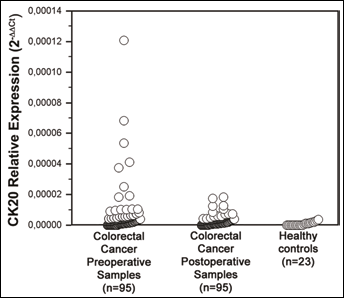
Expression of CK20 in blood samples of colorectal cancer patients and healthy volunteers. CK20 expression was analyzed by real-time RT-PCR and quantified by the ΔΔCT method using GAPDH as endogenous control and cDNA from the HT29 cell line as the calibrator sample. Preoperative and postoperative blood samples from 95 colorectal cancer patients and blood samples from 23 healthy volunteers were analyzed.
Ck20 Values Before And After Surgery In Relation To Disease Stage
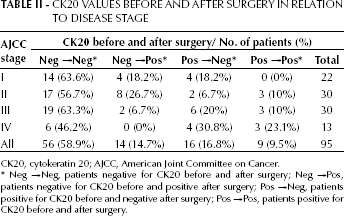
CK20, cytokeratin 20; AJCC, American Joint Committee on Cancer.
Neg → Neg, patients negative for CK20 before and after surgery; Neg → Pos, patients negative for CK20 before and positive after surgery; Pos → Neg, patients positive for CK20 before and negative after surgery; Pos → Pos, patients positive for CK20 before and after surgery
Three stage IV patients who received surgery also at the metastatic site at the time of blood sampling for analysis were negative for CK20 both before and after surgery. The proportion of positive blood samples was statistically significantly higher in both preoperative (Fisher's exact test, p=0.02) and postoperative (Fisher's exact test, p=0.04) CRC patients’ blood samples than in those of healthy volunteers.
There was no statistically significant difference between the results for CK20 in preoperative and postoperative blood samples from CRC patients (Liddell's exact test, p>0.99). The difference between the results for CK20 in preoperative and postoperative blood was also not statistically significant when analyzed for each clinical stage separately (Liddell's exact test: p=0.73 for stage I, p>0.99 for stage II, p=0.73 for stage III, p>0.99 for stage IV) (Tab. II).
Association with clinical stage and other demographic and clinical variables
No statistically significant association between the value of CK20 in preoperative blood samples and age (Mann-Whitney U test, z=-0.33, p=0.74), sex (chi-square test, ⚟ 2 =0.51, p=0.48), location of primary tumor (colon or rectum) (chi-square test, ⚟ 2 =0.94, p=0.33), and histopathological diagnosis (adenocarcinoma or mucinous adenocarcinoma) (Fisher's exact test, p=0.28) was observed. Also, no statistically significant association between the value of CK20 in postoperative blood samples and age (Mann-Whitney U test, z=0.06, p=0.96), sex (chi-square test, ⚟ 2 =2.60, p=0.11), location of primary tumor (colon or rectum) (chi-square test, ⚟ 2 =0.17, p=0.68), and histopathological diagnosis (adenocarcinoma or mucinous adenocarcinoma) (Fisher's exact test, p=0.44) was observed.
Clinical stage of the disease was determined according to the AJCC guidelines in all 95 patients included in the study (Tab. I). Expression of CK20 in preoperative blood samples was found in 4 out of 22 (18.2%) stage I patients, 5 out of 30 (16.7%) stage II patients, 9 out of 30 (30.0%) stage III patients, and 7 out of 13 (53.8%) stage IV patients (Tab. III). A statistically significant trend of increase in the proportion of CK20-positive preoperative blood samples with increasing stage of the disease was observed (Cochran-Armitage test for trend, ⚟ 2 =5.68, p=0.02). Also, the proportion of CK20-positive preoperative blood samples was statistically significantly higher in patients with distant metastases (AJCC stage IV) than in patients with no distant metastases (AJCC stages I to III) (Fisher's exact test, p=0.04) (Tab. III).
Distribution Of Ck20 Values Detected By Real-Time Rt-Pcr In Relation To Disease Stage
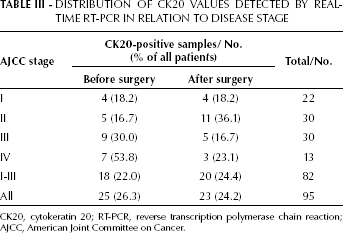
CK20, cytokeratin 20; RT-PCR, reverse transcription polymerase chain reaction; AJCC, American Joint Committee on Cancer.
Expression of CK20 in postoperative blood samples was found in 4 out of 22 (18.2%) stage I patients, 11 out of 30 (36.1%) stage III patients, 5 out of 30 (16.7%) stage III patients, and 3 out of 13 (23.1%) stage IV patients (Tab. III). No statistically significant trend of increase in the proportion of CK20-positive postoperative blood samples with increasing stage of disease was observed (Cochran-Armitage test for trend, ⚟ 2 =0.09, p=0.76). Also, no statistically significant association between the value of CK20 in postoperative blood samples and the presence of distant metastases was found (Fisher's exact test, p=1.00) (Tab. III).
At the time of analysis, the median follow-up time for the patients was 208 days (range: 12-465 days). Of the 95 patients, 12 (12.6%) had died of CRC at the time of analysis. Of those 12 patients, 3 had stage II disease, 3 stage III, and 6 stage IV.
Discussion
Detection of circulating tumor cells by standard or real-time RT-PCR and with CK20 or CEA as the most common markers has been investigated in patients with CRC as a potential prognostic tool to predict disease recurrence (4). However, different studies produced conflicting results regarding the specificity and prognostic value of this method (4). In this study 25 (26.3%) out of 95 CRC patients were positive for CK20 before surgery and 23 (24.2%) were positive for CK20 after surgery, which is in accordance with the results of other studies (4).
With our cutoff value, only 1 (4.3%) of 23 healthy control samples was positive for CK20. Furthermore, low expression of CK20 below our cutoff value was found in an additional 10 healthy controls. This illustrates a major advantage of real-time RT-PCR compared to standard PCR: real-time RT-PCR allows to define cutoff values of marker expression that can discriminate between low-level expression in control samples and higher-level expression in patients’ samples.
Positive results of markers used for RT-PCR detection of circulating CRC cells in blood samples from healthy volunteers and patients with inflammatory diseases are a significant limitation in the clinical use of these markers. It has been shown that the expression of CK19 and CEA can be induced in hematopoietic cells by cytokines, which can explain the positive results for these markers in patients with inflammatory diseases (36). However, none of these cytokines was able to induce the expression of CK20 in the same system (11). This suggests a potential advantage of CK20 over CK19 and CEA, 2 other extensively studied markers for RT-PCR detection of circulating CRC cells, which is why we analyzed CK20 in our study.
Several studies using both standard and real-time RT-PCR have shown positive results in healthy volunteers and patients with inflammatory diseases for CK20 also (5, 6, 10-17). One possible explanation for false-positive CK20 results could be the stable expression of CK20 that was shown in peripheral blood granulocytes (11). However, granulocytes can be removed from blood samples by Ficoll gradient density separation, the method employed in this study (11).
Considering the false-positive results reported in several studies it was suggested that one should be cautious in interpreting positive RT-PCR results for CK20 and CEA as they may reflect the presence of inflammatory cells rather than micrometastatic cancer cells (37).
In this study the association of the proportion of CK20-positive samples with clinical stage of the disease was studied as a surrogate indicator of the prognostic value of CK20 because disease stage correlates strongly with patient survival (32). In our study, a statistically significant trend of increase in the proportion of CK20-positive patients with increasing stage of disease was found for preoperative but not postoperative blood samples.
Several studies using standard and real-time RT-PCR have also shown statistically significant increases in the proportion of CK20-positive preoperative blood samples with increasing disease stage in CRC patients (7, 19, 21, 25, 38). However, some other studies failed to show such an association (6, 13, 20). The association of the proportion of CK20-positive patients with stage in postoperative blood samples was studied in a smaller number of studies with conflicting results (3, 12, 24).
The association with stage in preoperative but not postoperative blood samples shown in our study might mean that the presence of circulating cancer cells preoperatively reflects the tumor burden, while cancer cells detected in postoperative blood samples might also be the result of the shedding of cancer cells into the circulation due to surgical manipulation, which consequently results in a worse outcome (39). This indicates that, although detection of circulating CRC cells in postoperative blood samples is not associated with stage, it might still have prognostic value. The association with clinical stage is a surrogate indicator of prognostic value and only direct analysis of survival data from a follow-up study can give a reliable estimate of the prognostic value of any marker. If we exclude from the analysis patients who were negative before and positive after surgery, there is a statistically significant trend of increase in the proportion of CK20-positive postoperative blood samples with increasing stage of the disease in our study (Cochran-Armitage test for trend, ⚟ 2 =1.97, p=0.04), which supports such explanation.
Due to a short follow-up (208 days median) and small number of events (12 of 95 patients died of CRC) at the time of analysis, we could not reliably determine the prognostic value. Therefore, we are following patients from our study in order to subsequently directly analyze the prognostic value of CK20 expression analysis by RT-PCR in peripheral blood of our group of CRC patients. Follow-up studies in which the prognostic value of circulating CRC cells was directly studied yielded conflicting results. Several studies confirmed the association of circulating CRC cells with shorter overall and progression-free survival (5, 19, 22, 26-28). However, some other follow-up studies failed to confirm the prognostic value of detection of circulating CRC cells (13, 20, 29, 30).
In our study, 16 (64%) out of 25 patients who were positive for CK20 expression before surgery became negative after surgery, while 14 (20%) out of 70 patients who were negative before surgery became positive after surgery. Positive CK20 expression in preoperative blood but negative expression in postoperative blood of the same patient might reflect tumor cell clearance from the circulation after tumor resection associated with the curative success of surgery and therefore can be a marker of favorable prognosis. The results of several studies corroborate that hypothesis (10, 12, 40). For example, Uen et al found that 62% of patients positive for circulating CRC cells before surgery became negative after surgery and these patients had significantly better relapse-free survival than patients who were positive before and after surgery (40).
Negative CK20 expression in preoperative blood but positive expression in postoperative blood recorded in 15 patients of our study might also be of prognostic significance since, as mentioned, it can be the result of the shedding of cancer cells due to surgical manipulation. Ito et al analyzed expression of CEA as a marker for circulating CRC cells and found significantly increased expression in postoperative blood compared with preoperative blood and also an association with worse prognosis for positive results in postoperative but not preoperative blood samples, confirming this hypothesis.
However, discrepancy in CK20 results between preoperative and postoperative blood samples from the same patients can also partly be explained by stochastic mechanisms. It is possible that, similarly to what was proposed for circulating melanoma cells, CRC cells are shed into the bloodstream transiently, in a random, discontinuous manner (41). This explanation is supported by the results of a study in which 2 preoperative blood samples were taken with an interval of 3 days from each CRC patient: the proportion of CK20-positive patients increased significantly with the analysis of 2 blood samples compared with the analysis of a single blood sample (19).
In conclusion, the association with clinical stage shown in this study indicates that CK20 might have prognostic value as a marker for the detection of circulating CRC cells. Furthermore, this study has shown that surgical tumor resection can both reduce and induce the presence of circulating CRC cells.
