Abstract
Background
Circulating estrogens are associated with increased breast cancer risk, yet the role of estrogen metabolites in breast carcinogenesis remains unclear. This combined analysis of 5 published studies evaluates urinary 2-hydroxyestrone (2-OHE1), 16α-hydroxyestrone (16α-OHE1), and their ratio (2:16α-OHE1) in relation to breast cancer risk.
Methods
Primary data on 726 premenopausal women (183 invasive breast cancer cases and 543 controls) and 1,108 postmenopausal women (385 invasive breast cancer cases and 723 controls) were analyzed. Urinary estrogen metabolites were measured using enzyme linked immunosorbent assays. Study-specific and combined multivariable adjusted odds ratios (ORs) and 95% confidence intervals (CIs) were estimated based on tertiles of estrogen metabolites. Multinomial logistic regression models were fit according to hormone receptor status.
Results
Higher premenopausal 2:16α-OHE1 was suggestive of reduced breast cancer risk overall (study-adjusted ORIIIvsI=0.80; 95% CI: 0.49-1.32) and for estrogen receptor negative (ER-) subtype (ORIIIvsI=0.33; 95% CI: 0.13-0.84). Among postmenopausal women, 2:16α-OHE1 was unrelated to breast cancer risk (study-adjusted ORIIIvsI=0.93; 95% CI: 0.65-1.33); however, the association between 2-OHE1 and risk varied by body mass index (p-interaction=0.003).
Conclusions
Premenopausal urinary 2:16α-OHE1 may play a role in breast carcinogenesis; however, larger studies are needed. Our findings do not support reduced breast cancer risk with higher postmenopausal 2:16α-OHE1 overall, although obesity may modify associations with 2-OHE1.
Introduction
Endogenous estrogen exposure throughout a woman's lifetime is an established risk factor for breast cancer (1-3). The association between circulating estrogens and post-menopausal breast cancer is well established (3, 4), but among premenopausal women, menstrual cycle variations render this association less clear (2, 5, 6). Understanding the role of urinary estrogen metabolites in pre and postmenopausal breast cancer may elucidate mechanisms underlying breast carcinogenesis and offer potential strategies for prevention.
Estrogen metabolism occurs by the oxidative pathway (7). Circulating estrone is metabolized via 2 main pathways involving hydroxylation sites C2, C4, or C-16, leading to the formation of either A-ring or D-ring metabolites (7, 8). The main metabolites of the A-ring pathway include 2-hydroxyestrone (2-OHE1) and 4-hydroxyestrone (4-OHE), while 16-alpha-hydroxyestrone (16α-OHE1) and estriol result from the D-ring pathway (8, 9). Although both 2-OHE1 and 16α-OHE1 have estrogenic properties, their ability to bind to the estrogen receptor varies, as does the nature of their estrogenic action. The 16α-OHE1 metabolite is thought to be a more potent estrogen, able to bind covalently to the estrogen receptor (10) and induce cell proliferation (11, 12), whereas the 2-OHE1 metabolite has lower estrogenic activity, due in part to the reduced receptor affinity as well as reduced cell proliferative activity (13). Furthermore, these metabolites result from mutually exclusive pathways (14), and thus may have different genotoxic and mitogenic effects, rendering the ratio of 2:16α-OHE1 a potentially useful measure of exposure to active estrogen metabolites.
Few studies have evaluated the relationship between urinary 2:16α-OHE1 and breast cancer risk (15-22). While some studies suggest a reduction in breast cancer risk with higher urinary 2:16α-OHE1 among premenopausal (15, 18, 20) or postmenopausal women not on hormone therapy (17, 18), other studies report a modest inverse association or non-significant associations (15, 20-22), or do not report results separately by menopausal status (16, 17, 19). The objective of this analysis was to evaluate the individual levels of the urinary 2-OHE1 and 16α-OHE1 metabolites, and their ratio, in relation to the risk of breast cancer in both pre and postmenopausal women by performing a combined analysis of primary data from previously published studies.
Materials and Methods
Study identification
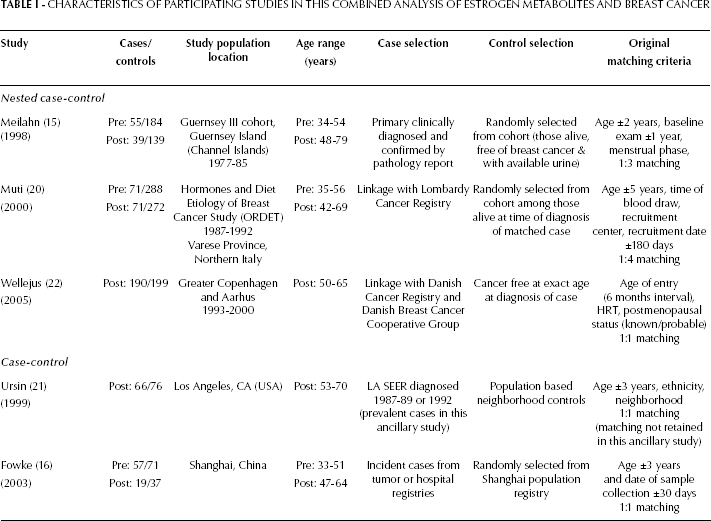
Original studies that evaluated estrogen metabolites in relation to breast cancer risk were identified through the National Library of Medicine and National Institutes of Health PubMed database using the following search strategy: (i) “estrogen metabolites” (n=410 articles) and (ii) “estrogen metabolites”, limited to females and English language (n=256 articles), (iii) “estrogen metabolites” and “breast cancer” (n=112 articles). Citations, abstracts and reference lists were reviewed to identify studies of urinary estrogen metabolites (as measured by ELISA) and breast cancer; 6 studies reported data separately for premenopausal women (15-20), as did 8 studies for postmenopausal women (15-22). This search is current as of March 26, 2012.
Articles were considered eligible if they met the following a priori inclusion criteria: (i) original research study, (ii) inclusion of pre or postmenopausal women, (iii) exposure measured as 16α-OHE1, 2-OHE1, and/or the ratio of these 2 main estrogen metabolites measured by ELISA assay, (iv) urine as the sample source, (v) breast cancer assessed as the main outcome and (vi) total sample size of at least 50 subjects. These criteria were chosen to increase comparability across studies.
Eligible studies
Investigators from each of these eligible studies were extended multiple invitations to participate in this combined analysis. The investigators who agreed, contributed data from 3 nested case-control studies conducted in Northern Italy (Muti et al, 20), Guernsey Island (Meilahn et al, 15), and Denmark (Wellejus et al, 22), and case-control studies conducted in Shanghai, China (Fowke et al, 16) and in the United States (Ursin et al, 21; Tab. I).
CHARACTERISTICS OF PARTICIPATING STUDIES IN THIS COMBINED ANALYSIS OF ESTROGEN METABOLITES AND BREAST CANCER
Data collection and extraction
Each study utilized a structured questionnaire to obtain information on demographics, reproductive history, lifestyle factors, anthropometric characteristics, and other breast cancer risk factors. Investigators submitted their de-identified study-specific data, study questionnaires, and variable descriptions. Each participating study was approved by the institutional review board or appropriate ethical committee at the respective institution, and participants provided informed consent. Each data file was checked for consistency with previously published results. Identified discrepancies were resolved with the study investigators. Policies for data publication and authorship were reviewed and accepted by participating investigators.
Exclusions
The following participants were excluded from the combined analysis: in situ breast cancer cases, due to limited numbers (n=7 premenopausal, n=6 postmenopausal); current users of oral contraceptives from Fowke et al (n=3 premenopausal, n=2 postmenopausal) and current users of postmenopausal hormone therapy (HT) from Wellejus et al (234 cases, 234 controls), to be consistent with the original exclusion criteria from other participating studies; cases from Fowke et al with post-treatment urine collection (n=14 premenopausal, n=18 postmenopausal); and women with missing metabolite data (n=5 premenopausal, n=13 postmenopausal). The final sample included 726 premenopausal women (183 cases and 543 controls) and 1,108 postmenopausal women (385 cases and 723 controls).
Completeness of data
All variables were coded with a common format with the exception of education and socioeconomic status (due to differences in classification systems). 2-OHE1 and 16α-OHE1 metabolite data were available for 99.3% of premenopausal and 98.7% of postmenopausal women. Data on estrogen receptor (ER) and progesterone receptor (PR) status were available for 62% and 60% of premenopausal cases, respectively, and for 80% and 38% of postmenopausal cases. In the Guernsey study, PR data were not available and ER status was unknown for the majority of cases. Similarly, PR was classified as missing for 75% of cases in Wellejus et al.
Information on alcohol consumption was available in only 3 studies (Wellejus et al, Fowke et al, Muti et al). Family history of breast cancer was not available in the studies from Wellejus et al and Ursin et al. Additionally, information on history of benign breast disease, smoking history, and oral contraceptive use was not available in the study by Ursin et al, although current smokers and current users of oral contraceptives were excluded from the original study. Due to incomplete information on tumor staging, metabolite levels were not assessed by stage of disease.
Postmenopausal status was defined as: no menstrual bleeding for at least 12 months (Muti et al); no hysterectomy and no menstruation in the 12 months prior to study entry, a bilateral oophorectomy, or age at last menstruation younger than age at hysterectomy (Wellejus et al); self-reported natural menopause, hysterectomy with unilateral oophorectomy, and age >55 years (Meilahn et al); Wellejus et al also defined probable postmenopausal status as menstruation during the 12 months prior to study entry and current use of HT, hysterectomy with unilateral (or unknown) oophorectomy, or age at last menstruation same as age at hysterectomy.
In premenopausal women, menstrual cycle phase at urine collection was categorized as follicular (≤14 days), luteal (≥15 days), or unknown, based on an assumed average cycle length of 28 days. Preliminary analyses revealed that additional adjustment for menstrual phase at the time of urine collection did not affect estimates, and menstrual phase at the time of urine collection was not a significant predictor in the study adjusted statistical models, so this variable was not included in subsequent models.
Specimen characteristics and assay methods
The following urine samples were collected: spot urine in Meilahn et al, Fowke et al and Wellejus et al; overnight urine in Muti et al; and first morning void in Ursin et al. Prior studies comparing levels of the 2:16α-OHE1 ratio in spot urine and 24-hour urine samples reported no significant differences (23). All participating studies measured urinary 2-OHE1 and 16α-OHE1 using a commercially available competitive solid-phase enzyme immunoassay kit (ESTRAMET) produced by Immuna Care Corp (Bethlehem, PA, USA). Four studies (Fowke et al, Muti et al, Ursin et al and Wellejus et al) utilized the more recent ELISA kit (24), whereas 1 study used the original assay developed by Klug et al (25). The main components of the later kit are similar to the original assay, except for modifications that allow for increased sensitivity among postmenopausal women (24, 26).
Statistical analysis
Published study-specific findings were replicated prior to conducting this combined analysis. The 2-OHE1 and 16α-OHE1 metabolites (ng/ml) were divided by the urinary creatinine concentration (mg/dl) in the studies by Fowke et al, Meilahn et al, Wellejus et al, and Ursin et al. In the Muti et al study, each estrogen metabolite value was divided by the total volume of urine. The large variability in average values of individual metabolites across studies was partially overcome by standardizing the 2-OHE1 and 16α-OHE1 according to a previously published formula (27):
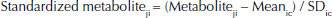
where Metaboliteji is the metabolite for person j in study i, and Meanic and SDic are respectively the mean and standard deviation of the metabolite in the control group of study i. The metabolites were analyzed using: (i) tertiles of the standardized 2-OHE1 and 16α-OHE1 metabolites based on the distribution among the control population (with the first tertile as referent group) and (ii) tertiles of 2:16α-OHE1, the ratio of the 2 individual estrogen metabolites prior to standardization, based on the control distribution (with the first tertile as referent group). The exposure categories presented are based on the tertile distribution of the combined control population (controls from all studies). The individual metabolites were standardized to account for differences in the methods used to adjust for urine concentration (creatinine levels or total volume of urine) and the type of sample collected (spot/overnight void/12 hour collection); the ratio was not standardized as it has the same value regardless of the urine concentration.
The distributions of covariates were compared between cases and controls using t-tests, Wilcoxon rank sum tests, or chi-square tests, as appropriate. Study-specific and combined odd ratios (ORs) and 95% confidence intervals (CIs) were estimated by conditional logistic regression (28) matched on 5-year age groups. The association between each metabolite (standardized 2-OHE1, standardized 16α-OHE1, and 2:16α-OHE1) and breast cancer was assessed using separate conditional logistic regression models: (i) adjusting for study only and (ii) adjusting for study and known breast cancer risk factors i.e. family history of breast cancer, (yes/no/unknown), history of benign breast disease (yes/no/unknown), body mass index (kg/m2), smoking (yes/no/unknown), oral contraceptive use (yes/no/unknown), age at menarche (<13 years, ≥13 years, unknown) and a combined parity variable (no children, age at first pregnancy <20, 20-29, or ≥30 years). Family history of breast cancer was not included in the postmenopausal models as all the women from Ursin et al and Wellejus et al were missing this information. Unknown categories for the covariates listed above were retained in the logistic regression models. Separate trend tests for each estrogen metabolite were performed using the midpoint for each tertile. We evaluated the main effect of study and interactions between study, BMI, smoking status, age, and each metabolite using multiparameter Wald tests.
Due to the observed differences in study-specific median metabolite levels and potential biological differences in estrogen metabolism by race and study design (urine samples collected pre or post diagnosis), sensitivity analyses excluded the 2 case-control studies, 1 of which was the Shanghai study. Subgroup-specific models were conducted using data from the 3 nested case-control studies to assess whether the association between estrogen metabolites and breast cancer varied by body mass index (BMI <25 or ≥25 kg/m2) or current smoking status (yes/no).
Separate study- and age-adjusted (premenopausal: ≤39, 40-44, ≥45 years; postmenopausal: ≤54, 55-59, 60-64, ≥65 years) multinomial logistic regression models were fit according to hormone receptor status using data from the nested case-control studies. The 4 outcomes for the multinomial ER models were: controls (reference), ER positive (ER+) cases, ER negative (ER-) cases, and ER status unknown (ER unk) cases. Comparable categories were used for the progesterone receptor (PR) models. Separate relative risk (RR) estimates compared each hormone receptor subtype to the control group, using the lowest tertile of each metabolite as the reference exposure category.
All analyses were performed using SAS (Version 9.1, Cary, NC, USA) or STATA 10. Tests of significance were 2-sided with no adjustment for multiple comparisons.
Results
Descriptive characteristics
The study sample was predominantly composed of white women. The average age for premenopausal cases and controls was 42 years (SD 4.9) (Tab. II) and 58 years for postmenopausal cases (SD 4.7) and controls (SD 5.2). Among premenopausal women, cases were more likely to report a history of benign breast disease (25.1%) compared to controls (22.3%) (p=0.04). Cases and controls were otherwise similar with regards to smoking status, alcohol use, age at first birth and 2:16α-OHE1. For postmenopausal women, history of benign breast disease differed significantly between cases (21.7%) and controls (14.8%) (p=0.001). Postmenopausal cases were somewhat more likely to consume alcohol and smoke, although these differences were not statistically significant. The distribution of covariates was otherwise similar between cases and controls within each menopausal status.
DESCRIPTIVE CHARACTERISTICS OF THE COMBINED ANALYSIS STUDY SAMPLE
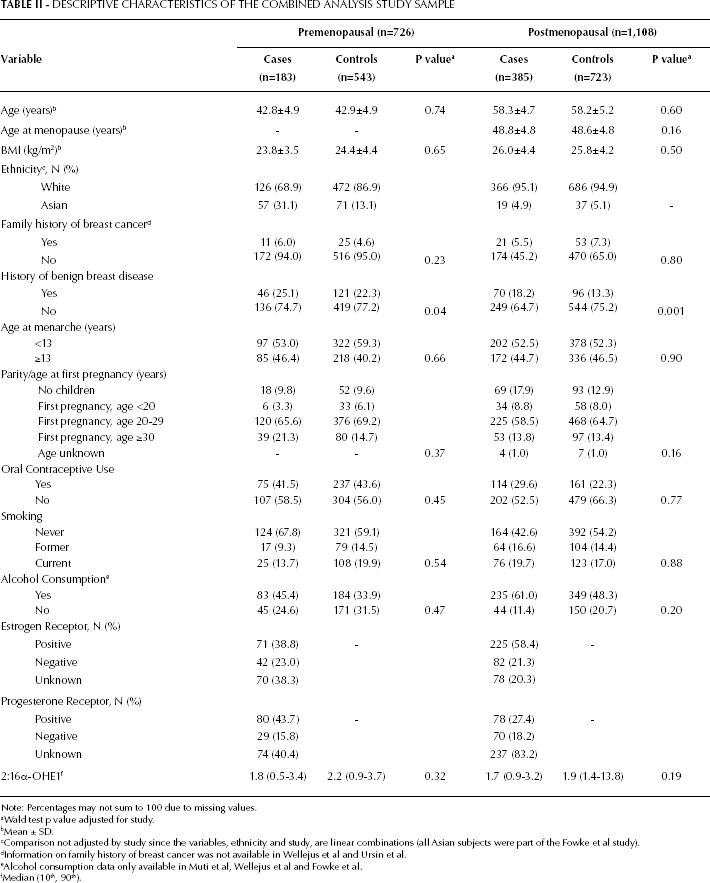
Note: Percentages may not sum to 100 due to missing values.
Wald test p value adjusted for study.
Mean ± SD.
Comparison not adjusted by study since the variables, ethnicity and study, are linear combinations (all Asian subjects were part of the Fowke et al study).
Information on family history of breast cancer was not available in Wellejus et al and Ursin et al.
Alcohol consumption data only available in Muti et al, Wellejus et al and Fowke et al.
Median (10th, 90th).
Premenopausal Women
Study-specific analyses
Among premenopausal women, the study-specific estimates for the associations between 2-OHE1, 16α-OHE1, and the 2:16α-OHE1 ratio and breast cancer (Tab. IIIa) were neither statistically different nor consistently monotonic across tertiles of each exposure.
STUDY-SPECIFIC UNADJUSTED ODDS RATIO ESTIMATES (ORs) AND 95% CONFIDENCE INTERVALS (CI): ASSOCIATION BETWEEN TERTILES a OF THE STANDARDIZED ESTROGEN METABOLITES (2-OHE1, 16α-OHE1) AND 2:16α-OHE1 AMONG PREMENOPAUSAL WOMEN AND BREAST CANCER
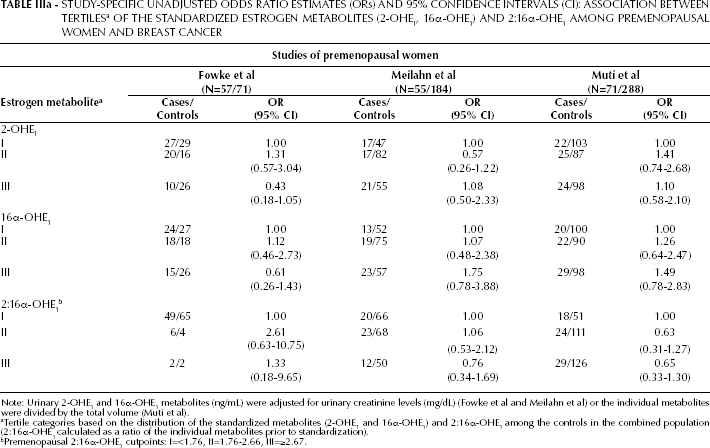
Note: Urinary 2-OHE1 and 16α-OHE1 metabolites (ng/mL) were adjusted for urinary creatinine levels (mg/dL) (Fowke et al and Meilahn et al) or the individual metabolites were divided by the total volume (Muti et al).
Tertile categories based on the distribution of the standardized metabolites (2-OHE1 and 16α-OHE1) and 2:16α-OHE1 among the controls in the combined population (2:16α-OHE1 calculated as a ratio of the individual metabolites prior to standardization).
Premenopausal 2:16α-OHE1 cutpoints: I=<1.76, II=1.76-2.66, III=≥2.67.
Combined analyses
No significant associations or linear trends were observed between breast cancer risk and the standardized 2-OHE1 or 16α-OHE1 metabolites, or the 2:16α-OHE1 ratio among premenopausal women (Tab. IV). Study-adjusted ORs for tertiles of 2:16α-OHE1 (ORIIIvsI=0.80; 95% CI: 0.49-1.32) and corresponding ORs adjusted for both study and known breast cancer risk factors (ORIIIvsI=0.74; 95% CI: 0.45-1.23) indicated a non-significant reduction in risk. The main effect of study was highly significant (p<0.001) and the ORs varied in different studies for the standardized metabolites 2-OHE1 (p-interaction=0.03) but not for 16α-OHE1 or 2:16α-OHE1 (p-interaction>0.10). No significant interactions were observed between age and the 2-OHE1, 16α-OHE1, or 2:16α-OHE1 metabolites (p>0.10, data not shown).
In study-adjusted analyses, using data from the 2 nested case-control studies (126 cases and 472 controls), premenopausal women in the second and third tertiles of 2:16α-OHE1 were less likely to be ER- cases (RR=0.40; 95% CI: 0.16-1.00 RR=0.33; 95% CI: 0.13-0.84, respectively; Tab. V). No significant associations were observed with the ER+ cases or between 2-OHE1, 16α-OHE1 or the 2:16α-OHE1 and breast cancer by PR (data not shown). No significant associations were observed in subgroup-specific analyses of BMI, nor were any interactions found between BMI and the metabolites (p>0.10) (BMI <25 kg/m2 and ≥ 25 kg/m2). However, among nonsmokers, higher 2:16α-OHE1 was suggestive of reduced breast cancer risk (ORIIIvsI=0.57; 95% CI: 0.31-1.06; p-interaction=0.46) while higher levels of standardized 16α-OHE1 were positively associated with breast cancer risk (ORIIIvsI =2.16; 95% CI: 1.19-3.94; p-interaction=0.08). Overall results from analyses restricted to the nested case-control studies were similar to those presented in Table IV There was no significant heterogeneity between the 2 prospective studies of white women nor was there any evidence of interaction between study and any metabolite (p>0.11 for each).
Postmenopausal Women
Study-specific analyses
Higher levels of the 2:16α-OHE1 ratio (tertile III: >2.46 vs tertile I: <1.53) were associated with a non-statistically significant reduction in breast cancer risk in the studies by Fowke et al (OR=0.38; 95% CI: 0.04-3.50), Meilahn et al (OR=0.66; 95% CI: 0.23-1.88), and Wellejus et al (OR=0.76; 95% CI: 0.42-1.39) (Tab. IIIb). In contrast, the study-specific estimates for Muti et al (ORIIIvsI =1.63; 95% CI: 0.64-4.12) and Ursin et al (ORIIIvsI=1.17; 95% CI: 0.47-2.90) were suggestive of either an increased risk of breast cancer with higher levels of the 2:16α-OHE1 ratio or did not show any association.
STUDY-SPECIFIC UNADJUSTED ODDS RATIO ESTIMATES (ORs) AND 95% CONFIDENCE INTERVALS (CI): ASSOCIATION BETWEEN TERTILES a OF THE STANDARDIZED ESTROGEN METABOLITES (2-OHE1, 16α-OHE1) AND 2:16α-OHE1 AMONG POSTMENOPAUSAL WOMEN AND BREAST CANCER
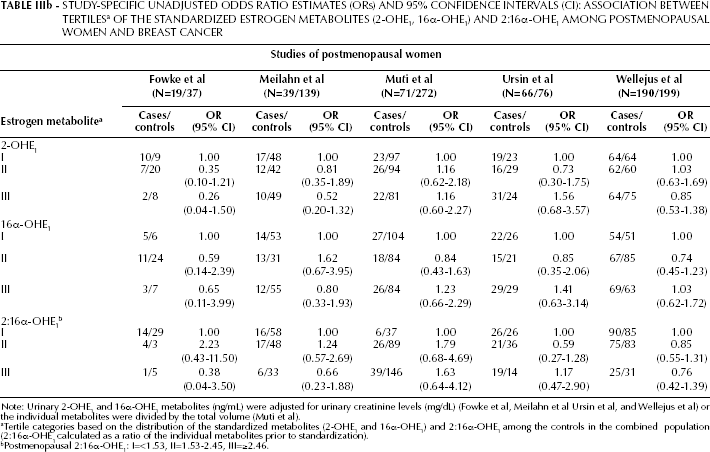
Note: Urinary 2-OHE1 and 16α-OHE1 metabolites (ng/mL) were adjusted for urinary creatinine levels (mg/dL) (Fowke et al, Meilahn et al Ursin et al, and Wellejus et al) or the individual metabolites were divided by the total volume (Muti et al).
Tertile categories based on the distribution of the standardized metabolites (2-OHE1 and 16α-OHE1) and 2:16α-OHE1 among the controls in the combined population (2:16α-OHE1 calculated as a ratio of the individual metabolites prior to standardization).
Postmenopausal 2:16α-OHE1: I=<1.53, II=1.53-2.45, III=≥2.46.
THE ODDS OF BREAST CANCER BY TERTILES OF STANDARDIZED URINARY ESTROGEN METABOLITES (2-OHE1, 16α-OHE1) AND 2:16α-OHE1
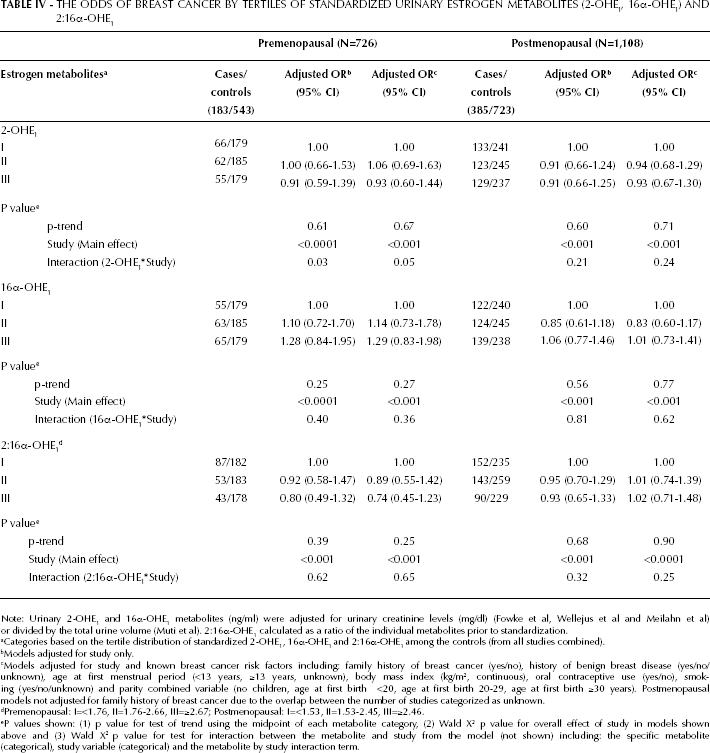
Note: Urinary 2-OHE1 and 16α-OHE1 metabolites (ng/ml) were adjusted for urinary creatinine levels (mg/dl) (Fowke et al, Wellejus et al and Meilahn et al) or divided by the total urine volume (Muti et al). 2:16α-OHE1 calculated as a ratio of the individual metabolites prior to standardization.
Categories based on the tertile distribution of standardized 2-OHE1, 16α-OHE1 and 2:16α-OHE1 among the controls (from all studies combined).
Models adjusted for study only.
Models adjusted for study and known breast cancer risk factors including: family history of breast cancer (yes/no), history of benign breast disease (yes/no/unknown), age at first menstrual period (<13 years, ≥13 years, unknown), body mass index (kg/m2, continuous), oral contraceptive use (yes/no), smoking (yes/no/unknown) and parity combined variable (no children, age at first birth <20, age at first birth 20-29, age at first birth ≥30 years). Postmenopausal models not adjusted for family history of breast cancer due to the overlap between the number of studies categorized as unknown.
Premenopausal: I=<1.76, II=1.76-2.66, III=≥2.67; Postmenopausal: I=<1.53, II=1.53-2.45, III=≥2.46.
P values shown: (1) p value for test of trend using the midpoint of each metabolite category, (2) Wald X 2 p value for overall effect of study in models shown above and (3) Wald X 2 p value for test for interaction between the metabolite and study from the model (not shown) including: the specific metabolite (categorical), study variable (categorical) and the metabolite by study interaction term.
Combined analyses
Among postmenopausal women, urinary 2:16α-OHE1 was not associated with breast cancer risk in models adjusted for study (OR=0.93; 95% CI: 0.65-1.33) or adjusted for both study and known breast cancer risk factors (OR=1.02; 95% CI: 0.71-1.48) (Tab. IV). Overall, no significant associations were observed with the standardized 2-OHE1 and 16α-OHE1 metabolites or the 2:16α-OHE1 ratio among postmenopausal women. Although the main effect of study was statistically significant (p<0.001 for all models), none of the interactions between the metabolites and either study (p-interaction>0.20) or age (data not shown) were statistically significant. Analyses limited to the 3 nested case-control studies (300 cases and 610 controls) showed similar results to those presented for the overall analysis (data not shown).
ESTIMATED RELATIVE RISK OF ESTROGEN RECEPTOR STAUS BY TERTILES OF STANDARDIZED ESTROGEN METABOLITES (2-OHE1, 16α-OHE1 AND 2:16α-OHE1) AS COMPARED TO CONTROLS a
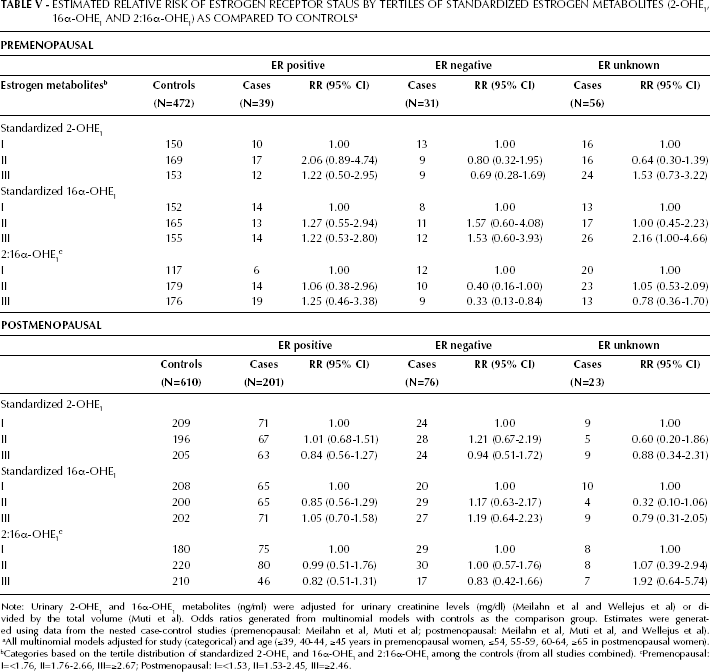
Note: Urinary 2-OHE1 and 16α-OHE1 metabolites (ng/ml) were adjusted for urinary creatinine levels (mg/dl) (Meilahn et al and Wellejus et al) or divided by the total volume (Muti et al). Odds ratios generated from multinomial models with controls as the comparison group. Estimates were generated using data from the nested case-control studies (premenopausal: Meilahn et al, Muti et al; postmenopausal: Meilahn et al, Muti et al, and Wellejus et al).
All multinomial models adjusted for study (categorical) and age (≤39, 40-44, ≥45 years in premenopausal women, ≤54, 55-59, 60-64, ≥65 in postmenopausal women).
Categories based on the tertile distribution of standardized 2-OHE1 and 16α-OHE1 and 2:16α-OHE1 among the controls (from all studies combined).
Premenopausal: I=<1.76, II=1.76-2.66, III=≥2.67; Postmenopausal: I=<1.53, II=1.53-2.45, III=≥2.46.
No significant differences were observed in the relative risk (RR) of ER+ or ER- tumors with 2-OHE1, 16α-OHE1, or 2:16α-OHE1 (Tab. V), with observed estimates in the same direction (ER+: RRIIIvsI=0.82; 95% CI: 0.51-1.31; ER-: RRIIIvsI=0.83; 95% CI: 0.42-1.66). Additionally, no significant associations were observed in the analyses by PR status (data not shown).
A significant interaction between 2-OHE1 and BMI was observed (p=0.003) among postmenopausal women. Higher levels of the standardized 2-OHE1 were associated with a reduction in breast cancer risk among women with a BMI ≥25 kg/m2 (ORIIIvsI=0.54; 95% CI: 0.34-0.87) vs those with a BMI <25 kg/m2 (ORIIIvsI=1.78; 95% CI: 0.97-3.22) (data not shown). No significant associations with either the standardized 16α-OHE1 or the 2:16α-OHE1 ratio were observed in the subgroup-specific analyses by BMI (<25 kg/m2 and ≥25 kg/m2) or smoking status (current vs non-current smoker), nor were any statistical interactions observed (Wald test p≥0.15 for each) (data not shown).
Discussion
This combined analysis suggests that higher urinary levels of the 2:16α-OHE1 ratio among premenopausal women may be inversely associated with breast cancer risk. While no associations were observed with either the standardized metabolites or 2:16α-OHE1 and breast cancer among postmenopausal women overall, findings from subgroup analyses suggest potential differences in the associations with standardized 2-OHE1 by BMI. Higher levels of the standardized 2-OHE1 were inversely associated with breast cancer risk among overweight women but not among normal weight women.
To date, 6 studies have evaluated premenopausal urinary estrogen metabolite levels (measured by ELISA) and breast cancer, with heterogeneous results reported across the studies (15-20). The original studies by Muti et al (20) and Meilahn et al (15) suggested non-statistically significant reductions in breast cancer risk among premenopausal women in the highest tertile of 2:16α-OHE1 More recently, Kabat et al (18) reported a significant protective association between higher levels of 2:16α-OHE1 and breast cancer among premenopausal women (OR ≥2.3 vs ≤1.4 = 0.50; 95% CI: 0.25-1.01). Although Kabat et al (2006) did not participate in this combined analysis, inclusion of this study likely would have strengthened the evidence supporting the inverse association observed with 2:16α-OHE1 among premenopausal women in this combined analysis. In addition to prior ELISA-based studies, a recent nested case-control study within the Nurses' Health Study (29), which measured urinary estrogen metabolites among premenopausal women using a newly developed liquid chromatography mass spectrometry assay (LC-MS/MS) (30), observed a nonsignificant inverse association with the 2:16α-OHE1 ratio. With regards to circulating estrogen metabolites, only 1 prior nested case-control study of premenopausal women (31) has been conducted, with results suggesting no association between serum 2:16α-OHE1 and breast cancer risk.
Twelve studies have evaluated the 2-OHE1 and 16α-OHE1 metabolites in relation to breast cancer among postmenopausal women, of which 6 were nested case-control studies in urine (15, 20, 22), serum (32, 33) or plasma (34), and 5 were case-control studies of urinary metabolites (16-19, 21). Findings from case-control studies have suggested either a potential reduction (17-19) in breast cancer risk with a higher 2:16α-OHE1 ratio or no association (21), while the nested case-control studies mostly suggested no overall association (among postmenopausal women not on HT), regardless of whether urinary or blood specimens were analyzed. Differences in the results between case-control and nested case-control studies may partly be explained by the inability to establish temporal inference in case-control studies. Additionally, in a recent prospective study by Fuhrman et al (35), which measured serum estrogen metabolites by LC-MS/MS (36), the ratio of 2-hydroxylation pathway to the 16-hydroxylation pathway was suggestive of an inverse association with postmenopausal breast cancer risk (HR=0.69; 95% CI:0.47-1.02). This suggests that the null findings reported in this combined analysis may also, in part, be due to limitations with the ELISA assay (26) or the specific metabolites measured (that is, individual hydroxylated metabolites vs comprehensive pathways).
Overall, this research area has been limited by the number of studies conducted and the relatively small number of cases included within those analyses, particularly the premenopausal cases. We attempted to address this limitation by carrying out this combined analysis of premenopausal women, nevertheless, the small number of eligible and participating studies limited our power to detect associations. Despite this, one cannot exclude the possibility that a null finding reflects a true lack of association between urinary estrogen metabolite levels (as measured by ELISA) and breast cancer.
Although hormonally-related factors may have different associations with ER+ tumors and ER- tumors (37-39), with estrogens more strongly associated with ER+ tumors than with ER- tumors (40), findings have been inconsistent. Furthermore, little is known regarding the relationship between estrogen metabolites and ER-specific subtypes among pre and postmenopausal women. In this combined analysis, we observed a reduced relative risk of ER- breast cancer among premenopausal women in the second and third tertiles of 2:16α-OHE1 compared to the lowest tertile. Although this finding was based on a fairly small number of cases and controls and was driven largely by the contribution of cases from the Muti et al study, it certainly warrants further research. To our knowledge, only 1 other study (18) has evaluated the association of urinary estrogen metabolites and invasive breast cancer among premenopausal women by ER status. In this study the authors observed significantly reduced odds of ER+ breast cancer with increasing levels of the 2:16α-OHE1 ratio (p-trend=0.02). Due to the stronger associations observed between circulating estrogen levels and ER+ breast cancer, one might expect a similar association between 2:16α-OHE1 and ER+ cases. However, since the 2-OHE1 and 16α-OHE1 metabolites operate through different pathways, with different potential genotoxic and mitogenic effects, our observed association with ER- tumors cannot be dismissed.
In our analysis of postmenopausal women, higher levels of urinary 2:16α-OHE1 were not significantly associated with either ER+ or ER- tumors and, further, our results suggest no difference in the overall effect of metabolites on ER-specific tumors given the similar risk estimates observed with both ER+ and ER- tumors. Results from prior case-control studies have been inconsistent, with results suggesting higher mean levels of the 2:16α-OHE1 ratio among postmenopausal women with ER+ tumors compared to ER- tumors (17) or possibly an inverse association between the 2:16α-OHE1 ratio and ER- breast cancer (18). Additionally, findings from the Nurses' Health Study suggest significant positive associations between plasma levels of 2-OHE1 and 2:16α-OHE1 and ER-/PR-tumors (34). With regards to PR status, the associations of each metabolite with breast cancer did not appear to vary by PR in either pre or postmenopausal women; however, our combined analysis was limited by the small number of cases classified by PR status. Overall, these inconsistent findings indicate the need for larger, more carefully designed studies with biologically well-characterized postmenopausal breast cancer cases.
Despite the standardization of the individual metabolites in this combined analysis and the apparent similarities across the studies that were combined, significant heterogeneity across studies was observed. Adjustment for study attenuated the protective associations observed in unadjusted models, suggesting that there are important, unaccounted differences across the studies including study design, laboratory variability, and the distribution of the estrogen metabolites overall and among various ethnic populations, particularly white and Asian women (41-45). Furthermore, in our analysis of premenopausal women, the statistically significant interaction observed between 2-OHE1 and study was no longer significant once analyses were restricted to the nested case-control studies, which excluded the case-control study of Asian women. While lower levels of 2-OHE1 and 16α-OHE1 among Asian women are expected, given the higher levels of endogenous estrogens reported among white women as compared to Asian women (43, 46), this significant study interaction highlights differences in estrogen metabolism among racial/ethnic populations.
Whether urinary estrogen metabolite levels vary with the timing of urine collection during different phases of the menstrual cycle remains unclear. Some studies suggest a potential difference (47) while others do not (23). In this analysis, adjustment for menstrual phase at the time of urine collection did not affect estimation. Given that urine samples in the ORDET study were collected during the luteal phase and menstrual phase was an original matching criterion in the Guernsey study, these original study design features may explain our observed lack of variability in estrogen metabolites by menstrual phase.
Strengths to this combined analysis include the use of primary individual level data, the ability to assess the relationship between estrogen metabolites and breast cancer using common cutpoints (48), the large number of combined breast cancer cases (which increases our overall power to detect associations), and the availability of information on relevant covariates. This analysis is a first attempt to try to reconcile the evidence on urinary estrogen metabolites and breast cancer by addressing this research question using a combined analysis. Furthermore, the inclusion of nested case-control studies provides the opportunity to assess metabolite levels not affected by disease status or by treatment.
Some limitations to this study should be considered when interpreting the findings. Genetic polymorphisms (49) and dietary factors (50) may influence the particular estrogen metabolic pathway favored, while additional estrogen metabolites, such as 4-hydroxyestrone, as well as quinone adducts may be important factors in breast carcinogenesis (51, 52); however, these components could not be assessed in this combined analysis. We attempted to collect detailed information on ER/PR status but information was limited given the availability of this assay at the time the studies were initiated (e.g. Guernsey). Despite multiple attempts to include all previously published eligible studies, data from a few studies could not be included, introducing potential selection bias. Additionally, the postmenopausal study population was restricted to women not currently on menopausal HT and thus, our results reflect the metabolism and excretion of spontaneous levels of circulating estrogen metabolites. For the higher levels of circulating estradiol achieved with the use of menopausal hormone therapy, the metabolic pathways as well as their importance may be different, as was reported by Wellejus et al (22). To date, it remains unclear whether a single measurement of estrogen metabolites is representative of long-term levels, or alternatively, whether estrogen metabolite levels measured at multiple time points, may be more informative in breast cancer studies. To our knowledge, 1 prior study has addressed this concern among premenopausal women: findings from the Nurses' Health Study II (53) suggest that baseline urinary estrogen metabolite measures may adequately represent at least 3 years of exposure among premenopausal woman.
The role of ovarian hormones, particularly estrogen, in breast carcinogenesis is complex, and the involvement of estrogen metabolites in this process has yet to be determined. Understanding the potential role of estrogen metabolites in the carcinogenic process will not only help elucidate the underlying mechanism of estrogen exposure but may also offer the potential to identify high-risk women on which to concentrate preventive efforts.
SUPPLEMENTAL TABLE - URINARY ESTROGEN METABOLITE LEVELS [MEDIAN (10th,90th)] BY STUDY AND CASE/CONTROL STATUS
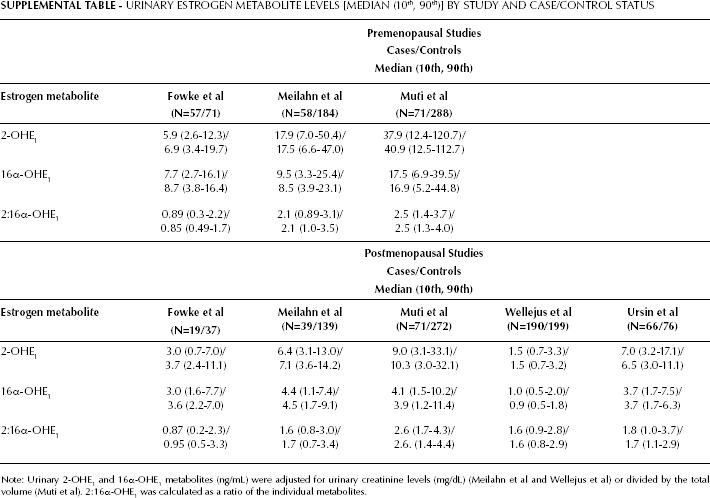
Note: Urinary 2-OHE1 and 16α-OHE1 metabolites (ng/mL) were adjusted for urinary creatinine levels (mg/dL) (Meilahn et al and Wellejus et al) or divided by the total volume (Muti et al). 2:16α-OHE1 was calculated as a ratio of the individual metabolites.
In summary, although we did not observe statistically significant associations overall, with either the standardized individual metabolites or with the ratio, this combined analysis suggests a possible reduction in breast cancer risk with higher premenopausal levels of the 2:16α-OHE1, and also that the association between 2-OHE1 and postmenopausal breast cancer may vary with BMI. Furthermore, this combined analysis approach allowed us to highlight the variability in metabolite levels across geographic and racial/ethnic populations along with the complexity in studying urinary estrogen metabolites in relation to breast cancer. Larger studies with estrogen metabolites measured at multiple time points or during different periods of life are needed in order to evaluate this potential association overall, within specific subgroups, and in relation to tumor hormone receptor status.
