Abstract
Nephropathy associated with BK virus has emerged as an important cause of allograft failure in renal transplant recipients. Here we exploited a recently developed novel monocyte based solid phase T cell selection system, in which monocytes are immobilized on solid support, for antigen-specific T cell purification. The underlying hypothesis of this new method is that antigen-specific T cells recognize, bind their cognate antigens faster than non-specific T cells and are concentrated on the surface after removing the non-adherent cells by washing. Moreover, activated antigen-specific T cells proliferate more rapidly than non-specific T cells, further increasing the frequency and purity of antigen-specific T cells. Optimal selection times for BK virus-specific T cells are studied. Our data demonstrated that T cell selection can usually increase the frequency of antigen-specific T cells by > 10 fold, whereas T cell expansion following the selection boost the frequency of antigen-specific T cells by another ~10 fold. This new T cell selection system is superior to traditional stimulation method (i.e. simply mixing antigen presenting cells and lymphocytes together) in generating antigen-specific T cells. This inexpensive and simple T cell selection system can produce large quantity of highly purified BK virus-specific T cells within 1–2 weeks after initial T cell activation.
Introduction
Polyomaviruses are small DNA viruses that typically establish persistent, but inapparent infection of their natural hosts. Among 12 polyomaviruses identified, BK virus (BKV) can cause latent infection and reactivate following immunosuppression in human. Particularly, nephropathy associated with BKV has emerged as an important cause of allograft failure in renal transplant recipients (Purighalla et al. 1995; Nickeleit et al. 1999; Nickeleit et al. 2000; Randhawa et al. 2000). Previous studies (Nickeleit et al. 2000; Randhawa et al. 2000; Hirsch et al. 2002; Ramos et al. 2002) have shown that reactivation of the virus occurs in 10% to 60% of renal transplant recipients, and 1% to 5% of renal transplant recipients develop BK nephropathy, with loss of allograft function occurring in about half the cases. There is no BKV specific antiviral therapy. Cautious decrease of immunosuppression appears to be the most commonly accepted initial approach to treatment (Nickeleit et al. 2000; Mathur et al. 1997) though this change may result in an increased risk of subsequent rejection (Mayr et al. 2001). Therefore new effective therapy is needed to eradicate BKV infection before establishment of BK nephropathy.
The link between spontaneous BKV reactivation and immunospression in renal-transplant recipients suggests that T-cell immunity is critical to control viral replication. In mouse models, it has been shown that cytotoxic T cells are critical for the clearance of acute polyomavirus infection (Ljunggren et al. 1994; Luckacher et al. 1998; Luckacher et al. 1999). In human, the presence of JC virus (JCV) VP1-specific T cells was demonstrated to be crucial in the prevention of progressive multifocal leukoencephalopathy progression (Koralnik et al. 2001; Koralnik et al. 2002). Furthermore, recent studies have demonstrated that adoptive cell immunotherapy is effective in treating Epstein-Barr virus (EBV)-related lymphoproliferative disease or Cytomegalovirus (CMV) associated interstitial pneumonia occurring in solid-organ transplant or hematopoietic stem cell transplantation recipients (Riddell et al. 1992; Rooney et al. 1995; Heslop et al. 1996). Although it is appealing to apply adoptive cell immunotherapy in BKV-related interstitial nephropathy, this approach requires knowledge of T cell responses to BKV antigens and an effective means to generate highly purified BKV-specific T cells simply, rapidly and inexpensively.
Of the proteins expressed by BKV, both T antigen (T-ag) and the capsid proteins (VP1, VP2 and VP3) are highly immunogenic. Two conserved antigens within BKV and JCV VP1 polypeptides restricted to human leukocyte antigen (HLA)-A*0201 have been described (Krymskaya et al. 2005; Chen et al. 2006). Within T-ag, our group previously reported several peptides as candidate major histocompatibility complex (MHC) class I and II T-cell epitopes (Li et al. 2006). Among these peptide epitopes, a 9mer peptide, LPLMRKAYL, was confirmed to be a super motif and restricted to HLA-B*0702 and -B*08 (Li et al. 2006). Furthermore, we developed a novel solid phase T cell selection system to purify antigen-specific T cells recently. This new method has been exploited to purify viral and tumor antigen-specific T cells (Li et al. 2006; Li et al. 2007; Li et al. 2008). In this system, antigen presenting cells such as peptide-loaded-monocytes were immobilized on solid support for antigen-specific T cell selection. The underlying hypothesis was that antigen-specific T cells recognize and bind to their cognate antigens faster than non-antigen specific T cells. Therefore antigen-specific T cells can be concentrated on the surface after removing the non-adherent cells by washing. Moreover, activated antigen-specific T cells proliferate more rapidly than non-specific T cells, further increasing the frequency and purity of antigen-specific T cells. In our previous study (Li et al. 2006) highly purified BKV-specific T cells have been prepared with this novel method for cytotoxicity study. Now we would like to present the complete data of BKV-specific T cell purification with monocyte based T cell selection system here. In this report, we demonstrate that our monocyte based T cell selection system is superior to traditional repeated stimulation method (i.e. simply mixing antigen presenting cells and lymphocytes together) to generate BKV-specific T cells. Large-scale production of highly purified BKV-specific T cells can be generated in 2–3 weeks for adoptive T cell immunotherapy against BKV nephropathy.
Methods
Study Population
Peripheral blood mononuclear cells (PBMCs) were harvested by leukapheresis from paid healthy donors at Thomas Jefferson University. Lymphocytes and monocytes that were seperated by Elutra® (Gambro) were processed by Ficoll-Hypaque, frozen and stored in liquid nitrogen until use. This study has been reviewed and approved by IRB at Thomas Jefferson University.
Peptides and Cell Lines
BKV T-ag candidate major histocompatibility complex class I T-cell epitope LPLMRKAYL (P27–35) restricted to human leukocyte antigen (HLA)-B*0702 and -B*08 was reported previously (Li et al. 2006). BKV VP1 candidate major histocompatibility complex class I T-cell epitope AITEVECFL (P44–52) was reported in the previous studies (Krymskaya et al. 2005; Chen et al. 2006). EBV HLA*B0702-binding peptide RPPIFIRRL (Khanna et al. 2000) and Human Papillomavirus 16 HLA*B8-binding peptide TLHEYMLDL (Oerke et al. 2005) were used as negative control for intracellular IFN-γ staining. These peptides (>90% purity) were purchased from Biosynthesis Inc.
Generation of EBV-Transformed B Cell Lines (LCL)
5–10 × 106 PBMC was incubated with concentrated supernatant of B95-8 cultures, in the presence of 1 μg/mL cyclosporin A (Sandoz) to establish an LCL cell line.
Monocyte Immobilization
10 × 106 monocytes were thawed and immediately added to one well of a 6-well-plate (BD Falcon™ Primaria®) in 2 mL of culture media (CM) consisting of RPMI 1640 medium (Mediatech, Inc) containing 10% heat-inactivated normal AB serum (Gemini) and DNAse I (960 U) (Worthington Biochemical Corporation). After 90 minutes incubation in cell culture incubator (37 °C, 5% CO2), the medium was removed and adherent cells were rinsed with warm (37 oC) phosphate-buffered saline (PBS, Mediatech, Inc), pulsed with 2 μM peptide in 3 ml CM for 90 minutes until use. In general, about 6 × 106 monocytes could be immobilized on the surface of a single well of the 6-well-plate.
Generation of Polyclonal BKV Antigen-Specific Cytotoxic T Cells with Traditional Stimulation Method
PBMCs were co-cultured with autologous monocytes that were previously pulsed with BKV peptide (2 μM) at a responder/stimulator ratio of 10:1. Starting on day 7, the responder cells were re-stimulated with peptide-loaded-monocytes at a responder/stimulator ratio 10:1. Also, on day 7 of culture and every 2–3 days thereafter, recombinant human IL-2 (rhIL-2; Proleukin; Chiron), final concentration 20 U/ml, was added to expand the proliferating T cells. On day 14, the frequency of antigen-specific T cells was analyzed by flow cytometric intracellular cytokine assay for IFN-γ production.
BKV Antigen-Specific T Cell Selection and Expansion (Fig. 1)
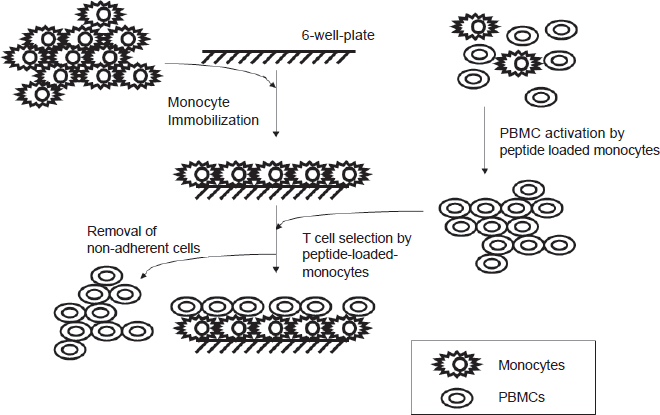
Lymphocytes (90 × 106 ± 10 in 3 mL CM, 37 °C) that were previously stimulated with BKV peptide-loaded-monocytes for 7 days were added onto immobilized monocytes in cell culture incubator. Various selection times (3, 5, 7.5, 10 and 12.5 minutes, respectively) were tested. Non-adherent cells were removed at the end of each selection times. The remaining adherent cells were gently washed with warm CM (37 °C) three times and again after 30 minutes incubation at 37 °C. The next day, adherent cells were removed by gentle agitation and transferred to a culture flask. RhIL-2 (20 U/ml) was added and thereafter every 2–3 days. On day 7, cells were analyzed by flow cytometric intracellular cytokine assay for IFN-γ production.
Intracellular Cytokine Staining (ICC)
Intracellular IFN-γ staining was performed as described (Li et al. 2006). Briefly, 2 × 106 lymphocytes were cultured with irradiated (25 Gy) peptide-loaded-monocytes or DCs expressing BKV antigens (lymphocytes:monocytes or DCs 1/1). After 2 hours, 10 μg/ml brefeldin A (Sigma) was added and cultures were incubated for a further 4 hours. After this period, cells were stained with CD3-PerCP and CD8-PE, fixed in FACS Lyse, permeabilized with FACS Perm2 and labeled with anti-IFN-γ–fluorescein isothiocyanate (all from Becton Dickinson) according to the intracellular-staining procedure protocol supplied by the manufacturer. Immunofluorence measurement was acquired on a FACScalibur flow cytometer and analyzed using CellQuest software. The frequency determined for the control (monocytes loaded with control peptide) was subtracted from the frequency for BKV peptides-stimulated population to determine the true antigen-specific T-cell frequency.
The procedure for intracellular IFN-γ staining of selected lymphocytes by immobilized monocytes on the solid support was modified from the procedure described above. After selecting lymphocytes with immobilized antigen presenting cells, non-adherent cells were removed at the end of each selection time. The remaining adherent cells were gently washed with 37 °C culture media. Washing was repeated after 30 minutes incubation at 37 °C. At 2 hours after selection, after removing culture media, 6 ml culture media containing 10 μg/ml brefeldin A was added and cultures were incubated for a further 4 hours. This was then followed by the procedure of cell staining with antibody, lysing and permeation described above. The frequency determined for the control (monocytes loaded with control peptide) was subtracted from the frequency for BKV peptides-stimulated population to determine the true antigen-specific T-cell frequency.
Tetramer Staining
PE-labeled BKV HLA*B07-tetramer (T-ag P27–35 LPLMRKAYL, Beckman Coulter) was used to detect P27–35-specific T cells in the CTL lines from Donor 6 (HLA*A0201+/*B07+). EBV HLA-A2-tetramer (LMP2 peptide CLGGLLTMV, Beckman Coulter) was used as tetramer control. Tetramer staining was done as described by the manufacturer. Briefly, CTLs (10 6 ) were incubated at room temperature for 30 min in (PBS)/1% fetal calf serum (FCS) containing the PE-labeled tetrameric complex. Samples were co-stained with anti-CD8 FITC and anti-CD3 PerCP. Stained cells were fixed in PBS containing 0.5% paraformaldehyde. Cells were acquired on a FACScalibur flow cytometer and analyzed using Cell Quest software.
Chromium Release Assay
The cytotoxic specificity of CTL line was analyzed in a standard 4-h chromium-51 release assay (Brunner et al. 1968). The following target cells were tested: autologous LCL pulsed with P27–35 vs. autologous LCL with effector/target ratio of 30:1, 10:1 and 3:1.
Results
One of the most commonly used methods to generate antigen-specific T cells is repeated stimulation of peripheral blood mononuclear cells (usually weekly) with antigen presenting cells pulsed with target peptide. This is followed by expansion in the presence of rhIL-2. In our new T cell purification method we exploited immobilized autologous monocytes to select BKV-specific T cells from previously activated PBMCs, followed by T cell expansion. We have performed T cell selection experiments with PBMCs from 8 healthy donors (3 times from each donor). Here we present data from healthy donor 2 (HLA*B08+) and 6 (HLA*B0702+) to illustrate the process of T cell selection. General scheme of solid phase T cell selection process was outlined in Figure 1.
The frequency of BKV T-ag P27–35-specific cytotoxic T cells (CTLs) was determined to be 0.1% (mean) by intracellular cytokine staining 7 days after PBMCs of healthy donor 2 (HLA*B08+) were stimulated with P27–35-loaded-monocytes. To confirm our hypothesis, we examined the frequency of P27–35-specific T cells selected at various selection times (ranging from 3 to 12.5 minutes) by immobilized peptide-loaded-monocytes on the support surface. After non-adherent cells were removed at the end of each selection time, intracellular IFN-γ staining of selected lymphocytes on the support surface was performed based on the modified procedure described in Method section. There were 1.9%, 2.1%, 2%, 2.1% and 2% (mean) IFN-γ producing cells on the solid support selected at 3, 5, 7.5, 10 and 15 minutes, respectively (Figs. 2A and B). Because monocytes might also present antigens other than P27–35 and induce IFN-γ production among selected T cells, we examined the quantity of IFN-γ production that might be contributed by non-specific antigens. There was no detectable IFN-γ production when P27–35-stimulated-PBMCs were selected with immobilized plain monocytes or monocytes loaded with irrelevant peptide (EBV HLA*B0702-binding peptide RPPIFIRRL) (selection time of 7.5 minutes, data not shown). Next we expanded the selected T cells right after selection (We did not expand lymphocytes selected at 3 minutes because of scarcity of selected cell population). On day 7, IFN-γ producing cells were increased to 24% (mean) in expanded lymphocytes selected at 7.5, 10 and 12.5 minutes (Figs. 2C and D), whereas there was only 2% (mean) P27–35-specific T cells generated through traditional repeated stimulation method (data not shown). There were 120, 140 and 160 × 106 cells (mean) among the final expanded cell population selected at 7.5, 10 and 12.5 minutes, respectively.
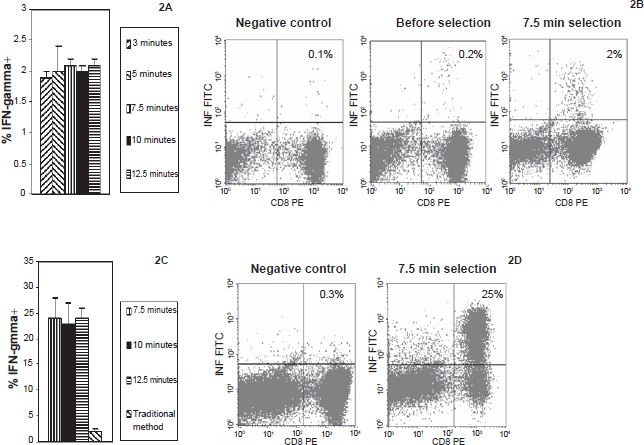
Selection of P27–35-specific T cells (Donor 2) by immobilized monocytes.
Similar T cell selection experiments were also performed with PBMCs from Donor 6 (HLA*B0702+). Data on P27–35-specific T cell selection from donor 6 were shown in Figure 3. In donor 6, the pre-selection frequency of P27–35-specific CTLs was 0.1% (mean) 7 days after PBMCs were stimulated with peptide-loaded-monocytes. After T cell selection with immobilized peptide-loaded-monocytes, IFN-γ producing cells on the solid support were increased to 2.7%, 2.6%, 3.1% and 2.9% (mean) selected at 3, 5, 7.5 and 12.5 minutes, respectively (Figs. 3A and B). Again, there was no detectable IFN-γ when these same activated lymphocytes were selected with immobilized plain monocytes or monocytes loaded with irrelevant peptide (Human Papillomavirus 16 HLA*B8-binding peptide TLHEYMLDL) (selection time of 7.5 minutes, data not shown). Expansion of these selected T cells (selection time 5, 7.5 and 12.5 minutes) for 7 days led to 25% (mean) frequency of P27–35-specific T cells in the final cell populations, whereas the frequency of P27–35-specific T cells was 2% (mean) in lymphocytes generated by traditional method (Figs. 3C and D). The total cell counts are 100, 150 and 180 × 106 (mean) among the final expanded cells of 5, 7.5 and 12.5 minutes selection time, respectively. Based on these findings, we performed VP1-specific T cell purification with PBMCs of Donor 10 (HLA*A0201+) with selection time of 7.5 min. After initial sensitization with peptide (VP1 P44–52), there was 0.8% T cells specific to VP1 P44–52. One week after selection and expansion, there were 120 million lymphocytes with frequency of 55% specific for VP1 peptide (data not shown).
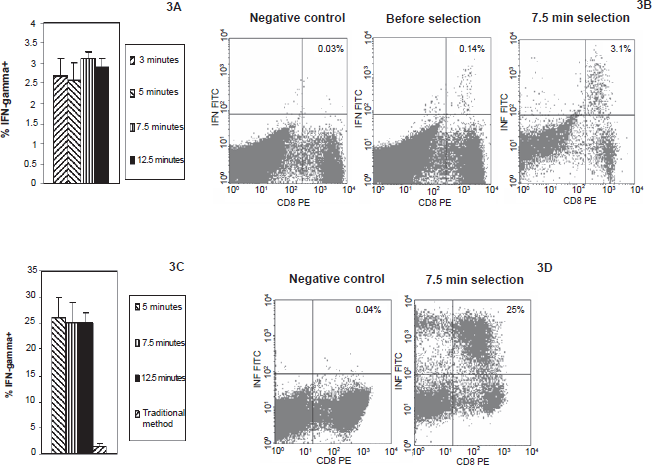
Selection of P27–35-specific T cells (Donor 6) by immobilized monocytes.
To further increase the frequency of P27–35-specific T cells, we decided to perform 2nd round T cell selection and expansion with lymphocytes generated from the 1st round T cell selection and expansion. Because there are significant quantity of P27–35-specific T cells (purity now in 20's percent) after 1st round selection and expansion, these lymphocytes can be subject to several consecutive selections (2nd round) to increase both purity and total number of P27–35-specific T cells. In experiments with these multiple consecutive selections (Fig. 4A), nonadherent lymphocytes removed from selection in 1st well are subject to selection in 2nd and so forth with a total of 3 selections (in 3 wells) were performed. 90 × 106 cells in 3 ml culture media generated from the 1st round selection and expansion were selected consecutively through 3 monocyte-coated wells (pulsed with peptide). Selection time was 7.5 minutes. After the selected cells were expanded for 7 days, the frequency of P27–35-specific T cells was increased to 60% and 55% (mean) for donor 2 and 6, respectively (Figs. 4B and 5A). There were 150 × 106 cells (mean) in the final cell products generated from each selection. We also analyzed these expanded cells (Donor 6) by tetramer staining, which yielded 53% (mean) of P27–35-specific T cells among cells generated from consecutive selections (Fig. 5B). Cytotoxicity study on the specific killings of BKV antigen expressing targets (here LCL pulsed with BKV peptide antigen) with purified T cells were demonstrated in Figure 6.
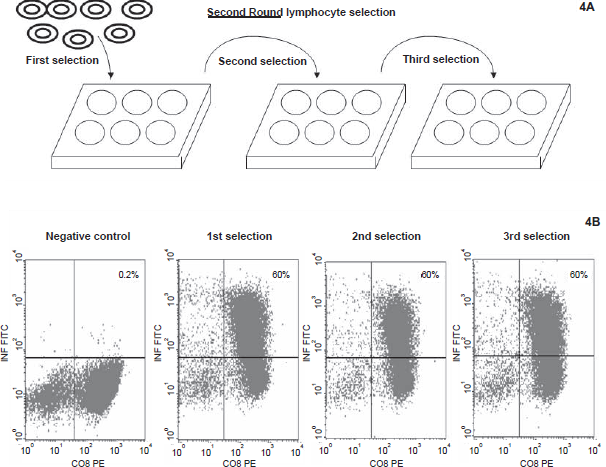
Effect of 2nd round P27–35-specific T cell selection of Donor 2.
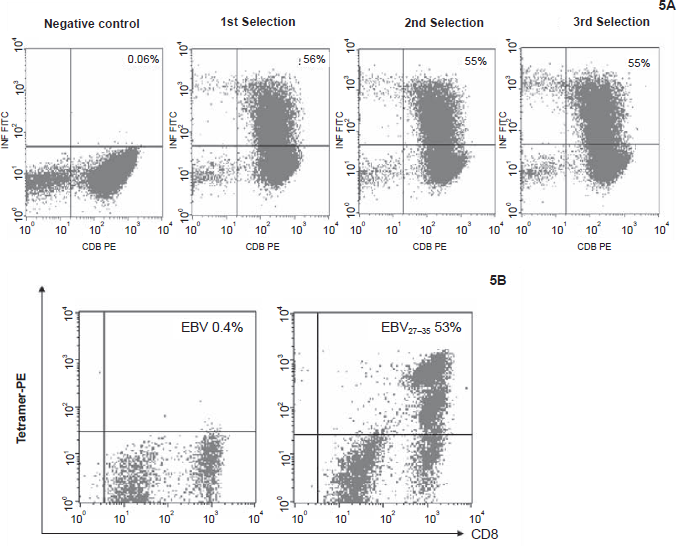
Effect of 2nd round P27–35-specific T cell selection of Donor 6.
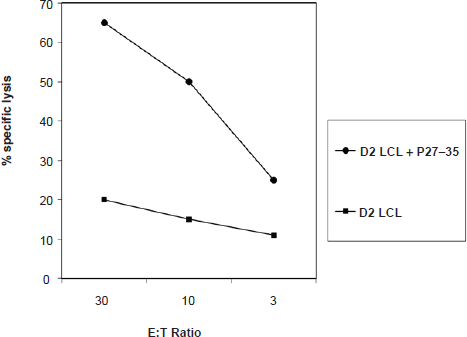
Purified T cells (donor 2, HLA-*B08) specific to BKV P27–35 lysed P27–35-loaded autologous LCL cell lines preferentially over unmanipulated autologous LCL cell lines. Each point represents the mean of data from triplicate experiments.
Discussion
In the past two decades, adoptive T cell therapy targeting malignancies such as melanoma and EBV-related lymphoproliferative disease or CMV associated interstitial pneumonia occurring in solid-organ transplant or hematopoietic stem cell transplantation recipients has shown promising potential. However, it has been a great challenge to produce functional antigen-specific T cells inexpensively, simply and rapidly. While several approaches to generate viral antigen and tumor-specific CTLs have been successfully introduced in clinical experimental protocols, the establishment of a broadly applicable process is still lacking. There are significant limitations in MHC-tetramer technology. They are costly and limited to a few HLA phenotypes. The IFN-γ capture assay has been shown to be inefficient and nonspecific in T cell selection. A protocol established by Comoli et al. (Comoli et al. 2003) to generate BKV-specific T cells requires preparation of DCs and use of BKV lysate. In addition it requires repeated weekly stimulation of PBMCs with antigen presenting cells. This would make timely therapy against BKV reactivation difficult. The difficulty of generating BKV T-ag specific T cells was further exacerbated by the presence of very low frequency of precursor memory T cells specific to BKV. In our previous study (Li et al. 2006), PBMCs from 17 healthy donors were screened directly by flow cytometry to detect T-cell responses to a BKV T-ag overlapping peptide library (171 15 mer peptides). No significant IFN-γ production was identified in any individuals, who were seropositive for BKV by ELISA. In accord to these findings, initial stimulation of PBMCs with T-ag epitope peptides usually yields very low frequency of T-ag specific T cells. Our novel antigen-specific T cell selection system provides a valuable tool to harvest antigen-specific T cells with very low frequency in PBMCs. Of the two components that are comprised of this T cell purification method, T cell selection can usually increase the frequency of antigen-specific T cells by > 1 log, whereas T cell expansion following the selection boost the frequency of antigen-specific T cells by another 1 log. These findings are consistent with results in our study on EBV LMP2-specific T cell and melanoma reactive T cell selection (Li et al. 2007; Li et al. 2008). Although the therapeutic doses of BKV-specific T cells is unknown currently, previous experiences of adoptive cell therapy against EBV and Cytomegalovirus indicates that it should be in the magnitude of several 10 8 (Rooney et al. 1998; Walter et al. 1995). Therefore, clinical scale of highly purified functional BKV T-ag specific T cells can be generated for adoptive cell immunotherapy in 2–3 weeks with our method.
In the process of T cell selection, there are significant amount of non-specific T cells even after 2 rounds of T cell selection and expansion. Part of the answers to the identities and characteristics of these T cells lie in the concept of TcR (T cell receptor)-binding degeneracy or TcR promiscuity (i.e. the fact that TcR recognition does not obey a one TcR-one peptide/MHC relationship). Previous studies have demonstrated that the antigen-binding surface of the TCR exhibits conformational flexibility, which may contribute to the high degree of promiscuity or cross-reactivity evident in TcR recognition of peptide-MHC (Rudolph et al. 2002a; Rudolph et al. 2002b). Theoretical considerations and experimental data suggest that a typical TcR can recognize in excess of 10 5 peptide-MHC complexes (Mason et al. 1998). Moreover, previous studies have also established a threshold-of-activation model for T-cell recognition and signaling (Sloan-Lancaster et al. 1996). The capacity of an extracellular stimulus to trigger activation to different thresholds is probably dictated by the amount of TcR cross-linking induced and the affinity of the ligand for the TcR. Thus, as a peptide analogue becomes more structurally similar to the agonist ligand, activation to higher thresholds will be accomplished and more effector functions will be stimulated (Sloan-Lancaster et al. 1993; Evavold et al. 1991; Evavold et al. 1993). In other words, agonists cause the full range of T cell activation phenotype from activation of signaling cascades to cytokine secretion or target cell killing and to proliferation. Partial agonists elicit only some of the features of T cell activation and antagonists can disable the signaling induced in response to an agonist ligand. The combination of TcR promiscuity and the threshold-of-activation model of T-cell recognition and signaling should explain why not every T cells selected by immobilized monocytes, which presents both BKV peptide and unknown antigens are able to secret IFN-gamma.
For BKV antigen-specific T cell selections, there are two types of kinetics depending on quantities of antigen-specific T cells and immobilized monocytes. In first scenario (e.g. Figs. 2B or 3B), after PBMCs were stimulated with peptide-loaded-monocytes, there were 0.1% (i.e. 9 × 104) of antigen-specific T cells among 90 × 106 cells used for T cell selection. This is much less than 6 × 106 immobilized monocytes and the kinetics of reaction 1 should follow first order reaction.
Therefore reaction 1 can be simplified to reaction 2 since the rate of reaction 1 is mainly determined by the quantity of antigen-specific T cells.
In 1st order kinetics, at each t1/2, 50% of floating antigen-specific T cells will be selected and removed by immobilized APCs. Although we do not have data on the value of t1/2 of BKV-specific T cells, examinations of selection of EBV LMP2-specific T cells with immobilized monocytes demonstrated t1/2 around 10 minutes (unpublished preliminary results). Because there was 50% of antigen-specific T cells (90 × 106 × 0.1% × 50% = 4.5 × 104) left after 1st selection, selection after first one with 2nd monocyte layer in 6-well-plate by one t1/2 can remove half of remaining antigen-specific T cells. In second scenario (e.g. in experiment Fig. 4), there are 25% of BKV-specific T cells after 1st T cell selection and expansion. Again, 90 millions lymphocytes are used for selection experiments. There are 25% × 90 × 106 = 22.5 × 106 BKV antigen-specific T cells. T cell selection should follow 2nd order kinetics (we do not have preliminary data on t1/2 of 2nd order kinetics at this moment). This large quantity of BKV-specific T cells makes it possible for multiple T cell selections in order to generate large quantity of BKV-specific T cells. Currently we are conducting studies to determine the t1/2 of T cell selection kinetics.
Human polyomaviruses are ubiquitous, and infection is widespread as indicated by seroprevalence rates of up to 90 percent worldwide (Knowles, 2001). In accord to these findings, previous study (Hirsch et al. 2002) suggested that BKV nephropathy constituted a secondary, rather than a primary, infection in most patients. These findings argued against the hypothesis that transplantation of kidneys from BKV-seropositive donors into BKV-seronegative recipients was a major cause of BKV nephropathy. However, the possibility that donor derived viral strains have a role could not be ruled out. Several risk factors associated with BKV nephropathy have been identified. One of them is the BK plasma viral load (Hirsch et al. 2002; Limaye et al. 2001). In one study (Hirsch et al. 2002), BK viremia was found to have 100% sensitivity, 88% specificity, 50% positive predictive value, and 100% negative predictive value. Although there have been no human studies of adoptive cell immunotherapy against BKV to confirm its efficacy, clinical observation that BKV replication may be controlled by reducing the level of maintenance immunosuppression implies that cellular immune response is critical to clear viremia. In addition, the success of adoptive cell immunotherapy in treating EBV-related lymphoproliferative disease or CMV associated interstitial pneumonia occurring in solid-organ transplant or hematopoietic stem cell transplantation recipients (Riddell et al. 1992; Rooney et al. 1995; Heslop et al. 1996) suggests that adoptive cell immunotherapy should be able to clear the viremia and restore cellular immunity. This was confirmed in mouse model that cytotoxic T cells are critical for the clearance of acute polyomavirus infection (Ljunggren et al. 1994; Luckacher et al. 1998; Luckacher et al. 1999). Therefore it is reasonable to believe that the clearance of viremia and restoration of cellular immunity should help to improve clinical outcome even in the setting of HLA-mismatched renal graft.
Our data presented here indicates that BKV T-ag specific T cells generated in this study could have immediate therapeutic implications in kidney transplant recipients who developed BKV viremia. In particular, a preemptive strategy is highly desirable in clinical scenarios where immunosuppression discontinuation is complicated by a high risk of rejection. Early phase clinical trial of adoptive cell immunotherapy with or without BKV vaccines containing T-ag peptides following cell infusion will be undertaken to determine its efficacy.
Financial Support
Department of Health, Pennsylvania.
Conflict of Interest
The authors have no commercial or other association that might pose a conflict of interest.
