Abstract
Aim
To assess serum HGF concentration in individuals with schizophrenia and investigate the efficacy of zinc and B-6 therapy on these levels.
Subjects and methods
Serum from 18 individuals diagnosed with schizophrenia and 19 age and gender similar controls (P = 0.18) were tested for HGF concentration using ELISAs, and tested for copper and zinc plasma levels using inductively-coupled plasma-mass spectrometry.
Results
HGF serum levels of individuals with schizophrenia, before zinc and B-6 therapy, were significantly lower than age and gender similar controls (P = 0.016), and significantly lower in schizophrenia patients pre-therapy compared to post therapy (P = 0.028). HGF levels normalized (reached levels similar to controls) post-therapy. Zinc levels in these same individuals also normalized, and perceived symptoms, particularly anxiety (P = 0.03), improved significantly after therapy.
Discussion
These results suggest an association between low HGF levels and schizophrenia and demonstrate that zinc and B-6 therapy may be associated with the normalization of HGF levels and perceived improvement in symptoms.
Introduction
Schizophrenia is a chronic, severe, and disabling brain disorder that has affected people throughout history. About 1 percent of Americans have this illness, 1 but it occurs in 10 percent of people who have a first-degree relative with the disorder. The risk is highest for an identical twin of a person with schizophrenia (40 to 65 percent chance of developing the disorder), suggesting a strong genetic link. 2
Symptoms of schizophrenia may include hallucinations, delusions and thought and movement disorders. 3 The disease affects men and women equally and occurs at similar rates in all ethnic groups around the world. Symptoms such as hallucinations and delusions usually start between ages 16 and 30, with men experiencing symptoms a little earlier than women. Most of the time, people do not acquire schizophrenia after age 45 4 and the disease rarely occurs in children, but awareness of childhood-onset schizophrenia is increasing.5,6
Although the etiology of schizophrenia remains elusive, there have been several genes found to be associated with an increased risk of schizophrenia, but no gene by itself is causative. 7
Dysfunction of the dorsolateral prefrontal cortex may be a central feature of the pathophysiology of schizophrenia, and this dysfunction may be related to alterations in gamma aminobutyric acid (GABA) neurotransmission. More specifically, axon terminals of GABA related chandelier neurons, as revealed by immunoreactivity for the GABA membrane transporter, are reduced substantially in the middle layers of the PFC in schizophrenic subjects. 8
Disrupted GABAergic circuitry has been reported in patients with schizophrenia and frontal lobe epilepsy, and may contribute to their associated impairments in behavioral flexibility. Recent research has shown that hepatocyte growth factor (HGF) modulates GABAergic inhibition and seizure susceptibility. 9 One type of behavioral flexibility, reversal learning, is dependent upon proper numbers of GABAergic interneurons and both the anatomical and functional deficiencies associated with behavioral impairment can be restored with exogenous postnatal hepatocyte growth factor (HGF) supplementation.10,11
Mice with a targeted mutation of the gene encoding urokinase plasminogen activator receptor (uPAR), a key component in HGF/SF activation and function, have decreased levels of HGF/SF and a 50% reduction in neocortical GABAergic interneurons at embryonic and perinatal ages, 12 which further supports a link between HGF and GABA
Hepatocyte growth factor (HGF), an 82 kDa, 674 amino acid residue heterodimeric glycoprotein, was originally isolated from rat platelets.13,14 This growth factor has also been called scatter factor, hepatopoietin A, and mammary growth factor. 15 It is one of a small family of factors lacking significant homology with other known growth factors, but including an HGF-like factor known as macrophage stimulating protein (MSP).16–19 HGF has mitogenic, morphogenic, and motogenic effects on hepatocytes, as well as endothelial, mesenchymal and hematopoietic cell types,18,20,21 and demonstrates noticeable species cross-reactivity. 22
HGF regulates cell growth, cell motility, and morphogenesis by activating a tyrosine kinase signaling cascade after binding to the proto-oncogenic c-Met receptor (translated by the MET gene). HGF is secreted by mesenchymal cells and, although it was first considered to exert biological effects only on specific target cells, it has since been demonstrated to mediate inflammatory responses to tissue injury, and regulate cell growth, cell motility, and morphogenesis in a wide variety of cells. Its ability to stimulate branching morphogenesis, cell migration, survival and proliferation gives it a central role in angiogenesis, tissue regeneration, as well as tumorogenesis.23–29
Signaling by HGF has also been found to have anti-inflammatory, antifibrotic, and pro-regenerative activity on various types of tissue. But it seems to be particularly active in the nervous system, where it has been found to have neurotrophic and angiogenetic activity on CNS neurons, promote both the survival of neurons and the regeneration of injured nerves, and function as a target-derived axonal chemoattractant, guiding axons to their target. As a result, it plays significant roles in the development of the CNS. 30
Because of the potential association between HGF and neurological development and differentiation, and its association with the etiology of neurological diseases, we tested patients with Schizophrenia for serum concentration of HGF and then compared those levels before and after zinc and B-6 supplementation therapy.
Materials and Methods
ELISA to measure serum HGF (ELISA Kit, R&D Systems, Minneapolis, Minn.)
All reagents and specimens were equilibrated to room temperature before the assay was performed. A 1:51 dilution of the patient samples was prepared by mixing 10 µl of the patient's sera with 0.5 ml of Serum Diluent. One hundred microliters of calibrators (20–200 Eu/ml antibodies), positive and Negative control serums, serum diluent alone, and diluted patient samples were added to the appropriate microwells of a microculture plate (each well contained affinity purified polyclonal IgG to HGF). Wells were incubated for 60 minutes (±5 min) at room temperature, then washed 4x with wash buffer. One hundred microliters of pre-diluter anti-human IgG conjugated with HRP was added to all microwells, incubated for 30 minutes (±5 min) at room temperature, then washed 4x with wash buffer. One hundred microliters of enzyme substrate was added to each microwell. After approximately 30 minutes at room temperature, the reaction was stopped by adding 50 µl of 1 M sulfuric acid, then the wells were read at 405 nm with an ELISA reader (BioRad Laboratories, Inc., Hercules, CA, USA).
Copper and zinc serum concentration
Assay to establish copper and zinc plasma concentration was performed by LabCorp, Inc. (Naperville, IL 60563) using inductively-coupled plasma-mass spectrometry.
Subjects
Experimental and controls
Serum from individuals with diagnosed schizophrenia (n = 18; 8 male; mean age 32.5 ± 10 years) and controls (n = 19; 17 male mean age 43.2 ± 12 years) was obtained from patients at the Health Research Institute/Pfeiffer Treatment Center.a Patient consent was obtained from all patients involved in this study.
The Pfeiffer Treatment Center is a comprehensive treatment and research center, specializing in the care of with neurological disorders, including shcizophrenia.
Controls were normal healthy volunteers, all employees of the Pfeiffer Treatment Center.
Severity of disease
A modified PANSS questionnaire 31 for schizophrenia assessment was used to determine the overall severity of schizophrenia. Patients or guardians were asked to rate behaviors such as irritability and anger, depression, hallucinations, delusions, suicidal tendencies, anxiety and paranoia. The patients were rated on a scale of 0–5 (5 being the highest) for each of these behaviors. We evaluated the overall severity of schizophrenia by establishing the mean of all of the scores for each patient, and we also compared the overall anxiety and overall depression rating of each patient. The overall schizophrenia assessment includes assessment of all the behaviors (above). The anxiety and depression rating includes assessment of just anxiety and depression behavior alone, respectively.
Serums
All serums, experimental and control serums were treated in an identical fashion–-frozen at -70C immediately after collection and cell/serum separation, then stored at -70C until thawed for use in ELISAs.
Zinc and Anti-oxidant Therapy
Individuals in this study who presented to the Pfeiffer Treatment Center with Schizophrenia were tested for zinc, copper and anti-oxidant levels. Based on deficiencies, they were then prescribed the appropriate dose of anti-oxidants. Pre-therapy patients represent those who were tested when they first presented and were not previously taking any zinc or anti-oxidants. Post-Therapy patients received anti-oxidant therapy (Vitamin C, E, B-6 as well as Magnesium, and Manganese if warranted), and zinc supplementation (as zinc picolinate), for a minimum of 8 weeks.
Statistics
All inferential statistics were derived from unpaired t-test and Mann-Whitney with 95% confidence intervals.
Results
Serum from 18 individuals diagnosed with schizophrenia and 19 age and gender similar controls were tested for HGF concentration using ELISAs, and copper and zinc plasma levels were established using inductively-coupled plasma-mass spectrometry (Labcorp, Naperville IL).
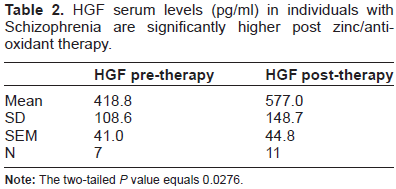
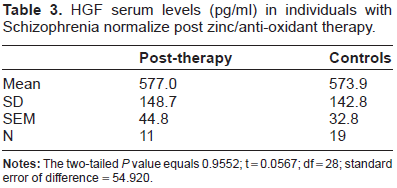
HGF serum levels of individuals with schizophrenia, before zinc and B-6 therapy, were significantly lower than age and gender similar controls (t-test P = 0.016; Mann-Whitney P = 0.015) (Table 1), and significantly lower in schizophrenia patients pre-therapy compared to post therapy (t-test P = 0.028; Mann-Whitney P = 0.046) (Table 2). HGF levels normalized (reached levels similar to controls) post-therapy (Table 3). Zinc levels in these same individuals also normalized (Fig. 1) (Normal; 90–150 mg/dL). Perceived severity of schizophrenia (scale of 0–5 (5 being the most severe)) was lower after zinc and B-6 therapy, particularly anxiety (P = 0.03) (Fig. 2).
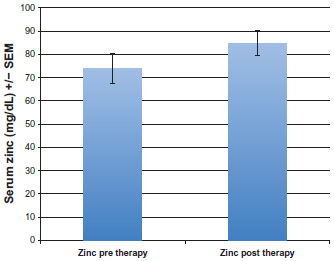
Zinc levels (mg/dL) increase after zinc and B-6 therapy.
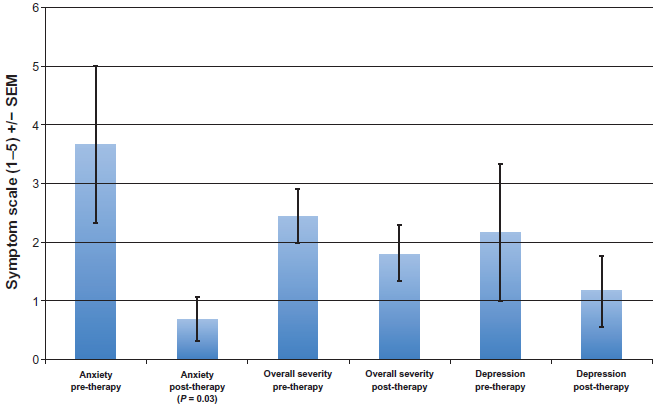
Perceived severity of symptoms (scale of 0–5; 5 being the most severe) was lower after zinc and B-6 therapy, particularly with respect to anxiety symptoms.
Serum HGF (pg/ml) in individuals with Schizophrenia pre-therapy is significantly lower than in healthy controls.
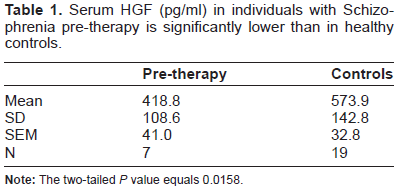
HGF serum levels (pg/ml) in individuals with Schizophrenia are significantly higher post zinc/anti-oxidant therapy.
HGF serum levels (pg/ml) in individuals with Schizophrenia normalize post zinc/anti-oxidant therapy.
Discussion
HGF has been found to be associated with a variety of diseases of the CNS. For instance, immunohistochemistry using anti-HGF antibody has revealed more intense immunolabeling in Alzheimer's disease (AD) than in control brains, and there appears to be a significant correlation between CSF HGF levels and white matter high-signal foci determined on brain magnetic resonance imaging (MRI) in AD patients. 32 In Amyotrophic lateral sclerosis (ALS), overexpression of hepatocyte growth factor (HGF) in the nervous system attenuates motoneuron death and axonal degeneration and prolongs the life span of transgenic mice overexpressing mutated Cu2+/Zn2+ superoxide dismutase. 33 Overexpression of HGF after gene transfer prevented neuronal death in a Parkinson's Disease rat model, 34 and decreased levels of HGF has been found in autistic children with GI disease. 35
The etiology and pathophysiology of schizophrenia remain unknown. However, almost half the altered proteins identified by proteomics in schizophrenia have been associated with mitochondrial function and oxidative stress responses, suggesting a role of oxidative stress. 36
Postmortem analysis shows that fast-spiking interneurons, expressing the calcium-binding protein parvalbumin and modulating cortical networks through their release of GABA, are depleted in the cortex of patients with schizophrenia.37,38
Ketamine and other NMDA-type glutamate receptor blockers induce psychotic and dissociative behaviors indistinguishable from those seen in schizophrenia patients. Ketamine exposure also depletes interneurons in culture of parvalbumin and GAD67 39 and exposure of mice to ketamine induced a persistent increase in brain superoxide due to activation in neurons of reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase. 40 Decreasing superoxide production prevented the effects of ketamine on inhibitory interneurons in the prefrontal cortex. This suggests that blocking NMDA signaling in GABAergic neurons causes a loss in GABA transmission, which in turn frees glutamatergic neurons from inhibitory innervation leading to heightened glutamate release and that oxidative stress may be an underlying mechanism.
Recently, Burdick et al demonstrated an association between a genetic variation of the MET gene and schizophrenia and general cognitive ability and suggested that MET variation influences schizophrenia risk and neurocognition, supporting a neurodevelopmental role across CNS-relevant phenotypes. 41 HGF, the only ligand for the cMET membrane protein, translated by the MET gene, has been shown to modulate GABAergic activity 42 and enhance NMDA currents in the hippocampus. 43
Our results demonstrate that HGF is significantly lower in schizophrenic patients compared to controls and we suggest a potential role for HGF deficiency in the etiology of schizophrenia by modulating NMDA and GABA deficiency.
Zinc is well known as one of the most important trace elements in the body. Dietary zinc deficiency is associated with a variety of physiological defects including anorexia, skin lesion, and growth retardation. 44 Mechanistic studies demonstrated that zinc deficiency affects a large number of hepatic genes involved in multiple cellular functions. In particular, zinc deficiency has been shown to down-regulate hepatic gene expression of metallothionein (MT), insulin-like growth factor I (IGF-I), insulinlike growth factor binding protein 1 (IGFBP1), cyclin D1, and HGF, which are involved in cell proliferation.45–47
Zinc supplementation has also been found to prevent liver cell injury through attenuation of oxidative stress, 48 and there is evidence suggesting that alcohol-induced liver damage initiates hepatocyte proliferation, and zinc supplementation accelerates liver regeneration, through up-regulating cell proliferation-related proteins such as HGF. 49
Vitamin B-6 is a co-factor for the conversion of glutamate to GABA.50,51 It has also been suggested that it may be efficient as the treatment for tardive dyskinesia and parkinsonism and successful as an add-on treatment in schizophrenia.52,53
The present data raise the intriguing possibility that HGF levels, low in schizophrenic patients before therapy, are raised by zinc and B-6 therapy and that these higher levels may be associated with improved symptoms, particularly anxiety. This suggests that HGF, through the modulation of GABA, may be playing a role in the etiology of the disease.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
We would like to thank Scott Filer, Executive Director and the entire staff of The Health Research Institute/ Pfeiffer Treatment Center, for their support of this research.
We would also like to thank Laurie Myers and Kyle Andrews for their technical assistance.
