Abstract
A Met residue is located adjacent to phosphorylation site 1 in the sequences of mitochondrial pyruvate dehydrogenase E1α subunits. When synthetic peptides including site 1 were treated with H2O2, the Met residue was oxidized to methionine sulfoxide (MetSO), and the peptides were no longer phosphorylated by E1α-kinase. Isolated mitochondria were incubated under state III or IV conditions, lysed, the pyruvate dehydrogenase complex (PDC) immunoprecipitated, and tryptic peptides analyzed by MALDI-TOF mass spectrometry. In all instances both Met and MetSO site 1 tryptic-peptides were detected. Similar results were obtained when suspension-cultured cells were incubated with chemical agents known to stimulate production of reactive oxygen species within the mitochondria. Treatment with these agents had no effect upon the amount of total PDC, but decreased the proportion of P-PDC. We propose that the redox-state of the Met residue adjacent to phosphorylation site 1 of pyruvate dehydrogenase contributes to overall regulation of PDC activity
Keywords
An inevitable consequence of aerobic metabolism is exposure of cells to reactive oxygen species (ROS), including H2O2 (hydrogen peroxide), O2- (superoxide), and OH (hydroxyl radical). Low concentrations of ROS can function in signal transduction leading to activation of defense responses, 1 while high concentrations cause oxidative damage of lipids, DNA, and proteins. 2 The side-chains of some amino acids (Pro, His, Arg, Lys, and Thr) are labile to oxidative stress, producing ketone or aldehyde derivatives.3,4 Herein we focus on how stress might result in the oxidation of Met to methionine sulfoxide (MetSO) and affect regulation of the pyruvate dehydrogenase complex (PDC) by reversible phosphorylation.
An emerging concept is that some effects of ROS are mediated by changes in protein phosphorylation. In some instances the changes have been attributed to activation of the MAPK (mitogen-activated protein kinase) pathways. 5 The mechanism of activation of MAPK by ROS has not been established. It is often assumed that the downstream clients of MAPK during oxidative stress are transcription factors, although none have yet been identified. Because protein Met oxidation is reversible, and converts a hydrophobic amino acid side-chain to a hydrophilic one, 6 it is of potential regulatory significance, and the impact on enzyme activity and protein:protein interactions has been noted. 7 For example, client recognition by some isoforms of calmodulin is altered by Met oxidation.8,9
The pyruvate dehydrogenase complex (PDC), a member of the α-ketoacid dehydrogenase complex family, 10 catalyzes the entry of carbon into the Krebs cycle. Occupying a key position in intermediary metabolism, activity of the mitochondrial PDC is regulated at several levels.11,12 The most spectacular regulatory mechanisms is reversible multi-site phosphorylation of the E1α subunit by an intrinsic kinase (PDK),13,14 and phosphatase. 10
The position of the E1α site 1 Ser residue phosphorylated by PDK, and sequences flanking this site, are highly conserved in mitochondrial PDC from diverse sources (Fig. 1). There is a conserved Met residue immediately adjacent to the site 1 Ser of PDC E1α. The experiments described herein were undertaken to explore the possibility that oxidation of this Met residue could specifically affect phosphorylation/regulation.

The amino acid sequences flanking phosphorylation site 1 of the pyruvate dehydrogenase complex E1α subunit.
Materials and Methods
Reagents
Synthetic peptide 1 was from Bethyl Laboratories (Montgomery, Texas); synthetic peptide 2 was from Chiron Mimotopes (Clayton, Australia). For Met oxidation, 40 μg of peptide were dissolved in 24 μL of 4 M urea followed by addition of H2O2 to 0.5% (v/v), and incubation at room temp for 30 min. Samples were passed through a C18 ZipTip (Millipore, Billerica, MA) to remove the urea, dried using a SpeedVac, then dissolved in PDK assay buffer. Alternatively the dried oxidized peptides were dissolved in 60% acetonitrile containing 10 mM ammonium phosphate, 0.2% trifluroacetic acid (Pierce Biotechnology, Rockford, IL), and 7 mg/mL α-cyano-4-hydroxycinnamic acid for analysis by MALDI-TOF MS. 15
Methoxychlor (1,1,1-trichloro-2,2-bis(4-methoxy phenyl)ethane was purchased from Chem Service (West Chester, PA). Unless otherwise noted, all other reagents were purchased from the Sigma Chemical Company (St. Louis, MO).
Plant materials
Mitochondria isolated from 10 day-old pea
We have previously developed an ELISA-based method to quantify both total PDC E1α and P-E1α. The method employs specific anti-E1α- 18 and anti-phosphorylation site 1 antibodies. 19 For immunoprecipitation of the PDC prior to MS analysis, the anti-E1α antibodies were cross-linked to POROS 20 protein G-beads (Applied Biosystems, Foster City, CA) essentially as previously described for cross-linking concanavalin A. 15 The POROS protein G-beads were incubated with 10 mM dimethyl pimelimidate (DMP) and 10 mM dimethyl suberimidate (DMS) in 20 mL phosphate-buffered saline (PBS), pH 8.5. Reaction was stopped by adding 10 mL of 100 mM Tris-HCl, pH 8.8. The matrix was then passivated overnight at 4 °C with 10 mL of 2 mg/mL BSA in 100 mM Tris-HCl, pH 8.8. After washing with three bed volumes of PBS, 500 μL of the beads were mixed with 2 mL of ascites fluid for 2 h at 4 °C. After binding, the matrix was washed with 5 bed volumes of PBS.
Purified pea seedling mitochondria 16 were diluted with PBS to 5 mg protein/mL, then disrupted using a Kinematica Polytron homogenizer (Brinkman Instruments). The broken mitochondria were centrifuged at 50,000 rpm for 1 h at 4 °C in a Beckman Coulter Optima MAX-E ultracentrifuge using a TL-100.2 rotor. Supernatants were incubated with 100 μL bed volume of immobilized-E1α antibodies for 2 h at room temp. The beads were then collected by centrifugation at 2000 rpm for 1 min at RT, then washed 3-times with 10 bed volumes of PBS. Bound proteins were eluted by two 150 μL applications of 200 mM Gly, pH 2.5. The pH of the combined elutes was adjusted to neutrality with 30 μL of 1 M Tris-HCl, pH 9.0. The proteins were either separated by SDS-PAGE and subjected to in gel tryptic-digestion, 15 or digested in solution prior to MS analysis.
Pyruvate dehydrogenase kinase
Recombinant
For qualitative assays, the reaction mixtures were analyzed by MALDI-TOF MS essentially as previously described. 15 The MS analyses were conducted using an ABI Voyager-DE Pro instrument (Applied Biosystems Inc, 850 Lincoln Centre Drive, Foster City, CA 94404 USA). For greater mass accuracy, spectra were collected in the reflector mode.
Results
Synthetic peptide 1, which includes the highly-conserved phosphorylation site 1 of the E1α subunit of PDC, was a good
The effect of Met oxidation upon the phosphorylation of synthetic peptide 1, which includes site 1, by recombinant

Data are representative of multiple independent analyses, each done in triplicate.
Synthetic peptide 1 was analyzed by MALDI-TOF MS before and after incubation with AtPDK. The native peptide yielded a diagnostic ion at 2098 m/z (Fig. 2) After incubation with ATP plus PDK, much of the MS signal is converted to a +80 species indicating that the site 1 Ser residue has been phosphorylated. Incubation with H2O2 resulted in two new ions, +16 and +32, indicating the presence of species with one or both Met residues oxidized to MetSO. When the H2O2-oxidized peptide was tested as a substrate for PDK, no +96 (+80 +16) or +112 (+80 +32) ions were observed (Fig. 2). Clearly our incubation conditions left some of the Met unoxidized, and this species was phosphorylated by AtPDK, but there is no indication that the fully-oxidized peptide is subsequently phosphorylated. Synthetic peptide 1 includes two Met residues, and it is possible that the distal MetSO prevents phosphorylation of the site 1 Ser rather than the proximal MetSO. This was tested using a peptide that contains only a single Met residue, proximal to site 1. Oxidized peptide 2 was not phosphorylated by AtPDK (data not presented), supporting our assumption that the Ser+1 MetSO is responsible.
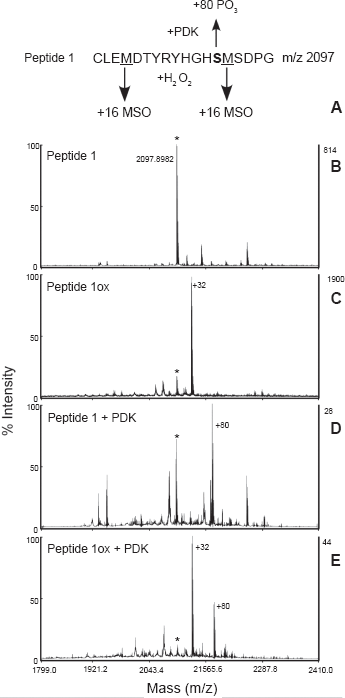
Analysis of phosphorylation/oxidation of peptide 1 by MALDI-TOF MS. In panels C and E the peptide was oxidized with H2O2. In panels D and E the peptide was incubated with recombinant AtPDK plus ATP.
In order to test for Met oxidation in a context more biological than in vitro incubation of a synthetic peptide with a recombinant kinase, we incubated isolated intact mitochondria under conditions that favor the production of H2O2 in the matrix. The mitochondria were then lysed and the PDC immunoprecipitated. The subunits were separated by SDS-PAGE, and the E1α subunit was excised then subjected to in gel-tryptic digestion followed by MALDI-TOF MS analysis. When the mitochondria were incubated either under state IV conditions or State III (5 mM succinate plus 25 μM ADP) plus 10 μM methoxychlor, tryptic-peptides were detected corresponding to both the Met and MetSO species (Fig. 3). When the mitochondria were incubated under State III conditions without methoxychlor, analysis by MS revealed neither Ser-phosphorylation nor Met-oxidation (data not shown). This result was anticipated, because the concentration of ATP present would not be high enough to activate PDK, 17 and ROS production would not be expected under these conditions.
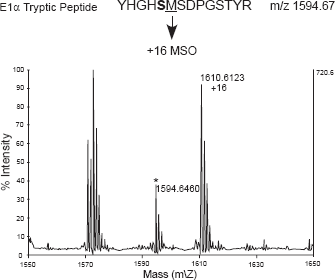
Isolated intact pea (
Additionally, tobacco BY-2 suspension cells were incubated with selected compounds known to induce ROS production (Table 2). The treated cells were then analyzed to quantify the phosphorylation state of mtPDC. Reagents known to promote ROS production in the mitochondrial matrix, menadone, methoxychlor, and t-BHP, all resulted in a decrease in the proportion of P-PDC (Table 2). In contrast, direct treatment with H2O2 had no effect. We assume that this is a distinction between H2O2 generation in the immediate proximity of the PDC and extracellular application of H2O2 which is likely degraded long before reaching the mitochondrial matrix. When the H2O2 was added to the cultures there was a foaming on the tissue surface, presumably H2O2 being decomposed by the cell wall-associated peroxidases.
The effect of ROS-generating reagents on the phosphorylation-state of the mitochondrial PDC from tobacco
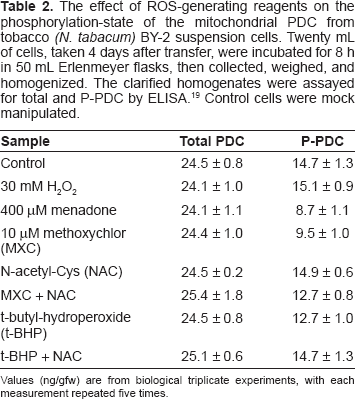
Values (ng/gfw) are from biological triplicate experiments, with each measurement repeated five times.
Discussion
A generalization emerging from studies of both prokaryotes and eukaryotes is that protein oxidative damage is selectively targeted rather than randomly occurring. In plant cells, chloroplasts, 21 peroxisomes, 22 and mitochondria 2 are recognized as the major sites of ROS production. Many highly oxidized proteins have been found in these subcellular compartments, as well as in the nucleus/cytoplasm. 23 Despite recent advances, the specific residues oxidized and the nature of the modifications remain largely unaddressed. The oxidation of Pro, His, Arg, and Lys side-chains to ketone or aldehyde derivatives is a an irreversible consequence of exposure to ROS.3,4
In contrast, the oxidation of Met to MetSO is reversible. Typically, but not exclusively,
24
MetSO is reduced by MetSO reductase,25–28 with thioredoxins as the likely physiological electron donors. The reversibility of Met oxidation allows the possibility that this modification is of regulatory importance. It has been reported that
The change in physical properties, Met is hydrophobic while MetSO is hydrophilic, 6 could affect kinase client protein recognition or binding, or phosphate transfer. The proximity of Met/MetSO to the phosphorylation site appears important in terms of modifying the extent of phosphorylation. In mtPDC E1α, the Met residue is immediately proximal to the phosphorylation site. However in acetyl-CoA synthetase, a Met is five residues upstream of the phosphorylation site and oxidation to MetSO is without effect (SC Huber, unpublished).
Initial studies were conducted in vitro in a non-biological context; synthetic peptides and the recombinant kinase. Our analyses were subsequently extended to include isolated functional pea seedling mitochondria, and suspension-cultured tobacco BY-2 cells. The mitochondria were incubated under conditions where ROS (and specifically H2O2) would be generated within the matrix; state IV respiration or treatment with the organochlorine pesticide methoxychlor that causes apoptotic cell death via generation of H2O2 in the mitochondrial matrix.32,33 In addition to methoxychlor, the BY-2 cells were incubated with menadone and t-butyl-hydroperoxide which are also known to induce mitochondrial ROS. 34 In all cases, the results are consistent with the proposal the Met oxidation inhibits phosphorylation of mtPDC. Our results with t-butyl-hydroperoxide are consistent with those previously reported from studies of perfused rat liver. 34 In those studies, the dephosphorylated form of PDC increased from 46% to 72% during t-butyl hydroperoxide treatment. The inhibition of mtPDC phosphorylation by methoxychlor or t-butyl-hydroperoxide was prevented/reversed by concurrent incubation with N-acetyl-Cys (NAC). These results are consistent with the reports that this compound protects prevents oxidative damage.35,36
When posttranslational modifications such as Met oxidation are considered in a biological context, there is often concern about artifacts that arise during sample isolation, preparation, and analysis. It has recently been reported that Met can be oxidized to MetSO during electrophoretic separations. 37 To minimize this possibility, we included 10 mM sodium bisulfite in the PAGE running buffers. 38 In control assays we observed no evidence for artifactual Met-oxidation in our analyses.
Is there any physiological rationale for Met oxidation/regulation of mitochondrial PDC? In our very simplified model of regulation, mitochondrial PDC is phosphorylated/off in the light.10,12 In illuminated plant cells ATP is supplied by photosynthesis/photophosphorylation, 39 and high cellular ATP concentrations lead to high PDK activity/low PDC activity. 40 Furthermore, NH4+, produced during photorespiration, is the primary positive regulator of PDK activity. 41 It is feasible to envision instances, however, when there might be specific spatial or temporal demand for an intermediate or product of the Krebs Cycle in the light. Met-oxidized PDC could accommodate this need by inhibiting phosphorylation of PDC E1α by PDK. Experiments to simultaneously measure Ser-phosphorylation and Met-oxidation states by ELISA are in progress.
It seems unlikely that the mitochondrial PDC is the only enzyme where regulation involves a complex interaction among reversible phosphorylation and reversible Met-oxidation. It is becoming increasingly evident that the occurrence of both MetSO and the enzyme systems necessary to reduce MetSO are ubiquitous.42–44 We propose that the sites of regulatory phosphorylation will be statistically enriched in nearby Met residues that will be reversibly oxidized.
Disclosure
The authors report no conflicts of interest.
