Abstract
Macular edema is the leading cause of visual impairment in patients with retinal vein occlusion. Limited improvements may be obtained with laser photocoagulation or intravitreal triamcinolone. However, according to the data provided by randomized clinical trials, intravitreal injections of ranibizumab (Lucentis; Genentech, South San Francisco, CA) constitute a new effective and safe option for the management of these vision-threatening diseases. The aim of the present review is to summarize the clinical evidence of ranibizumab for macular edema due to retinal vein occlusions.
Introduction and Background
Retinal vein occlusions (RVO) are the second most frequent major retinal vascular disease after diabetic retinopathy. 1 The affected vein can be the central retinal vein (central retinal vein occlusion, CRVO) (Fig. 1), or more commonly one of its retinal branches (branch retinal vein occlusion, BRVO), mainly the superior temporal BRVO (Fig. 2). 2 Venous thrombus formation leads to poor venous drainage, dilation and tortuosity of the large retinal veins, and increased retinal capillary pressure. These changes result ultimately in exudation of blood, fluid, and lipid into the retina, leading to the development of macular edema (ME). 3 ME is the leading cause of visual impairment in patients with RVO. 4
Macular grid laser photocoagulation has been considered the gold standard therapy for ME due to RVO through the last three decades. The photocoagulation of the photoreceptors reduces the oxygen consumption of the outer retina and allows oxygen to diffuse from the choroid to the inner retina, where it relieves hypoxia. 5 7 Clinical trials demonstrated a moderate efficacy of macular photocoagulation in eyes with branch BRVO, 8 but not in cases with central CRVO. 9
The SCORE trial (Standard Care versus Corticosteroid for Retinal Vein Occlusion) was a phase III multicenter clinical trial that compared standard care versus 1 or 4 mg preservative-free intravitreal triamcinolone (IVTA) for ME due to CRVO and BRVO. In CRVO patients, IVTA was superior to observation, and the 1 mg dose had a better safety profile. In BRVO patients IVTA was not superior to grid photocoagulation and was linked to high negative side effects. 10
More recently, a sustained delivery biodegradable dexamethasone implant (DEX implant; Ozurdex; Allergan, Inc., Irvine, CA) has been shown to be an effective therapy for ME due to RVO. However, over 12 months, significant cataract progression occurred in 29.8% of phakic patients receiving a second DEX implant. On the other hand, only 15.4% of patients developed a significant intraocular pressure increase, which otherwise was transient and controlled with either topical medication or observation. 11 This means that, with regard to triamcinolone, the new dexamethasone intravitreal implant improves relatively the safety profile when considering IOP raise, but there is still a safety concern with cataract progression after the second and further retreatments. Perhaps the only clear indication for the intravitreal DEX implant may be pseudophakic vitrectomized patients.
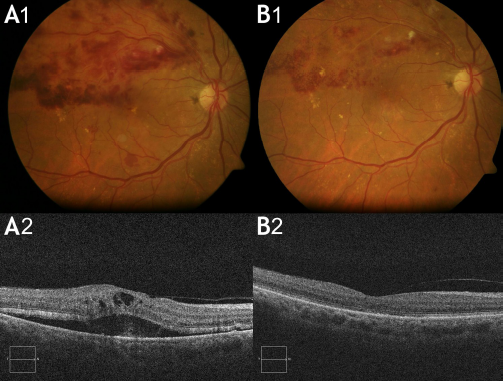
Brach retinal vein occlusion. (

Central retinal vein occlusion. (
The huge improvement of visual outcomes achieved by repeated intravitreal injections of ranibizumab (Lucentis; Genentech, South San Francisco, CA) in macular edema due to RVO has changed the therapeutic perspective of this entity. The purpose of the present manuscript is to review the major efficacy and safety concerns of the treatment of macular edema in RVO with ranibizumab.
Rationale for the Use of Antiangiogenic Agents in the Management of Macular Edema due to Retinal Vein Occlusion
Following RVO, functional and structural changes in the retinal capillaries, in addition to reduced retinal blood flow, lead to the development of hypoxia. These changes trigger upregulation and intraocular release of several inflammatory factors (mainly interleukin 6 and 8, monokine induced by interferon γ, monocyte chemotactic protein 1, inducible protein 10), and especially a key mediator: vascular endothelial growth factor (VEGF). High levels of VEGF-A have been detected in the ocular fluids of patients with RVO. 12 14
VEGF is a disulfide linked homodimer composed of two 23-kDa subunits. VEGF plays a key role in the pathogenesis of ME secondary to retinal vascular diseases as far as it increases the vascular permeability, 15 and promotes inflammation. 16 Thus, inhibition of VEGF is reasonable and clearly justified when managing ME due to RVO.
Until the approval of ranibizumab for ME due to RVO, intravitreal bevacizumab (Avastin; Genentech, San Francisco, CA) has been widely used for this indication in an off-label fashion, and with a lack of randomized clinical trials ensuring its systemic safety profile. Anyway, we must remember that ranibizumab and bevacizumab are different molecules, 17 and a key difference between them is the systemic effect of bevacizumab, which induces a significant reduction in VEGF plasma levels following a single intravitreal injection.18,19
Summary of the main ocular changes in vision and central subfield thickness in the main clinical trials.

There is still controversy over the potential role of ranibizumab in cases with ischemic forms of RVO. While some preclinical studies have hypothesized a harmful effect of this treatment in cases with retinal ischemia, others conclude that there is no increase in the avascular area of the ischemic retina. In our experience, cases with ischemic BRVO and CRVO evidenced an analogous anatomical response in comparison to cases with perfused RVO. Obviously, the visual outcome may be limited to the associated ischemic damage in such cases.
Summary of ocular and nonocular adverse events in the main clinical trials.


Ranibizumab for Macular Edema due to Retinal Vein Occlusion: Summary of Clinical Evidences
The terms “retinal vein occlusion” and “ranibizumab” retrieved 51 publications in Pub Med. However, an electronic search limited to “clinical trials” was performed in order to review those manuscripts offering the first level of evidence for the present review. All available data from the selected articles were extracted and tabulated with respect to each study's identity, its design (prospective or retrospective, randomized or not randomized), protocol treatment, time of follow-up, baseline and final visual acuity, baseline and final central subfield thickness, mean number of intravitreal injection, number of ocular adverse events, number of systemic adverse events.
Twelve clinical trials focusing in ranibizumab for ME due to RVO have been reported.3,20–30 Overall, the clear conclusion of them is that ranibizumab is an effective and safe therapeutic approach. The main results are summarized in Table 1.
In those trials reporting results after 24-month follow-up,3,24–27 the mean visual acuity improved in +15.3 letters (Table 1). Such an impressive visual outcome is achieved both in fixed monthly injections protocols and also in pro re nata retreatment protocols. However, no comparative study between these two regimens has yet been published. Initially, after a single injection of ranibizumab, there is a rapid increase in visual acuity parallel to a significant decrease in the retinal thickness as measured by optical coherence tomography. Further retreatments achieve small visual gains after this initial one, but contribute to stabilize the ME and the visual acuity.
The proportion of local or systemic adverse events related to the treatment with ranibizumab varies from 0,009 to 0,11. These data evidences the safety of ranibizumab even in cases with associated cardiovascular systemic risk factors, which is the case of patients with RVO (Table 2).
Although no definite recommendation may be delivered with the actual data available, the classical initial management of waiting at least three months to treat patients with ME due to RVO seems to be clearly obsolete due to the results from these trials.
Conclusion
Nowadays, ranibizumab is the most efficient and safe therapy in the management of ME secondary to both BRVO and CRVO. The intravitreal injection of 0.5 mg of ranibizumab in these cases leads to a rapid decrease of the macular thickness and to significant improvements in visual acuity.
It is important to underline that this review focus on ranibizumab, and the results provided by this treatment must not be extrapolated to bevacizumab. These are two different molecules 17 which share the inhibition of VEGF-A as their main mechanism of action; but bevacizumab has not proven its safety in randomized clinical trials, whereas ranibizumab has evidenced an excellent safety profile in several trials of patients with RVO.
Further studies are warranted in order to provide the guidelines that may enable the retinal physicians to optimize the visual outcomes in cases of ME due to RVO, and to establish retreatment protocols looking for less number of injections and visits to the hospital. Until then, we can clearly state that the standard of care for these patients is the intravitreal injection of ranibizumab.
Author Contributions
Conceived and designed the experiments: RGP, MDL. Analysed the data: RGP, RDM, CML. Wrote the first draft of the manuscript: RGP. Contributed to the writing of the manuscript: RGP, RDM, CML, MDL. Agree with manuscript results and conclusions: RGP, RDM. Jointly developed the structure and arguments for the paper: RGP, RDM. Made critical revisions and approved final version: RGP, RDM, CML, MDL. All authors reviewed and approved of the final manuscript.
Funding
Roberto Gallego-Pinazo has received lecture fees from Novartis and research support from Allergan, Alcon, Carl Zeiss Meditec, and Novartis.
The rest of co-authors have no financial conflict to disclose.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
