Abstract
Background:
Diabetes mellitus is a risk factor for central retinal vein occlusion (CRVO); however, it is unclear whether certain commonly used medications among diabetics or glycemic control impact visual outcomes in diabetic eyes with CRVO.
Purpose:
To evaluate the effect of select systemic medications and glycemic control on presenting features, treatment burden, and outcomes in patients with diabetes who develop a central retinal vein occlusion (CRVO).
Methods:
Retrospective longitudinal cohort study at a single tertiary academic referral center from 2009–2017 investigating eyes of patients being treated for diabetes mellitus at CRVO onset. Eyes with a prior history of anti-vascular endothelial growth factor (anti-VEGF) therapy or laser treatment within the year prior to CRVO onset were excluded. Main outcomes and measures were visual acuity (VA), central subfield thickness (CST), cystoid macular edema (CME), and number of intravitreal injections and laser treatment throughout follow-up.
Results:
We identified 138 eyes of 138 participants who were diabetic at CRVO onset. Of these, 49% had an ischemic CRVO. Median follow-up time was 25.5 months. Fifty-five eyes (40%) had a HbA1c within 6 months of CRVO presentation. HbA1c was positively correlated with both presenting CST (p = 0.04) and presence of CME (p < 0.01). In all 138 eyes, mean presenting VA was 20/246, and mean final VA was 20/364. Better-presenting VA was significantly associated with aspirin 325 mg use (p = 0.04). Lower CST at presentation was significantly associated with metformin use (p = 0.02). Sitagliptin use at CRVO onset was associated with a lower prevalence of CME at final follow-up (p < 0.01). Lower final CST was significantly associated with glipizide use at CRVO onset (p = 0.01). There were no significant associations between systemic medications or HbA1c and treatment burden or final VA (p > 0.05).
Conclusion:
Although aspirin 325 mg, metformin, sitagliptin, and glipizide were associated with better-presenting VA, lower-presenting CST, lower prevalence of macular edema at final visit, and lower final CST, respectively, none of these systemic agents or glycemic control were associated with decreased treatment burden or improved visual outcomes in diabetics with CRVO.
Introduction
Retinal vein occlusion (RVO) is the second most common vision-threatening retinal vascular disease after diabetic retinopathy.1–3 Central retinal vein occlusion (CRVO) makes up 15% of all RVO cases and has a reported worldwide prevalence of 0.8%. 4 The pathogenesis of CRVO includes various theories that implicate anatomic abnormalities, vascular pathology, and thrombotic events. 5 In these eyes, venous occlusion leads to increased intravenous pressure proximal to the occlusion and weakening of the blood–retinal barrier in the affected venous distribution, leading to intraretinal blood and fluid. 3
Diabetes mellitus is a known risk factor for CRVO development.6,7 In diabetic individuals with poor glycemic control, vascular abnormalities including endothelial cell dysfunction and hyperactive platelets contribute to poorer cardiovascular health, leading to an increased risk of CRVO.8–10 However, the precise interaction between diabetes and CRVO continues to be investigated. 8 It remains unclear whether these patients have poorer clinical outcomes after the development of a CRVO.
Several systemic glucose-lowering medications including metformin, sodium glucose cotransporter-2 inhibitors, and glucagon-like peptide-1 have been associated with a decreased risk of cardiovascular morbidity and mortality.11,12 In addition, metformin, sulfonylureas, thiazolidinediones, and sitagliptin have also been shown to reduce platelet hyperactivity associated with diabetes and CRVO.13,14 In contrast, basal insulin and sulfonylureas appear to be neutral on cardiovascular disease outcomes.15,16 Given the association between cardiovascular risk and CRVO, these studies raise the important question as to whether systemic medications commonly used by diabetics may affect clinical features and outcomes in CRVO.
Understanding how various commonly administered systemic medications, including hypoglycemics and aspirin, and overall glycemic control affect the presentation and clinical course of CRVO may provide meaningful clinical information to guide decision-making. The purpose of this study is to evaluate how commonly used systemic medications as well as glycemic control affect the presentation, treatment burden, and outcomes in diabetic individuals with CRVO.
Methods
This study was approved by the Duke University Institutional Review Board (approval number Pro00075701) and was conducted in accordance with the Declaration of Helsinki. The need for informed patient consent was waived for this retrospective study. A retrospective review of medical records was performed for all patients with diabetes mellitus who were newly diagnosed with a CRVO by a retina specialist at a single tertiary referral center between January 2009 and June 2017. Patients were considered to have diabetes if it was listed as a diagnosis in the electronic medical record at the time of CRVO diagnosis. Patients were not excluded if their presenting hemoglobin A1c (HbA1c) was below 6.5, as long as they were on at least one medication for glycemic control at the time of CRVO diagnosis. Eyes were excluded if there was a history of anti-vascular endothelial growth factor (anti-VEGF) therapy or laser treatment for diabetic macular edema (DME) in the year prior to the diagnosis of CRVO.
The use of the following systemic medications at the time of CRVO onset was reviewed: insulin, metformin, sulfonylureas, thiazolidinediones, dipeptidyl peptidase 4 (DPP4) inhibitors, and aspirin (81 and 325 mg). In addition, demographic information, eye examination findings, time since CRVO diagnosis, medical and ophthalmic comorbidities, treatment course, and follow-up duration were recorded. The presence of non-proliferative diabetic retinopathy (NPDR) or proliferative diabetic retinopathy (PDR) in either eye was recorded as noted by the retina specialist at the time of presentation with CRVO. HbA1c level obtained within 1 year of the presenting visit was recorded. If there were multiple values, the one in closest proximity to the presenting visit date was recorded. Best-corrected visual acuity (VA) using an Early Treatment Diabetic Retinopathy Study (ETDRS) chart was converted to the logarithm of the mean angle of resolution (logMAR) scale for statistical analyses. Central subfield thickness (CST), cystoid macular edema (CME), and subretinal fluid (SRF) were abstracted from spectral domain optical coherence tomography (OCT) at the presenting and final visits.
CRVO eyes were defined as ischemic when the subject had counting fingers vision not attributable to a media opacity and either of the following: (1) physician confirmed afferent pupillary defect or (2) evidence of anterior or posterior segment neovascularization not attributable to another disease. The CRVO was considered to be ischemic if these criteria were met at any point during the first year of follow-up.
Data were analyzed via R statistical software, version 3.5.1. A one-way analysis of variance (ANOVA) was performed to evaluate differences in follow-up duration between medication groups. Wilcoxon rank-sum tests were used to determine whether there were significant differences in follow-up duration between patients on a selected medication and patients not on that selected medication. To identify medications of interest, we fit a series of univariate linear or logistic regression models with a single independent variable (the selected medication) and single dependent variable (the outcome of interest) separately for each time point collected. Outcomes included logMAR VA at presentation and final follow-up, presence of CME at presentation and final follow-up, presence of SRF at presentation and final follow-up, CST measurement at presentation and final follow-up, number of intravitreal anti-VEGF injections at 1 year and final follow-up, CRVO type (ischemic vs nonischemic), and presence of an enlarged foveal avascular zone on fluorescein angiography at presentation. Linear and logistic regressions were used to evaluate any association between presenting HbA1c and the above measured outcomes. Multivariable regressions were used to account for race in the associations found to be significant between selected medication and functional or anatomic features on presentation and outcome. A p value less than 0.05 was defined as statistically significant.
Results
In this study, 138 individuals with diabetes mellitus and unilateral CRVO were identified and met inclusion criteria. The median follow-up was 25.5 months (range: 1–189). As described in Table 1, the mean age at CRVO onset was 66.9 ± 12.1 years, and 50% of the cohort was female. The racial distribution included 57% White, 30% Black, <1% multiracial, and 12% other. Fifty-one percent of the cohort had a nonischemic/perfused CRVO. Of the 138 eyes in the total cohort, 55 eyes (40%) had a HbA1c recorded within 6 months of CRVO onset. For this subcohort, median and mean HbA1c was 6.9% and 7.1%, respectively (Table 2).
Demographics of diabetic individuals presenting with CRVO.
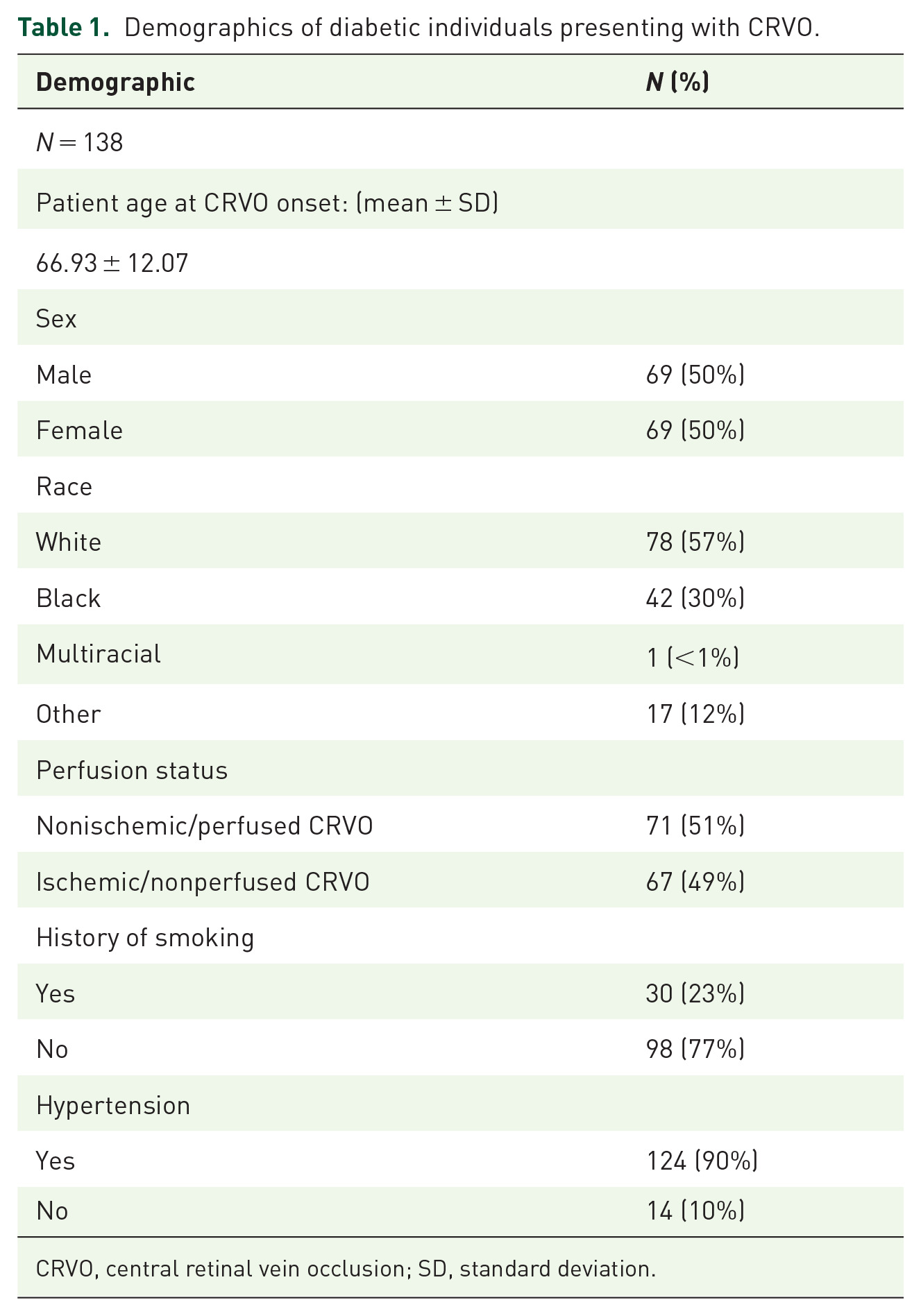
CRVO, central retinal vein occlusion; SD, standard deviation.
Clinical characteristics of eyes with CRVO in individuals with diabetes mellitus.

Of the 138 eyes in the total cohort, 55 had a hemoglobin A1c recorded within 6 months of CRVO onset. Only these are eyes are represented in statistics provided here. CRVO, central retinal vein occlusion; logMAR, logarithm of minimal angle of resolution.
For the entire cohort of 138 eyes, the median number of total glucose-lowering medications at CRVO onset was 1. At presentation with CRVO, 49% were taking metformin, 38% were taking a sulfonylurea, 26% were taking insulin, 9% were taking thiazolidinediones, and 7% were taking a DPP4 inhibitor. At 1 year, 52% of patients were taking metformin, and 11% were taking thiazolidinediones. The first treatment for CME at presentation of CRVO was intravitreal bevacizumab (Avastin, Genentech) in 50%, intravitreal ranibizumab (Lucentis, Genentech) in 12%, intravitreal triamcinolone acetonide (Triesence, Alcon) in 7%, intravitreal aflibercept (Eylea, Regeneron) in 4%, intravitreal tissue plasminogen activator (tPA) in 2%, and posterior sub-Tenon’s triamcinolone acetate (Kenalog, Bristol Myers Squibb) in 1%. Panretinal photocoagulation (PRP) was performed in 3%.
Treatment course
For the 81 eyes that had >1 year of follow-up (mean follow-up 51.9 months), the mean number of injections was 6.89 (range: 0–44) at the final visit. The distribution of intravitreal injections by medication is depicted in Figure 1.

The distribution of intravitreal injection therapies in eyes with central retinal vein occlusion in individuals with diabetes through the final follow-up. Mean (max, min) of intravitreal injections are as follows: triamcinolone [0.59 (0, 16)], ranibizumab [2.47 (0, 24)], bevacizumab [2.83 (0, 25)], aflibercept [1.66, (0, 31)].
Visual acuity and anatomic outcomes
Among all eyes with a median follow-up of 25.5 months, the mean presenting VA in the CRVO-affected eye was 20/246, and the mean final VA was 20/364. Ischemic CRVO eyes had a poorer mean final VA of 20/1157 compared with nonischemic CRVO eyes with a mean final VA of 20/136 (p < 0.01). Mean presenting CST was 541.87 μm (range: 126–1550 μm), and the mean final CST was 354.72 μm (range: 141–1332 μm). Prevalence of CME was 78% at presentation, and this decreased to 51% by final visit. The percentage of eyes with SRF at presentation was 34%, decreasing to 7% by the final visit (Table 3).
Imaging characteristics of eyes with CRVO in individuals with diabetes mellitus.

CME, cystoid macular edema; CRVO, central retinal vein occlusion; CST, central subfield thickness; OCT, optical coherence tomography.
Systemic diabetic medication use and OCT parameters
Univariate analysis revealed that lower-presenting CST was significantly associated with metformin use at CRVO onset (p = 0.02). In the 55 eyes with a recorded HbA1c within 6 months of CRVO onset, lower HbA1c level was also correlated with lower CST at presentation (p = 0.04). Lower CST at final follow-up was significantly associated with the use of sulfonylureas at presentation (p = 0.01) and the total number of diabetic medications taken at CRVO onset (p < 0.01). In analyzing the change in CST at the final visit, a greater decrease in CST was significantly associated with total number of diabetic medications taken and the specific use of metformin. Among the cohort, there was a 131.4 μm decrease in CST from presentation to the final visit for each additional diabetic medication taken (range: 0–3; p < 0.01). The 71 (51%) subjects who were not taking metformin at presentation with CRVO did not show this favorable trend; there was a 129.8 μm increase in CST from presentation to the final visit (p < 0.01).
Sitagliptin, a DDP-4 inhibitor, use at CRVO onset was significantly associated with a lower prevalence of CME at final follow-up (p < 0.01). HbA1c was significantly associated with CME at baseline (p < 0.01) in the cohort of eyes with HbA1c within 6 months of CRVO onset. There were no significant correlations between final choroidal thickness measurements or prevalence of SRF on OCT and the use of systemic medications at CRVO presentation. Similarly, HbA1c within 6 months of presentation was also neither associated with choroidal thickness nor prevalence of SRF on OCT.
Systemic diabetic medication use and visual acuity
Aspirin use (325 mg) at CRVO onset in diabetic individuals was significantly associated with better-presenting VA (p = 0.04), but was not associated with final VA. Use of aspirin (81 mg) at CRVO onset did not correlate with either presenting or final VA. There were no significant correlations between the use of diabetic medications and presenting or final VA in CRVO. Similarly, HbA1c was also not significantly associated with final VA in the 55 eyes with HbA1c recorded within 6 months of CRVO onset.
Additional analyses
Patient age at the time of CRVO presentation was negatively correlated with insulin use at CRVO onset (p = 0.04). Patients taking insulin at CRVO onset had a mean age of 66.3 years at presentation compared with 68.2 years for those not taking insulin at CRVO onset.
We analyzed treatment burden at 1 year and at final follow-up and found no significant correlations between the use of systemic medications investigated in this study and treatment burden. In the subcohort of 55 eyes with a HbA1c recorded within 6 months of CRVO onset, linear regression between HbA1c and number of intravitreal injections also revealed no significant correlation.
Multivariable regressions between race, select medications (aspirin 325 mg, metformin, sitagliptin, and glipizide), and presentation features or outcomes (better-presenting VA, lower-presenting CST, lower prevalence of macular edema at final visit, and lower final CST, respectively) revealed that race was not significant (all p < 0.05).
Discussion
Within the CRVO literature, many reports describe an increased CRVO risk among individuals diagnosed with diabetes and other systemic vascular diseases.7,17–19 One previous Taiwanese study has suggested that metformin may provide a protective effect against development of any RVO in diabetic patients, decreasing the adjusted CRVO hazard ratio from 3.66 to 2.38. 19 However, no further studies have investigated the effect of other commonly prescribed systemic medications in other diabetic RVO cohorts. Here, we investigated whether the use of select systemic medications was associated with a difference in CRVO outcomes. We found that HbA1c was directly correlated with CST and CME at presentation. Better-presenting VA was associated with taking aspirin 325 mg at CRVO onset. The use of sitagliptin at CRVO onset was associated with lower final CST. A greater number of total diabetic medications at CRVO onset while taking metformin was associated with greater reduction in final CST. Sitagliptin use at CRVO onset was significantly associated with decreased CME at final follow-up. Despite these imaging findings, there were no significant correlations between glycemic control measured by HbA1c within 6 months of CRVO presentation or the use of these agents and treatment burden or final VA.
Within our entire cohort, the average VA worsened from presentation to final visit with a final median VA of 20/200 despite decreased mean CST over the same period. Previous literature has found that CST measurements may not adequately predict VA.20,21 In CRVO, this is likely due to a combination of structural damage from CME and macular ischemia. This is consistent with our study as eyes with ischemic CRVO had much worse VA at final visit compared with those with nonischemic CRVO despite treatment. Hayreh et al. investigated the visual outcomes in CRVO based on perfusion status. The CRVO perfusion status affected VA outcomes, such that nonischemic CRVO had better visual outcomes, whereas ischemic CRVO had worse visual outcomes, even with resolution of CME. 22 In this study, 49% of our cohort had an ischemic/nonperfused CRVO compared with 16% of the CRVO cohort in the study by Hayreh et al. The proportion of ischemic eyes was higher in our study, likely because diabetics are more likely to develop an ischemic CRVO than a nonischemic CRVO, 23 driving the overall mean VA of the cohort to worsen from presentation to final visit.
Upon investigating the effects of glycemic control in CRVO, we found that HbA1c within 6 months of CRVO presentation was positively correlated with CST and prevalence of CME on presentation but was not associated with visual or anatomic outcomes. It is possible that CRVO patients in this study had a component of DME though we tried to account for this by excluding patients with a recent history of anti-VEGF therapy. Higher HbA1c is a well-known risk factor for DME; intensive treatment with insulin to lower HbA1c had been shown to decrease the prevalence of macular edema in diabetics. 24 While the increased prevalence of macular edema may also be driving the increase in CST, previous studies have found a correlation between increased CST and HbA1c even in eyes where DME is not clinically evident. 25 While these imaging findings were found to be significant at presentation, HbA1c within 6 months of CRVO presentation was not significantly correlated with CST or CME prevalence or VA at the final visit.
In our analysis of the effects of individual systemic medications, individuals who were on insulin presented with CRVO at a younger age. It may be that insulin use serves as an indicator for poorer diabetic control, resulting in greater severity of retinal vascular disease and its sequelae. It is also possible that a significant proportion of patients on insulin were type 1 diabetics who, on average, develop diabetes at a younger age than type 2 diabetics. Still, this information is useful for type 1 diabetics who, on average, present with ocular complications at a younger age, have a greater duration of diabetic disease, and have a higher risk for developing neovascularization. 23 Further analysis of systemic medication effects in these subsets separately would aid in risk assessment for CRVO.
Aspirin is commonly prescribed in diabetics who have an increased cardiovascular risk. Low-dose aspirin (81 mg) did not show a significant correlation with either presenting or final VA. Higher-dose aspirin use (325 mg) in diabetics, however, was significantly associated with better-presenting VA. This positive finding in high-dose aspirin use could result from prevention of platelet aggregation and stabilization of vascular epithelium. 26 These effects could result in reduced likelihood of an ischemic perfusion status at CRVO onset, which is both more likely in the diabetic population and more likely to present with poorer VA on presentation. 22 Our group previously investigated the role of aspirin in CRVO more broadly, not limited to the diabetic population, and found that low-dose aspirin was associated with foveal hemorrhage at CRVO presentation but was not associated with any changes in outcomes similar to the findings in this study. 27
We did not find any significant associations between systemic medications and treatment burden or final visual outcomes. However, our study did find a significant association between the change in CST and the total number of diabetic medications that individuals were taking at CRVO onset. This could highlight a potential synergistic or potentiation effect among metformin and other glucose-lowering agents (as metformin is often administered as a first-line agent). Conversely, subjects who were not taking metformin at CRVO onset had an increase in CST from baseline to final visit. This yields discussion of a potential protective effect of metformin against structural changes due to CRVO, although the mechanism remains unclear. One suggested mechanism is the role of metformin in reducing cardiovascular risk. Studies have not only reported that RVO is associated with atherosclerotic and thrombophilic cardiovascular risk factors, but have also observed metformin to lower mortality in diabetic patients with such risk factors.28–32 Additional studies suggest a mechanism whereby metformin improves oxidative stress, preserves antioxidant function, and retrains platelet activation and aggregation – all factors which have been shown to contribute to advanced atherosclerosis in type 2 diabetes. 33 Collectively, these mechanisms could explain why these patients may have a better initial presentation and structural recovery than individuals who did not take metformin at CRVO onset.
Our study has limitations inherent to its retrospective nature. Treatment protocols varied among subjects and there is a large variation in drug effect on individuals. Time to CRVO presentation and the duration of follow-up also varied. As all patients had diabetes, it is possible that a component of DME contributed to CST; however, we did exclude individuals who had any anti-VEGF therapy or laser in the year prior to the diagnosis of CRVO. Given that diabetic retinopathy has been associated with increased risk of CRVO, 34 the presence of NPDR and PDR was noted during the presenting exam. However, progression of diabetic retinopathy was not further abstracted throughout the follow-up period and thus limits our ability to analyze the effect of diabetic retinopathy on CRVO outcomes.
Overall, we found that in diabetic individuals with CRVO, specific systemic medications such as metformin, sitagliptin, glipizide, and aspirin 325 mg were associated with lower-presenting CST, lower final prevalence of macular edema, lower final CST, and better-presenting VA, respectively; however, hypoglycemic agents and aspirin were not associated with decreased treatment burden or superior visual outcomes. Although poorer glycemic control was significantly associated with increased prevalence of CME and higher CST on presentation, HbA1c was not correlated with visual outcomes or treatment burden. Together, these findings suggest that some commonly prescribed medications in diabetics may confer some benefit in presenting features or anatomic outcomes after CRVO, even if there was no corresponding benefit to visual outcomes. Further research is warranted to determine whether some medications are preferred over others in diabetics at higher risk of CRVO or at CRVO diagnosis.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
