Abstract
Dietary acidity is a likely contributor to the development of osteoporosis. Dietary acidosis in an ovine model has effects on trabecular bone that have been previously shown to mimic human osteoporosis. Effects on cortical bone using this model have not been investigated. The objective of this study was to examine the effects of dietary acidosis on cortical bone mineral density and material properties.
Skeletally mature ovariectomized (OVX) sheep consumed either a normal diet (ND) or a metabolic acidosis diet (MA) for 6 or 12 months. Whole femoral and cortical bone beam BMD was determined using dual energy x-ray absorptiometry (DEXA). Beams were then subjected to three point flexure monotonically to failure to determine strength and modulus and then ashed to determine percent mineralization.
Femoral BMD in adult OVX ND 6 mo sheep was significantly greater than those in the non-OVX ND group. The BMD in the MA groups was lower than the control non-OVX ND group. Cortical beams had significantly decreased modulus in all MA and OVX groups when compared with the non-OVX ND group and a tendency towards decreased strength in all groups with significance only in the OVX ND 6 mo sheep. Percent mineralization increased in MA and OVX groups when compared to the non-OVX ND group and was significantly increased in the OVX ND 6 mo and OVX MA 12 mo groups. A significant correlation was seen between BMD of the beam and breaking strength and modulus. Dietary acidity impacts cortical bone and results in reduced material properties that may contribute to failure.
Introduction
Osteoporosis is a multifactorial disease with high morbidity. Dietary acidity is likely an important confounder in the development of osteoporosis.1–4 Humans typically consume an acidic diet that is high in sulfur containing proteins and poor in fruits and vegetables containing calcium and potassium. It is unknown whether people consuming an acidic diet develop identical changes in cortical bone to those seen with senile and postmenopausal osteoporosis.
Metabolic acidosis induces increased urinary calcium excretion, yet whether increased urinary excretion leads to increased bone calcium loss in humans remains controversial.5–7 Compensated metabolic acidosis occurs when the balance of ions in the diet favors anions over cations, more specifically, where chloride, sulfur and phosphorus are present in larger amounts than sodium, potassium, calcium, and magnesium. This occurs when the diet is high in sulfur containing proteins, and poor in potassium rich fruits and vegetables. It is hypothesized that to maintain electroneutrality, calcium is mobilized from bone.8,9 Therefore, over time excess calcium ions are excreted in the urine resulting in osteopenia and potentially osteoporosis. Previous studies in our laboratory using an ovine model support this proposed mechanism by documenting decreased bone mineral density in areas of the skeleton dense in trabecular bone as measured by DEXA examination in sheep consuming an acidifying diet. 10 Dietary acidosis also causes increased osteocalcin levels, a biomarker that increases during periods of increased bone turnover.11,12 We have also documented increased fragility of cancellous bone, trabecular thinning and loss of connectivity within the cancellous envelope with dietary metabolic acidosis induced bone resorption. 13 Analysis of bones from our laboratory ovine model by Fourier Transform Infrared Spectros-copy (FT-IR) examination revealed increased crystallinity to maturity indices that mirror human osteoporosis. 14
The mechanical properties of bone reflect the material properties of its constituents. Isolation of the cortical bone allows completion of materials testing which is capable of evaluating how this particular subset of bone tissue responds to mechanical loading. Stress-strain data allows investigators to characterize structural properties of a representative specimen of basic bone tissue, without the complication of individual variation in geometry of whole bone tissues.
The purpose of this experiment was to examine the effect of dietary acidity on mineral density, mineral content, percent mineralization, and material properties of cortical bone. We hypothesized that dietary acidosis leads to compensatory changes in ovine cortical bone mineral density similar to that seen in human osteoporosis. A sheep model was chosen to evaluate cortical bone changes in response to dietary acidity because of our experience with this model and its ability to mimic osteoporosis in the trabecular bone envelope.
Experimental Subjects
All animals utilized in this research project were cared for according to the policies and principles established by the Animal Welfare Act and the NIH Guide for Care and Use of Laboratory Animals and were approved by the institutional animal care and use committee.
Skeletally mature Ramboulliet × Columbia female sheep, age 4 years, were studied with a mean weight of 72 kg (SD 6.9 kg). All sheep had a normal physical examination upon inclusion. Four treatment groups were evaluated and varied in ovariectomy status, diet consumed, and duration of dietary pressure. Sheep evaluated in this report participated in 2 separately funded and approved experiments in our laboratory. One group of 12 ewes was treated with ovariectomy alone for 6 months (OVX ND 6 mo), one group of 12 ewes was treated with ovariectomy plus an acidifying diet for 6 months (OVX MA 6 mo), one group of 13 ewes was treated with ovariectomy plus an acidifying diet for 12 months (OVX MA 12 mo), and the final group was composed of 15 control ewes (ND). All sheep were allowed paddock turnout and exercise was not restricted.
Materials and Methods
Diet
Sheep consumed an acidifying diet or normal diet depending on treatment group. The diets were analyzed to determine the dietary cation-anion difference (DCAD) by quantifying strong ion content and DCAD was estimated using the equation: DCAD mEq of dry matter = (Na + K + (0.15)Ca + (0.15)Mg) - (Cl + (0.2)S + (0.3)P). Both diets were designed to provide nutrients in excess of and calories appropriate for this species based on values established by the U.S. Board of Agriculture, National Research Council, with the exception that sodium was below NRC requirements for the acidosis diet formulation. However, all sheep had free access to supplemental salt (sodium chloride) during the study period. The amount of supplemental salt consumed was not quantified.
Metabolic acidosis (MA) was induced primarily by limiting the amount of potassium and adding sulfur and chloride to the diet in the form of grain with added ammonium chloride. Details of the diet have been published previously. 10 The normal diet (ND) consisted of free choice grass hay and the MA diet consisted of a specially formulated pellet that provided adequate amounts of all nutrients, including calcium in addition to a limited amount of grass hay. The ND had a DCAD of approximately +308 mEq/kg dry matter, which is normal for this species. The sheep consumed approximately 3,000 kcal/day and their daily intake DCAD was approximately +1,000 mEq/day. The MA diet group consumed a special grain that had a DCAD of −465 mEq/kg dry matter. For an intake of approximately 3,000 kcal/day, which included both the special DCAD grain and the grass hay fed to each treatment group, sheep in the acidic dietary treatment groups consumed a daily intake DCAD of +160 mEq/day.
All sheep were humanely euthanized using an intravenous overdose of pentobarbital at the end of their predetermined study period. Both femurs from each sheep studied were harvested at necropsy, wrapped with saline soaked towels, and frozen at −20 Celsius until evaluation.
Bone mineral density
Bone mineral density (BMD) evaluation of whole femurs was performed ex vivo using dual energy x-ray absorptiometry (DEXA). a The central cranial cortex of the femoral mid-diaphysis was then sectioned using a bone saw b into beams of approximate dimensions 1.0 mm × 1.0 mm × 50.0 mm. Unique identification of each beam included group number, sheep number, and orientation. Each beam was measured to determine center width, height, and length using a digital caliper capable of measuring accurately to 0.01 mm. c Beams were labeled in pencil following preparation, wrapped in 0.9% saline soaked gauze, and placed in a plastic bag and refrigerated until evaluation within hours of creation. Beams were then evaluated for bone mineral density using DEXA examination recorded in g/cm2. Bone mineral density was corrected for measured height of the individual beam by dividing density by height in cm, which transformed to units g/cm3 and allowed comparison among varying heights of beams. Following DEXA examination of beams, they were rewrapped in saline soaked gauze and frozen until used for mechanical testing. DEXA examination was performed with the femurs and bone beams surrounded by a soft tissue platform that was also included in the machine calibration.
Hologic Delphi QDR dual-energy X-ray Absorptiometer and Software version 11.1. Hologic, Inc. Bedford, MA.
EXAKT Technologies, Oklahoma City, OK.
Absolute Digimatic CD-4”CS, Mitutoyo Corporation, Japan.
Mechanical testing
Mechanical testing was performed by loading beams monotonically to failure in three-point bending. Beam specimens were mounted in a servo-hydraulic testing machine d using a specially designed jig. Actuator ramp rate was 1mm displacement per minute. Sampling acquisition was performed every 0.01 seconds, with 1600 points of data per second and 16,000 buffer size. The actuator was positioned just above and was not in contact with the beam at the start of the test (0 mm) and was set to run for a total of 6 mm in an axial direction. Following breakage, each end of the specimen was measured adjacent to the site of failure using a digital caliper capable of measuring accurately to 0.01 mm. Measurements were then averaged and used to determine cross sectional area at the site of failure (mm2).
Model 805, MTS Corp., Eden Prairie, MN.
Breaking force and displacement were measured at the time of failure. Breaking force was defined as the load reached by the specimen at failure. Displacement was defined as the distance extended by the specimen on the failure load. Displacement was measured electronically starting from the point of actuator contact. The following material properties were calculated by normalization to the cross-sectional area of the beam. Breaking strength was defined as the stress reached by the specimen at failure. Breaking strength was calculated using the equation: σ = 3FL/2 CSA where σ was the bending stress (MPa) equivalent to breaking strength, F was the breaking force (N), L was the distance between the outer contact points of the testing jig (mm), and CSA was the cross-sectional area (beam width × beam height) measured at the site of failure (mm2). Modulus of elasticity in bending was calculated in the elastic region of the stress deflection curve. For the purposes of material testing, femoral cortical bone beams machined from the cranial cortex were assumed to be composed of a uniform material. Modulus was calculated using the equation: E = L3F/4wh3δ where E was the flexural modulus (MPa), L was the distance between the outer contact points of the testing jig (mm), F was the breaking force (N), w was the beam width measured at the site of failure (mm), h was the beam height measured at the site of failure (mm), and δ was the displacement or deflection of the beam at failure (mm).
Percent mineralization
Bone ashing was performed on femoral cortical beams following DEXA examination and mechanical testing. Beams were weighed in air and in normal saline solution to determine volume and apparent wet density. Specimens were then dried for 16 hours at 100 °C under 60 kPa vacuum, and weighed to determine apparent dry density. Finally, they were ashed at 800 °C in a muffle furnace for 24 hours, and weighed to determine apparent ash density. Percent Mineralization was then calculated as 100 (ash density/dry density).
Statistical analysis
Statistical analysis was performed using commercial software. e Type 3 tests and least squares means were used to evaluate influence of treatment group on outcome, significance was set at p < 0.05. In addition to direct comparisons, statistical analysis was performed to evaluate correlations between variables, with level of significance set at p < 0.05. Variables evaluated for correlation were BMD in relation to breaking strength and modulus. Furthermore, BMD of cortical beams evaluated by DEXA examination was compared to % mineralization of cortical beams evaluated by bone ashing.
SAS Institute, Cary, NC.
Results
Bone mineral density
Whole femoral BMD in g/cm2 was evaluated by DEXA (Fig. 1). Whole femoral BMD for OVX sheep consuming a ND for 6 months was significantly higher than the controls (OVX ND 6 mo: 1.0319 ± 0.01781 g/cm2 vs. ND: 0.9785 ± 0.01593 g/cm2, p = 0.0294). The whole femoral BMD for OVX adult sheep consuming the MA for 6 and 12 months did not differ significantly from the controls (OVX MA 6 mo: 0.9625 ± 0.01781 g/cm2 and OVX MA 12 mo: 0.9363 ± 0.01711 g/cm2).
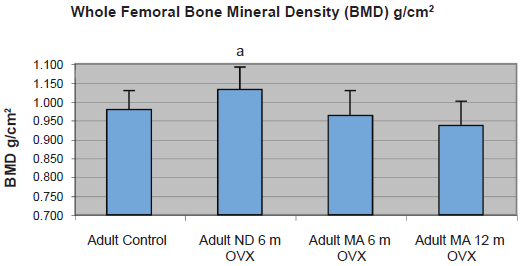
Graph shows whole femoral bone mineral density in g/cm2 as assessed by DEXA examination. Significant difference p < 0.05 as compared to control values denoted by ‘a’.
Femoral cortical beam BMD evaluated by DEXA examination was not discriminating between treatment groups and controls. Beam BMD was 1.1031 ± 0.07796 g/cm3 in the OVX ND 6 mo group, 1.2276 ± 0.07728 g/cm3 in the OVX MA 6 mo group and 1.2969 ± 0.07561 g/cm3 in the OVX MA 12 mo group, and BMD 1.2037 ± 0.07908 g/cm3 in the control group.
Mechanical testing
OVX significantly reduced breaking strength at 6 months compared to controls (OVX ND 6 mo 178.36 ± 11.5966 MPa vs. ND 231.04 ± 11.3123 MPa) (p = 0.0004). However, OVX sheep consuming the MA diet for 6 months and 12 months were equivalent to controls (OVX MA 6 mo 219.62 ± 11.6415 MPa and OVX MA 12 mo 214.18 ± 11.4952 MPa).
All treatments resulted in a significant decrease in modulus. Cortical beams from control sheep had a modulus of 25226 ± 1500.64 MPa which was significantly greater then the OVX ND 6 mo group (16510 ± 1540.62 MPa, (p < 0.0001)) the OVX MA 6 mo group (21178 ± 1546.60 MPa (p = 0.0348)) and the OVX MA 12 mo group (19352 ± 1527.56 MPa (p = 0.0026).
Percent mineralization
Bone percent mineralization was significantly lower in the adult control group (71.86 ± 0.06%) than in the OVX ND 6 mo group (72.46 ± 0.06% (p = 0.0494)) and the OVX MA 12 mo group (73.10 ± 0.07% (p = 0.0002)). Ovariectomized sheep consuming the MA diet for 6 months were equivalent to the control (71.86 ± 0.06%).
Statistical correlation
A significant positive correlation was seen between BMD of the beam and the material properties breaking strength (p = 0.0074) and modulus for all groups (p = 0.0011). Beam mineral density determined by DEXA examination and ashing procedures also had a significant positive correlation in all groups (P = 0.0012), emphasizing the degree of association of these two different measurement techniques (Fig. 2).

Graph of beam mineral density determined by DEXA examination compared to that determined by ashing procedures.
Discussion
Previous work in our laboratory has shown that MA diets result in decreased lumbar BMD, decreased vertebral trabecular thickness, connectivity, and crystallinity.10,12–14 The purpose of this study was to gather preliminary data on the effects of the MA diet and/or OVX on cortical bone. In treated groups, we did not demonstrate variability in the density of the whole femoral bone, cortical strength, or cortical percent mineralization.
Despite the limitations of this preliminary investigation, we present some novel information regarding an ovine animal model of dietary induced MA, OVX and the resulting changes seen in cortical bone. Certainly the interactions of OVX and MA diets are complex and have been suggested in other studies to have opposing, cumulative, or regional effects on bone.10,15 Minimal change was seen with MA dietary pressure within the cortical bone envelope. This different than what is typically reported to occur in trabecular bone where a steady decrease in BMD is typically documented. Structural changes in cortical bone may be taking place secondary to the biomechanical stresses imposed by decreased trabecular BMD. The lack of uniformity in the decreases in strength and modulus support this hypothesis.
There is little evidence in this report that the ovine model of MA results in bone with similar qualities as that seen in human osteoporotic bone. Only in OVX sheep consuming a ND was the decrease in breaking strength similar to that seen in aging humans, nearly 20%. 16 Evaluation of modulus revealed significant decreases in adult sheep compared to age matched controls for each treatment group. Interestingly, normal sheep femurs had a similar modulus to human normal tibia reported to be 27 Gpa 17 and ovariectomy has been shown to decrease modulus in radii from sheep treated by OVX alone and dietary induced MA.15,18,19
Limitations of this study include imbalanced groups, failure to study groups of non-OVX sheep consuming the MA diet at 6 and 12 months and OVX sheep consuming the ND diet for 12 months, and failure to perform the study with additional longitudinal time points. In order to address the limitations of the study, statistical analyses applied were rigorous in an effort to accommodate for imbalance of sample size.
In conclusion, we did not document alterations in cortical density and strength after 6 months of exposure to a MA diet. Normal strength and architecture may be maintained to compensate for biomechanical stresses on the bone induced by loss of trabecular bone. Additional study may elucidate the overall effect of acidifying diets on both tra-becular and cortical bone and the mechanical adaptations that result from those changes.
Disclosure
The authors report no conflicts of interest.
