Abstract
We present an overview of some applications of coherence transfer experiments in spatially resolved NMR, with examples from imaging and volume localized spectroscopy. While the major preoccupation of spatially resolved NMR experiments is normally with the dominant component (e.g. water) of heterogeneous multi-component systems, the interest in minor components (e.g. metabolites) of such systems is a strong motivation to develop and apply special techniques. Unlike water, these components typically involve scalar coupled spin systems. They lend themselves therefore to investigations based on the correlated evolution of coupled spins. Specifically, we briefly describe in this contribution the spatially resolved version of multiple quantum experiments, indirect detection experiments and spin correlation experiments.
Keywords
Introduction
Nuclear spins I are associated with magnetic moments µ (= γħI) that precess in an external magnetic field B0 which is applied along the z direction and is termed the Zeeman field. The constant of proportionality between magnetic moment µ and spin Iħ is γ, the magnetogyric ratio, ħ being Planck's constant h divided by 2π.
Spin angular momenta I are, further, quantized, implying that they are stable in only a finite number of orientations in a Zeeman field: a nucleus with spin quantum number I can take only 2I + 1 orientations, corresponding to z spin components m = -I, -I + 1, …, I-1, I. As an important example, spin-1/2 nuclei ( 1 H, 19 F, 31 P, 13 C, 15 N, 29 Si, …) may take only 2 orientations. Each orientation is associated with a different energy (E m = -γħmB0), and the supply of the right quantum of energy permits the nuclear spin to reorient among the allowed energy levels.
Nuclear Magnetic Resonance (NMR) deals with the excitation and detection of such transitions. The transition or resonance frequency ν is proportional to the intensity of the Zeeman field applied and falls typically in the radiofrequency (rf) region: ν = γB0/2π. Transitions, which occur by the direct absorption of a photon of the right frequency, are generally subject to additional conditions: the orientation of a spin may change only to a ‘neighboring’ energy level which differs in energy by a single quantum (i.e. Δm = ± 1); and no more than one spin in a coupled spin cluster may change its orientation. Further, in order to effect such transitions the magnetic component of the resonant radiation field, of amplitude 2B1, must be polarized perpendicular to the quantizing Zeeman field B0.
At thermal equilibrium an ensemble of spins in a Zeeman field develops a net polarization: a non-zero z component of nuclear magnetization is established (longitudinal magnetization M z , which is a form of spin order). At equilibrium, transverse (xy) magnetization M xy vanishes, corresponding to random phases of precession of the spins in the ensemble. The effect of the resonant radiation field is to induce a coherent precession of spins, generating a non-zero component of spin magnetization M xy transverse to the Zeeman field. This non-equilibrium state may be detected by the same resonator that generates the excitation rf field, although surface coils are often employed for detection, owing to their increased sensitivity. States that involve coherent precession of spins may be generically termed coherences; these include not only transverse magnetization components as noted above, but also unobservable states corresponding to multiple quantum transitions (Δm ≠ ± 1, vide infra). Coherences are non-equilibrium states of spin ensembles, which gradually return to thermal equilibrium by non-radiative relaxation processes, which are most often exponential in time, with a time constant T2.
When the basic NMR experiment is performed in the presence of static or rf magnetic field gradients G, spatial information is encoded in the phase and/or frequency, or amplitude of precessing spin magnetization. This enables the measurement of spatially resolved NMR.
Spatially resolved NMR is generally performed employing resonators that enclose the object being investigated, for purposes of signal excitation and detection. However, as noted above, surface coils that–-as the name implies–-are mounted on the surface of the object being investigated may also be employed for these purposes (Ackerman et al. 1980). Surface coils have a ‘depth profile’ characteristic, in the sense that their maximum efficiency at excitation and detection lies at a depth below the surface that equals their radius.
The language of diffraction experiments is often employed to describe spatially resolved NMR experiments, in particular the reciprocal or k space description. In the NMR context, this mathematical description is associated with the definition of k, the spatial frequency, as the integral over time of the product γG/2π (i.e. the product of the magnetogyric ratio with the gradient).
Why Coherence Transfer?
The principal NMR characteristic of most molecules–-unlike water–-is that in a highly homogeneous field they exhibit not one, but a number of NMR resonance frequencies. This is caused by minute differences in the local magnetic field sensed by the nucleus, as governed by the electron density distribution in its neighborhood, an effect known as the chemical shift. Further, nuclear spins also interact among each other, directly through space, as well as indirectly through bonds. It is the latter spin-spin interaction, of magnitude J, that survives in solution state, the former averaging out to zero. Chemical shifts and scalar spin-spin couplings, then, govern the high resolution NMR spectrum of a nuclear spin in solution. In turn, the high resolution NMR spectrum serves as a molecular fingerprint.
Clearly, if spatially resolved NMR information could carry the molecular fingerprint, one would have an extraordinarily powerful diagnostic tool at one's disposal, in characterizing heterogeneous multi-component systems.
The unambiguous registration of the molecular imprint in the NMR spectrum, in turn, relies on experiments that take advantage of the spin coupling network of the system to effectively ‘label’ NMR transitions, and this is commonly accomplished by coherence transfer (Ernst et al. 1987; Chandrakumar et al. 1987). Coherence transfer involves transferring coherences or magnetization components between coupled spins, keeping track of the source and destination spin precession frequencies. Over the decades, the science of spin manipulation has matured into a robust technology.
Motivation
It is often of interest in spatially resolved NMR experiments to probe the spatial distribution of minor components of multi-component systems, as noted earlier. Such is the case for example in metabolite imaging experiments.
One approach would be to employ 1 H chemical shift imaging (CSI), especially with water suppression, as well as outer volume suppression where appropriate. A possible four dimensional pulse sequence implementation is shown in Figure 1. This experiment renders a voxel-wise display of the chemical shift spectrum (Maudsley et al. 1983). Alternatively, the dataset may be displayed as a series of images, at each of a set of selected chemical shifts. The CSI experiment (Callaghan, 1991; Kimmich, 1997) does have issues related to inter-voxel bleeding owing to the point spread function, besides being relatively time consuming because k-space is scanned pointwise.
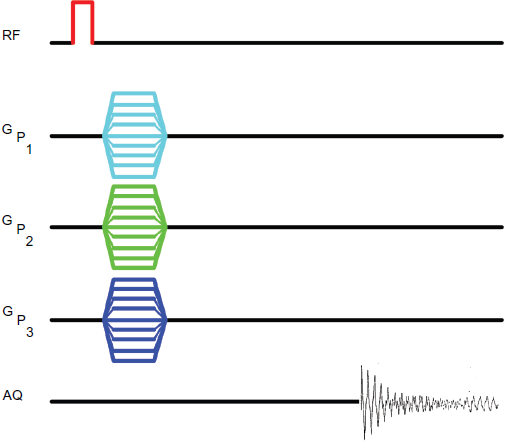
Pulse sequence for 4D CSI. A hard pulse initiates the experiment, no slice selection being involved. Three simultaneous phase encode gradients in the three orthogonal directions are independently incremented in successive experiments, the signal being acquired in the absence of a read gradient, in the manner characteristic of spectral acquisition.
If, however, one were not interested in the entire range of metabolites that are present, but instead in a specific species, an alternative approach could involve the 1 H chemical shift selective imaging (CSSI) of the desired component alone (Joseph, 1985; Callaghan, 1991; Kimmich, 1997). This could for example be achieved with a spin echo type imaging sequence where the excitation could be chemical shift selective, the refocusing being slice selective, as shown in Figure 2. This approach would however be limited by the limited 1 H chemical shift separation between the desired component and others, especially at low fields. Further, selective suppression of the strong water response in vivo would be an exacting task, more so if the desired signals to be imaged are in the frequency neighborhood of the water proton signal.
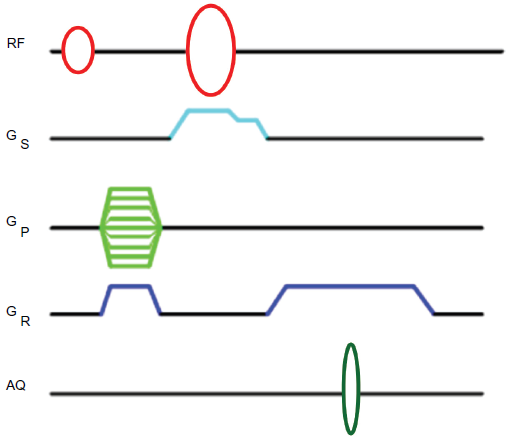
Pulse sequence for CSSI. The first shaped pulse generates transverse magnetization at the chosen chemical shift, which is then phase encoded and read dephased. Slice selection is then achieved by the following shaped pulse which is issued in the presence of a gradient. The signal is acquired in the presence of the read gradient.
The two experimental approaches introduced above to carry the molecular fingerprint in the imaging experiment are fundamentally different from the next two to be introduced, viz., multiple quantum imaging and indirect imaging, in that the latter involve editing based on transformations of spin order, generally by coherence transfer that is mediated by couplings in the spin system. [Note however that transformations of spin order may also be effected under certain conditions by relaxation, especially non-exponential relaxation processes (Bendall et al. 1983; Jaccard et al. 1986; Müller et al. 1987).] The spin transformations involved in multiple quantum imaging may be represented by the schematic diagram of Figure 3.
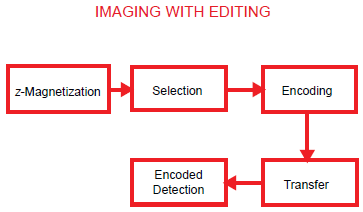
Schematic representation of an imaging experiment involving ‘editing’. For the multiple quantum (MQ) experiment, the ‘selection’ module comprises, for example, a multiple quantum preparation sandwich with preparation time typically ca. (2J)-1, which may be used to select a species of interest, starting from a state of thermal equilibrium. MQC may then be phase encoded in the subsequent ‘encode’ module, converted to observable single quantum coherence (SQC) in the next ‘transfer’ module and finally detected under frequency encoding.
One possibility is the excitation and phase encoding of multiple quantum transitions (e.g. of 1 H) characteristic of the coupling network of the molecular species of interest. This would then be followed by conversion to single quantum transitions, which are finally frequency encoded and detected, as shown in Figure 4. While permitting line scans in k-space, this approach also enables image magnification in the phase encode direction(s), the field of view being reduced by a factor equal to the order of the multiple quantum coherence being encoded.
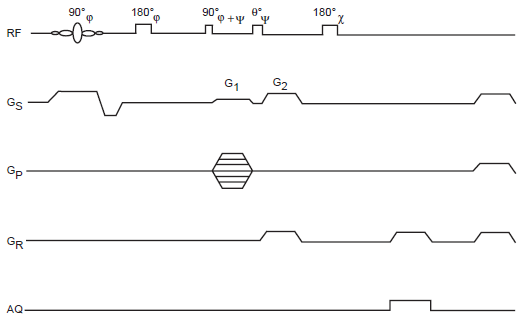
Pulse sequence for 2D multiple quantum imaging with line scans in k space (MUSQ). Slice selective excitation of MQC is performed by the initial three pulse sandwich of pulses, followed by phase encode and coherence pathway selection, refocused reconversion and frequency encoded spin echo detection. The pulse phases to be selected depend on the choice of parity of MQC: of odd order or even order. Selection of odd order MQC's requires the phase of the last pulse of the preparation sandwich to be in quadrature (i.e. 90° out of phase) with respect to the initial pulse. Selection of even order MQC's on the other hand requires the phase of the last preparation pulse to be the same as (or 180° out of phase wrt) the first preparation pulse.
Alternatively, one could take recourse to indirect imaging of nuclei such as 13 C, 15 N, etc., which have attached protons. Such X nuclei have the advantage of excellent chemical shift discrimination, but the disadvantage of poor detection sensitivity. Indirect detection–-via coupled spins I–-is well known to offer in such a case the advantage of improved signal intensities to a maximum possible extent of γ I 3 /γ X 3 in NMR spectroscopy. In the context of spatial resolution, an additional factor emerges in favor of indirect detection, viz., the fact that all X nuclei (except 3 H!) have a smaller γ than the proton, which therefore results in reduced frequency/phase dispersal per unit applied field gradient–-and therefore reduced spatial resolution in the direct imaging of X nuclei.
The indirect imaging approach corresponds in reality to selective 1 H imaging, with efficient suppression of all unwanted 1 H resonances including that of water. The spin transformations involved in indirect imaging may be represented by the schematic diagram of Figure 5. Such an approach also offers scope to employ a tracer strategy to visualize processes in the metabolic steady state, by way of 13 C or 15 N labeling.
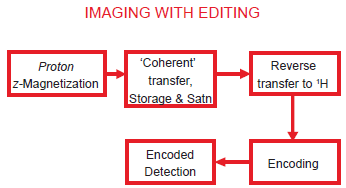
Schematic representation of an imaging experiment involving ‘indirect’ detection. Couplings between abundant spins and rare spins are exploited to perform molecule selective imaging: commencing with equilibrium 1 H magnetization, for example, coherence transfer is performed to the coupled rare spin (such as 13 C or 15 N) and the resulting enhanced magnetization of the rare spin is stored longitudinally. This then permits efficient ‘saturation’ of irrelevant magnetization of the abundant spins including that from other molecules. Following the saturation module which typically comprises a string of RF pulses on the abundant spins alternating with gradients, the enhanced magnetization of the rare spins stored longitudinally is brought back to the transverse plane and reverse transfer is performed to the coupled abundant spin. This may then be followed by slice selection, phase encoding and readout on the abundant spin channel.
As another option, one may perform image guided correlation spectroscopy experiments, which directly carry the molecular imprint. In this approach, volume element(s) of interest is (are) chosen from the basic 1 H image of the dominant component in the system and the molecular information in the chosen voxel(s) is then registered by experiments that employ suitable coherence transfer protocols. It may be recalled that efficient multi-voxel experiments may be performed by employing Hadamard encode-decode strategies (Müller, 1992).
In this contribution, we review some experimental implementations of coherence transfer for NMR imaging and volume localized correlation spectroscopy.
A Note on Transformations of Spin Order
Population differences of spin energy levels at thermal equilibrium correspond, as noted earlier, to an ordering of spins and to the establishment of longitudinal bulk magnetization (‘polarization’) of the spin ensemble. Longitudinal magnetization is proportional to the expectation value of the z-component of spin, I z .
Equilibrium population differences may be transformed into non-equilibrium states, e.g. coherences or other kinds of spin order, by the action of resonant radiofrequency pulses. Relaxation processes on the other hand drive the return of the spin system to thermal equilibrium.
States of the spin ensemble may be described in terms of density operators, which may be visualized as vectors in spin operator space (‘Liouville space’). The effects of internal spin interactions as well as the interactions of spins with external magnetic fields and rf fields correspond to unitary transformations that are equivalent to rotations of vectors in Liouville space.
Spin order, incidentally, may be formally defined in terms of the length (‘norm’) of the density operator, i.e. the trace of the square of the density operator–-and is conserved under unitary transformations.
On the other hand, relaxation processes do not correspond to rotations: they do not conserve the norm of vectors in Liouville space.
Over the last fifteen years, considerable attention has been devoted in the literature to the design of optimal spin transformation strategies, i.e. strategies by which one may maximize the transformation of a given initial spin state into the desired final spin state (Nielsen et al. 1996). One possibility is to construct pulse sequence modules that result in optimal spin transformations, frequently ignoring relaxation effects in such a design strategy. A more recent alternative is to employ optimal control theory to effect desired spin transformations, subject to a number of conditions that one may choose to impose, e.g. rf power deposition restrictions, relaxation, etc. (Khaneja et al. 2001). Optimal sequence design considerations may be reasonably expected to become ever more influential in designing spatially resolved NMR experiments.
Multiple Quantum Studies
Multiple quantum imaging relies on the creation of multiple quantum coherences (MQC's), i.e. transitions between energy levels whose magnetic quantum numbers m differ by a quantity other than ±1 unit (Drobny et al. 1978; Braunschweiler et al. 1983; Weitekamp, 1983). It is usually the case that couplings between spins are required to prepare such coherences. While they would normally be expected to be resolved couplings in solution state, unresolved couplings could nevertheless function similarly, owing to their influence on relaxation. In any event, the preparation of MQC's generally involves a two pulse sandwich (three pulses including a refocusing pulse), as depicted in Figure 4 and Figure 6. One may visualize this broadly as the absorption of a photon by the spin cluster, followed by spin interaction during evolution, which prepares it to absorb another. The selection of desired signals on each scan may be achieved conveniently by gradient controlled coherence pathway selection. As an example, two gradient pulses with amplitude ratio 1: n issued respectively before and after the transfer module would enable the selection of the pathway that leads from n quantum coherence before the transfer period to -1 quantum coherence after the transfer. It is also possible to use the read dephase gradient as the first coherence pathway selection gradient issued before the transfer period.
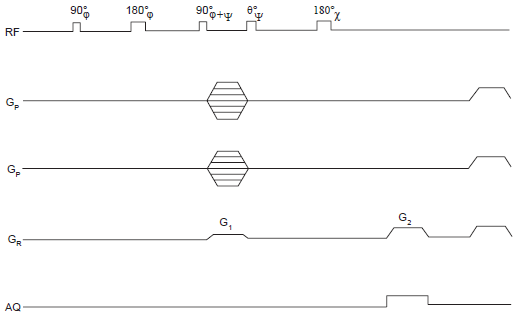
Pulse sequence for 3D multiple quantum imaging with line scans in k space (MUUSIQ). The essential elements are similar to those in Figure 4, except that there is now no slice selection, and two orthogonal phase encodes are involved with independent incrementation. The specific implementation shown combines the read dephase and frequency encode functions with coherence pathway selection.
Early multiple quantum imaging procedures were performed on solids where the linewidth is too large to permit standard frequency encoding (Garroway et al. 1984; Günther et al. 1990). Accordingly, those experiments involved a spectral-spatial strategy, with point scans in k-space. However, multiple quantum imaging may be performed with procedures that correspond to line scans in k space if the single quantum linewidth is small enough to permit frequency encoding (Chandrakumar et al. 1993a). This is normally the case when dealing with liquid like phases, or in general with phases that have a low order parameter (Wimperis et al. 1991). An early demonstration of multiple quantum imaging with line scans, carried out on a phantom object is shown in Figure 7. The phantom comprised three long cylindrical tubes–-two containing ethanol and one containing methanol–-stacked together in an empty larger outer tube. Under standard conditions, methanol does not exhibit spin couplings owing to the rapid exchange of hydroxyl protons, while ethanol does exhibit spin couplings between the three protons of the methyl group and the two protons of the methylene group. In principle, the ethyl group could sustain upto 5-quantum coherence. In the experimental results shown, 3-quantum coherence was generated, phase encoded and reconverted to single quantum coherence for readout. The molecule specificity as well as reduction in FOV per unit applied field gradient may be noted. It may be noted that in such homonuclear spin systems, n quantum coherence behaves as though γ were increased n-fold.
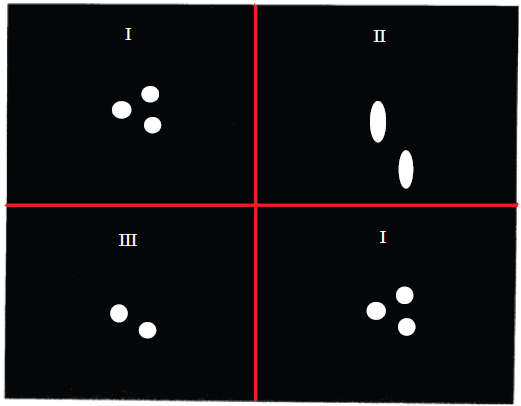
Slice selective, two dimensional triple quantum-single quantum imaging at 300 MHz on a three-tube phantom described in the text. Three different images have been overlaid to compose the figure. They are all slice selective 128 x 128 axial (xy) images, the slice being perpendicular to the long axis (z) of the tubes. Top left image I (reproduced for convenience on bottom right): standard single quantum spin echo image; Top right image II: 3Q-SQ MUSQ image with the same phase encode gradient increment as in I; Bottom left image III: 3Q-SQ MUSQ image with phase encode gradient increment reduced by a factor of 3. The phase encode direction in each case is the vertical axis of the figure. The experiments were performed with a 15 mm RF insert with an actively shielded Bruker microimaging probehead in an 8.9 cm bore magnet, the gradient strengths being about 7 G cm-1. A sinc pulse was employed for slice selection, the slice thickness being about 0.67 mm. [Adapted from Chandrakumar et al. 1993a].
The optimization of the efficiency of these experiments is being currently explored by us. It may be noted that optimized experiments of this class could in principle be harnessed for 31 P ATP imaging, and for magnified imaging of capillarities (with potential MRA applications), to mention some possibilities. Other authors have employed multiple quantum filtering for imaging selected species, but in these cases, both phase and frequency encode were performed on single quantum coherences (Pekar et al. 1986; Pekar et al. 1987; Reddy et al. 1994).
Indirect Imaging
As noted earlier, molecule specificity may be readily achieved if one were to attempt imaging of X nuclei, such as 13 C or 15 N. However, in addition to the customary problem of detection sensitivity, one has to contend here with an additional problem of reduced sensitivity to gradients. While specialized hyperpolarization studies (Ardenkjaer-Larsen et al. 2003; Johannesson et al. 2004) may be performed to improve detection sensitivity, indirect imaging appears to be a general all round solution, addressing both the issues of detection sensitivity as well as sensitivity to gradients (Heidenreich et al. 1998a).
The maximum efficiency may be approached in a strategy that employs 1 H-coupled X nuclei. Equilibrium proton polarization may be transferred to the coupled X nucleus (forward coherence transfer) and stored longitudinally. Undesired proton magnetization including that of water and other irrelevant species may then be efficiently saturated. Following this, the enhanced X nucleus magnetization stored longitudinally may be brought into the transverse plane and a reverse coherence transfer may be performed back to the coupled protons. The resulting selected proton magnetization may then be subjected to standard imaging protocols, such as gradient echo, spin echo or EPI. Figure 8 shows a pulse sequence for this application, in spin echo mode. This sequence employs J cross polarization (JCP) for the forward and reverse coherence transfer, which has advantages in terms of motion insensitivity under spin lock.
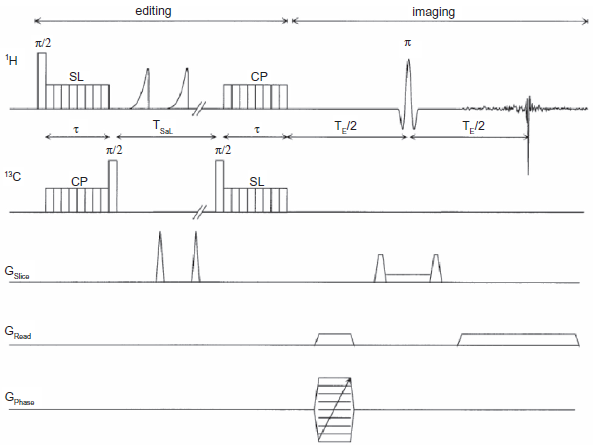
Pulse sequence for ‘indirect’ 13 C imaging in spin echo mode. Each of the modules of Figure 5 may be readily recognized. Selective coherence transfer from 1 H to 13 C is accomplished by J cross polarization with a highly windowed sequence, PRAWN, to ensure low rf power deposition; this is followed by longitudinal storage on the 13 C channel and a saturation comb of alternating rf pulses (applied to 1 H) and gradients. Thereafter the enhanced 13 C magnetization stored longitudinally before saturation is brought back into the transverse plane and reverse transferred to the coupled 1 H spins. This is then followed by phase encode and read dephase, slice selection and frequency encoded readout.
A point to note is that the standard gradient echo would not work in this mode since the echo time TE is often of the order of (2J)-1, J being the H-X coupling constant. This would then cause the generation of essentially equal but opposed magnetization vectors (‘anti-phase’ magnetization), which would result in nulling the signal detected in the presence of the read gradient. This effect of spin coupling during a gradient echo sequence may be annulled by introducing a refocusing pulse on any one nucleus (conveniently, the X nucleus) at the middle of TE. Equivalently, in an EPI procedure, X nucleus refocusing pulses may be issued at every zero crossing of the read gradient, just before or after the blip gradient.
A further feature needs mention: in XAn systems (such as XA2 and XA3) that have multiple protons (A) attached to a given X nucleus, saturation cannot in principle randomize all states other than the desired magnetization that has been stored longitudinally. In particular, forward transfer by cross polarization on such systems generates not only the desired magnetization but also other states that include homonuclear zero quantum coherence and homonuclear longitudinal multispin order (LOMO; Chandrakumar et al. 1993b) of protons that are coupled to the X nucleus. The standard saturation procedure of a comb of 90° rf pulses each followed by a gradient does not succeed in randomizing such states, which survive the saturation and contribute to proton magnetization following reverse transfer. This contribution may be readily computed and is displayed in Figure 9 for the XA2 system and in Figure 10 for the XA3 system. On a scale where each proton spin A contributes unit signal intensity in the standard 1 H spectrum, the final signal intensity in the two cases would be expected to be 1 unit (XA2) and 1.11 units (XA3), not taking into account the reverse transfer of these multispin states that survive the saturation. When the contribution of these terms are taken into account, however, it is seen that the final signal intensities could in fact be upto ~1.33 units (XA2) and ~1.26 units (XA3), assuming that relaxation of these multispin states is negligible during the saturation period.
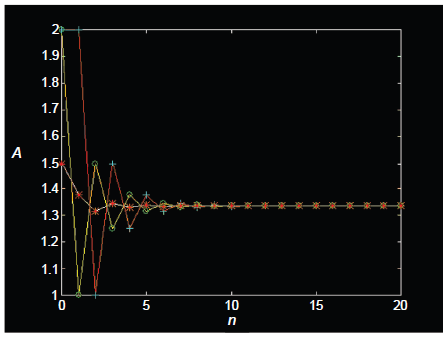
Amplitudes A of zero quantum coherences (open circle) and longitudinal zz terms (plus sign) after a JCP forward transfer (A2→X) in an XA2 system, following n repetitions of a saturation comb (90x° pulse on the A spins, followed by gradient). Net reverse transfer amplitudes (asterisk) are also shown (X→A2), scaled to unit intensity per A spin.
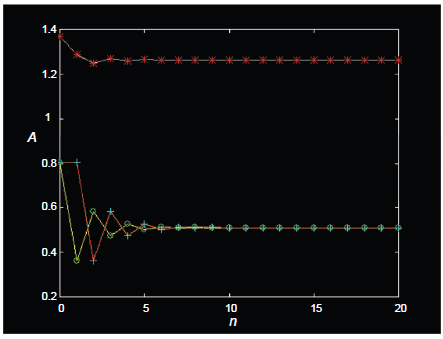
Amplitudes A of zero quantum coherences (open circle) and longitudinal zz terms (plus sign) after a JCP forward transfer (A3→X) in an XA3 system, following n repetitions of a saturation comb (90x° pulse on the A spins, followed by gradient). Net reverse transfer amplitudes (asterisk) are also shown (X→A3), scaled to unit intensity per A spin.
An application of indirect 13 C imaging to the imaging of the hypocotyl region of plant seedlings in vivo is shown in Figure 11. In this application (Heidenreich et al. 1998b), a castor bean seedling was imaged, whose cotyledons were stripped of their endosperm and dipped in a bath of 50 mM 13 C1 glucose and 50 mM 13 C1 fructose. The seedling synthesizes sucrose from its hexose intake from the nutrient bath. The F1 site of sucrose was selected for imaging. It may be noted that 13 C chemical shifts permit discrimination of the fructose ring of sucrose from that of free fructose. Images are run as a function of time after dipping the cotyledons in the nutrient bath. The spin echo images–-in combination with EPI based experiments (Hudson et al. 2002) that were about six times faster–-clearly demonstrate the selective unloading of sucrose in the cortex parenchyma of the hypocotyl region, as opposed to the pith.
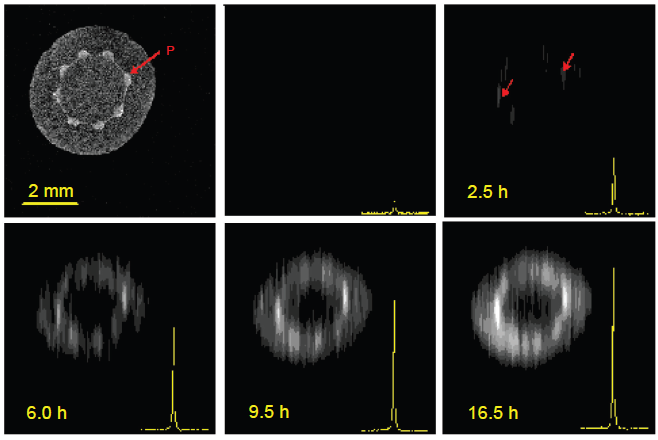
1 H-detected 13 C images of castor bean seedling, obtained at 400 MHz as a function of time after the cotyledons stripped of their endosperm were dipped in a bath of 13 C enriched fructose and glucose. Red arrows point to the vasculature as seen from the water proton image (top left) and from the 1 H detected image of sucrose (top right). [Adapted from Heidenreich et al. 1998a].
It may be noted that these cross polarization based experiments that employ windowed pulse sequences (PRAWN; Chandrakumar et al. 1999) for selective JCP are typically run with a peak rf amplitude of the spin lock (SL)/contact pulses (CP) of ~1 kHz with an rf duty cycle of ~20%, corresponding to an average rf amplitude of ~200 Hz, which is of the order of the 13 C- 1 H coupling, J. It may be recognized however that even average rf amplitudes down to J/4–J/5 Hz permit the transfer to occur with little loss of efficiency (Chandrakumar, 2004).
Proton detected indirect imaging of 15 N has also been reported, producing images of the distribution of urea in the kidney (Freeman et al. 1993).
Volume Localized Spectroscopy Involving Coherence Transfer
Here, we briefly discuss four applications:
Heteronuclear:
Homonuclear:
Volume Localized X Nucleus Spectroscopy with Polarization Transfer
Experiments such as INEPT (Morris et al. 1979) have been implemented for sensitivity enhanced, volume localized detection of X nuclei, such as 13 C. The pulse sequence is shown in Figure 12 (Watanabe et al. 1998).
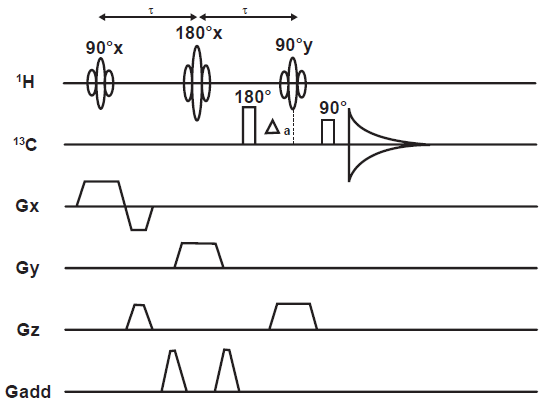
The LINEPT pulse sequence. Δ a is set to (4J)-1 to generate 1 H transverse magnetization anti-phase with respect to the J CH coupling.
The approach is to perform volume localization on the 1 H channel of a typical PT sequence, then transfer polarization to the coupled 13 C spins. It may be recalled that the INEPT mode of PT involves transformation of proton longitudinal magnetization to two-spin longitudinal order of the 1 H- 13 C coupled pair, passing through an intermediate state of anti-phase 1 H transverse magnetization; 13 C transverse magnetization is then generated from the state of heteronuclear two-spin order.
These experiments are clearly valuable as they permit the volume localized 13 C spectral registration of metabolites. An issue with such ‘broadband’ heteronuclear experiments however is the adequate coverage of the 13 C chemical shift range with 90° and 180° pulses issued on resonators of the required size, especially at higher fields. Suitably tailored adiabatic pulses go some way in tackling this.
Volume Localized Spectroscopy of 1 H Attached to 13 C
The exclusive detection of protons attached to 13 C may be readily accomplished by exploiting the effects of J modulation, as is done in the POCE (Proton Observe Carbon Edited) experiment (Rothman et al. 1985). As shown in Figure 13, this involves basically a two scan procedure that commences with generation of 1 H transverse magnetization, followed by a free evolution period (echo time 2τ) during which it is refocused by a π pulse and either of two actions apply in addition: (A) 13 C decoupling is applied during the echo time but is gated off during the first half of this period on alternate scans; or (B) during the middle of the echo time, carbon flip (by a π pulse) is accomplished on alternate scans. As a consequence, the phase of 13 C coupled 1 H magnetization is opposed to that of 1 H magnetization from the 12 C isotopomer in one scan but matches it in the other; subtraction of the second scan from the first then eliminates proton signals from the 12 C isotopomer. The carbon gated decoupling method requires the echo time to be set to (2/J), while the carbon flip method requires that the echo time be set to (J)-1.
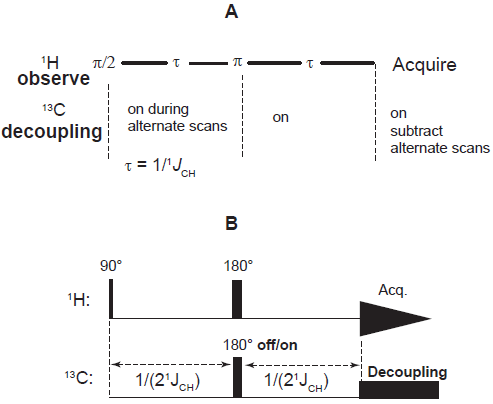
Alternative pulse sequences for Proton Observe Carbon Edited (POCE) spectroscopy: (A) gated decoupling; and (B) carbon flip.
This approach has been extended to include volume localization in both the PRESS and STEAM environments (Chen et al. 1998; Pfeuffer et al. 1999), the latter for example in the ACED STEAM experiment, shown in Figure 14.
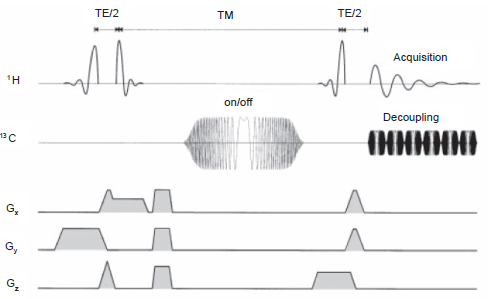
The Adiabatic Carbon Edit and Decouple (ACED) STEAM pulse sequence. The functioning of the sequence is described in the text.
This sequence involves a TE/2 setting of (2J)-1 and an adiabatic inversion pulse on the 13 C channel on alternate scans during the mixing period TM. The mechanism of the sequence is basically similar to that of INEPT, except for the absence of a refocusing pulse during the first TE/2 period; transverse anti-phase 1 H magnetization is partially transformed at the end of TE/2 into two-spin longitudinal order of the 1 H- 13 C coupled pair. The adiabatic inversion pulse on 13 C toggles the sign of the two-spin longitudinal order. When accompanied by receiver phase alternation, this enables suppression of 1 H signals that are not 13 C coupled. The final TE/2 period, also of duration (2J)-1, refocuses the anti-phase 1 H magnetization generated by the last 1 H 90° pulse, permitting 13 C decoupled 1 H spectral acquisition.
Volume Localized 2D Zero Quantum Spectroscopy in the STEAM Environment
The schematic of the STEAM sequence is shown in Figure 15 and a pulse sequence implementation in Figure 16 (Granot, 1986; Kimmich et al. 1987; Frahm et al. 1987).

Block diagram of a general volume localization experiment. Three orthogonal planes selected by a combination of suitable selective rf pulses and gradients intersect at a voxel from which the desired echo signal is read out.
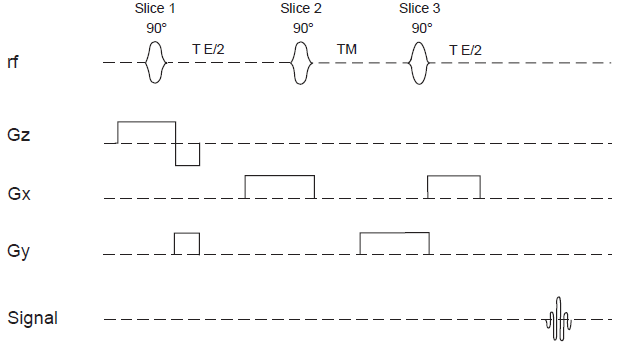
STEAM sequence for volume localized spectroscopy.
The STEAM sequence functions by generating transverse magnetization from a slice selected by the first pulse, allowing it to evolve, then storing longitudinally a part of this magnetization arising from the rod of intersection with the orthogonal slice selected by the second pulse. The third slice selective pulse then brings the longitudinal magnetization into the transverse plane, leading to an echo (the so-called stimulated echo) arising from the voxel of intersection of the three orthogonal slices.
This sequence may be readily adapted to two-dimensional spectroscopy as shown in the schematic of Figure 17, the virtual time dimension corresponding to a variable TM, during which the evolution of zero quantum (ZQ) coherences occurs. It may be noted that homonuclear zero quantum coherences do not sense field gradients–-and therefore remain intact during a gradient dephase following the second rf pulse of the STEAM sequence.
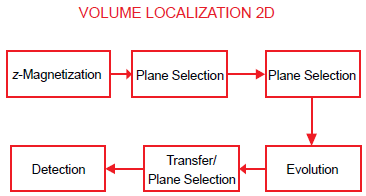
Block diagram of a volume localized 2D zero quantum experiment. Zero quantum coherence of coupled spins is generated in the rod of intersection of the slices selected by the first two rf pulses; these ZQC's evolve during a systematically incremented TM, and echo as SQC in a voxel following the third slice selective STEAM pulse; this is then detected. Systematic repetition of the experiment with incremented TM generates a 2D dataset that delivers the zero quantum spectrum in F1 and the single quantum spectrum in F2 on double Fourier transformation.
This experiment permits identification of coupled spins, displaying the difference of their chemical shifts in the virtual dimension F1 and the normal (single quantum) chemical shifts in the acquisition dimension F2. Considering the example of lactate, for instance, the difference of the methyl and methine proton shifts occur in F1 and their respective chemical shifts occur in F2. This is useful for assignment purposes, especially at low fields that are typical for MRI. The methine proton resonance occurs too close to that of water to permit unambiguous detection in standard 1D mode. Opening up a second frequency dimension and spreading the peaks in the two dimensional frequency plane offers significant advantages in solving the assignment problem. An early 2D ZQ correlation spectrum of rat brain is shown in Figure 18 (Chandrakumar, 1996) and was obtained on a 6 mm cube voxel of an intact rat brain after it was killed with an injection. The lactate peaks that develop owing to anaerobic glycolysis following death are clearly visualized; however owing to the strong ridge at F1 = 0, peaks from species with closely spaced chemical shifts, such as taurine, are less easy to identify.
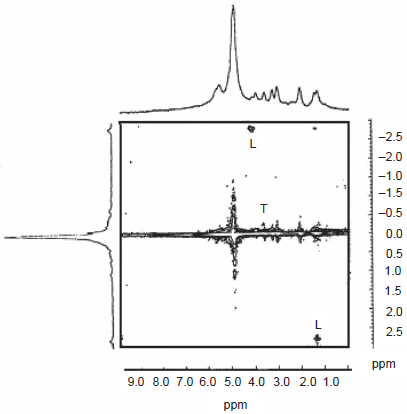
2D ZQ correlation spectrum of in-tact rat brain, performed at 100 MHz on a cubic voxel with edge 6 mm. [Adapted from Chandrakumar, 1996].
Spin Echo Correlation Spectroscopy with Suppression of the Pseudo-Diagonal (DISSECT)
The spin echo correlation spectroscopy (SECSY) experiment is well known in high resolution NMR (Nagayama et al. 1979) as an alternative implementation of the basic correlation spectroscopy experiment, COSY (Aue et al. 1976). The principal difference in the operating mode of SECSY vis-à-vis COSY is that the 90° mixing pulse is issued at the middle of the evolution period, at the end of which data acquisition commences from the top of the spin echo that is generated. For coupled spins, SECSY generates peaks with half their chemical shift differences along F1 and the normal shift of each of the coupled spins along F2. Clearly, there is some resemblance here with zero quantum spectroscopy. Like the latter, SECSY also is troubled by a strong ridge at F1 = 0, which corresponds to magnetization that was not transferred between coupled spins during the experiment. For this reason we may term the ridge at F1 = 0 as the pseudo-diagonal and the peaks on the ridge as ‘pseudo diagonal’ peaks. A volume localized version of SECSY has been found to be useful for spatially resolved NMR because acquisition from the echo top is attractive in light of the inhomogeneities inherent in the in vivo context (Ziegler et al. 1993); however this also suffers from the strong pseudo-diagonal.
A modified version of SECSY, which we term DISSECT (DIagonal Suppressed Spin Echo Correlation SpecTroscopy) is successful at strongly suppressing the ridge at F1 = 0. The pulse sequence is shown in Figure 19, results on a phantom in Figure 20, and a slice selective version of DISSECT in Figure 21 (Kickler, 2002).
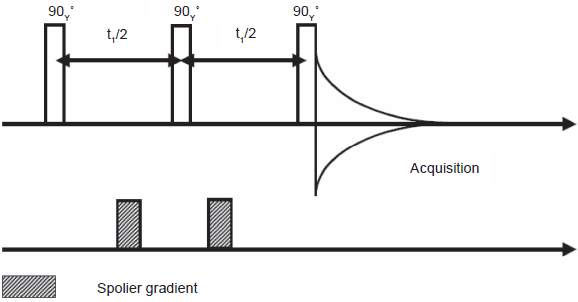
The DISSECT sequence. This three-pulse sequence includes a pair of gradients around the second 90° pulse to ensure echo selection.
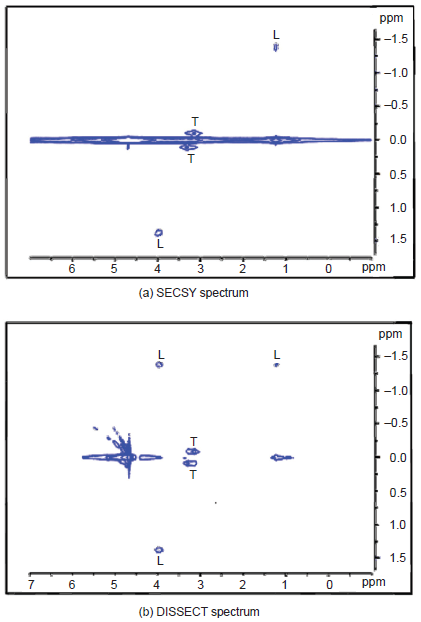
20 mM taurine and lactate in D2O, registered with a surface coil using SECSY and DISSECT. [Adapted from Kickler, 2002].
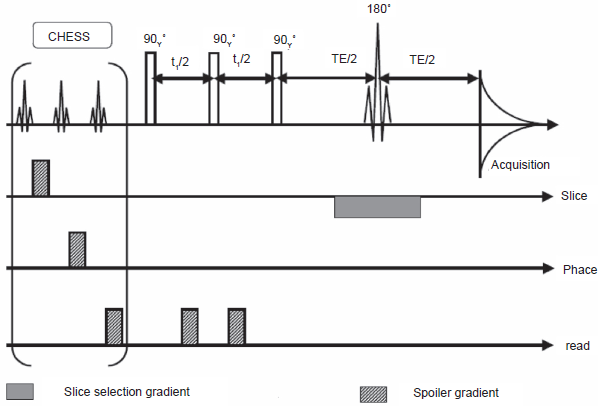
Slice selective DISSECT sequence. This version includes a chemical shift selective suppression module as front-end (CHESS), and is followed by the three-pulse DISSECT sequence, and finally a slice selection module.
The sequence involves a third 90° pulse of appropriate phase and functions essentially by storing undesired ‘diagonal’ peak signal contributions longitudinally, while transforming some anti-phase magnetization components to unobservable multiple quantum coherences, leaving other anti-phase contributions to echo beyond the acquisition window.
Application of DISSECT for volume localized correlation spectroscopy on real world ‘objects’ is in progress in our Laboratory.
Disclosure
The authors report no conflicts of interest.
Footnotes
Acknowledgments
I have great pleasure in acknowledging a number of collaborators and students who were involved in segments of our own work that have been included in this overview. I would like to acknowledge, in particular, Dr. S. Sendhil Velan (MQI); Dr. J. Frahm, Dr. K-D. Merboldt, Dr. M.L. Gyngell, Dr. G. Helms (2D STEAM ZQS); Prof. R. Kimmich, Dr. W. Köckenberger, Dr. M. Heidenreich, Dr. R. Bowtell (CYCLCROP); Dr. V. Visalakshi (reverse transfer JCP calculations with inclusion of zz and ZQ terms); Dr. V. Vijayan (calculations of JCP with weak rf fields); Dr. M. Décorps, Dr. A. Ziegler and Dr. N. Kickler (DISSECT). I owe special thanks to Ms. Christy George for her time and efforts in patiently and skilfully upgrading the presentation quality of the manuscript.
