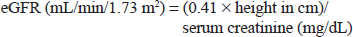Abstract
Gadobutrol is a 1-molar gadolinium-based contrast agent with well-characterized safety and efficacy for magnetic resonance imaging (MRI) in adults and children ≥ 2 years old. This observational study assessed gadobutrol-enhanced MRI in children < 2 years of age. Sixty infants (mean age 11.1 months) underwent MRI using gadobutrol at standard dose of 0.1 mL/kg (0.1 mmol/kg) body weight. MRI examinations included brain, spine, and neck (n = 24), subcutaneous soft tissues (n = 14), chest, abdomen, and pelvis (n = 12), musculoskeletal system (n = 7) and vascular system (n = 3). No patients experienced adverse events related to gadobutrol injection. In 57 patients with confirmed diagnoses, gadobutrol-enhanced MRI provided findings consistent with confirmed pathologies. This study indicates that gadobutrol at a standard dose for MRI is safe in patients aged < 2 years and provides diagnostic information for multiple pathologies.
Introduction
Magnetic resonance imaging (MRI) is widely considered the optimal imaging modality for diagnosing a range of congenital and acquired disorders in pediatric patients. 1 Gadolinium-based contrast agents (GBCAs) are frequently utilized with MRI to enhance the identification and characterization of pathology and to assist in selecting the most appropriate management.
There is growing evidence to characterize the efficacy and safety of GBCAs in children above 2 years of age.2–5 For children below this age, study data on contrast-enhanced MRI (in particular, on safety) are more limited, being restricted largely to case studies or to small numbers of patients within larger studies.6–10
A primary requirement for the use of contrast agents in patients less than 2 years of age is optimal dosing for safety and efficacy. Dosing of GBCAs in all age groups is typically adjusted according to body weight. For very young children, renal immaturity has the potential to influence the clearance of agents that are excreted predominantly by glomerular filtration, including GBCAs. This has led to recommendations to use caution when administering GBCAs to neonates and infants. 11
While there is morphological evidence of renal immaturity in the neonate (ie, up to 1 month old), renal function does appear to be responsive to the changing physiological demands in the growing infant.12,13 An estimated glomerular filtration rate (eGFR) value normalized to body surface area has been established in clinical practice for the assessment of renal function, offering the advantage of standardization and permitting comparison between individuals of different sizes. The average body surface area, calculated from height and weight, is generally taken to be 1.73 m2 for an adult. eGFR values normalized to body surface area are lower in young pediatric subjects than in adults. This is because the body surface area/body weight ratio is higher in pediatric subjects, as small size has a relatively higher body surface area. Body surface area-corrected eGFR values rise with increasing age. Thus, eGFR values normalized to body surface area have to be interpreted in pediatric patients bearing in mind the appropriate reference range. A body surface area-corrected eGFR of 25–30 mL/min/1.73 m2 (±2 standard deviations) can be considered physiologically normal in the term neonate, and is not indicative of impaired renal function, as it would be in adults.14,15 Body surface area-corrected eGFR values attain adult levels by around 6–12 months of age.14,16 Pharmacokinetic studies of renally excreted drugs in this age group describe kinetics comparable with older children and adults. 17
These observations suggest that it is important to investigate the optimal dosing of GBCAs in children below 2 years of age, including appropriate interpretation of eGFR values.
Gadobutrol (Gadovist®/Gadavist™; Bayer HealthCare, Berlin, Germany) is a GBCA that uniquely combines a 1-molar (1-M) concentration of gadolinium chelate with high T1 relaxivity, yielding the highest effect on T1 relaxation times, which is associated with favorable diagnostic performance in adult studies of central nervous system (CNS), liver, and kidney MRI and MR angiography.18–22 Gadobutrol is indicated for use in contrast-enhanced MRI in adults and in children aged 2 years and above in the USA, Canada, Europe, and other countries. The main indication is CNS imaging, although in many countries MR angiography, kidney, liver, and whole body imaging are approved as well. Gadobutrol is not currently approved for use in children aged below 2 years.
Gadobutrol has a macrocyclic structure, which contributes to the high stability of the gadolinium chelate. 23 As shown by in vitro experiments, macrocyclic GBCAs release no detectable free gadolinium ions into human serum, whereas gadolinium release is observed for agents with a linear structure. 24 It is the prevailing theory that release of gadolinium ions is associated with the development of nephrogenic systemic fibrosis (NSF; also known as nephrogenic systemic sclerosis), which is reported in rare cases in adults, and more rarely in older children, with severely impaired renal function.25,26 GBCAs with a linear structure (gadodiamide, gadopentetate, and gadoversetamide) are contraindicated in patients with impaired renal function, while the use of ‘low-risk’ macrocyclic GBCAs (gadobutrol, gadoteridol, and gadoterate meglumine) is permitted in patients with impaired renal function, with the recommendation to screen patients at risk for NSF and to estimate kidney function in patients at risk for chronically reduced renal function.27–29 To date, no cases of NSF have been reported in children under 2 years old following administration of any GBCA.
A recent study of gadobutrol in children aged 2 to 17 years concluded that its safety profile in this age group is similar to that in adults. 2 Laboratory parameters relevant to renal safety, including serum creatinine and eGFR, revealed no clinically significant changes following gadobutrol administration. Pharmacokinetic analyses indicated that no dose adjustment of gadobutrol was required compared with adults, other than standard body weight-adjusted dosing (ie, 0.1 mmol/kg body weight). Based on the similar pharmacokinetic profile of gadobutrol in children aged 2 to 17 years compared with adults, imaging efficacy would be predicted to be the equivalent across these age groups for all approved indications. 30 The pharmacokinetics of gadobutrol in subjects under 2 years of age has not yet been evaluated in clinical trials. However, it is estimated that more than 7000 administrations of gadobutrol have now been performed in children under 2 years old worldwide. 31
The current study describes the first extensive observational experience of gadobutrol-enhanced MRI in patients under 2 years of age. Safety and efficacy data were collected from assessments performed according to the specific clinical requirements of each patient, following locally developed protocols.
Methods
Study design
A single-center, prospective, observational study of the safety and efficacy of gadobutrol-enhanced MRI in patients under 2 years of age was conducted between December 2009 and July 2010.
The study was approved by the local Human Research Ethics Board and was performed in accordance with the current amendment of the Declaration of Helsinki and the International Conference on Harmonization/Good Clinical Practice Guidelines.
Study population
All study patients presented to a single division of pediatric radiology at a service that investigates approximately 20 children under age 2 each month. Contrast-enhanced MRI was performed according to local protocols for specified pathologies. Gadobutrol was used in all consecutive patients, based on a departmental policy change to adopt this specific contrast agent. Exclusion criteria for contrast agent administration included previous reactions to any GBCA or a history of renal impairment.
MRI protocols
MRI protocols, including methods for patient preparation, imaging sequences, and data acquisition, followed local guidelines. Patients underwent MRI with a 1.5-Tesla (T) system (Siemens Avanto 1.5) and standard coils. MRI imaging pre- and post-contrast used routine turbo spin-echo (SE) sequences. A typical MRI protocol was used: T1-weighted (repetition time [TR] = 400–430 ms, echo time [TE] = 12–13 ms) and T2-weighted (TR = 5900 ms, TE = 105 ms) SE acquisitions and fluid-attenuated inversion recovery MR scans (TR = 7850 ms, TE = 117 ms) of the head before administration of the contrast agent (slice thickness = 5 mm; matrix = 307 × 384; field of view = 200–230 mm; inversion recovery = 2200; and number of excitations = 2). T1-weighted SE acquisition was performed within 2–5 minutes (min) of administering the contrast agent. The same imaging system, planes of view, and parameters were used for both pre- and post-contrast examinations in each patient, and care was taken to ensure that image location and angulation were identical for both examinations.
Gadobutrol was administered as a single intravenous bolus of 0.1 mL/kg body weight (equivalent to 0.1 mmol/kg body weight), which is the recommended standard dose in adults and pediatric patients aged above 2 years. 2 No dose adjustment was made based on whether the child was term or preterm. The contrast agent was injected into a peripheral vein by hand at approximately 1 mL/second, followed by about 5 mL saline flush. We also injected into central lines of selected patients, utilizing the same injection rate and 10 mL of saline flush. The central lines were then injected with 5 mL of heparin into an implanted venous access device (IVAD) central line or 3 mL of heparin into a Broviac central line. Studies were supervised and interpreted by 2 experienced pediatric radiologists with 15 years and 13 years of practice experience, respectively.
Safety
Adverse events were assessed by review of the inpatient and outpatient medical charts in hospitals across the region by one of the investigators for up to 120 days after the MRI procedure. Adverse events were rated as potentially contrast agent-related at the discretion of the investigator. Examples of monitored adverse events included immediate effects such as retching, vomiting, itching rash, hives, urticaria, and bronchospasm. We were unable to assess certain local adverse symptoms, such as the subjective feeling of heat. Delayed reactions over 120 days included admission to hospital or a visit to the emergency department for electrolyte abnormalities, renal abnormalities, skin disorders, cardiac disorders, or hematologic impairment.
Renal function was assessed based on the serum creatinine concentrations. Serum samples were collected, stored, and analyzed according to local laboratory standards. Renal insufficiency was defined as a serum creatinine concentration > 60 μmol/L (0.68 mg/dL).
32
eGFR standardized to body surface area was calculated retrospectively using the revised Schwartz equation
33
; (an online calculator is available at: http://nkdep.nih.gov/professionals/gfr_calculators/idms_schwartz.htm):
Physiological eGFR values were defined as follows: age 4 to 28 days: 26 to 68 mL/min/1.73 m2, 1 to 6 months: 39 to 114 mL/min/1.73 m2, 6 to 12 months: 49 to 157 mL/min/1.73 m2, and 12 to 19 months: 62 to 191 mL/min/1.73 m2, gradually reaching values of around 165 mL/min/1.73 m2 by the age of 2 years. 34
Efficacy
The efficacy of gadobutrol-enhanced MRI was assessed using corresponding diagnoses obtained from MRI and the final, confirmed diagnoses established from clinical, pathological, or follow-up imaging studies. The images were assessed by the investigators.
Statistical analyses
Descriptive statistics were employed for analysis of safety and efficacy outcomes in this observational study. Serum creatinine and eGFR assessments were performed on the total population and on subgroups categorized by age. Efficacy analyses identified the proportions of patients for whom the MRI diagnosis was confirmed by the final diagnosis.
Results
Study population
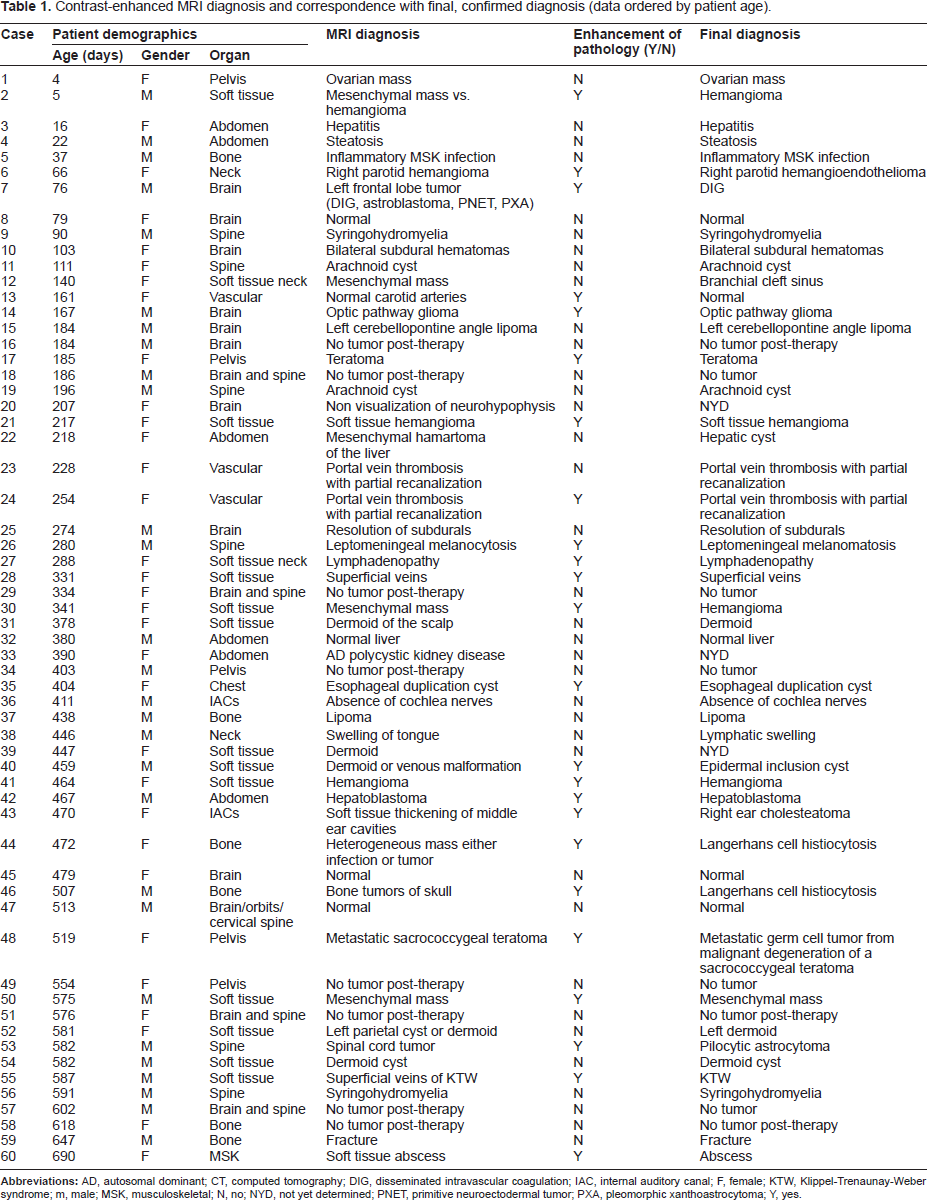
Contrast-enhanced MRI diagnosis and correspondence with final, confirmed diagnosis (data ordered by patient age).
The organ systems assessed for pathologies included the brain, spine, and neck (n = 24 patients), subcutaneous soft tissues (n = 14), chest, abdomen, and pelvic organs (n = 12), musculoskeletal system (n = 7), and vascular system (n = 3). These organ systems, grouped by patient age, are indicated in Figure 1.
Study population: organ systems assessed; patients grouped by age.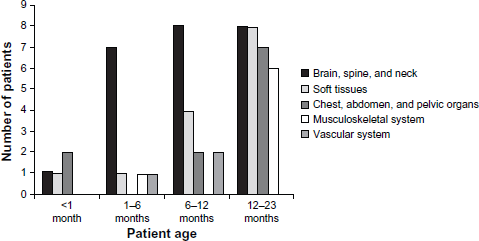
Safety
No patients experienced an adverse event considered to be related to gadobutrol administration during the 120-day observation period post-MRI. There were no episodes of extravasation of contrast during injection in any of the patients.
Serum creatinine concentrations were measured in 33 patients pre-contrast injection and in 25 patients at a mean of 25 days (range, 1–107 days) post-injection. Twenty-one of the 25 patients assessed post-injection were also assessed pre-injection. The lowest detectable serum creatinine concentration at assay was 10 μmol/L. All assessed patients had a normal creatinine concentration (ie, <60 μmol/L) pre-contrast, with the exception of 1 patient (aged 37 days at MRI), whose serum creatinine concentration was 65 μmol/L pre-contrast injection (Case number 5). This serum creatinine measurement was obtained when the child came to the hospital dehydrated 26 days prior to MRI. The child was rehydrated, became stable, and was sent home from emergency. At the time of MRI, the child was hemodynamically stable. The need to assess for osteomyelitis using contrast outweighed concerns regarding impaired renal function, given the state of the patient when the serum creatinine was measured, approximately 4 weeks earlier.
Serum creatinine concentrations post-gadobutrol injections were in the normal range in all assessed patients. Changes in serum creatinine concentration in patients who were assessed both pre- and post-injection (n = 21) were variable. The greatest change in creatinine concentration was a decrease in the patient with above-normal levels pre-injection. There were no clear differences in the change in creatinine concentration between patients who were categorized by age (Fig. 2A), gender, or organ system investigated. A review of the medical records of patients without creatinine measurements confirmed the absence of electrolyte abnormalities.
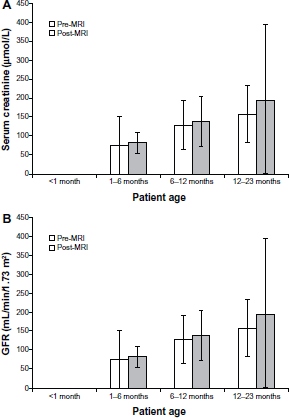
eGFR values were broadly within normal ranges pre-contrast injection and showed a trend, as predicted, to higher values in older patients, with large intra-individual variations. Changes in eGFR values between pre- and post-injection times were also variable in assessed patients (n = 19), with no clear relationship to age (Fig. 2B), gender, or organ system investigated.
Contrast-enhanced MRI data in patients who underwent two gadobutrol examinations (data ordered by patient age).
Efficacy
The final diagnosis was confirmed by clinical, pathological, or follow-up imaging (ultrasound or MRI) in 57 of the 60 patients.
Enhancement of pathology by gadobutrol was observed in 24 of 60 patients (40%) overall (Table 1). The 24 cases that showed enhancement of pathology had a final diagnosis that is normally associated with enhancement following MR contrast injection. Three of the 36 cases in whom no enhancement was seen post-gadobutrol injection had no final diagnosis at the time of data analysis. Of the remaining 33 patients, the final diagnosis matched the well-established MR imaging findings of no enhancement with contrast agents.
Illustrative cases of gadobutrol-enhanced MRI in a range of pathologies are described in Figures 3–7.
Two-month-old male with left frontal lobe desmoplastic infantile ganglioglioma (Case 7). Pre-contrast transverse T2-weighted 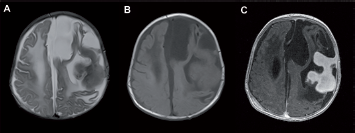
Discussion
This prospective observational study reports the first extensive experience of contrast-enhanced MRI in patients aged less than 2 years using the 1-M contrast agent gadobutrol.
Gadobutrol administered at a dose of 0.1 mL/kg (0.1 mmol/kg) body weight was associated with no adverse events that was considered to be related to contrast medium injection. This experience in children under 2 years is consistent with previous studies of gadobutrol, which described low rates of adverse events in older children and adults.2,35 Based on a prior study of gadobutrol in patients aged 2–17 years with a drug-related adverse event rate of 5.8%,2,35 we would have expected to see 3 to 4 patients with adverse events, whereas we observed none. Low incidences of adverse events have also been reported for other GBCAs in pediatric patients.3,9
The exact time interval required before administration of a second dose of gadobutrol is unknown. Eleven patients required a repeat gadobutrol examination at 3–6 months after the first examination. Hahn et al's
2
study in 138 children aged between 2 and 17 years indicated that 77% of the dose was excreted renally within 6 hours post-injection. Simulated data indicate that the serum gadolinium plasma concentration plateaus and approaches zero by 12 hours post-injection. Our practice is not to inject a second dose of gadolinium into an infant until at least 24 hours has elapsed.
Two-month-old female with a proliferating right parotid hemangioma (Case 6). Coronal STIR T2-weighted 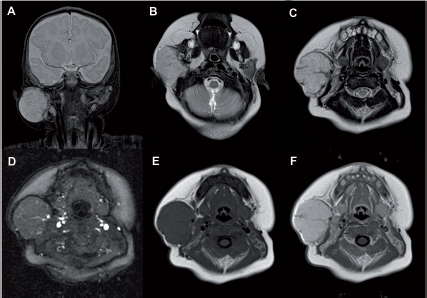
Serum creatinine concentrations indicated no adverse responses to gadobutrol administration. In no cases did the serum creatinine concentration exceed normal reference levels post-contrast injection. Changes in serum creatinine concentrations in patients assessed both pre- and post-MRI were highly variable, which may reflect the contribution of one or more of the numerous factors known to influence creatinine levels in the very young, including age, gender, ethnicity, lean body mass, maternal creatinine levels, hydration status, timing and type of food intake, and the presence of renal or other diseases.36–38 eGFR values, calculated retrospectively from serum creatinine concentrations, similarly showed high intraindividual variability that remained, however, within age-related physiological limits.
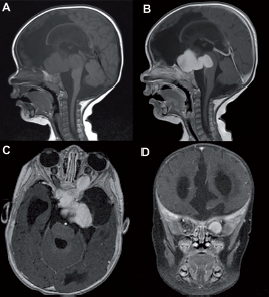
Five-month-old male with an optic pathway glioma (Case 14). Sagittal T1-weighted images pre-
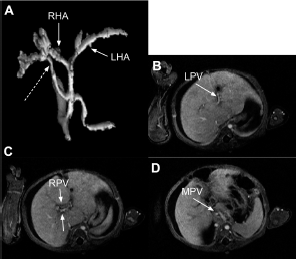
Seven-month-old female previously operated on for a hepatoportoenterostomy and eneteroenterostomy for biliary atresia returns with ascites and no flow in the main, left, and right portal veins on ultrasound (Case 23). She was treated with heparin for 5 days. A maximal intensity projection image from the arterial phase of axial VIBE images post–gadobutrol shows an accessory right hepatic artery originating from the celiac axis (dashed arrow,
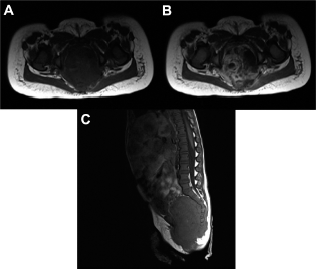
Twenty-month-old female was found to have a pelvic mass on ultrasound (Case 48). A pelvic MRI shows a heterogeneous pre-sacral mass with extension into the spinal canal
In the 57 patients in whom a final diagnosis was obtained, the gadobutrol-enhanced MRI findings matched accepted contrast-enhanced MRI findings for the pathologies investigated. The 24 cases that showed contrast enhancement had a final diagnosis for which contrast enhancement would have been expected, while the 33 cases that showed no enhancement had diagnoses that are consistently associated with a lack of enhancement. 39 These efficacy outcomes are comparable to the results obtained in patients over the age of 2 years, and support the use of gadobutrol-enhanced MRI in children aged less than 2 years in whom a clinical need exists for such imaging studies.
Limitations associated with the current study include the small series of patients recruited from one center, the observational study design, and the lack of serial serum creatinine measurements pre- and post-contrast administration in all patients from which to derive eGFR values. These limitations suggest the need for a controlled, clinical follow-up study of gadobutrol use in this age group.
Conclusion
Contrast-enhanced MRI using the 1-M agent, gadobutrol, demonstrated a favorable safety profile in this study of patients aged less than 2 years, similar to that observed in adults and older children. Standard weight-adjusted dosing of gadobutrol (0.1 mmol/kg body weight) appeared to be appropriate for the under 2-years age group. At this dose, gadobutrol-enhanced MRI demonstrated excellent efficacy in terms of diagnostic accuracy. Assessment of the risk-benefit balance for contrast-enhanced MRI in pediatric patients requires a number of considerations specific to this age group, taking into account developmental changes, pathologies characteristic of this population, and modifications in MR technique related to body size. The current observational study indicates that gadobutrol-enhanced MRI has a favorable safety and efficacy profile in patients less than 2 years of age, based on dosing adjusted to body weight. It is hoped that the experience of gadobutrol-enhanced MRI reported in this observational study may form a basis for further investigation of MRI protocols in very young children.
Funding
The observational study received no funding. Editorial assistance was funded by Bayer HealthCare. Bayer HealthCare is the manufacturer of gadobutrol (Gadovist®/Gadavist™).
Author Contributions
Conceived and designed the experiments: RB, MN. Analyzed the data: RB, MN. Contributed to the writing of the manuscript: RB. Agree with manuscript results and conclusions: RB, MN. Jointly developed the structure and arguments for the paper: RB, MN. Made critical revisions and approved final version: RB, MN. All authors reviewed and approved of the final manuscript.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Footnotes
Acknowledgments
Editorial support was provided by PAREXEL MMS.
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.