Abstract
Cranberry has been shown useful in the prevention of urinary infection by E. coli. In this study, we examined the changes in the hydrophobicity of P fimbriated E. coli and biofilm formation after incubation with commercial cranberry syrup extract at various concentrations. After incubating a bacterial suspension with cranberry at dilutions of 1:100 and 1:1000, tests of haemagglutination inhibition, surface hydrophobicity and biofilm formation were carried out. The surface hydrophobicity of E. coli decreased significantly after incubation with cranberry and this effect was not modified by the culture medium. Biofilm formation was inhibited after incubation with cranberry syrup and this effect was dependent on the culture medium. Thus, in some circumstances cranberry can modify nonspecific adhesive properties of E. coli. In previous studies, cranberry has only been implicated in the inhibition of P-fimbriated E. coli, but our observations show that it acts by modifying adhesive properties under P-related fimbriae.
Introduction
Cranberries contain two chemical constituents that have been associated with the prevention of bacterial adherence: fructose (a monosaccharide) and proanthocyanidins (PACs). Fructose inhibits the adherence of mannose sensitive fimbriae to urinary epithelial cells. 1 Various studies have shown that cranberry PACs inhibit the adherence of P fimbriated E. coli to the urinary epithelium, while having only slight effect on Type 1 fimbriae.1,2
The adherence of E. coli to the epithelia is considered the first step in the development of infection, and so the relation between its pathogenic effect and the tissue's attachment capacity is of great importance for subsequent disease development. 3 Oligosaccharide chains containing mannose are frequently found in cell surface glycoproteins, and thus many bacteria have developed fimbriae to adhere to these molecules (Type 1 fimbriae), which can be blocked when mannose is added. Together with the expression of Type 1 fimbriae, some E. coli strains express a specific lectin (α-Gal(1 →4) β-Gal) associated with P fimbriae, which mediate the specific adherence of E. coli to the urinary epithelial cells. 1 Both Type 1 (mannose-sensitive) and Type P (mannose-resistant) fimbriae of E. coli are regulated by genes that are localised in the bacterial chromosome, and their expression can be modified depending on the characteristics of the culture medium or of the host's internal medium. Some authors 4 have suggested that Vaccinium macrocarpon syrup may induce conformational changes in the surface macromolecules of P fimbriae, reducing both their length and their density. Thus, the expression change of the lectins on the bacterial surface is a determinant factor, on the one hand, in the specific adherence to epithelial cell receptors and, on the other, in modifying certain physical characteristics of the bacteria (hydrophobicity) that favour or hamper its approach to the epithelial surface, and hence its adherence. 5
Mannose-resistant fimbriae share many characteristics that have previously been related to the colonization factor antigen (CFA), and more recently with P fimbriae. Several studies1,6,7 have shown that Vaccinium macrocarpon syrup inhibits the adherence of uropathogenic E. coli to epithelial cells, an effect that has been related with the functionality of P fimbriae (the virulence factor) and, to a lesser degree, with Type 1 fimbriae. 1 Bacterial attachment to a substratum is the first step in biofilm formation. The contribution of type 1 fimbriae and P to biofilm formation has been studied by several authors and seems to depend on the characteristics of the culture medium, the strain and the surface characteristics. 8 Less information is available on the effects of cranberry on the hydrophobic characteristics of E. coli and their ability to form the biofilm. 9
To date, and to the best of our knowledge, no studies have been made assessing the capacity of cranberry to modify the adherent properties of bacteria that are not related to P fimbriae. The fundamental aim of the present study was to assess whether cranberry may affect the “non-specific” adherence properties of E. coli. In addition, we wished to determine whether the inhibition of haemagglutination by cranberry is associated with changes in nonspecific adherence properties vs surface hydrophobicity, adherence to nitrocellulose or biofilm formation by E. coli.
Material and Methods
Bacterial strains, media and growth conditions
Nine strains of uropathogenic E. coli (695, 787, 471, 472, 593, 595, 760, 629 and 607) were used, obtained from patients with acute pyelonephritis, together with 4 strains of E. coli obtained from the Spanish Type Culture Collection (CECT): CECT 424 (F- thr- leulacY mtl- thi- ara gal ton 2 malA xyl. Resistant to phages T1, T2 and T6.); CECT 4076 (Serovar. O157:H7, originally isolated from haemorrhagic colitis); CECT 417 (SupE44(am). mutant tRNA); and CECT 743 (Serovar. O142 K86B:H6. Isolated from children with diarrhoea).
In order to enhance the activity of the Type 1 fimbriae, 10 the strains were grown in TSB culture medium at 37 °C for 48 h, then centrifuged at 2000 g for 10 min. The supernatant was then discarded, and the strains suspended in PBS (pH 7.4). This washing process was performed twice. Finally, the bacterial suspension was adjusted to 109 bacteria/mL (OD of 1.0 to 542 nm). To enhance the activity of the P fimbriae, the strains were incubated for 16 h on CFA agar, 11 extracted from the surface of the agar after washing with 5 ml PBS, and the suspension was then centrifuged at 3000 rpm for 10 min. The supernatant was then discarded, the washing process was repeated, and finally the bacteria were suspended in PBS with 1% mannose to obtain a concentration of 109 bacteria/mL(OD of 1 to 542 nm). All measurements were performed in duplicate and the mean values of the two measurements were used.
Haemagglutination
The blood used was obtained from donors with blood group A Rh+, and collected in tubes with 100 μl of trisodium citrate as anticoagulant, at 3.8%, to which 1 ml of blood was added. For the haemagglutination techniques of the mannose-sensitive strains, the blood was diluted to 1:4 with PBS. For haemagglutination of the mannose-resistant strains, the blood was diluted in PBS with methyl-α-D-mannopyranoside 0.1 mM. Over a U-bottom microwell plate, 50 μl of bacterial suspension and 50 μl of diluted fresh blood (1:4) were added. The haemagglutination results became visible after 45–60 min at room temperature.
Cranberry syrup: procedure
The cranberry syrup was provided by Pharmatoka Lab., (France) using a dispersion of 2.8% cranberry extract Gykacran® in glucose syrup (France) and characterised by the Department of Analytical Chemistry at the University of Granada. 12 Serial dilutions of cranberry syrup were made in PBS (pH 7.4) to 1:10 and 1:100, and the pH was adjusted to 7.0 with NaOH. 100 μl of each dilution of the cranberry extract was incubated for 30 min at room temperature in a rotary shaker with 1 ml of the bacterial suspension (OD 1 to 542 nm), producing a final dilution of the cranberry syrup extract of approximately 1:100 and 1:1000. The final dilution of 1:1000 was chosen because this was the lowest dilution that inhibited haemagglutination. Each suspension was then centrifuged at 3000 rpm for 10 min and resuspended in an equal volume of ammonium sulphate (salt aggregation) or PBS (adherence to nitrocellulose or biofilm formation).
Ammonium sulphate aggregation test
The technique used was as described by Lindahl et al. 13 Briefly, solutions of ammonium sulphate were prepared, with osmolarities ranging from 0.2M to 4M, using sodium phosphate as a dilutant. Taking 20 μl of bacterial suspension, an equal volume of ammonium sulphate solution was added. The presence of aggregation was observed after 30 seconds’ gentle manual rotation, at room temperature, over a glass slide, and the lowest concentration of ammonium sulphate that produced visible aggregation was noted down. Aggregation with the 4M solution was interpreted as a hydrophobicity of 0%, while aggregation with the 0.2M solution was interpreted as 95% hydrophobicity.
Adherence to nitrocellulose filters
The procedure described by Lachica and Zink 14 was performed. Briefly, a bacterial suspension in a PUM buffer with ammonium sulphate 0.2M (1:2, v:v) was adjusted to 105 bacteria (OD 0.6 to 540 nm). The initial absorbance (Abs1) was determined, and 3 ml of this bacterial suspension was passed through an 8 μm-pore nitrocellulose filter (Millipore). The absorbance of the filtrate (Abs2) was then determined and the results expressed as a percentage of the adherence to the nitrocellulose filter.
Biofilm formation 15
The strains were incubated at 37 °C for 24 h in glass tubes with 2.5 ml of TSB or CFA broth. 0.5 ml of the latter and 50 mcL of cranberry syrup at dilutions 1:10 and 1:100 were placed in Eppendorf tubes, resulting in final cranberry syrup dilutions of 1:100 and 1:1000. An Eppendorf tube containing an equal amount of TSB or CFA broth without inoculum was used as a negative control, and 0.5 ml of the bacterial suspension in Eppendorf tubes as a positive control. After incubation for 24 hours, the contents of each tube were aspirated carefully with a Pasteur pipette, washed three times with 250 mcl of PBS, and gently shaken to remove non adherent bacteria. The tubes were then air dried and added to 200 mcL of 99% methanol as a fixative, and left for 15 minutes. The excess methanol was then discarded and the tubes air dried. 200 mcL of 2% Hucker's crystal violet dye was added and the tubes submerged in distilled water for 5 minutes, with the remaining tubes being discarded. This procedure was repeated three times. Subsequently, bacteria were solubilized in each tube, in 250 mcL of 33% acetic acid. The optical density of the suspension of each tube was measured at 540 nm and the zero adjusted to 33% acetic acid. Non-inoculated TSB broth was used as a negative control. The results obtained are expressed as follows: OD ≤ OD control: Non adherent bacteria. OD control ≤ OD ≤ 2 × OD control: slightly adherent bacteria. 2 × OD control ≤ OD ≤ 4 × OD control: moderately adherent bacteria. OD ≤ 4 × OD control: Highly adherent bacteria.
Statistical analysis
SPSS 15.0 statistical software was used to perform the Friedman and Wilcoxon test for the comparison of dependent variables, and the Mann-Whitney-Wilcoxon test for the comparison of independent variables. Kendall's W was used for the comparison of paired variables. In addition, Kendall's coefficient of inter-variable concordance was calculated.
Results
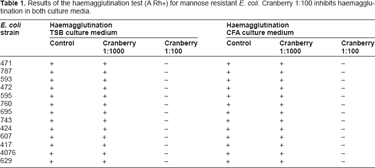
Table 1 shows that the addition of methyl-α-D-mannopyranoside 0.1 mM to blood group A, Rh+ does not inhibit the haemagglutination of any of the 13 strains of E. coli utilised in this study (mannose-resistant strains). The incubation of strains of E. coli with cranberry syrup extract at a final concentration of 1:1000 did not inhibit haemagglutination, while incubation with the cranberry at a final concentration of 1:100 inhibited it, in every case, for strains grown either in TSB or CFA culture medium.
Results of the haemagglutination test (A Rh+) for mannose resistant E. coli. Cranberry 1:100 inhibits haemagglutination in both culture media.
No differences were observed in the surface hydrophobicity of E. coli following its growth in TSB culture medium (Z = 0.35; pNS). However, incubation of the bacterial suspension with cranberry syrup at a final concentration of 1:1000 or 1:100 produced significant reductions in surface hydrophobicity, depending on the concentration of cranberry, after growth in either TSB or CFA medium (Fig. 1).
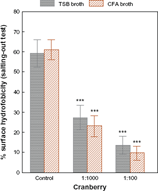
Mean values and standard error of the mean of surface hydrophobicity for E. coli after incubation in CFA and TSB broth. There is a statistically significant decrease in surface hydrophobicity after incubation with cranberry syrup at final dilutions of 1:1000 and 1:100. Note: ***P < 0.001.
The percentage of adherence to nitrocellulose was not significantly modified after growth on CFA agar or in TSB culture medium (Z = 0.73; pNS). In both culture media, the percentage of adherence to nitrocellulose of E. coli, after incubation for 20 min with cranberry syrup, remained unchanged. Kendall's test of concordance revealed the existence of concordance between the surface hydrophobicity values of E. coli and the percentage of adherence to nitrocellulose following growth in TSB culture medium (T = 0.50; P = 0.02), and an absence of such concordance following growth on CFA agar (T = 0.09; P = NS).
After culture in CFA broth, biofilm formation was unchanged by incubation with cranberry syrup at a final concentration of 1:1000 or 1:100 (W = 0.138, P = NS). However, in TSB medium, biofilm formation was inhibited after incubation with cranberry syrup (W = 0.93, P < 0.001), and the formation of biofilm in the absence of cranberry syrup was significantly higher after culture in TSB (Z = 2.31, P = 0.02), as shown in Figure 2.
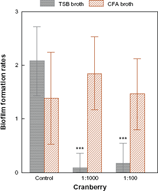
Mean values and standard error of biofilm formation rates for E. coli after incubation in CFA and TSB broth. growth in TSB produces a statistically significant decrease in adherence after incubation with cranberry syrup at final dilutions of 1:1000 and 1:100. Note: *P < 0.05; ***P < 0.001.
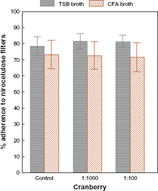
Mean values of adherence to nitrocellulose for E. coli before and after incubation with cranberry syrup at final dilutions of 1:1000 and 1:100.
Discussion
Our study shows that cranberry syrup can modify the surface hydrophobicity and the biofilm formation of E. coli on polystyrene (microtitre plates), the latter effect being dependent on the characteristics of the culture medium. The greater expression of P fimbriae, as a response to the culture medium, was not found to be related with the greater formation of biofilm. The first step in the colonisation of the epithelium by E. coli is determined by its capacity to adhere to the host cells; this capacity is determined, initially, by the micro-organism's electric surface charge and its surface hydrophobicity, 16 and subsequently by other factors, such as the formation of diverse types of fimbriae, and of specific adhesins.17–19 Miyazaki et al 20 showed that fimbriae types 1, P and S are not essential factors in the adhesion and later colonisation of the urogenital epithelium by E. coli. These adhesins may, in general, be considered features of the virulence of extra-intestinal E. coli, but they are not essential for E. coli to become uropathogenic, which accounts for current research interest into the extent to which cranberry extract may affect the non-specific adherence properties of E. coli. Cross-talk among pilus operons within a bacterial cell, probably triggered by environmental cues, can result in a switch in expression from one pilus type to another, a process known as phase variation. The variable expression of the fimbriae in a single strain of E. coli increases the probability of adherence and invasion of the uroepithelium. 21
Cranberries contain different amounts of fructose, PACs, flavonoids and anthocyanins. PACs isolated from cranberry fruit consist of predominantly epicatechin units with at least one A-type linkage. The A-linkage in cranberry PACs may represent an important structural feature for bacterial anti-adherence activity. The type A cranberry PACs prevent adherence of P-fimbriated uropathogenic E. coli to uroepithelial cells in vitro. 4 The cultivation of E. coli in CFA broth favours the expression of P fimbriae. 11 We observed that the incubation of E. coli in CFA broth with cranberry syrup does not alter the formation of biofilm, from which we infer that the increased expression of P fimbriae is not related to the formation of biofilm. The latter finding differs from that observed after culture in TSB broth, implying that biofilm formation can be inhibited by cranberry syrup, depending on the characteristics of the culture medium, while the expression of P fimbriae is not an important element in the formation of biofilm. Hancock et al 8 showed that the expression of P fimbriae significantly decreases biofilm formation under different growth conditions.
The incubation of E. coli with cranberry syrup extract produces a reduction in the surface hydrophobicity, dependent on the concentration of the extract, and which is independent of the different culture media (Fig. 1). This effect may be different from the inhibition of P fimbriae of cranberry PACs, since according to our findings and those of Hancock et al, 8 P fimbriae have little influence on the formation of biofilm. Further studies are currently being conducted, involving a partitioning of the components of the cranberry syrup and a study of the effects thus produced. With respect to surface hydrophobicity, the incubation of E. coli with a 1:1000 dilution of cranberry extract, which does not affect the haemagglutination mediated by Type P fimbriae, does produce significant reductions in the surface hydrophobicity, thus showing that extremely low levels of this extract are capable of modifying the non-specific adherence properties of E. coli. According to our own results, the adherence of E. coli to nitrocellulose is not related to the predominant expression of Type P or Type 1 fimbriae, and incubation with cranberry syrup extract does not affect adherence rates.
We conclude that, depending on the culture medium, cranberry can modify the non-specific adherence properties of E. coli and alter its ability to form biofilm.
Abbreviations
Tryptic Soy Broth;
Colonization Factor Antigen;
Phosphate buffered saline.
Footnotes
Acknowledgements
This study was funded by the Carlos III Institute of Health (Spain).
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
