Abstract
Viability of
Introduction
Bifidobacteria, a predominant genus in the stools of breast-fed infants has come a long way. 1 It is widely used as probiotic dietary adjuncts with health benefiting properties. Bifidobacteria are extensively incorporated into yogurts, cultured milk drinks, cheese or as dietary supplements in the form of dried product. 2
For probiotic to exert health benefits to the host, viability of the strain is of utmost importance.3,4 The recommended minimum count is of 106 live organisms per g of food at the point of consumption.3,4 The conventional technique of using stock cultures with multiple subculturing steps for the preparation of bulk starters are time consuming with higher risk of contamination. With advances in cell mass production technology, the development of concentrated starter cultures in freeze-dried and spray-dried forms for direct product vat inoculation has proved to be a better alternative. 2
Dehydration is a common practice to preserve biological materials so that they are stable in the long run. 5 Among the many cell preservation methods, spray-drying is widely used in the industry because it is economical, especially on a large-scale. 6 Despite the fact that spray-drying is more cost-effective, many microorganisms cannot tolerate the drying process due to the high heat involved. Some of the many factors affecting survival during spray-drying are the strain, growth phase, protective medium used, outlet temperature of spray-drier and pre-adaptation treatment of the culture.7–10 Heat-adaptation, also known as heat-shock treatment, has been reported to increase thermotolerance of bacterial cells during spray-drying.8,11 Thus, the application of heat-adaptation technique prior to spray-drying has gained popularity.
Freeze-drying, the sublimation of ice from frozen preparations is a popular method for the preservation of lactic acid bacteria (LAB). 5 Although freeze-drying is commonly used, microorganisms are also susceptible to various stresses (such as freezing and osmotic stress) and lead to cell injury (such as membrane and cell wall damage. Therefore, cryoprotectants are commonly added to minimize cell damage. Skim milk is a popular drying medium and protectant because it contains protein that prevents cellular injury 12 and facilitates rehydration by creating a porous structure in the freeze-dried powder. 13 Many other compounds have also been tested to improve survival of LAB during freeze-drying such as disaccharides, polyols, vitamins and proteins. 12
This research was conducted to identify a suitable preservation method for
Materials and Methods
Microorganism and preparation of culture
Heat-adaptation of culture prior spray-drying
Harvested cell pellets were resuspended in glass bottles (SCHOTT DURAN®, Mainz, Germany) containing 1000 ml volume of 10% (w/v) sterile skim milk (NZMP medium heat skim milk powder, New Zealand). The samples in glass bottles were transferred to temperature controlled water baths set at 45 °C and 60 °C. The bottles were submerged such that the water level was at the neck of the bottles and were held here for 30 min. Samples which were not subjected to heat adaptation were considered as control.
Spray-Drying at different air outlet temperature
The samples were spray-dried in a laboratory-scale spray-drier (Anhydro, Denmark), with centrifugal atomization. Hot drying air was introduced into the chamber at a constant air inlet temperature of 160 °C. This hot air was mixed continuously with the mist of atomized liquid followed by instantaneous evaporation. The flow rate of the feed solution was regulated to obtain air outlet temperature of 75 ± 2 °C and 85 ± 2 °C. Spray dried powders were collected and were mixed thoroughly with a sterile spatula. The dried powders were stored in tightly sealed sterile glass bottles.
Addition of different protective medium prior freeze-drying
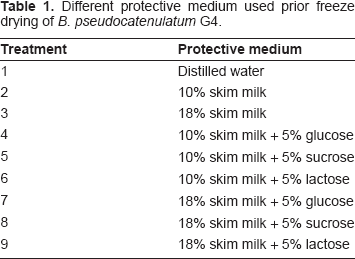
Harvested cells were resuspended in sterile protective medium consisting of sterile skim milk (10% or 18%, w/v) supplemented with different sugar solutions (glucose 5%, lactose 5% or sucrose 5%, w/v). Distilled water was used as control. The protective media were sterilized by autoclaving at 121 °C for 15 min. The treatment combinations are described in Table 1.
Different protective medium used prior freeze drying of
Freeze-drying condition
The cultures in different cryoprotectants were first placed in different sterile metal trays and evenly spread to obtain approximately 1 cm thickness. The cultures were then frozen at -80 °C and subsequently lyophilized in a freeze-drier (Christ® Epsilon 1–80, Germany) at 35 °C for 24 h at a pressure of 1.65 mbar. During the final drying stage, the pressure was dropped to 0.01 mbar to ensure that all the moisture and the gas were completely removed. After freeze-drying, the dried powders were evenly mixed using a sterile spatula and finally stored in tightly sealed sterile glass bottles.
Rehydration and enumeration of B. pseudocatenulatum G4
1 g of dried powder was resuspended in 9 ml skim milk (10%, w/v) to rehydrate the samples. The samples were maintained at room temperature for 10 min as reported earlier by Valdez et al. 17 Serial dilutions of each sample were made using 0.1% (w/v) sterile peptone water and plated on TPY agar in duplicate. This was followed by anaerobic incubation at 37 °C for 48 h. Viable cells were enumerated before drying (initial count) and immediately after the drying process.
Percent survival was calculated according to method of Hyun and Shin:
10
N∘ represents the initial viable count before FD or SD (cfug-1) FD represents freeze-drying SD represents spray-drying.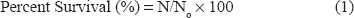
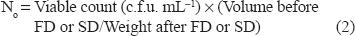
Moisture content analysis
The moisture content of spray-or freeze-dried powders was determined by oven drying at 102 °C. 18 Moisture content was analyzed by determination of the difference in weight before and after drying, expressed as a percentage of the initial powder weight.
Statistical analysis
Statistical analysis was performed using MINITAB version 13 (Minitab Inc., United States). The experiments were repeated twice. Significance differences between values are at P < 0.05 levels using T-test.
Results and Discussion
Spray-drying
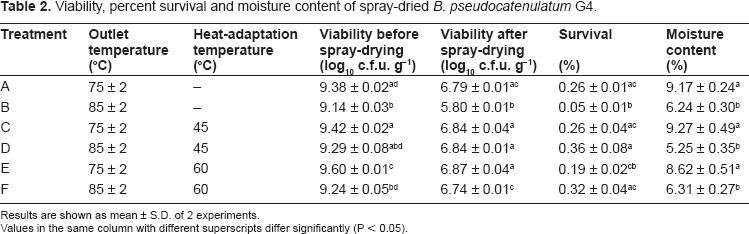
Table 2 shows the viability, percentage survival and moisture content of spray-dried
Viability, percent survival and moisture content of spray-dried
Results are shown as mean ± S.D. of 2 experiments.
Values in the same column with different superscripts differ significantly (P < 0.05).
The survival of organisms during spray drying depends on their heat resistance and the applied temperature. 19 Therefore, spray-drier air outlet temperature is an important factor that affects the viability of the organism. Various range of air outlet temperatures have been used by researchers: 95–105 °C; 20 85–90 °C 21 75–95 °C 6 and 70–100 °C. 22 Since temperature above 90 °C resulted in cell death due to protein denaturation and destruction of DNA, 19 outlet temperature of 75 ± 2 °C and 85 ± 2 °C were used in this study.
At outlet temperature of 75 ± 2 °C, the spray-dried powder had residual moisture content ranging between 8.62%–9.27%. Significantly lower moisture content between 5.25%–6.31% were obtained when it was spray-dried at higher outlet temperature (85 ± 2 °C). This was in accordance with study by Ananta et al where the residual moisture content was inversely proportional to outlet temperature. 23 Since the moisture content of dried culture is a crucial factor in determining the stability and shelf life of the culture, it is important to obtain a proper ratio between the outlet temperature and the residual moisture content. 21
Bacteria constantly face adverse environmental changes and they have to adapt to the new conditions. It has been hypothesized that bacteria respond to these changes through metabolic reprogramming that may subsequently increase their resistance towards environmental stress. 22 Their resistance develops upon exposure to sublethal stress, which induces greater tolerance against the same stress later on. 24 The enhanced thermotolerance of many organisms have been linked to the production of heat shock protein (Hsps) upon exposure to elevated temperature condition. 25 Heat shock proteins act as molecular chaperones in protecting cells against heat damage by binding to cellular proteins in such a way that maintains their original conformation and reduce denaturation. 9 This has led to the intensified research on heat-adaptation to increase survival of bacteria cells during spray-drying.
The use of heat-adaptation technique had been successfully applied on spray-drying of
Freeze-drying
In comparison, freeze-dried cells exhibited higher viability than spray-dried cells regardless of the protective media used (Table 3). The initial viable cell count prior freeze-drying was 109 cfu/g. After freeze-drying, viability remained in the same log unit, meeting the recommended minimum count of 106 live organism per g of probiotic product.3,4 However, for the control treatment, substantial drop in viable count was observed when distilled water was used as the protectant.
Viability, percent survival and moisture content of freeze-dried
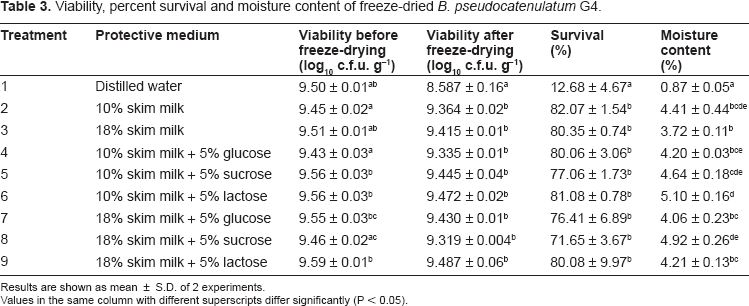
Results are shown as mean ± S.D. of 2 experiments.
Values in the same column with different superscripts differ significantly (P < 0.05).
In terms of percentage survival, high survival rates of 71.65%–82.07% were obtained when different combinations of skim milk and sugar were used as protectant. However, the values were not significantly different from one another. Therefore, the addition of sugar did not contribute to higher cellular protection against freeze-drying. Hence, we recommend the use of 10% skim milk as the cryoprotectant for this strain. Generally, the freeze-drying performance of this strain was considered satisfactory as high percentage survival was obtained after the drying-process. In comparison, lower percent survival of freeze-dried bifidobacteria (43.1%–51.9%) had been reported by Wang et al in fermented soymilk.
26
In another study on
In terms of moisture content, freeze-dried powder had lower percentage values ranging between 3.72%–5.10% (Table 3). Although microorganisms generally show higher survival rate under low moisture condition, excessive drying may be harmful to the survival of organisms. Extreme drying causes removal of three types of cellular water; free, intermediate and structured water, which risks damage to cellular proteins.
27
A minimum amount of water must remain in the dried cultures for a higher rate of survival, which in turn depends on the drying medium and freeze-drying condition. Zayed and Roos
28
reported that the optimum moisture content for freeze-dried
It is recognized that organisms preserved by drying techniques undergo various cellular stresses that may lead to cell injury or even cell death. The rate of survival is strain-specific and depends on the drying method used. Our finding was in accordance with study by Wang et al in which they reported that
The addition of sugars as cryoprotectant for the inhibition of free radical production is highly recommended by many researchers.31,32 This is because sugars are food-grade, easily available and cost effective. Sugars increase the stability of cellular protein by forming hydrogen bonds with them, thus reducing the risk of exposure to the environment.
12
They should be present on both sides of the membrane to be able to maintain the cell protein structure in the dry state.
33
Meanwhile, Carvalho et al demonstrated that the cryoprotective effect of sugars in the drying medium depends on the sugar previously found in the growth medium.
34
However in this case, no significant differences in percent survival were observed when different combinations of skim milk and sugars were used. This may be due to the presence of naturally occurring protein and lactose in milk, which offer the necessary protection to this strain. Therefore, the inclusion of additional sugars in skim milk did not result in increased survival during freeze-drying. King and Su
35
also reported a similar finding, where no significant difference in the protective effect of 10% nonfat dry milk solids added with sucrose and other compounds on freeze-dried
Conclusions
In order to be successfully incorporated into functional food applications, the culture should be able to withstand the harsh conditions during cell preservation. This study demonstrated that
Publish with Libertas Academica and every scientist working in your field can read your article
Available to your entire community free of charge
Fairly and quickly peer reviewed
Yours! You retain copyright
Footnotes
Acknowledgement
This project was supported by the IRPA Grant from the Ministry of Science, Technology and Environment, Malaysia.
The authors report no conflicts of interest.
