Abstract
Efficient delivery of genetic material to cells is needed for tasks of utmost importance in laboratory and clinic, such as gene transfection and gene silencing. Synthetic cationic lipids can be used as delivery vehicles for nucleic acids and are now considered the most promising non-viral gene carriers. They form complexes (lipoplexes) with the polyanionic nucleic acids. A critical obstacle for clinical application of the lipid-mediated DNA delivery (lipofection) is its unsatisfactory efficiency for many cell types. Understanding the mechanism of lipid-mediated DNA delivery is essential for their successful application, as well as for rational design and synthesis of novel cationic lipoid compounds for enhanced gene delivery. According to the current understanding, the critical factor in lipid-mediated transfection is the structural evolution of lipoplexes within the cell, upon interacting and mixing with cellular lipids. In particular, recent studies with cationic phosphatidylcholine derivatives showed that the phase evolution of lipoplex lipids upon interaction and mixing with membrane lipids appears to be decisive for transfection success: specifically, lamellar lipoplex formulations, which were readily susceptible to undergoing lamellar-nonlamellar (precisely lamellar-cubic) phase transition upon mixing with cellular lipids, were found rather consistently associated with superior transfection potency, presumably as a result of facilitated DNA release subsequent to lipoplex fusion with the cellular membranes. Further, hydrophobic moiety of the cationic phospholipids was found able to strongly modulate liposomal gene delivery into primary human umbilical artery endothelial cells; superior activity was found for cationic phosphatidylcholine derivatives with two 14-carbon atom monounsaturated hydrocarbon chains, able to induce formation of cubic phase in membranes. Thus, understanding the lipoplex structure and the phase changes upon interacting with membrane lipids is important for the rational design and successful application of cationic lipids as superior nucleotide delivery agents.
Introduction
Recent advances in the genetic understanding of various cellular processes and disease pathogenesis associated with the culmination of the Human Genome Project (Venter et al. 2001) made possible the identification and therapeutic targeting of numerous genes involved in diseases. The diseases that have been approached with gene therapy are widely diversified, including autosomal recessive single gene disorders (cystic fibrosis, hemophilia, severe combined immune deficiency due to adenosine deaminase), autosomal dominant disorders, many forms of cancer, HIV, and other infectious diseases, inflammatory conditions and intractable pain (Flotte, 2007). Important therapeutic procedures, such as gene transfection and gene silencing, require efficient delivery of genetic material to cells. Gene delivery systems include viral and non-viral vectors. Viral vectors are the most effective (Hendrie and Russell, 2005; Giacca, 2007), but their application is limited by their immunogenicity and oncogenicity. Synthetic cationic lipoids, which form complexes (lipoplexes) with polyanionic DNA, are presently the most widely used non-viral gene carriers (Feigner and Ringold, 1989). Common problem for the clinical application of cationic lipids as gene carriers is their toxicity–-it has been reported that some of them can induce irreversible damages to cultured cells and can be responsible for fatality in animal experiments (Dass, 2004). Thus, efforts are made to design and synthesize biocompatible cationic lipids of lower toxicity such as phospholipids (Solodin et al. 1996; MacDonald et al. 1999a,b). Other approach to address the toxicity issue include, e.g. taking advantage of non cationic phospholipids in association with multivalent cations to achieve the complexation of DNA (Bruni et al. 2006; Tresset et al. 2007).
Apart from toxicity, a critical obstacle for clinical application of lipid-mediated delivery (lipofection) of gene-based drugs is its unsatisfactory efficiency for many cell types. Progress in enhancing lipofection efficacy has been impeded because its mechanism is still largely unknown (Song et al. 1997; Jaaskelainen et al. 1998; Subramanian et al. 2000; Ghosh et al. 2002; Gaucheron et al. 2002; Niculescu-Duvaz et al. 2003; Lobo et al. 2003). Understanding the mechanism of lipid-mediated DNA delivery is essential for its successful application, as well as for rational design and synthesis of novel cationic lipoid compounds for enhanced gene delivery.
Polar lipids are known for their ability to form an impressive variety of polymorphic and mesomorphic phases–-lamellar and nonlamellar–-when dispersed in aqueous media (Fig. 1.I.) (Luzzati and Tardieu, 1974; Seddon and Templer, 1995; Koynova and Tenchov, 2008). Phase state has been recognized as potentially important for the transfection activity of lipoplexes (Koltover et al. 1998; Smisterova et al. 2001; Simberg et al. 2001; Zuhorn and Hoekstra, 2002). Typically, lipoplexes are arranged as multilayer structures in which DNA is intercalated between the lipid bilayers (Fig. 1.II.) (Radler et al. 1997; Boukhnikachvili et al. 1997; Lasic et al. 1997; MacDonald et al. 1999a). Some earlier studies suggested that the inverted hexagonal phase leads to more efficient transfection efficiency than does the lamellar phase (Koltover et al. 1998; Smisterova et al. 2001). Recent experiments dispute this suggestion, however, and there is now considerable experimental evidence invalidating direct general correlation between lipoplex structure and transfection activity (Ross et al. 1998; Rakhmanova et al. 2000; Lin et al. 2003; Simberg et al. 2003; Caracciolo et al. 2003; Congiu et al. 2004; Caracciolo and Caminit, 2005; Wang et al. 2006; Koynova et al. 2008). Furthermore, a viewpoint now emerging is that the critical factor in lipid-mediated gene delivery is the structural evolution of lipoplexes upon interacting and mixing with cellular lipids (Tarahovsky et al. 2004; Zuhorn et al. 2005; Koynova et al. 2005).
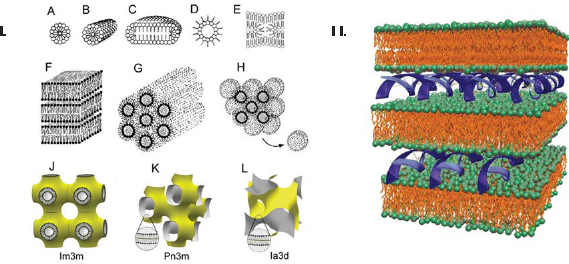
The unbinding of DNA from cationic lipid carrier when lipoplex gets inside the cell has been identified as one of the key steps in lipofection. According to the current understanding, the unbinding is a result of charge neutralization by cellular anionic lipids; indeed, experiments have revealed that addition of negatively charged liposomes to lipoplexes results in dissociation of DNA from the lipid (Zelphati and Szoka, 1996; Xu and Szoka, 1996; Ashley et al. 1996; MacDonald et al. 1999a; Tarahovsky et al. 2004; Zuhorn et al. 2007). A set of significant recent findings suggest that the structure of cationic lipid carriers changes dramatically upon interaction with cellular lipids, and furthermore that such changes may critically affect the delivery efficiency. Recent studies showed that the phase evolution of lipoplex lipids upon interaction with membrane lipids appears to be decisive for transfection success (Koynova et al. 2005, 2006; Wang et al. 2006). Noteworthy is that such a concept can, in principle, also account for the considerable differences in the transfection potency of lipoplexes with different cells.
Thus, studies on the structure of lipoplexes and the lipid phase changes upon interacting with membrane lipids, in search for correlations with the lipoplex transfection efficiency, are reviewed herein.
At present, a growing list of synthetic lipid transfection reagents is commercially available. Each one has its own success on specific cell line. The present review is mainly focused on a particularly attractive cationic lipid class, O-substituted phosphatidylcholine derivatives, in which the phosphate oxygen of phosphocholine is substituted (generating a triester), thus converting the head group zwitterion into a cation (Solodin et al. 1996; MacDonaldet al. 1999b). These lipids are especially attractive because they are slowly metabolized and have remarkably low toxicities (MacDonald et al. 1999a,b). Representatives of this cationic phospholipid class have been found to exhibit high transfection activity, both in vitro and in vivo, in antitumor and anti-cystic fibrosis pharmaceuticals (Gorman et al. 1997; McDonald et al. 1998; Noone et al. 2000; Das and Niven, 2001; Faneca et al. 2004, 2007, 2008).
Exemplary structures of the cationic phosphatidylcholine derivatives used are shown in Figure 2. Triflate salts were synthesized as described (MacDonald et al. 1999b), or were purchased as the chloride salt from Avanti Polar Lipids (Alabaster, AL):
Structures of exemplary cationic phosphatidylcholine derivatives.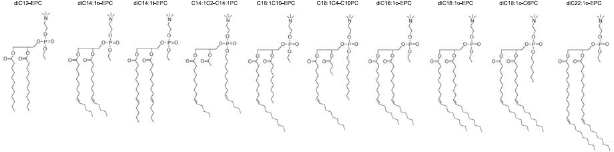
1,2-dioleoyl-sn-glycero-3-ethylphosphocholine (diC18:1-EPC; EDOPC), 1,2-dilauroyl-sn-glycero-3-ethylphosphocholine (diC12-EPC; EDLPC), 1,2-dimyristoyl-sn-glycero-3-ethylphosphocholine (diC14-EPC; EDMPC), 1,2-dipalmitoyl-sn-glycero-3-ethylphosphocholine (diC14-EPC; EDPPC), 2-distearoyl-sn-glycero-3-ethylphosphocholine (diC18-EPC; EDSPC), 1,2-diphytanoyl-sn-glycero-3-ethylphosphocholine (diC16:4me-EPC; EDPhyPC), 1-oleoyl-2-decanoyl-sn-glycero-3-ethylphosphocholine (C18:1/C10-EPC), 1-stearoyl-2-decanoyl-sn-glycero-3-ethylphosphocholine (C18:0/C10-EPC), and 1,2-dierucoyl-sn-glycero-3-ethylphosphocholine (diC22:1-EPC)–-this class of cationic phospholipids has been extensively characterized and found to form lamellar lipoplexes, which exhibit from high to superior transfection activity (MacDonald et al. 1999a, b; Koynova et al. 2006; Tenchov et al. 2008).
Other O-substituted phosphatidylcholine derivatives
1,2-dioleoyl-sn-glycero-3-stearylphosphocholine (diC18:l-C18PC), 1,2-diphytanoyl-sn-glycero-3-palmitylphosphocholine (diC16:4me-C16PC)–-these effectively triple-chain lipids exhibit strong hexagonal-phase preferences (Wang et al. 2006); 1,2-dioleoyl-sn-glycero-3-hexylphosphocholine (diC18:1-C6PC), 1,2-didecanoyl-sn-glycero-3-octylphosphocholine (diC10-C8PC), 1,2-didecanoyl-sn-glycero-3-myristyl-phosphocholine (diC10-C14PC) form bilayer cubic phases at physiological temperatures (Wang et al. 2006; Koynova et al. 2008); diC22:1-EPC and diC10-C14PC exhibit large temperature hysteresis and could form either lamellar or cubic phases at room or physiological temperature, depending on the thermal prehistory (Wang et al. 2006; Koynova et al. 2008). These compounds thus provide a remarkable opportunity to, with minimal ambiguity, examine the effect of the initial lipid phase on lipoplex properties.
The recent approach of fine tuning lipoplex properties by using mixtures of cationic lipids (Wang and MacDonald 2004, 2007; Wang et al. 2006; Koynova et al. 2007a,b) provided the opportunity to highly improve the transfection activity by properly formulating mixtures of cationic lipids. Thus, for example, for the diC 12-EPC/diC18:1-EPC (EDLPC/EDOPC, 60:40) mixture this procedure has been found to provide up to 30x increase in transfection (Wang and MacDonald, 2004) (see below).
Model Membrane Lipids
Interactions of lipoplexes with cellular membranes have been approached starting from simple lipid systems, with progression to complex mixtures modeling membrane lipid compositions and native membrane extracts, as follows:
Anionic membrane lipids
Synthetic: dioleoyl- derivatives of phosphatidylglycerol (DOPG), phosphatidylserine (DOPS), phosphatidic acid (DOPA); tetraoleoylcardiolipin (TOCL); From natural sources: brain phosphatidylserine (PS), bovine liver phosphatidylinositol (PI), heart cardiolipin (CL).
Zwitterionic membrane lpids
Synthetic: dioleoylphosphatidylethanolamine (DOPE), cholesterol, lysophosphatidylcholine; recent data suggest that negatively charged lipids are not a sine qua non for DNA release from lipoplexes (Gordon et al. 2005); in a sense, this is consistent with the concept about the paramount importance of phase properties/intrinsic curvature/bilayer frustration–-thus the potency of zwitterionic lipids for releasing DNA needs to be tested.
Membrane models
Mixtures of anionic/zwitterionic lipid (phosphatidylcholine): anionic lipids listed above have been mixed with dioleoylphosphatidylcholine (DOPC) at, e.g. 8:2 ratio–-the approximate common zwitterionic/anionic lipid ratio of plasma and nuclear membranes; endosomes are reported to have composition similar to that of the plasma membrane (White DA, 1973; Gennis RB, 1989). Cholesterol is known as strong modulator of membrane biophysical properties; 20% cholesterol added to the above mixtures mimics its content in nuclear and plasma membranes (Koynova and MacDonald, 2007). Phosphatidylethanolamine is a major non-lamellar forming membrane lipid; it is thus important to assess the effect of cationic lipid carriers on its phase behavior (Tenchov et al. 2008). Membrane mimicking compostion (MM = DOPC: DOPE:DOPS:Chol, 45:20:20:15, w/w) has been found to be a very efficient DNA releaser (Koynova et al. 2006; Koynova and MacDonald, 2007).
Polar and total lipid extracts from bovine liver, brain, and heart are commercially available from Avanti Polar Lipids (Alabaster, AL) and are useful in assessing the lipoplex-membrane interactions (Koynova and MacDonald, 2007).
Lipoplex Structures
Lamellar lipoplexes, lipid and DNA spacings
Small angle X-ray diffraction (SAXD) patterns have revealed that the lipoplexes formed by the majority of the cationic phospholipids are arranged in lamellar arrays, as shown by the sets of sharp reflections in the diffraction patterns (Fig. 3). Noteworthy, even cationic lipids forming nonlamellar structures per se, often form lamellar lipoplexes (Rakhmanova et al. 2000; Koynova et al. 2008). The lamellar repeat period of the lipoplexes is typically ~1.5 nm higher than that of the pure lipid bilayers, as a result of the DNA intercalation between the lipid layers. The presence of the DNA strands between the phospholipid bilayers, similarly to other cationic lipids (Radler et al. 1997; Boukhnikachvili et al. 1997; Lasic et al. 1997) has been verified by the electron density profiles of the lipoplexes (MacDonald et al. 1999a).
Smal-angle X-ray diffraction profiles of EDOPC and EDLPC/EDOPC (60:40) lipoplexes, at 4:1 lipid/DNA weight ratio (arrow points to the peaks originating from the DNA–DNA in-plane correlation); diffraction data were collected for 1 sec at 37 °C; Inset: Thin-section electron microscopy of (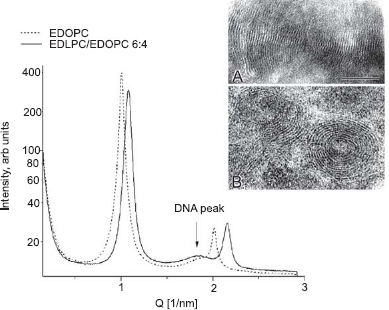
In addition to the sharp lamellar reflections, a low-intensity diffuse peak was also present in the lipoplex diffraction patterns. Such a peak has been interpreted as reflecting the in-plane packing of the DNA strands intercalated between the lipid lamellae (Salditt et al. 1997; Radler et al. 1997; Lasic et al. 1997; Koynova and MacDonald, 2003a). Its position is dependent on the lipid-DNA stoichiometry.
DNA arranges into rectangular columnar superlattice between lipid bilayers in the low-temperature gel phase of the lipoplexes of the saturated cationic lipids (Artzner et al. 1998; Koynova and MacDonald, 2004). This is evidenced by two or three diffuse reflections in addition to the sharp lamellar reflections; these are attributed to the DNA ordering both within the smectic layers between the lipid bilayers, as well as across bilayers, from one DNA layer to another (Fig. 4A) (Koynova and MacDonald, 2004). The positions of these reflections in the lipoplexes of the cationic O-ethylphosphatidylcholines index to a centered rectangular columnar lattice, S
hk
= √[(h/a)2+ (k/b)2], with lattice constants a and b; the constant b is determined by the lamellar spacing d (b = 2d). The DNA scattering peaks index as (1,1) and (1,3), respectively (and in some cases (1,5)).
(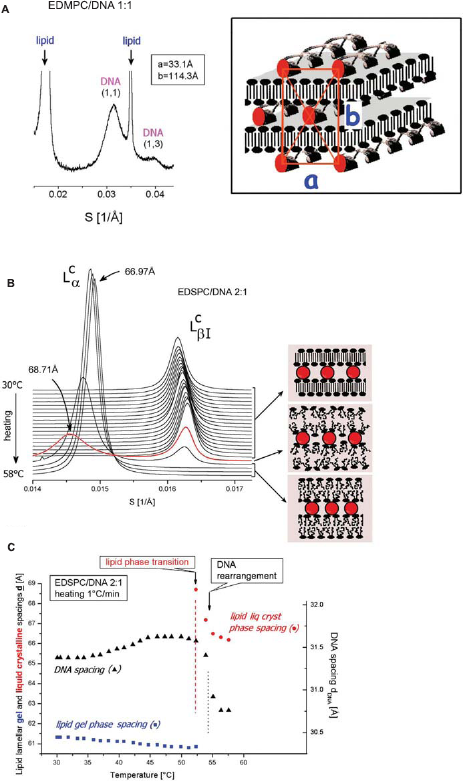
Aqueous dispersions of the saturated-chain representatives of the O-ethylphosphatidylcholines exhibit gel–-liquid crystalline phase transitions at temperatures close to those of their parent phosphatidylcholines; addition of DNA does not change significantly the lipid phase transition parameters (MacDonald et al. 1999a; Koynova and MacDonald, 2003a). Thus, upon heating, the lipid layers of the lipoplexes undergo a phase transition from the interdigitated gel phase to a liquid crystalline lamellar Lcα phase, at temperatures close to those of the pure lipid dispersions: 23 °C, 41 °C, and 52 °C, respectively, for the EDMPC, EDPPC, and EDSPC complexes. The transition temperatures are independent of the amount of DNA This transition is associated with an expansion of the lamellar repeat distance d by 4–6 å (Fig. 4B, C). The lipid phase transformation was immediately followed by a rearrangement of the DNA lattice, specifically, a decrease of the DNA in-plane distance by ~1 å and a loss of the interlamellar correlation, the latter revealed by the disappearance of the multiple DNA reflections from the SAXD patterns. Thus, at temperatures above the lipid phase transition, a single broad DNA reflection has been detected, attributed to the in-plane, strand-to-strand positional correlation of the DNA strands.
Hexagonal phase lipoplexes
Certain cationic phospholipids such as C18-DOPC, C16-DiPhyPC, C6-DOPC (effectively triple-chained) form lipoplexes substantially different from the lamellar lipid/DNA sandwiches described above. Their structure consists of DNA coated by cationic lipid monolayers and arranged on a two-dimensional hexagonal lattice. This arrangement is identified by their small-angle X-day diffraction pattern, with diffraction maxima of reciprocal spacings fitting the ratio 1:√3:√4 (Fig. 5). The lower intensity of the diffraction peaks from the 11 and 20 planes in the presence of DNA relative to the patterns without DNA (Fig. 5A) is a likely result of the higher electron density of DNA relative to water (Francescangeli et al. 2004). It is thus an indication of the presence of DNA in the core of the hexagonal phase cylinders, i.e. for the formation of columnar inverted hexagonal phase lipoplexes (as shown in the cartoon on Fig. 5). Remarkably, hexagonal phase lipoplexes formed by cationic phospholipids consistently exhibited lower transfection activity that the lamellar ones (Wang et al. 2006).
SAXD patterns of (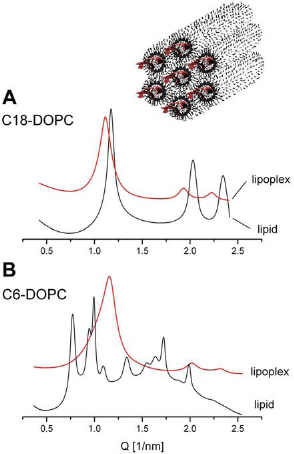
In certain lipoplexes made of cationic lipid mixtures, coexistence of lamellar and hexagonal phase is observed. Such are some C18-DOPC/EDOPC and C16-DiPhyPC/EDOPC mixtures (Fig. 6). Noteworthy, these are compositions consistently corresponding to a minimum in the transfection efficiency vs. composition curves (Wang et al. 2006). Although the diffraction patterns of these mixtures clearly show the presence of two phases, structural ambiguities remain since it is hard to specify the topological modes of phase coexistence. For example, Lα and HII phases might coexist either in separate aggregates or in the same aggregate, and the distinction might be critical for DNA unbinding and release. Supplemental information on the lipoplex organization could be hence sought through size measurement; it is specific for the HII phases to be highly aggregated and hence their dispersions tend to have large particles. Really, the pure C18-DOPC lipoplexes, which are HII structures (see above), were significantly larger than those of the lamellar phase EDOPC lipoplexes; in contrast, the C18-DOPC/EDOPC lipoplexes with intermediate lipid compositions were similar in size to those of EDOPC (Wang et al. 2006). These observations, as well as the fact that the coexisting Lα and HII phases are virtually epitaxially matched (ahex= 2dhex/√3 ≍ dlam; Fig. 6) has been interpreted as suggesting that the mixed-phase lipoplexes may have a topology like that depicted in Figure 6. In addition to the topological relationship between the two phases, another question that arises with respect to samples having two coexisting phases is whether both phases contain DNA Analysis of the structural parameters shows that the presence of DNA in both phases seems likely (Wang et al. 2006).
Coexisting lamellar and inverted hexagonal phases in lipoplexes of EDOPC/ diC18:1-C18PC 60:40 at 37 °C as revealed by their SAXD patterns (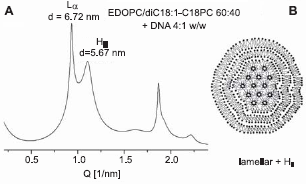
Another example of lipoplexes exhibiting phase coexistence are mixed lipoplexes undergoing a solid-liquid crystalline (melting) transition. Remarkably, such lipoplexes have been found to demonstrate higher transfection activity than the single-phase ones (see below).
The cationic diC18:1-C6PC, diC10-C14PC, and diC22:1-EPC have found to form cubic phases–-either in the entire temperature range from -10 °C to 90 °C (diC18:l-C6PC) (Koynova et al. 2008), or to exhibit an irreversible lamellar-cubic transition on heating, between 50–70 °C for diC22:1-EPC (Koynova et al. 2008) and at 25–30 °C for diC10-C14PC (Fig. 7). The lipoplexes formed by C6-DOPC arrange into hexagonal phase, while the lipoplexes of di22:1-EPC and diC10-C14PC are lamellar (Koynova et al. 2008) All three lipids exhibit lower transfection activity than the lamellar-forming 1,2-dioleoyl-sn-glycero-3-ethylphosphocholine (EDOPC).
SAXD patterns recorded upon heating and cooling scans of diC10-C14PC dispersions: the heating phase sequence is Lβ $Ar Lα $Ar Qla3d $Ar QPn3m; the cooling sequence is QPn3m $Ar Lβ.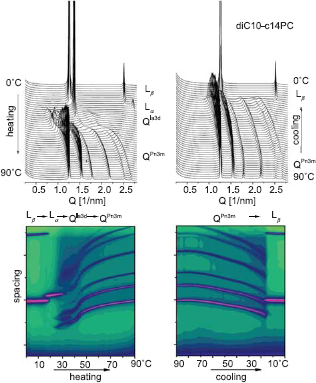
According to current understanding, the basic steps of lipid-assisted transfection include adsorption of lipoplexes to the cell surface, endocytosis, release of DNA and then transport of the released DNA to the nucleus for transcription (Legendre and Szoka, 1992; Zhou and Huang, 1994; Zabner et al. 1995). The release of DNA from the cationic lipid carrier when the lipoplex gets inside the cell has been identified as one of the key steps in lipofection. Unbinding of DNA from the cationic lipid is believed to be a result of charge neutralization of the latter by cellular anionic lipids. Cationic lipid-DNA interactions are strong (Pozharski and MacDonald, 2003) and the only apparent possibility for release of DNA under cellular conditions (which preclude high temperatures and high concentrations of competing counterions) is by neutralization of the cationic lipid charge with cellular anionic lipids. Indeed, experiments have revealed that addition of negatively charged liposomes to lipoplexes results in dissociation of DNA from the lipid (Zelphati and Szoka, 1996; Xu and Szoka, 1996; Ashley et al. 1996; MacDonald et al. 1999a; Tarahovsky et al. 2004). Thus, intermixing of membrane lipids with lipoplex lipids is presumed to be a necessary step in transfection.
Lipoplexes with mean diameters of about 400–500 nm and lamellar repeat distances of ~5–6 nm, as typically observed with the studied systems, would contain some ~20 membrane bilayers. It is thus obvious that lipid exchange between a multilamellar lipoplex and surrounding single bilayer of endosomal membrane is certainly not sufficient to neutralize the cationic charge necessary for DNA unbinding and release from lipoplexes, and much more extensive intermembrane interactions are required for more complete DNA release. Numerous contacts visualized by electron microscopy between lipoplexes and various cellular membranes (Koynova et al. 2007a) may therefore support a concept of gradual lipoplex peeling and DNA release. Moreover, recent data clearly indicate that some step after membrane fusion is rate-limiting for transfection (Wang et al. 2006).
The process of lipid intermixing is most often discussed in terms of membrane fusion. However, in principle, monomer lipid exchange between aggregates could produce generally similar intermixing. The presumptive higher CMC of the charged lipids may significantly accelerate the lipid molecular transfer. The higher concentration of monomers was indeed found to significantly facilitate molecular transfer between aggregates, even when the aggregates are all positively charged (Koynova and MacDonald, 2005).
Given the need for intermixing of membrane lipids with lipoplex lipids, a set of recent findings, discussed below, takes on particular significance. These findings suggest that the structure of cationic lipid carriers might change dramatically upon mixing with cellular lipids, and furthermore, that such changes may critically affect DNA delivery efficiency.
Structural Evolution of the Lipid/ DNA Complexes upon Interaction with Cellular Lipids–-the Phase Evolution Concept
Cationic phospholipid/membrane lipid mixtures form nonlamellar phases
It has been reported that mixtures of cationic lipids with anionic lipids of the type found in cell membranes are unusually prone to form nonlamellar phases, even when the pure components form only lamellar phase: even small amounts of anionic lipid added to certain cationic lipids generate virtually the entire panoply of possible lipid arrays (Tarahovsky et al. 2000; Lewis and McElhaney, 2000; Koynova and MacDonald, 2003b). For example, a regular progression of polymorphic phase behavior was observed for mixtures of the anionic phospholipid, cardiolipin, and the cationic EDOPC. Whereas the two lipids separately assume only lamellar phases, their mixtures exhibit a whole sequence of nonlamellar phases. The inverted hexagonal phase HII forms at net charge neutrality. When one type of lipid is in significant excess, a bicontinuous cubic structure is observed. Increasing the excess of cationic or anionic charge leads to the appearance of membrane bilayers with numerous interlamellar contacts, i.e. sponge structures (Tarahovsky et al. 2000).
The finding that cationic lipid/membrane lipid mixtures are prone to form nonlamellar phases is of significant importance for understanding the relationship between lipid phases and lipid-mediated gene delivery (lipofection). Since escape of DNA from the lipoplexes must involve neutralization of cationic lipid by cellular anionic lipids, most likely by fusion of cell membranes with lipoplexes, a wide variety of nonlamellar arrays can potentially appear in treated cells. Indeed, phase diagrams of mixtures of the cationic ethylphosphatidylcholine with representatives of membrane lipid classes (phosphatidylcholine, phosphatidylethanolamine, phosphatidylglycerol, cholesterol) revealed a large variety of polymorphic and mesomorphic structures (Koynova and MacDonald, 2003b). Marked enhancement of the tendency to form nonlamellar phases has been observed in mixtures with phosphatidylethanolamine (Fig. 8A) and cholesterol, as well as with the anionic phosphatidylglycerol (Fig. 8B). Because of the potential relevance to transfection, it is noteworthy that such phases form at close to physiological conditions (temperature, anionic lipid content) and in the presence of DNA. Since mixing of cationic and membrane lipids must occur during transfection, a large assortment of non-lamellar arrays can potentially be generated in treated cells. It now seems quite likely that lipid interactions giving rise to different phase preferences when cationic lipids mix with membrane lipids could well explain the otherwise baffling differences in transfection by different agents of different cell lines.
Phase changes in the cationic EDPPC upon admixing phosphatidylethanolamine, PE (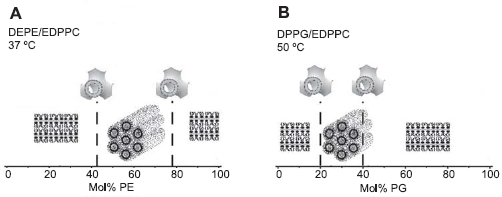
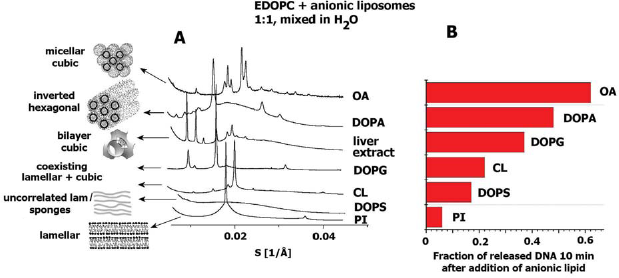
As already discussed, the only possibility for release of DNA under cellular conditions seems to be by neutralization of the cationic lipid charge with cellular anionic lipids (Zelphati and Szoka, 1996; Xu and Szoka, 1996). The rate of DNA-cationic lipid separation has been found to depend on the lipid composition of the anionic liposomes (Tarahovsky et al. 2004). X-ray diffraction experiments have revealed correlation between the releasing capacities of the anionic lipids and the mesomorphic structures they form when mixed with the cationic phospholipid EDOPC (Tarahovsky et al. 2004). Anionic lipids that were more efficient in releasing DNA formed nonlamellar phases of high negative curvature; conversely, the anionic lipids for which only inefficient release of DNA was observed, formed mostly lamellar phases (Fig. 9).
(
Further, correlation between delivery efficiency of the cationic lipid DNA carriers and the mesomorphic phases they form when interacting with negatively charged membrane lipids has been established (Koynova et al. 2005). Specifically, formulations that are particularly effective DNA carriers, form phases of highest negative interfacial curvature when mixed with anionic lipids, whereas less effective formulations form phases of lower curvature (see section 7.1 below). Thus, transfection outcome appears correlating with the propensity of the cationic/anionic lipid mixture to evolve into highly curved mesomorphic structure, when they interact The propensity of lipid bilayers to form nonlamellar phases has been described by the concept of bilayer “frustration” brought about by the imbalance of forces in the bilayer, and which imposes a nonzero intrinsic curvature of the two opposing monolayers (Gruner, 1985; Anderson et al. 1988; Seddon and Templer, 1995). In terms of this concept those cationic lipid formulations which tend to easily transform into curved nonlamellar arrays when mixed with anionic lipid, presumably retain higher degree of “frustration”. The stored curvature elastic energy in a “frustrated” bilayer seems to be comparable to the cationic lipid-DNA binding energy: the same magnitude of ~1–2kBT has been estimated for both the binding free energy of lipoplexes (Bruinsma, 1998; Pozharski and MacDonald, 2002, 2003), and the curvature elastic energy stored in a flat phosphatidylethanolamine-dominated membrane (Attard et al. 2000; Botelho et al. 2002). The release of stored curvature elastic energy upon lamellar-nonlamellar phase transition, and the balance between these energy terms could therefore play a significant role in lipoplex-membrane interactions and DNA unbinding energetics. This result corroborates with the hypothesis that structural evolution of lipoplexes upon interaction with cellular lipids is the controlling factor in lipid-mediated DNA delivery. Here we recall that, for effective delivery, DNA must be retained until the lipoplex is inside the cell–-thus initial lamellar lipoplex structures are likely responsible for the successful import of DNA into cells.
Thus, a strategy for optimizing lipofection can be deduced: lamellar lipoplex formulations with compositions close to the lamellar-nonlamellar phase boundary, which could easily undergo phase transition upon mixing with cellular anionic lipids, could be expected to be especially efficient because they would be equally successful in transporting DNA into cells (since lamellar phase optimally protects DNA (Tarahovsky et al. 2004)), and in releasing it subsequently.
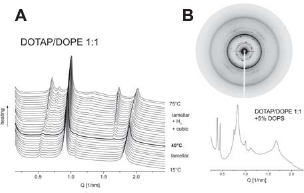
Indeed, X-ray diffraction experiments have provided support for this “efficiency formula”: one of the effective and widely explored lipid transfection formulations–-DOTAP/DOPE 1:1 (mol/mol)–-exhibits precisely this kind of behavior: at room temperature it forms lamellar phase, whereas upon heating, lamellar$Arinverted hexagonal phase transition begins just above physiological temperature, at ~39–40 °C (Fig. 10A). Addition of a minor proportion (≤5%) of the anionic lipid phosphatidylserine was enough to convert the lipoplex structure into a predominantly cubic bicontinuous Pn3m phase at physiological temperature (Fig. 10B). Thus, after uptake into the cell, these lipoplexes would very easily be converted into nonlamellar arrays that allow the DNA release, which is the likely reason for their effectiveness.
(
Electron and confocal fluorescence microscopy have revealed that after the internalization and accumulation in perinuclear region, lipoplexes of lower transfection potency remained in compact perinuclear endosomes, while the lipoplexes of superior activity (such as, e.g. the EDLPC/EDOPC 60:40 formulation (Wang and MacDonald, 2004), see below) interacted with the nuclear and other membranes and released DNA both into the cytoplasm and into the nucleus (Fig. 11). Thin section electron microscopy revealed extensive fusion of endocytosed EDLPC/EDOPC 6:4 lipoplexes with membranes of endoplasmic reticulum, mitochondria and nucleus (Koynova et al. 2007a). It is believed that the excessive fusogenic ability of EDLPC/EDOPC 60:40 lipoplexes allows plasmid DNA to circumvent the cytoplasmic barriers and escape nuclease degradation.
Confocal microscopy of HUAEC cells fixed 24 h after treatment with lipoplexes that contained rhodamine-labeled lipid (A and B), and fluorescein-labeled DNA (C and D). Cells were treated with EDOPC lipoplexes (A and C) and EDLPC/EDOPC 60:40 lipoplexes (B and D). EDOPC lipoplexes (A, C) remained in compact perinuclear endosomes, while EDLPC/EDOPC 60:40 lipoplexes (B, D) interacted with cellular membranes and released DNA into both cytoplasm and nucleus. (Reproduced with permission from (Koynova et al. 2007a); copyright (2007), Elsevier).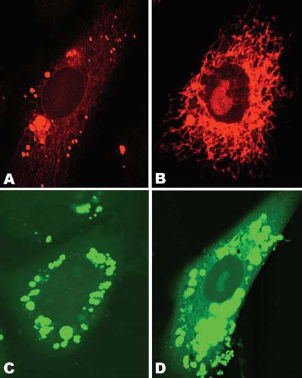
Another persuasive example of a correlation of the phase properties of the carrier lipid/cellular lipid mixtures and transfection success is provided by the C18:1/C10-EPC and C18:0/C10-EPC cationic phospholipids, the former one being 50x more effective as a DNA transfection agent (HUAEC cells) than the latter, despite thei0similar chemical structure and virtually identical lipoplex organization (Koynova et al. 2007b). A likely reason for the superior effectiveness of CI8:1/C10-EPC relative to C18:0/C10-EPC was suggested by the phases that evolved when these lipids were mixed with negatively-charged membrane lipid formulations. The saturated C18:0/C10-EPC remained lamellar in mixtures with a biomembrane-mimicking lipid formulation DOPC/DOPE/DOPS/Chol 45:20:20:15, w/w; in contrast, the unsaturated C18:1/C10-EPC exhibited a lamellar-nonlamellar phase transition in such mixtures, which took place at physiological temperatures, ~37 °C (Fig. 12).
SAXD patterns of mixture of C18:1/C10-EPC with MM (MM = DOPC:DOPE:DOPS:Chol, 45:20:20:15, w/w), recorded upon temperature heating scan at 1 °C/min. Lamellar-cubic phase transition takes place at close to physiological temperatures ~37 °C. (Reproduced from (Koynova et al. 2006); copyright (2007) National Academy of Sciences, U.S.A).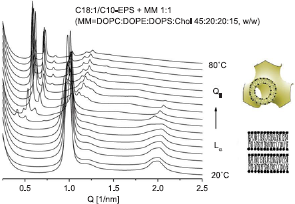
A relationship between membrane fusion and a lamellar-nonlamellar (specifically, lamellar-cubic) phase transition has long been a prominent feature in the literature, and has been well elaborated with respect to both molecular mechanism and energetics (Siegel and Epand, 1997; Siegel, 2005). Simple topological considerations also indicate that lamellar-nonlamellar phase transformations should include some form of a bilayer fusion step; correspondingly, membrane fusion should necessarily proceed with formation of nonlamellar motifs and, in fact, a prospective nonlamellar membrane fusion intermediate structure has been experimentally observed (Yang and Huang, 2002). Thus the recorded disposition of the C18:1/C10-EPC to form highly curved cubic arrays in mixtures with membrane lipids is the suggestive reason for its high fusogenicity, promoting high transfection activity.
Hence, C18:1/C10-EPC lipoplexes are likely to easily fuse with membranes and, as a result of lipid mixing, the resultant aggregates should exhibit extensive phase coexistence and heterogeneity, thereby facilitating DNA release and leading to superior transfection efficiency (Fig. 13). These results highlight the phase properties of the carrier lipid/cellular lipid mixtures as a decisive factor for transfection success and strengthen the concept that the structural evolution of lipoplexes upon interaction with cellular lipids appears to be a controlling factor in lipid-mediated DNA delivery.
Lamellar cationic lipid carriers which form nonlamellar structures upon contacting the membrane lipids easily release DNA and exhibit optimum transfection efficiency. (Reproduced from (Koynova et al. 2006); copyright (2007) National Academy of Sciences, U.S.A).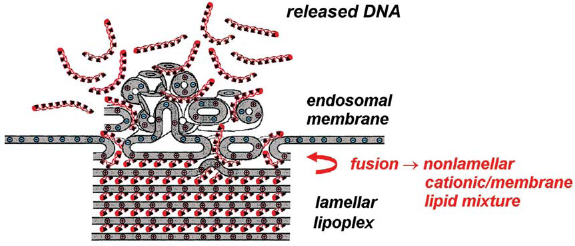
The primary approach to improving transfection potency of cationic lipids has been to synthesize cationic amphiphiles with new versions of positively charged polar groups, with essentially no attention given to variations in the hydrophobic portion of the lipid molecules. Recent data provide evidence for a relationship between transfection success and lipid phase behavior (Tarahovsky et al. 2004; Zuhorn et al. 2005; Koynova et al. 2005, 2006). As it is well known, the latter is strongly modulated by the hydrophobic portion of the lipid molecules. Indeed, data demonstrate that hydrophobic moiety of cationic phosphatidylcholine derivatives strongly modulates liposomal gene delivery into primary human umbilical artery endothelial cells (HUAEC). Superior transfection activity was found for cationic phosphatidylcholines with two 14-carbon atom monounsaturated hydrocarbon chains (myristoleoyl or myristelaidoyl) hydrocarbon chains: diC14:1c-EPC, diC14:1t-EPC and C14:1C2:0-C14:1PC (Tenchov et al. 2008).
The lipid structural characteristic which exhibits distinct change around the chain length of 14C-atoms is the lipid ability to form bicontinuous inverted cubic phases QII. Indeed, it is a rule of thumb that QII phases tend to be found for diacyl or dialkyl phospholipids and glycolipids that have chain lengths of C14 or shorter; the longer-chain lipids typically exhibit a direct lamellar–inverted hexagonal Lα-HII phase transition (Marsh, 1990; Seddon and Templer, 1995; 2000; Koynova and Tenchov, 2008). The reasoning behind this is that longer hydrocarbon chains can relieve packing frustration caused by the geometric packing of circular cylinders creating ‘voids’, thus being able to more easily form the HII phase (Seddon and Templer, 1995), whereas the shorter chain lengths are unable to extend easily to fill the voids, and the inverse bicontinuous cubic phases, with lower packing frustration, dominate–-i.e. the stabilization of the HII phase with respect to the bicontinuous cubics at longer chain lengths occurs because of the reduction in the packing frustration (Anderson et al. 1988; Templer et al. 1998; Shearman et al. 2006).
Indeed, studies on the effect of a set of cationic phospholipids on the phase behavior of a lipid representative of the most abundant class of nonlamellar-forming membrane lipids–-the phosphatidylethanolamines, and which has been used as a benchmark for an Lα-HII transition–-dipalmitoleoylphosphatidylethanolamine (DPoPE), showed that while the long-chain weak transfection agents mostly preserve the HII phase, when admixed to DPoPE, the superior transfection agents with 14C-atom monounsaturated hydrocarbon chains strongly promoted the formation of an inverted cubic phase, replacing the HII phase in a wide temperature range (Tenchov et al. 2008).
Thus, the optimization strategy for lipofection could be further specified and elaborated: cationic lipids able to induce a lamellar-cubic rather than lamellar-H II transition when mixed with membrane lipids seem to be the most successful transfection agents (Tenchov et al. 2008).
Synergy in Lipofection by Cationic Lipid Mixtures
In line with the concept that lipid hydrophobic moiety is able to strongly modulate transfection activity, combinations of two cationic lipid derivatives having the same headgroup but tails of different chain lengths has been shown to have considerably different transfection activity than do the separate molecules. Thus, cationic lipid mixtures have been suggested as a new strategy for finely tuning lipoplex properties and especially their transfection potency (Wang and MacDonald, 2004).
Medium chain/long chain mixtures
Particularly strong “mixture” synergy has been observed with the combination of dilauroyl (12C chains) and dioleoyl (18C:1 chains) homologues of O-ethylphosphatidylcholine (EDLPC/EDOPC mixture). This mixture, at the EDLPC/EDOPC 60:40 composition, transfected DNA into HUAEC more than 30-fold more efficiently than either compound separately (Wang and MacDonald, 2004).
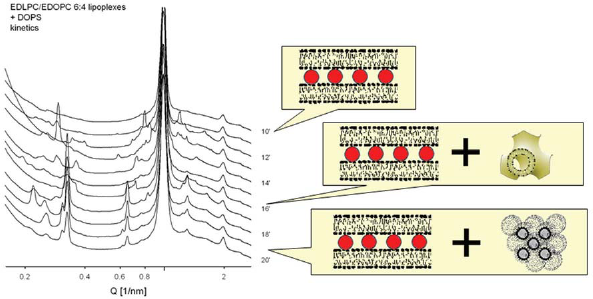
Both the cationic lipid dispersions and the lipid/DNA complexes of the EDLPC/EDOPC mixture have been found to arrange into lamellar arrays at all compositions; no correlation was established between the structural parameters of the lipoplexes and their transfection activity. When anionic membrane lipids interact with the lipoplexes however, variety of nonlamellar phases were formed: thus, EDLPC/EDOPC 60:40 (the composition that exhibited particularly high transfection efficiency) in mixture with the anionic cardiolipin formed the inverted micellar cubic phase, Fd3m (Fig. 14), while either components and mixtures of other compositions all formed phases of lower curvature (Koynova et al. 2005). The micellar cubic phase exhibits even higher interfacial curvature than the inverted hexagonal phase (Seddon and Templer, 1995). In the case of the anionic cardiolipin, it was identified by 11 diffraction maxima with reciprocal spacings fitting the ratio: √8: √11: √12: √16: √24: √32: √35: √36: √48: √51: √56, characteristic for the cubic aspect #15 (Kasper and Lonsdale, 1985). The anionic lipid DOPG similarly formed micellar cubic phase when mixed with the most efficient EDLPC/EDOPC 60:40 formulation. Another anionic membrane lipid, DOPS, was found to form the micellar cubic phase transiently when mixed extempore with the EDLPC/EDOPC 60:40 formulation, as revealed by our kinetic synchrotron X-ray experiments (Fig. 15).
Transfection efficiency (red bars) of EDLPC/EDOPC lipoplexes quantified by expression of β-galactosidase in HUAEC(Wang and MacDonald, 2004); the formulation of maximum efficiency (60:40 w/w) formed micellar cubic phase when mixed with the anionic lipid cardiolipin (diffraction pattern and cartoon structure shown in the inset). (Reproduced with permission from (Koynova et al. 2005); copyright (2005) American Chemical Society). Kinetics of phase changes in the EDLPC/EDOPC (60:40) lipoplexes upon addition of the anionic lipid DOPS, in the time interval 10–30 min after the addition (as indicated on the right side of the diffraction patterns): bilayer cubic Pn3m phase forms initially in parallel to the lamellar phase; later, micellar cubic Fd3m could be distinguished. The charge ratio of the cationic/anionic lipids in the sample is 1:1. (Reproduced with permission from (Koynova et al. 2005); copyright (2005) American Chemical Society).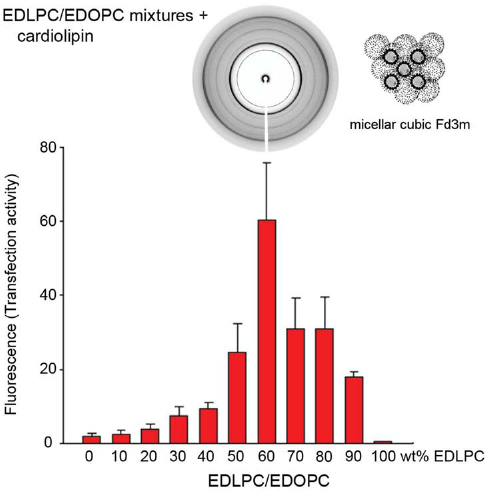
Cationic lipid mixtures exhibiting solid-liquid crystalline phase transition provide another example of mixture synergism, i.e. mixed formulations exhibiting higher transfection activity than do the separate molecules. By juxtaposing the temperature-composition phase diagrams of the mixtures with their transfection activity, it has been found that among a group of more than 50 mixture formulations, the compositions with maximum delivery activity resided unambiguously in the solid-liquid crystalline two-phase region at physiological temperature. Thus, the transfection efficacy of formulations exhibiting solid-liquid crystalline phase coexistence is more than 5x higher than that of formulations in the gel (solid) phase, and over twice that of liquid crystalline formulations; phase coexistence occurring at physiological temperature thus appears to contribute significantly to mixture synergism.
EDMPC/EDPPC mixture
Temperature-composition phase diagram of the EDMPC/EDPPC mixture show, that mixtures with 70–80 mol% EDPPC reside within the two phase region of the phase diagram, between the solidus and liquidus lines, and are thus expected to exhibit solid-liquid crystalline (S + L) phase coexistence at 37 °C (Fig. 16A). Precisely at those compositions lipoplexes were found to exhibit maximum transfection efficiency (Fig. 16B) (Koynova et al. 2007b).
(
DiC14DAB/diC18DAB mixture
According to the temperature-composition phase diagram of the diC14DAB/diC18DAB binary mixture, compositions with ≤45 mol% diC18DAB are in the liquid crystalline phase, whereas mixtures with ≫90 mol% diC18DAB are in the solid (gel) phase at physiological temperature (37 °C). 1 with 45–90 mol% diC18DAB reside within the solid-liquid crystalline phase coexistence region; thus, solid and liquid crystalline domains are expected to coexist in the lipid bilayers of these samples at 37 °C (Fig. 16C). At these compositions, lipoplexes were found to exhibit maximum transfection efficiency (Fig. 16D) (Koynova et al. 2007b).
Other cationic lipid mixtures
Similarly, superior transfection activity was established for sets of compositions residing within the solid-liquid crystalline two-phase region also for the mixtures of: EDOPC/diC14DAB, EDOPC/diC18DAB, EDOPC/DMTAP (Koynova et al. 2007b).
This relationship between delivery activity and physical property can be rationalized on the basis of the known consequences of lipid phase transitions, namely the accumulation of defects and increased disorder at solid-liquid crystalline phase boundaries. Packing defects at the borders of coexisting solid and liquid crystalline domains, as well as large local density fluctuations, could be responsible for the enhanced fusogenicity of mixtures. This study leaded to the important conclusion that manipulating the composition of the lipid carriers so their phase transition takes place at physiological temperature can enhance their delivery efficacy.
Conclusion
In order to achieve their goal, lipoplexes need to release DNA after they has been transported into cells. Escape of DNA from the lipoplexes needs to involve neutralization of cationic lipid by cellular anionic lipids; thus, intermixing of membrane lipids with lipoplex lipids is presumed to be a necessary step in transfection. Because of the strong propensity of the cationic/anionic lipid mixtures to form nonlamellar phases, even when the components form only lamellar phase, a wide variety of nonlamellar arrays can appear in treated cells. The primary viewpoint of the present review is that the structural evolution of lipoplexes upon interaction with cellular lipids is a controlling factor in lipid-mediated DNA delivery. Thus, lamellar lipoplex formulations with compositions close to the lamellar-nonlamellar phase boundary, specifically prone to form cubic phases, and which could easily undergo phase transition upon mixing with cellular anionic lipids, seem to be especially efficient This insight is important for the rational design and successful application of cationic lipids as superior nucleotide delivery agents.
Footnotes
Acknowledgment
The author highly appreciates the expert guidance and friendly support of Professor Robert C. MacDonald throughout her work on cationic phospholipids. The present work was supported by the Center for Cancer Nanotechnology Excellence (CCNE), Northwestern University, initiative of the National Institutes of Health's National Cancer Institute under Award Number U54CA119341, and by the NIH grant GM074429 provided through the STTR program of the National Institute of General Medical Sciences to Avanti Polar Lipids.
The author reports no conflicts of interest.
