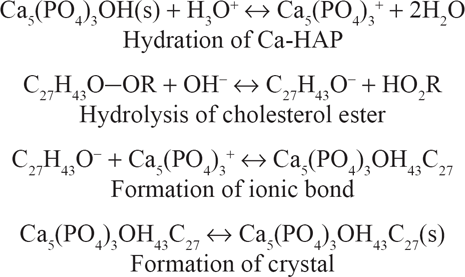
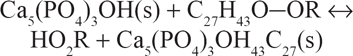Abstract
This paper proposes that atherosclerosis is initiated by a signaling event that deposits calcium hydroxyapatite (Ca-HAP). This event is preceded by a loss of mechanical structure in the arterial wall. After Ca-HAP has been deposited, it is unlikely that it will be reabsorbed because the solubility product constant (Ksp) is very small, and the large stores of Ca+2 and PO4−3 in the bones oppose any attempts to dissolve Ca-HAP by decreasing the common ions. The hydroxide ion (OH–) of Ca-HAP can be displaced in nature by fluoride (F–) and carbonate (CO3−2) ions, and it is proposed that anions associated with cholesterol ester hydrolysis and, in very small quantities, the enolate of 7-ketocholesterol could also displace the OH– of Ca-HAP, forming an ionic bond. The free energy of hydration of Ca-HAP at 310 K is most likely negative, and the ionic radii of the anions associated with the hydrolysis of cholesterol ester are compatible with the substitution. Furthermore, examination of the pathology of atherosclerotic lesions by Raman and NMR spectroscopy and confocal microscopy supports deposition of Ca-HAP associated with cholesterol. Investigating the affinity of intermediates of cholesterol hydrolysis for Ca-HAP compared to lipoproteins such as HDL, LDL, and VLDL using isothermic titration calorimetry could add proof of this concept and may lead to the development of a new class of medications targeted at the deposition of cholesterol within Ca-HAP. Treatment of acute ischemic events as a consequence of atherosclerosis with denitrogenation and oxygenation is discussed.
Introduction
All living organisms must possess structure to separate themselves from their surroundings. Plants use cellulose, bacteria have a cell wall, invertebrates have calcium carbonate shells, and vertebrates have skeletons of primarily calcium hydroxyapatite Ca5(PO4)3OH (Ca-HAP). In humans, Ca-HAP is deposited within a matrix that is responsive to the stress demands on the bone (Wolff's Law).1,2 However, the deposition of Ca-HAP is not exclusive to bone. The process also occurs in muscle, neural tissue, granulomas, neoplasms, cardiac valves, and many other tissues. One proposal of this paper is that deposition of Ca-HAP is a response of the organism to mechanical stress and/or the need to provide structure to form a partition within the tissues.
Evidence to Support the Proposition that Ca-HAP Deposition Occurs Throughout the Human Body Because of Structural Needs
Diffuse idiopathic skeletal hyperostosis (DISH) is associated with bridging bone formations fusing vertebral bodies called syndesmophytes (located by arrows) that provide structure for severe degenerative intervertebral discs (Fig. 1).3,4
DISH with Ca-HAP hyperostosis (syndesmophtyes).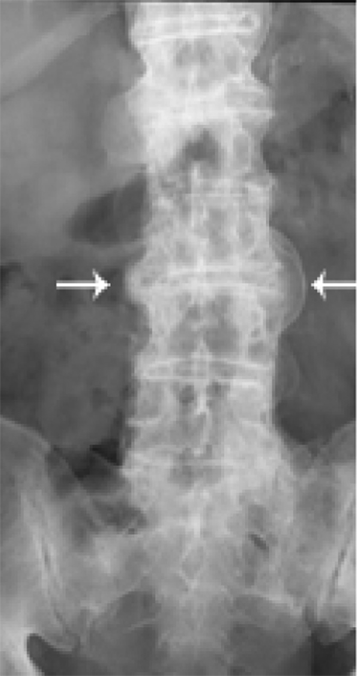
Patients suffering from myositis ossifications traumatica deposit Ca-HAP in muscle after trauma that is associated with premature stress loading (Fig. 2).
5
Myositis ossificans traumatica with calcification within muscle of likely Ca-HAP (arrow).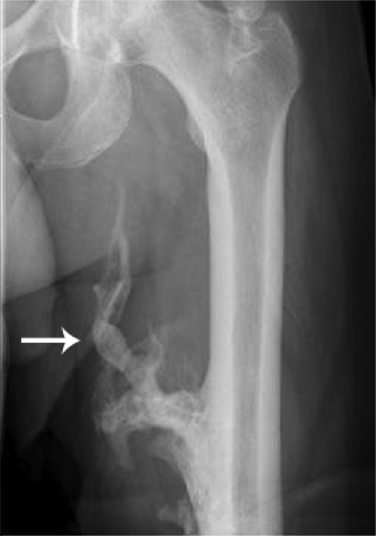
Osteoarthritis is characterized by depositions of Ca-HAP that is commonly associated with structural deterioration of the joint from trauma and loss of cartilage. 4
Pancreatic, granulomatous and neoplastic diseases develop partitions of calcium within the tissue to possibly separate disease and non-disease zones (Fig. 3).
6
Intraductal carcinoma of the breast with calcium deposit (arrows) forming a partition between malignant cells and stroma.
Deposition of Ca-HAP in Arterial Vessels as a Response to Mechanical Stress
In humans, calcification can occur in the intima, media, and, uncommonly, in the adventitial layers of the artery. It is probable that local factors influence where the Ca-HAP deposition occurs, and this may be related to the efficiency of structural reinforcement.
The arteries most commonly associated with severe disease include the carotid, aortic, superficial femoral and coronary, many of which are associated with lengths of low shear stress in the vicinity of a bifurcation. 7 As has been demonstrated in coronary bifurcations, plaque in these low shear stress areas exhibit a higher proportion of dense calcium and lower proportion fibrous tissue consistent with the need to provide structural support by the deposition of Ca-HAP. 8
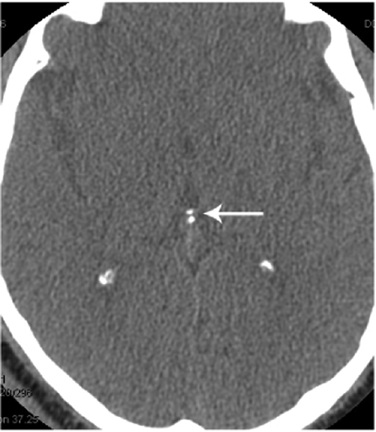
Arteries from organs associated with extremely high blood flow compared to tissue mass such as the kidney and pineal gland are also frequently calcified. In addition to the high flow, the weak capillary structure of the pineal gland may account for pineal calcification in 70% of adults along with accumulation of fluoride (Fig. 4).9–11
Arteries and arterioles of diabetic patients may have abnormal vascular tone caused by augmentation of Ca+2 sparklet activity from hyperglycemia. This elevation in Ca+2 sparklet activitiy is associated with an increase in vascular tone producing resistance stress on the vessels.14,15 This may account for the more diffuse findings of calcium deposition in their arterial vessels in addition to deposition in critical low shear stress vessels.
Hypertension either essential or secondary to renal disease, endocrine disorders (pheochromocytoma or Cushing's disease), or vasoconstriction from rheumatic disease such as rheumatoid arthritis, Raynaud's disease or phenomenon and systemic sclerosis produces resistance stress on the arterial wall predisposing to arterial calcification.16,17
Hypertension from medications or toxins such as cigarette smoke produces chronic changes in vascular resistance increasing the stress on the arterial wall. 18
Although not exhaustive, the above examples provide evidence that Ca-HAP is a key compound providing structure throughout the body and that deposition is not a stochastic event but rather related to mechanical forces coupled with a signaled response to reinforce the arterial wall.
Evidence that Genetic Expression Causes Deposition of Ca-HAP
The signals that initiate Ca-HAP are not well understood, but the genetic expression is probably present in many cell types as observed in idiopathic non-arterioscelerotic cerebral calcification, Fahr's disease, characterized by diffuse hereditary depositions of calcium in the brain usually in the regions of the basal ganglia, cerebellum and sometimes also found in the putamen, caudate nucleus, thalamus and white matter. This condition was first described by Karl Theodor Fahr in 1930, is usually progressive and associated with dementia, psychosis, movement disorders, pyramidal signs, and frontal lobe and cerebellar dysfunction. The analysis of brain tissue reveals that the calcium is primarily in the form of CA-HAP and the calcification process does not originate from the blood vessels.19,20 Therefore Ca-HAP can be, but is not usually, deposited in tissues not subjected to mechanical stress or requiring a partition (Fig. 5).
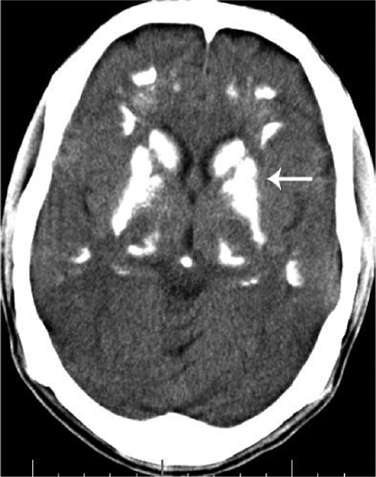
Ca-HAP deposition within the basal ganglia in Fahr's disease. (arrow) 58–75 percent of the lesions probably consist of Ca-HAP. 19 This work by radiopaedia.org/image/539084 is licensed under a Creative Commons Attribution 3.0 Unported License.
Other genetic causes of Ca-HAP deposition include the ACDC condition and idiopathic infantile arterial calcification, both of which affect the arteries (Fig. 6). 21 In the ACDC condition, or arterial calcification due to CD73 deficiency individuals with ACDC have calcium buildup in their arteries below the waist and in the joints of their hands and feet, but not in the arteries of their heart. Affected individuals can experience claudication. 22 In idiopathic infantile arterial calcification the calcium deposits have been shown to be Ca-HAP. 23
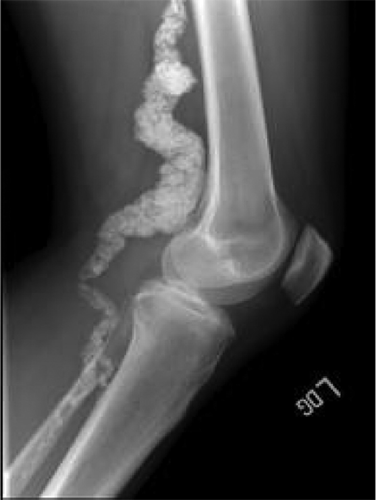
ACDC condition. National Human Genome Research Institute.
In addition to the structural argument presented in this paper, various mechanisms have been proposed to explain the deposition of Ca-HAP in the arterials vessels, including:
A response to endothelial inflammation 24
Metabolic changes related to calcium and phosphate concentrations 24
Chemical mediation from osteopontin, vitamin D, or other promoters, or lack of inhibitors of mineralization25,26
Cellular mechanisms from osteoblastic activity. 27
Clinical and laboratory evidence to support the hypothesis that deposition of Ca-HAP is a key step in the early formation of atherosclerotic plaques:
Cardiac calcium computerized tomography detects calcium in early lesions of coronary atherosclerosis and this investigation has been recommended by the American Heart Association for high-risk patients.28,29
In pathologic specimens, Ca-HAP is incorporated with cholesterol and, to a lesser extent, with 7-ketocholesterol (7-keto) in atherosclerotic plaques.30,31
Ca-HAP is deposited diffusely in arterial vessels and in vessels of low shear strength in diabetics and in patients with stage 3 and 4 kidney disease.32–35
Lanthanum, administered as LaCl3, is an element with similar chemical properties as Ca+2 and suppresses atherosclerosis. 36 Lanthanum binds to cellular Ca+2 sites in a less reversible manner than Ca+2. 36 Lanthanum carbonate has been shown to reduce progression of vascular calcification in a cohort of patients who require hemodialysis. 37
Homocysteinemia, a high-risk factor for atherosclerosis, produces early calcification of the arterial vessels. Although tempting to define this calcification as the reactive sulfhydral group in homocysteine binding to CA-HAP, the chemistry, particularly the large ionic radius of sulfur compared to oxygen, does not support the development of an ionic bond. Probably, the mechanism for increased atherosclerosis relates to the generalized weakening of the vascular wall from homocysteine and subsequent deposition of structural Ca-HAP. This may explain “Homocysteine: the Rubik's cube of cardiovascular risks factors.” 38
Fatty lesions including fatty streaks may precede the development of calcium deposition. The biochemistry and morphology have been studied extensively including the development of foam cells with lipid accumulation. These structural changes probably have some effect on the integrity of the vessel wall which may signal for Ca-HAP deposition. Fatty streaks are reversible but Ca-HAP deposition is probably irreversible. 39
Fate of Ca-HAP Deposited in the Arterial Wall and Subsequent Development of the Ca-HAP Cholesterol Complex
Once deposited, Ca-HAP is unlikely to dissolve. The Ksp of Ca-HAP in aqueous solution is extremely small and approximately [Ca]5[PO4]3[OH] = 2.03 × 10−59. 40 Since an equilibrium exists between [Ca-HAPartery] ↔ [Ca-HAPplasma] ↔ [Ca-HAPbone] and the bone contains huge stores of Ca-HAP, it is unlikely that therapeutic attempts to change [Ca+2] and/or [PO4−3] by chelation or decreasing a common ion through medications will be effective.
In nature, the ionic bond between Ca2+ and OH–allows for various anionic substitutions including fluoride and carbonate. 41 Incorporation of F– into Ca-HAP has been postulated to occur by a number of mechanisms including substitution for the OH–group. 41 The ionic radius of an anion predicts whether it will fit into the crystal matrix of Ca-HAP. The chloride ion, present in plasma, has an atomic radius of 1.81 Å and will rarely form an ionic bond with Ca-HAP; however, O– and F– have comparable ionic radii (1.44 Å and 1.33 Å) which favors the substitution. 42 The ionic radius of the OH– is reported as 152 Å. 43 The OH– group (leaving as H2O) is located on the crystal surface, which could permit a cholesterol anion formed during ester hydrolysis to bind without steric problems (Fig. 7). In this concept, cholesterol ester is hydrolyzed producing a reactive intermediate anion that can displace the OH– of Ca-HAP with H2O as the leaving group (Fig. 8). Even without the aid of computerized molecular modeling it is apparent that the anion of cholesterol ester hydrolysis is much too large to fit into the crystal lattice structure of Ca-HAP in a manner similar to the ionic substitution found in F–, however the surface substitution of OH– for the anion of cholesterol ester hydrolysis is quite probably, can be modeled, and may have some similarities to the substitution of carbonate that is found in nature. Furthermore, Ca-HAP column chromatography has been used to separate anions of lipoproteins and proteins. 44
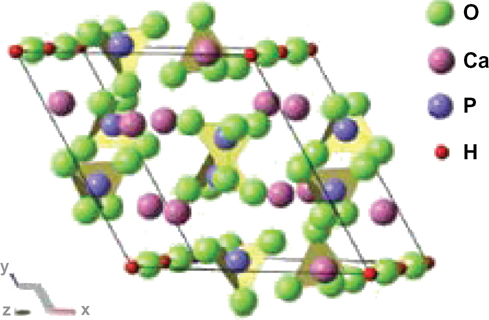
Structure of a Ca-HAP unit cell–-The OH– groups are located along the edges, which would allow for surface substitutions by cholesterol ester anion intermediates. Reprinted with permission from Snyders R, Music D, Sigumonrong D, Schelnberger B, Jensen J, Schneider JM. Experimental and ab initio study of the mechanical properties of hydroxyapatite, Appl Phys Lett. 2007;90:193902–2. American Institute of Physics.
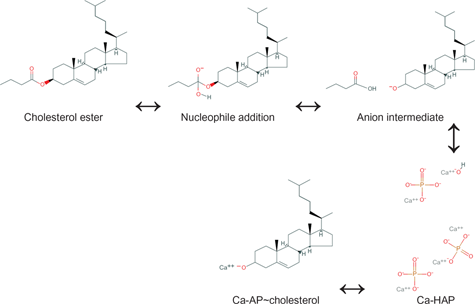
Proposed mechanism of formation of Ca-AP~cholesterol at pH 7.4.
Reaction for Formation of Ca-Ap~Cholesterol Complex from Cholesterol and Ca-HAP a
Simplifying:
Stoichiometric formula.
Thermodynamic Considerations of Proposed Reaction of Ca-HAP with Cholesterol
The enthalpy of hydration (δH) of one mole of CA-HAP at pH 7.0 is -406 kj/mole. 45 The change in entropy (δS) of this reaction is not reported. However, the δS of the reaction at 310 K is most likely positive as a more disordered state is produced (solid to liquid). Therefore, the free energy (δG) for formation of the calcium hydroxyapatite ion (Ca-AP+) independent of temperature is probably negative, and the reaction is spontaneous. The ester hydrolysis of cholesterol catalyzed by plasma esterases is known to proceed in humans and may be a risk factor for atherosclerosis. 46 Unless there are conformational constraints or competitive inhibition for the cationic site, the Ca-AP~cholesterol complexes should exist.
Special Consideration of 7-keto
The concentration of oxysterols in plasma is many orders of magnitude less than cholesterol, but they may have some importance in the development of atherosclerosis.47,48 Oxidized cholesterols fed to animals are associated with a more rapid progression of atherosclerosis. 49 Based on the plasma concentration, 7-keto is found in fatty streaks in a concentration greater than would be expected. 47 The mechanism for deposition of 7-keto may take the same route as other cholesterol esters by substitution of OH– with the anion intermediate of ester hydrolysis or additionally by substitution with the 7-keto enolate. Of note, in 1954 Rosenkrantz and Gut showed that the enol tautomer of 7-keto did not form in solvents, but they did not investigate the reaction in biological fluids. 50
Additional support for the mechanism of formation of the Ca-AP~cholesterol complex discussed above:
Raman microspectroscopy supports the coexistence of calcium and cholesterol in microenviroments.51,52
Magic angle spinning NMR supports the existence of cholesterol mono crystals in close proximity to Ca-HAP. 30
Confocal microscopy studies support cholesterol associated with the surface of Ca-HAP seeds. 53
Electron microscopy studies show Ca-HAP and non-esterified cholesterol associated in a crystal agglomerate. 54
The Ca-AP~cholesterol complex is found in locations where lipoprotein carriers of cholesterol come in contact with Ca-HAP. These locations include arteries and the pineal gland where the capillary structure is fragile. Cholesterol deposition is not a common feature of DISH, osteoarthritis, granuloma, or Fahr's disease because the lipoprotein carriers cannot easily cross the endothelial wall.4,19 However, a cellular mechanism with endo- and exocytosis or active transporters utilizing high energy sources could transport the lipoproteins.
Ca-HAP, Cholesterol, Lipoproteins
It has been proposed that cholesterol has an affinity for Ca-HAP and that the extent of ionic binding depends upon the lipoprotein carrier of cholesterol. Of the lipoproteins, HDL has the greatest affinity for cholesterol, and the Ca-AP~cholesterol complex is less likely to form compared to formation with LDL or VLDL. 55 Proof of this concept could be achieved by using isothermic titration calorimetry to compare binding affinities of in situ hydrolyzed cholesterol esters of HDL, LDL, and VLDL with Ca-HAP. If this is true, medications could possibly be developed to antagonize these molecular attractions.
Diabetes and Atherosclerosis
The systemic complications of diabetes, which include but are not limited to retinopathy, neuropathy, nephropathy, myopathy, and gastropathy, may be related to attenuated sensing of shear stress, impairment of NO synthase, and elevation of vascular tone that probability affect the structural properties of the arterial wall.35,56 Atherosclerotic lesions in diabetes are frequently found in the media of the arterial wall and diffusely located throughout the arterial tree. The biochemical mechanisms responsible for the occurrence of atherosclerosis in diabetes must still be elucidated.
Alzheimer's Disease and Atherosclerosis
Atherosclerosis has long been recognized as a risk factor for Alzheimer's disease and other forms of dementia. Impaired nutrient supply to the brain associated with Ca-HAP deposition could alter diffusion at the pre-capillary level. In the Baltimore Longitudinal Study of Aging, 136/200 participants had evidence of intracranial atherosclerosis that increased the odds for dementia independent of brain infarcts; however, the atherosclerosis did not correlate with Alzheimer's pathology. 57
Renal Disease and Atherosclerosis
It is widely accepted that impaired renal function as measured by glomerular filtration rate (GFR) is closely related to arterial calcification and subsequent development of atherosclerosis. 33 The etiology of these phenomena has been speculated by a number of authors without a common resolution.32,34,58 Hypertension caused by increased activity in the renin-angiotensin system commonly found in stages 3 and 4 of kidney failure could increase the stress on the arterial wall thus signaling deposition of Ca-HAP.
Options for Treatment
As previously discussed, unless there is a mechanism to structurally enhance stressed arterial vessels, it is unlikely that Ca-HAP depositions will dissolve. Preventing further deposition of Ca-HAP may be possible with:
Exercise that increases bone density and shifts the concentration of Ca-HAP away from vascular deposition. 59
Decreasing arterial wall tension by controlling hypertension. 59
In diabetic patients, improved regulation of glucose may decrease oxidative stress within the arterial smooth muscle. 60
Discontinuation of vasoconstrictors such as cigarette smoke. 61
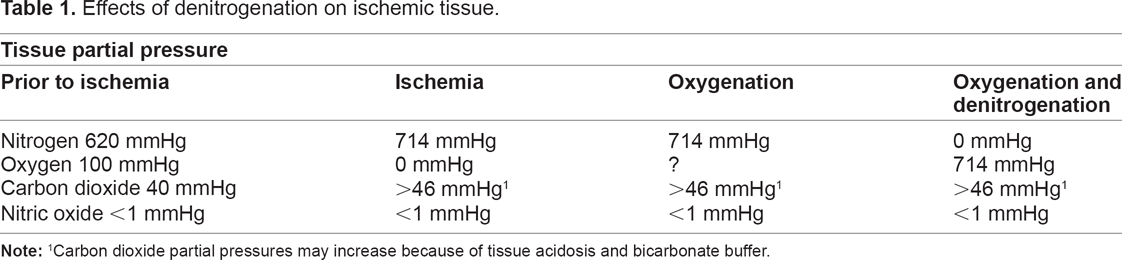
Acute therapies for patients suffering from various forms of ischemia from atherosclerosis, such as hemorrhagic, embolic, and low-flow events, may include denitrogenation as well as traditional oxygenation. This concept was first put forth by VanDeripe.62,63 As tissues become ischemic, the partial pressures of nitrogen, carbon dioxide, and nitric oxide increase as the oxygen tension falls. Denitrogenation and administration of high concentrations of oxygen preferably with an inert gas such as helium will displace nitrogen and increase the partial pressure of oxygen in ischemic tissues (Table 1). With a simple apparatus and a high oxygen flow, a large percentage of the nitrogen stored in the body can be expelled within 1 hour. 64 If such a device is not available, a non-rebreathing face mask with one-way expiratory valves that is attached to high oxygen flows should facilitate denitrogenation.65,66 There is no single well defined threshold below which oxygen toxicity will not occur. However, at normal atmospheric pressure the development of oxygen toxicity is mainly a function of the inspired oxygen concentration above 50% and the time of exposure. 67 When an inert gas cannot be mixed with oxygen, the risk of oxygen toxicity from reactive oxygen species increases, but the 1 hour exposure to 100% oxygen for denitrogenation may be tolerated. 68 The long and safe history of administering high concentrations of oxygen for short periods of time during general anesthesia attests to this practice. This treatment could be instituted by paramedics and other emergency personnel and may salvage tissue that was once considered non-viable, especially if the body has time to adapt to the acute ischemic event. If indicated, this relatively low-risk procedure may be coupled with higher-risk forms of thrombolytic therapy.
Effects of denitrogenation on ischemic tissue.
Treatment of chronic accumulation of plaque has been outlined in numerous publications.69–71 Preventive therapies include control of blood glucose, hypertension, body weight, and stress. Exercise, proper diet, and abstinence from tobacco products can be very helpful. Medication management includes cholesterol-lowering agents, anticoagulation of varying degrees, and lipid modification.
Possible Flaws in the Concepts Presented
This paper fails to discuss the underlying molecular mechanisms of Ca-HAP deposition, but rather proposes, on the basis of clinical and empirical observations, that accumulation of Ca-HAP is a response to reinforce structure. This may be an oversimplification that could lead to erroneous conclusions. Reflecting on the proposed chemistry, the hydrolysis of an ester under basic conditions is known to produce an anion intermediate, but the clinical significance of the presumed short-lived intermediate of cholesterol hydrolysis could be overestimated. The AG of the proposed reactions is negative, proceeding spontaneously, but the rates could be so slow that they are also not clinically significant. Finally, denitrogenation for the treatment of ischemia, as described, would need to undergo a clinical trial as, often in medicine, concepts on paper fall short of expectations when applied to human physiology.
Conclusion
This author believes and presents supporting evidence to suggest that atherosclerosis is initiated by a signaling event that results in deposition of Ca-HAP. This event is preceded by a loss of mechanical structure in the arterial wall. Once Ca-HAP is deposited, it is unlikely to be reabsorbed because the solubility product constant Ksp is very small, and the large stores of Ca+2 and PO4−3 in the bones oppose any attempts to dissolve Ca-HAP due to the common ion effect. Investigating the differences in affinity for hydrolysis products of cholesterol ester intermediates between Ca-HAP and lipoproteins such as HDL, LDL, and VLDL, using isothermic titration calorimetry, could lead to the development of a new class of medications targeted at the deposition of cholesterol within Ca-HAP. In addition to preventive therapies, consideration should be given to denitrogenation coupled with high-flow oxygenation for the treatment of acute ischemic events. It is hoped that his paper will provide some useful insights not previously addressed about an underlying mechanism of atherosclerosis and novel therapies.
The author has no conflicts of interest to report pertaining to the authorship or publication of this manuscript.
Footnotes
Acknowledgement
The author would like to thank Julie Rosato of Duke University for assistance with designing and formatting the illustrations and Kathy Gage of Duke University for editorial assistance.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.