Abstract
The ability of α-tocopherol to prevent the degradation of the polyunsaturated fatty acids, docosahexaenoic acid (DHA) and linolenic acid (ALA) at elevated temperature was investigated in a series of laboratory experiments. DHA is known to promote nervous system function and health, however, DHA and other highly unsaturated fatty compounds are readily oxidized with loss of bioactivity. Mixtures of DHA or ALA and α-tocopherol were incubated at 70 °C. Samples of these mixtures were analyzed by gas chromatography. After 72 hours at 70 °C mixtures with 0.25 mg α-tocopherol/mg retained 79.6% of the initial amount of DHA and 94.2% of the ALA. When α-tocopherol was present at 2.5 mg/mg the amount of DHA decreased to 62.5% and ALA decreased to 87.4%. Control samples that did not contain α-tocopherol retained 43% DHA and 52.6% ALA. These results indicate that α-tocopherol can limit the degradation of these polyunsaturated fatty acids at elevated temperature. However, the greatest protective effect was not obtained with the highest level of α-tocopherol. This relationship needs to be considered when formulating feeds where elevated process temperatures are likely to occur.
Introduction
The health benefits associated with polyunsaturated fatty acids such as docosahexaenoic acid (DHA) has generated interest in their use as dietary supplements and additives in animal feeds.1–4 Benefits include improved nervous system development and cardiovascular health.5–7 Fish oils represent a rich source of polyunsaturated fatty acids and especially the more highly unsaturated fatty acids such as DHA. However, when fish oils are used in the feed formulations of laying hens to improve the nutritional value of the eggs there is some decrease in sensory quality reported as fishy off-flavors.8–10 This can be alleviated by using marine algae as the primary source of the polyunsaturated fatty acids rather than the fish oil. 11 Similar studies with flaxseed as a source of α-linoleic acid (ALA) have shown that the polyunsaturated fatty acids do accumulate in the eggs, however, there are anti-nutritional compounds in the flaxseed that have negative effects on production. 12 When isolated ALA was fed to Shaver hens a reduction in hatchability was observed that could be off-set by supplementing the diet with soybean oil. 13 The addition of polyunsaturated acids to poultry feeds provides a straightforward technique to improve the nutritional quality of the poultry eggs. The highly unsaturated structure of these lipid compounds makes them susceptible to oxidative degradation which results in a loss of bioactivity and the development of volatile odiferous compounds. 14
Antioxidants can limit the oxidative degradation of unsaturated compounds and thereby extend the shelf-life of products formulated with these compounds. One of the most active naturally occurring antioxidants is α-tocopherol. It has the ability to donate electrons to lipid radicals and reduce peroxidation. 15 It is one member of a group of four closely related structures, eg, α, β, γ and δ-tocopherol, together with four structural analogs, α, β, γ and δ-tocotrienol, that comprise the vitamin E family of compounds. These compounds are synthesized by plants and obtained as a byproduct from oilseed processing.16,17 This commercial vitamin E product is a mixture of the eight compounds and contains only the bioactive stereoisomers produced by plants. The observed activity of these compounds is related to both their primary chemical structure, eg, the number and location of methyl groups attached to the ring which gives rise to the α, β, γ or δ designation of the tocopherols, and the particular spatial orientation of atoms within the compound that produces stereoisomerism or chirality of the structure. The configuration of each chiral center is labelled R or S to denote such differences in spatial orientation. In biological systems the chirality of a substrate can strongly influence its activity due to enzyme-mediated processes, for example, which discriminate between the various isomers. This can result in large differences in the absorption, transport, and reaction rates of the substrate or nutrient. Among the possible isomers of α-tocopherol the all R or R, R, R structure is considered the most significant based on bioavailability and dietary factors. When α-tocopherol is prepared by chemical synthesis the product contains a mixture of active and inactive stereoisomers of α-tocopherol that exhibits approximately half the activity of the natural form, R, R, R- α-tocopherol. This variation in bioactivity due to source, natural or synthetic, is accounted for in nutritional labelling to provide an equivalent basis of comparison.
Clinical studies have examined the benefit of diets supplemented with vitamin E or one of the individual tocopherol compounds. Two studies examined the connection between vitamin E and coronary heart disease and concluded that long-term use of dietary supplements (100 IU/day) was associated with a reduced risk of heart disease in both men and women.18,19 In contrast, a study that investigated the use of vitamin E supplements and lung cancer concluded that there was a small increased risk with long-term use at high doses, ie, 400 mg/day. 20 In other studies related to prostate cancer there was shown a benefit of a diet supplemented with α-tocopherol in combination with selenium, however, follow-up investigations have not supported the early positive findings.21,22 Mechanistically, the ability of antioxidants to deactivate free radicals that could be responsible for mutagenesis and subsequent cancer development appears to have preventive value. However, as noted above the action of α-tocopherol changes from antioxidant to prooxidant depending on concentration.
This study was undertaken to explore the efficacy of synthetic α-tocopherol (the racemic mixture of active and inactive isomers) to limit the oxidative degradation of DHA and ALA that can occur at elevated temperatures. The combination of α-tocopherol with these two polyunsaturated fatty acids can lead to higher quality feeds and improved production.
Materials and Methods
Materials
Methyl docosahexaenate (98%), methyl linolenate (99%) and (±) α-tocopherol (95%) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Methyl palmitate (99%) was obtained from Nu-Chek Prep, Inc. (Elysian, MN, USA). Acetone (HPLC grade) was purchased from Burdick and Jackson (Muskegon, MI, USA).
Accelerated oxidation
Working solutions of the fatty acid methyl esters, DHA and ALA, were prepared in acetone and diluted to a final concentration of 4 mg/mL. Methyl palmitate was used as an internal standard at 0.1 mg/mL. Tocopherol was added at 1, 10, or 20 mg/mL levels to provide mixtures of 0.25 mg tocopherol/mg DHA, 2.5 mg tocopherol/mg DHA, or 5 mg tocopherol/DHA. Experimental controls contained no tocopherol. The accelerated oxidation was performed by placing 1 mL volumes of each mixture into 2 mL glass screw-cap vials. The vials were heated with a dry block heater and maintained at a temperature of 70 °C. Samples were collected after 24 and 72 hrs. The experiments were run in duplicate. The values presented are averages of the duplicated experiments. Single factor ANOVA was used with an alpha value of 0.05.
Chromatographic analysis
Samples, 0.1 mL, were removed from the glass vials by syringe and diluted in acetone for analysis by gas chromatography. Analysis was performed with the Agilent 6890 Gas Chromatograph equipped with a flame ionization detector. Helium was used for the carrier gas at a total flow of 30 mL/min. Injections were made with a 50:1 split. The injection volume was 1 μL. Samples were separated on a SP-2380 column (Supelco, Bellefonte, PA) that measured 30 m × 0.25 mm × 0.2 micron. The inlet temperature was 160 °C and the oven was programmed from 140 °C to 250 °C at 5 °C/min. The detector temperature was 260 °C. Standard solutions of the polyunsaturated fatty esters were prepared at concentrations from 0.05 mg/mL to 1.0 mg/mL and used to quantify the amount of DHA and ALA remaining in the mixtures.
Results and Discussion
The results of accelerated oxidation experiments are presented in Table 1. In the control samples that did not contain tocopherol the amount of DHA has been reduced to 78.2% and ALA reduced to 89.2% of the starting amounts after 24 hours at 70 °C. At 0.25 mg tocopherol/mg a small increase is observed for DHA, 78.2% versus 79.4%, and a larger increase for ALA, 89.2% versus 99.1%. As the amount of tocopherol is increased to 2.5 mg/mg the protective effect is larger with 87.4% DHA remaining. At 5 mg tocopherol/mg the additional increase in DHA is not significant. However, at this level of tocopherol the protective effect on ALA shows a decrease to 90.4%.
Amount of lipid remaining after 24 and 72 hours at 70 °C.
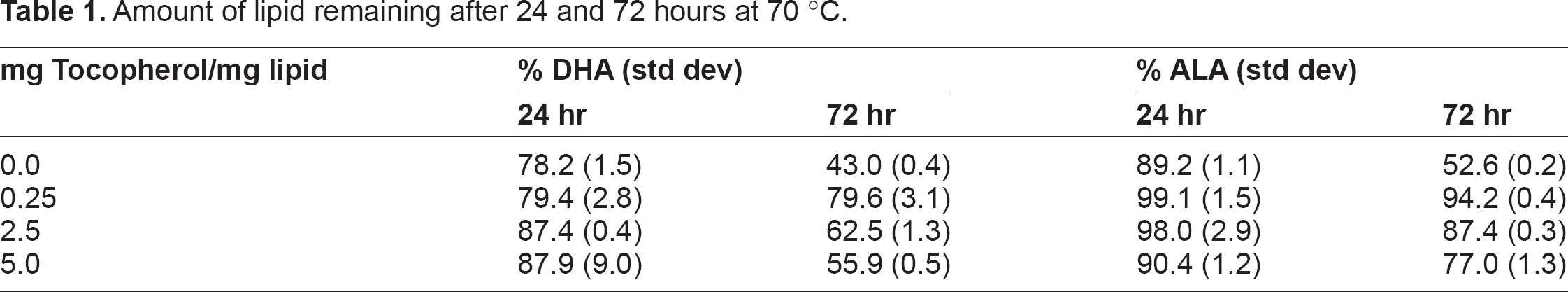
After heating at 70 °C for 72 hours there is a decreasing trend observed with increasing amounts of tocopherol. The controls show the lowest amounts of DHA and ALA with 43% and 52.6% remaining, respectively. The greatest protective effect of the tocopherol is seen to occur with the lower level of 0.25 mg/mg for both DHA and ALA. The protective effect decreases at the higher levels of tocopherol although the amounts of DHA and ALA still exceed the values for the control samples. This trend was not expected and suggests a change in the ability of tocopherol to provide antioxidant activity at the higher concentrations. The generally accepted mechanism of antioxidant power in tocopherol is given by the ring structure which acts as a donor to stabilize the peroxy radicals that otherwise lead to lipid oxidation. The mixtures of tocopherol and polyunsaturated fatty acids used in these experiments do not have the ability to regenerate the active form of tocopherol after it stabilizes a peroxy radical. Therefore the active form of tocopherol is consumed stoichiometrically and it would be expected that the higher levels of tocopherol would provide greater antioxidant power. This is seen for DHA after 24 hours with 0.25 tocopherol mg/mg and 2.5 mg tocopherol but not for ALA. These results suggest that feed formulations containing tocopherol and polyunsaturated acids should be carefully evaluated to preserve the benefits of each compound if exposure to elevated temperatures is anticipated.
Footnotes
Acknowledgements
The expertise of L.B. Ito was greatly appreciated in performing chromatographic analysis.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
