Abstract
Glycosylation is essential to the synthesis, folding, and function of glycoproteins in eukaryotes. Proteins are co- and posttranslationally modified by a variety of glycans in the endoplasmic reticulum (ER); modifications include C- and O-mannosylation, N-glycosylation, and the addition of glycosylphosphatidylinositol membrane anchors. Protein glycosylation in the ER of eukaryotes involves enzymatic steps on both the cytosolic and lumenal surfaces of the ER membrane. The glycans are first assembled as precursor glycolipids, on the cytosolic surface of the ER, which are tethered to the membrane by attachment to a long-chain polyisoprenyl phosphate (dolichol) containing a reduced α-isoprene. The lipid-anchored
Introduction
Eukaryotic cells are subdivided into organelles by lipid bilayers, with a variety of unique functions and lipid compositions.
1
In addition, the lipid components in the plasma membrane are distributed asymmetrically between the two monolayers with the aminophospholipids, phosphatidylserine and phosphatidylethanolamine, located on the cytoplasmic leaflet, and the choline-containing lipids, sphingomyelin and phosphatidylcholine, in the exoplasmic leaflet. As the cytoplasmic surface of the endoplasmic reticulum (ER) is the site of synthesis of the majority of eukaryotic phospholipids, at least 50% of the newly synthesized phospholipids must flip to the lumenal monolayer to support the uniform expansion of the membrane bilayer. The observed final asymmetric distribution of phospholipids in the plasma membrane is the net result of competing bidirectional, energy-independent flip/floppases and ATP-requiring, phospholipid pumps catalyzing the unidirectional transport of phospholipids across the membrane bilayers. Current models suggest that energy-independent flip/floppases mediate the transmembrane movement of phospholipids in biogenic (self-assembling) membranes, such as the ER, and the P-type and ATP-binding cassette transporters function to maintain the transbilayer phospholipid asymmetry in membranes, such as the plasma membrane.2–5 The glycosyl donors employed in protein N-glycosylation, C- and O-mannosylation, and glycosylphosphatidylinositol membrane (GPI) anchoring are assembled on both sides of the ER membrane and require the participation of the bidirectional, energy-independent class of flippase proteins to enter the lumen of the ER and participate as glycosyl donors.5–7 While more properly functioning as
Protein N-glycosylation requires the synthesis of a precursor oligosaccharide, containing three glucose (Glc), nine mannose (Man), and two
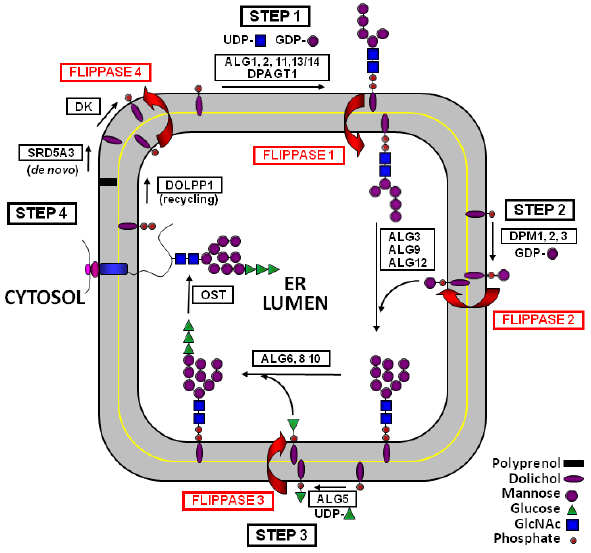
Topological model for DLO synthesis in the ER. M5-DLO (Step 1), Man-P-Dol (Step 2), and Glc-P-Dol (Step 3) are synthesized on the cytosolic surface of the ER membrane from appropriate sugar-nucleotide donors and Dol-P and diffuse transversely to the lumenal monolayer where they participate in the synthesis of full-length G3-DLO. The polar head groups of the dolichol-linked intermediates do not spontaneously cross the ER membrane but are escorted across the highly hydrophobic lipid bilayer by a specialized class of membrane transporters referred to as flippases, numbered arbitrarily as indicated (Flippase 1: M5-DLO Flippase; Flippase 2: Man-P-Dol Flippase; Flippase 3: Glc-P-Dol Flippase; and Flippase 4: Dol-P Flippase). The G3 oligosaccharide is transferred to protein nascent chains releasing a molecule of Dol-P-P, which is dephosphorylated by DolPP1; Dol-P is then returned to the cytosolic monolayer by Flippase 4 (Step 4) for utilization in further rounds of DLO synthesis.
Man-P-Dol also provides the mannosyl units for protein C- and O-mannosylation and GPI anchor biosynthesis, as illustrated in Figure 2. 15 Similar to DLO assembly, this Man-P-Dol is synthesized on the cytosolic surface of the ER membrane and must be transported to the lumenal monolayer to function as mannosyl donor. Presumably, the Dol-P molecules released by the mannosyltransferase reactions are returned to the cytosolic surface for reutilization by the same mechanism as the Dol-P formed from Dol-P-P and the Man-P-Dol and Glc-P-Dol mediated reactions in G3-DLO synthesis on the lumenal leaflet. 6
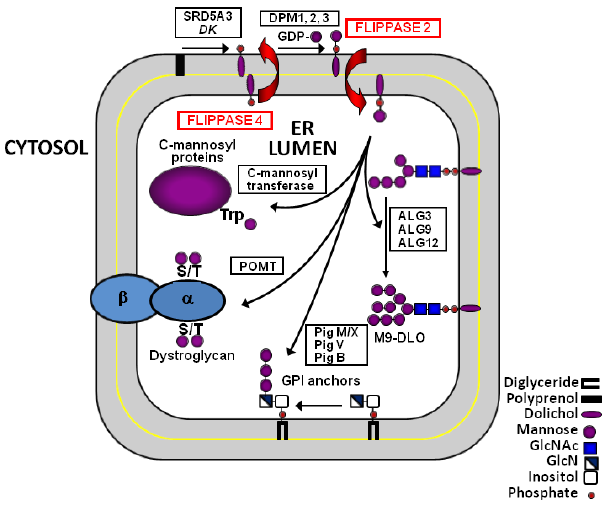
Man-P-Dol acts as mannosyl donor for the synthesis of M9-DLO, GPI anchors, and C- and O-mannosylated proteins. Man-P-Dol is synthesized on the cytosolic monolayer of the ER membrane from GDP-Man and Dol-P and flips (Flippase 2) to the lumenal monolayer where it functions as mannosyl donor for four mannosyltransferases in the DLO pathway and three mannosyltransferases in GPI anchor assembly. At least seven O-mannosyltransferases have been characterized in yeasts and mammals. The number of C-mannosyltransferases remains unknown. Following mannosyl transfer, Dol-P is released and returned to the cytosolic monolayer by Flippase 4.
There is substantial evidence supporting the hypothesis that the transbilayer diffusion of the dolichol-linked intermediates is facilitated by specialized membrane transporters, termed “flippases”. The highly hydrophobic nature of the inner core of the lipid bilayer presents a substantial biophysical barrier to the spontaneous passage of the hydrophilic head group of the amphipathic dolichol-linked intermediates.5,16–18 Although all of the glycosyltransferases required to assemble the DLO intermediates have been described, the identities of the proteins that catalyze the transmembrane movements of the dolichol-linked intermediates and the precise roles of two proteins involved in DLO assembly (Lec35 and RFT1) have not yet been clearly defined.
The description of this enigmatic class of membrane transporters remains a high priority. It is likely that currently undiagnosed congenital disorders of glycosylation (CDG)19–24 will be traced to defects in flippases in the protein glycosylation pathways. CDG is a rapidly expanding family of genetic diseases attributed to the defective synthesis of glycoproteins and glycolipids, and there are presently over 100 genetic diseases resulting from mutations in glycosylationrelated genes.
23
CDG patients present with a wide range of clinical symptoms varying from relatively mild, with primarily gastrointestinal effects, to very severe, involving multi-organ dysfunctions, skeletal abnormalities, and mental and psychomotor delay, which often result in embryonic lethality or infant mortality.
21
As many of these disorders are extremely rare, it is not presently known if the range of clinical symptoms can be considered diagnostic of a specific disorder or are due to the
Despite continued keen interest in the putative flippases of the protein glycosylation pathways of the ER, they remain undescribed due, primarily, to technical difficulties inherent in assaying their biochemical activity. Current analytical approaches are based on localizing a specific DLO intermediate to one leaflet of the bilayer by using chemical modification with a membrane-impermeant reagent,5,26,27 or lectin capture,5,28 or transport of water-soluble analogs containing short isoprenyl chains.29–32 The existing evidence suggests that the DLO flippases are energy independent and bidirectional and catalyze an extremely rapid equilibration of DLO intermediates between the monolayers, with half-times in the range of a minute or less. These properties, along with the inherent fragility of transbilayer asymmetry, pose formidable experimental challenges to using classical biochemical methodologies. This article reviews the information that is currently available concerning the flippase activities of the protein glycosylation pathways of the eukaryotic ER. Results and observations from some previously unpublished studies are included. All studies were conducted in accordance with federal guidelines and approved by the Institutional Animal Care and Use Committee of the University of Kentucky.
Man-P-Dol Flippase
As noted earlier, Man-P-Dol is synthesized on the cytosolic surface of the ER from GDP-Man and dolichol phosphate by Dpm1 in
The molecular description of the proteins involved in the transmembrane diffusion of Man-P-Dol has been a subject of interest for a number of years.35,36 However, due to the absence of a convenient biochemical assay for Man-P-Dol flippase activity, progress has been slow. Genetic approaches have also failed to identify likely flippase candidate proteins, including two recent screens in Hap1 cells (haploid mammalian cell lines) for defects in GPI assembly. Both of these screens identified all of the proteins known to participate in GPI formation but failed to identify any proteins that could plausibly be involved in the transmembrane flipping of either Man-P-Dol or glucosamine-PI.37,38 There are a number of potential explanations for this observation, including the possibility, as is true in yeast, that an essential protein is synthesized by a flippase requiring pathway, in this case an essential GPI-anchored protein,39,40 or that multiple genomic copies of flippase genes are present, making it difficult to eliminate all of the active transcripts. It is possible that flippases from other pathways possess overlapping substrate specificity, allowing complementation of the defect by other flippases, or the flippase activity may reside in a multitasking protein with another essential function as a second
Two strategies for the in vitro measurement of Man-P-Dol flippase activity have been developed. One approach, relies on the use of a water-soluble version of Man-P-Dol containing citronellyl phosphate (Cit-P), a short-chain analog of Dol-P with a 10 carbon isoprenyl chain and a saturated α-isoprene. This assay relies on the assumption that recognition of the polar head group and its movement across the bilayer are the most critical aspects of the flipping process and measures Man-P-Dol flipping as a simple transport process into sealed rat liver ER vesicles.29,32,43 This is a variation of the approach used by Bishop and Bell to assay flip-flopping of phosphatidylcholine in ER vesicles. 44
The selection of Man-P-Cit as an appropriate analog for Man-P-Dol is strongly supported by the observation that Man-P-Cit is a suitable substrate for all the Man-P-Dol-mediated mannosyltransferases in the DLO pathway 45 and in GPI assembly, 46 in O-mannosylation, 47 and in C-mannosylation (J. S. Rush, C. J. Waechter, and J. Hofsteenge, 2008, unpublished observation). Furthermore, Man-P-Cit was able to allosterically stimulate GlcNAc-P-P-Dol synthesis, 48 similar to native Man-P-Dol, and was able to cross the ER membrane and function as a mannosyl donor in streptolysin-O-permeabilized Lec35 cells under conditions in which the Man-P-Dol utilization defect was preserved. 34 Lec35 cells exhibit a defect in Man-P-Dol and Glc-P-Dol utilization that, apparently, does not involve dolichol-intermediate flipping. 34
Man-P-Cit is transported into the lumen of intact rat liver ER vesicles by a process that is time dependent, reversible, saturable, protease sensitive, stereoselective, enriched in the ER, sensitive to incubation with diethylpyrocarbonate (DEPC), and energy independent. 29 The activity was not observed in intact erythrocytes or synthetic liposomes 29 but was observed in proteoliposomes reconstituted from octyl glucoside and Triton X-100 soluble detergent extracts of rat liver ER membrane proteins. 43 Furthermore, the soluble detergent extracts could be fractionated by ion exchange and partially purified. 43 It is observed that when Man-P-Cit uptake in intact rat liver ER vesicles is measured over a range of temperatures, an Arrhenius plot of the rate of transport reveals an inflection point at ~16°C, correlating exactly with a corresponding inflection point in the rate of increase in the incorporation of [3H] mannose units into [3H]Man6–9-DLO from GDP-[3H]Man (which requires the participation of [3H]Man-P-Dol as an intermediate), performed in a parallel incubation (J. S. Rush and C. J. Waechter, 2007, unpublished observation). In this experiment, the increase in the initial rate of incorporation of [3H]mannose into [3H]Man-P-Dol is linear with respect to temperature, as would be anticipated for a process occurring on only one side of the membrane bilayer. This observation is consistent with Man-P-Cit uptake and Man-P-Dol flipping occurring by a transport process, which is affected similarly by membrane fluidity and supports the conclusion that Man-P-Cit transport accurately reflects flippase activity.
An alternative approach utilized native [3H]Man-P-Dol symmetrically reconstituted into sealed synthetic proteoliposomes and a membrane-impermeant oxidant (carboxy-2,2,6,6-tetramethylpiperidine 1-oxyl NO+) to selectively modify the [3H]Man-P-Dol molecules located in the outer monolayer of the synthetic vesicles.5,26 In this method, 50% of the [3H]Man-P-Dol in phospholipid vesicles lacking an active flippase will be oxidized. The remaining 50% is sequestered within the inner monolayer of the vesicle and is protected from reaction with the oxidant. However, in vesicles equipped with an active flippase, the [3H]Man-P-Dol facing the lumen of the proteoliposome is able to equilibrate into the outer monolayer and be oxidized. The result is that a larger fraction of the [3H]Man-P-Dol will be subject to oxidation. This study demonstrated rapid transbilayer movement of Man-P-Dol that is protein dependent, energy independent, DEPC sensitive, and highly specific for both the anomeric configuration of Man and a reduced α-isoprene unit. 26
Further studies showed that Man-P-Dol flippase activity could be resolved from flippase activities that mediate the transmembrane movements of M5-DLO (see Man5GlcNAc2-P-P-Dol flippase section) and glycerophospholipid flippase by concanavalin A (ConA) affinity and ion-exchange chromatography. 26 Limiting dilution of these detergent-soluble extracts into phospholipid vesicles leads to the conclusion that the Man-P-Dol flippase constitutes ~1% of the proteins in the detergent extract. 26
Flippase assays based on water-soluble analog transport have the advantages of simplicity and rapidity and allow near-initial rate estimations in the absence of detergents.29,32 The alternative method employing full-length Man-P-Dol avoids potential concerns arising from the use of structural analogs,5,26 but relies on the reconstitution of detergent-solubilized membrane preparations, and is potentially limited by the time constraint imposed by reaction with the mannolipid, which may significantly exceed the extremely rapid time course of transmembrane diffusion, even at 0°C (τ < 7 minutes).5,26 In this regard, it is important to note that the properties of the Man-P-Cit transport activity in rat liver ER vesicles were identical to the activity reconstituted from detergent extracts, so the probability that some essential intrinsic property of the flippase system is affected by detergent solubilization and reconstitution is very low. Although neither method has yet yielded a full molecular description of the Man-P-Dol flippase proteins, some preliminary properties have been identified and it is reassuring that the apparent properties of the flippase protein determined by the two methods are very similar. The complementary analytical methods identify an activity that is energy independent, bidirectional, stereoselective, sensitive to proteolysis and protein alkylation by DEPC and NEM, and amenable to fractionation by a variety of chromatographic approaches.
Glc-P-Dol Flippase
Glc-P-Dol is synthesized by Glc-P-Dol synthase (ALG5) using UDP-Glc as glucosyl donor and Dol-P as the lipid acceptor on the cytosolic surface of the ER but functions as a glucosyl donor in the synthesis of G3-DLO on the lumenal monolayer.49,50 The topological reorientation of Glc-P-Dol has been studied in sealed, ER-enriched microsomal vesicles from pig brain and rat liver by following the transport of a water-soluble Glc-P-Dol analog, Glc-P-citronellol (Glc-P-Cit). 30 Similar to the properties of the Man-P-Dol flippase reported earlier, Glc-P-Cit uptake in rat liver ER vesicles is time dependent, saturable, bidirectional, trypsin sensitive, and completely dependent on an intact permeability barrier. Furthermore, Glc-P-Cit is not taken up by synthetic liposomes or erythrocytes and Glc-P-Cit transport activity fractionates with heavy microsomes (ER-enriched) in brain. 30 The molecular description of the Glc-P-Dol flippase protein remains a high priority, as severely affected human patients with CDG attributable to defects in the enzymes that catalyze attachment of Glc units to the DLO intermediate (ALG6 and ALG8) have been identified and genetic defects in Glc-P-Dol flipping would be expected to have similar pathologies. 51
Man5GlcNAc2-P-P-Dol Flippase
M5-DLO is synthesized from Dol-P, UDP-GlcNAc, and GDP-Man on the cytosolic surface of the ER (Fig. 1) by two multienzyme complexes, consisting of DPAGT1, ALG13, and ALG14 that form GlcNAc2-P-P-Dol 10 and ALG1, ALG2, and ALG11 that add one β-mannosyl and four α-mannosyl units to form M5-DLO. 11 The transmembrane diffusion of the M5-DLO intermediate is particularly interesting due to the presumed difficulty involved in the passage of this large, extremely hydrophilic head group through the hydrophobic core of the ER membrane.28,52,53 The identification of the yeast RFT1 protein as a candidate M5-DLO flippase, primarily based on genetic evidence by Helenius et al, was an exciting development. 54 RFT1-depleted yeast cells accumulate large quantities of M5-DLO and are severely defective in protein N-glycosylation despite possessing normal levels of Man-P-Dol synthase and no apparent defect in the Man-P-Dol: M5-DLO mannosyltransferases (ALG3, ALG9, and ALG12). 54 However, direct biochemical assay of M5-DLO flippase activity by two independent methods indicates that although RFT1 is clearly required for proper assembly of the full-length DLO intermediate, it is dispensable for M5-DLO flipping in vitro.28,55–57
M5-DLO flipping has been measured directly in reconstituted proteoliposomes, employing a ConA binding assay. 26,28,55,56 In this assay, proteoliposomes are reconstituted from Triton X-100 soluble cellular extracts and exogenous phospholipids with traces of [3H]M5-DLO equally distributed between the two monolayers. When incubated with soluble ConA, the radiolabeled DLO in the outer leaflet of the proteoliposome is bound by ConA and cannot be extracted into organic solvent, whereas since ConA cannot spontaneously cross the membrane bilayer, the DLO in the inner leaflet is sequestered from the ConA and, as a result, is fully extractable. Ideally, in proteoliposomes lacking an active flippase, only 50% of the [3H]M5-DLO is extractable. In proteoliposomes containing an active flippase, unbound M5-DLO is able to equilibrate with the outer monolayer and become bound by ConA. Consequently, a smaller fraction of the intermediate is extractable in liposomes containing a flippase activity. Using this assay, M5-DLO flipping was shown to be extremely rapid (a rate constant of > 2 × 10-3 s-1), protein dependent, energy independent, bidirectional, and protease sensitive. Significantly, the M5-DLO flippase activity could be resolved from an unrelated phospholipid flippase activity by several chromatographic methods. 56 Furthermore, the M5-DLO flippase activity preferred the natural M5-DLO isomer over a catabolic M5-DLO intermediate, with an alternative branching pattern, and a biosynthetic M7-DLO intermediate demonstrating a high degree of substrate specificity. 56 A further report showed that M5-DLO flipping was inhibited by the inclusion of GlcNAc2-P-P-Cit, a water-soluble M5-DLO analog, and that whereas M5-DLO was rapidly flipped with a time constant τ < 2 minutes, the triantennary intermediate M3-DLO flipped much more slowly with τ < 200 minutes. DLOs larger than M5-DLO also flipped slowly with τ values ranging from 10 to > 200 minutes. 28 Importantly, this activity was present in Triton X-100 soluble extracts derived from cells in which RFT1 had been depleted and was not immunoprecipitated with specific antibody to epitope-tagged RFT1.28,55 In addition, M5-DLO flippase activity could be resolved from the epitope-tagged RFT1 protein by velocity sedimentation, ion-exchange, and blue dye resin chromatography.28,55
In another set of studies, sealed ER vesicles from RFT1-depleted yeast cells were fully capable of elongating [3H]GlcNAc2-P-P-Dol, synthesized in situ, to form [3H] M9-DLO in vitro when incubated with nonradioactive GDP-Man. 57 As this elongation reaction cannot occur without translocation of the DLO intermediate to the lumenal compartment, this is strong evidence that the RFT1-depleted microsomes are still capable of flipping M5-DLO. GlcNAc2-P-P-Dol has been shown to be flipped, presumably by the M5-DLO flippase in microsomes from a variety of tissues and yeasts.58–63 To measure M5-DLO flipping directly in sealed yeast ER vesicles, the transport of [3H]GlcNAc2-P-P-Dol15, a water-soluble analog of GlcNAc2-P-P-Dol with a 15-carbon α-reduced isoprenol chain, was measured. RFT1-depleted, ER-enriched yeast microsomes transported water-soluble [3H]GlcNAc2-P-P-Dol15 into the lumen of the sealed vesicles at an even faster rate than intact vesicles from undepeleted cells. 57 Furthermore, when the sealed RFT1-depleted microsomal vesicles were incubated with a membrane-permeant N-glycosylation acceptor peptide and [3H]GlcNAc2-P-P-Dol15, the peptide was glycosylated with [3H]GlcNAc2, presumably by the lumenal oligosaccharyltransferase (OST), demonstrating that the water-soluble analog has access to the lumenal compartment. 57 Finally, in an indirect assay for the recycling of Dol-P, 14 it is observed that when sealed RFT1-depleted yeast microsomal vesicles are incubated with a membrane-permeant N-glycosylation acceptor peptide, a very large amount of newly formed Dol-P is generated (J. S. Rush and C. J. Waechter, 2008, unpublished observation). This assay was previously used to successfully establish the biochemical mechanism, by which Dol-P derived from Dol-P-P, generated by the N-glycosylation reaction, is returned to the cytosolic monolayer for further rounds of DLO synthesis 14 and is strong evidence that a large portion of the accumulated M5-DLO can enter the lumen of the ER in vesicles prepared from RFT1-depleted cells and become accessible to OST.
This biochemical evidence strongly suggests that RFT1 is not an essential subunit of the M5-DLO flippase in ER vesicles isolated from disrupted cells. Recently, this conclusion has been further supported by the observation that mutant
Dol-P Flippase
Dol-P is formed on the lumenal surface of the ER membrane from several biosynthetic processes, including (1) cleavage of Dol-P-P generated during the OST reaction of protein N-glycosylation, (2) the three glucosyltransferase reactions utilizing Glc-P-Dol, and (3) by all of the Man-P-Dol-mediated mannosyltransferase reactions (the four DLO mannosyltransferases, the three GPI anchor mannosyltransferases and C- and O-mannosylation; see Fig. 2) utilizing Man-P-Dol as mannosyl donor.6,14 This lumenally oriented Dol-P must be returned to the cytosolic surface of the ER in order to participate in further rounds of glycan synthesis. The recycling of Dol-P substantially contributes to the maintenance of biologically relevant rates of protein N-glycosylation, as a significant reduction of glycosylation is observed in the yeast DolPP1 mutant, CWH8, even though this mutation presumably affects only the recycling of Dol-P from DLO and not the substantial number of Dol-P molecules arising from the other glycosyltransferase reactions. 14 Despite the clear significance of the recycling of the lipid anchor, the molecular and biochemical details of this process remain virtually unknown. It will be worth exploring the possibility that CWH8 (DolPP1) plays a role in the translocation of Dol-P back to the cytosolic surface of the ER.
Presumably, Dol-P is returned to the cytoplasmic monolayer by a flippase-mediated process, as the amphipathic lipid anchor is unable to spontaneously cross the hydrophobic interior of the membrane bilayer. However, the identity of this flippase has not been established. Indeed, it is not known if multiple, unique flippases are associated with the various glycosyltransferases and the DolPP1 involved in the production of lumenal Dol-P mentioned earlier. A membrane-associated activity that transports Cit- 32 P (a water-soluble analog of Dol-P) into the lumen of sealed vesicles has been detected (J. S. Rush and C. J. Waechter, 2006, unpublished observation), as shown in Figure 3. When sealed rat liver ER vesicles are incubated with Cit- 32 P, the analog is rapidly transported into the lumen of the vesicle with an apparent half-time of uptake of ~23 seconds (Fig. 3, panel A). When the ER vesicles are first loaded with Cit- 32 P by preincubation and then diluted into isotonic buffer at room temperature, the internalized Cit- 32 P effluxes from the vesicles with a half-time of exit of ~30 seconds, showing that the uptake process is fully reversible (Fig. 3, panel A). Cit- 32 P does not spontaneously leak out of protein-free phospholipid vesicles, as shown in Figure 3, panel B (half-time of exit ~94 hours). Preliminary studies indicate that as with the other ER-associated flippase activities, this activity is membrane protein dependent, time dependent, protease sensitive, and energy independent (J. S. Rush and C. J. Waechter, 2006, unpublished observation). The identity of the protein mediating the return of Dol-P to the cytosolic monolayer has not yet been determined, but recycling by an antiport mechanism via one of the other flippase activities seems unlikely, as Cit-P does not have any appreciable effect on the uptake or efflux of any of the other water-soluble analogs employed in flippase assays.
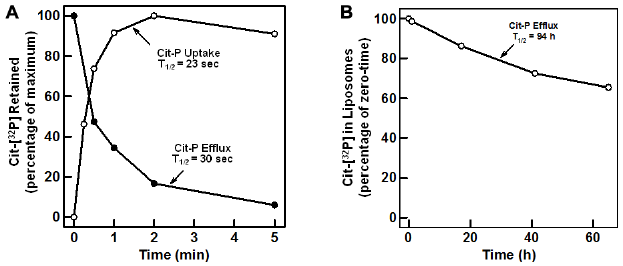
Transport of 32 P-Cit into the lumen of intact rat liver ER vesicles. Panel A, open circle (●), 32 P-Cit was incubated with sealed rat liver ER vesicles at 22°C for the indicated periods of time. Following incubation, the transport reactions were diluted with 0.5 mL ice-cold buffer (10 mM Tris-CL, pH 7.4, 0.25 M sucrose) and filtered through a Millipore 0.45 µm HA filter. The filters were washed with an additional 10 mL ice-cold buffer and assayed for radioactivity, as described earlier. 29 Panel A, closed circle (•), sealed rat liver ER vesicles were pre-incubated with 32 p-Cit for 10 minutes and diluted with 0.5 mL, 10 mM Tris-Cl, pH 7.4, and 0.25 M sucrose all at 22°C. At the indicated times, the diluted reactions were filtered through a 0.45 µm HA filter, washed with an additional 10 mL ice-cold buffer, and assayed for radioactivity. Panel B, large unilamellar synthetic phospholipid vesicles were reconstituted from octyl glucoside soluble phospholipids in the presence of 32 P-Cit, as described elsewhere.43,67 The vesicles containing 32 p-Cit entrapped in the lumen were incubated with 10 mM Tris-Cl, pH 7.4, and 0.25 M sucrose at 22°C for the indicated periods of time and chromatographed on a 30 mL column of Sephadex G-50 equilibrated in 10 mM Tris-Cl, pH 7.4, and 0.25 M sucrose. Fractions of 1 mL were collected and monitored for radioactivity by scintillation spectrometry. The percentage of the radioactivity eluting in the void volume of the column is plotted versus time of incubation.
Concluding Comments
The transbilayer diffusion of Man-P-Dol in the ER membrane was first investigated by Haselbeck and Tanner using an elegant enzyme-coupled assay over 40 years ago.35,36 Despite continued interest in the mechanism of Man-P-Dol flipping in the ER, the proteins that participate in this essential step involved in C- and O-mannosylation, DLO synthesis, and GPI anchor formation in the ER remain unidentified. The fragility of the topological asymmetry of the lipid bilayer and the apparent physical properties of the putative flippases have presented substantial experimental difficulties to the direct measurement of flippase activity in vitro. The biochemical assays reviewed here are important advances and have led to the identification of a number of physical properties of the prospective flippases. In common with the glycerophospholipid flippases present in biogenic membranes, the ER dolichol-intermediate flippases are bidirectional and energy independent and catalyze an extremely rapid reequilibration of the intermediates between the two monolayers. Hypothetical models for flippase action have been proposed by Kol et al, 65 Sanyal and Menon,5,26 and Ernst and Menon. 66 It is possible that the translocation events are energy independent for lipid intermediates because the flipping is driven by mass action, as the intermediates are rapidly utilized at the lumenal sites. It is also reassuring that although the experimental approaches employed in these assays differ significantly, the individual studies identify very similar physical properties, which are also consistent with observations from coupled in vitro enzymatic systems. The development of reliable methods for the solubilization, fractionation, and functional reconstitution of the flippase activities coupled to proteomic approaches paves the way for the purification, identification, and characterization of these final undescribed steps in protein glycosylation in the ER.
Author Contributions
Conceived and designed the experiments: JSR. Analyzed the data: JSR. Wrote the first draft of the manuscript: JSR. Developed the structure and arguments for the paper: JSR. Made critical revisions: JSR. The author reviewed and approved of the final manuscript.
Footnotes
Acknowledgments
I am grateful to Skip Waechter (University of Kentucky College of Medicine) for his support and encouragement and for the opportunity to participate in a portion of the work reviewed in this article and to Dr. Dean C. Crick (Colorado State University), Dr. Hudson H. Freeze (Burnham Institute, La Jolla, CA), and Dr. Anant K. Menon (Weill Cornell Medical College) for critical comments.
