Abstract
The GABA-A receptor has been strongly implicated in the organization and function of cortical sensory circuits in the adult mammal. In the present work, changes in the expression patterns of select GABA-A subunits were examined as a function of development. The RNA expression profiles for three subunit types were studied, α1, β2/3 and δ at four developmental time points, (p0, p15, p30 and p90). The o1, β2/3 subunits were present at birth and following a modest increase early in life; mRNA expression for these subunits were found at stable levels throughout life. The expression pattern for the δ subunit showed the most dramatic changes in the number of positive cells as a function of age. In early life, p0 through p15 expression of mRNA for the δ subunit was quite low but increased in later life, p30 and p90. Together these data suggest that much of the potential for inhibitory connectivity is laid down in the pre and early post-natal periods.
Introduction
Research directed at understanding the development of central sensory pathways must frequently tackle the extent to which experiential factors and genetically-imprinted mechanisms determine the connectivity and functional organization of brain circuits. Particular molecular scripts are engaged by development with such reliability that their expression is synonymous with the arrival of certain post-natal dates, whereas the expression schedule of other gene products appears to have a strong relationship with sensory experience. Understanding the pressures that shape the final, adult organization of auditory pathways is believed to reside in the crossroads of these two dominant developmental forces.
To date, evaluating the progression of structural-functional changes of the ascending auditory pathway has placed a heavy emphasis on how excitatory signals of frequency-related information are transmitted and used in auditory processing. It is however, widely appreciated that inhibitory networks that are present at all levels of the auditory central nervous system are essential to the functional refinement and stability of these circuits 13 and may be key to enabling these circuits to extract higher order information from crudely encoded auditory signals. 4 An important part of this refinement process is the addition or replacement of select subunits in the pentameric structure of GABA-A receptors.5,6 Changes in subunit composition and/or the appearance of these subunits can lead to pronounced functional changes in channel performance, including alterations in the probabilities of opening or duration of open times, to dramatic reassignment of a channel from excitatory to inhibitory.7,8
Here, we examined changes in the expression patterns of selected GABA-A receptor subunits in the mouse auditory cortex as a function of postnatal development. Three classes of sub-units α, β, and δ were chosen with the most representative and/or most abundant form of sub-unit used for this investigation. The α1 subunit was chosen for the a sub-unit class given its abundance in sensory cortex and the potential for identification of this sub-unit in all cortical layers. Despite an average 70% homology between the a subunit and other sub-units in this class, it is known that exchanging this sub-unit is associated with development and produces detectable functional differences in the GABA receptor. 5 In the selection of the β2/3 sub-unit, the choice in variants is narrowed as this is the structure that is associated with GABA binding and therefore must be largely conserved. The β2/3 sub-unit was chosen for this study because its inclusion is essential for a functional GABA channel and as such provided a natural ceiling measurement of the numbers of cells expressing GABA-A receptors in our different age groups. Finally, the δ sub-unit, as opposed to other single form sub-units was chosen for one main reason, this sub-unit is the location of neurohormone based neuromodulation, a feature that promises to dramatically impact cell function by its presence in auditory circuits. 9
Our findings show that above p0, there is relatively stable expression of the α1 and β2/3 subunits throughout post-natal development, whereas the δ subunit mRNA becomes strongly apparent between p15 and p30, and remains highly expressed into adulthood. These findings suggest that inhibitory transmission is central to shaping the development and function of the mammalian auditory cortex.
Material and Methods
We looked at the expression patterns of α1, β2/3 and the δ subunits at post-natal day 0 (P0), P15, P30 and P90 (n = 3/age group). The ages examined were chosen to determine if and how differences in expression patterns coincides the key developmental stages of the central auditory pathways, both before and after hearing onset. All procedures were approved by the University Committee on Animal Resources of the University of Rochester, and are in accordance with NIH guidelines.
The expression of different receptor subunits was determined by fluorescence in situ hybridization (FISH); our methodology has been described in detail previously. 10 Briefly, purification of plasmids containing fragments of the mouse α1, δ, and β2/3 genes was carried out from bacteria with a Qiagen miniprep kit. Plasmids were linearized by digestion with appropriate restriction enzymes. Purification of cDNAs enabled their use as templates for the generation of digoxigenin (DIG)-labeled antisense and sense strand riboprobes, via in-vitro transcription, using standard methods that we have previously published.10 Brain sections mounted on charged slides were first rapidly incubated in a 4% paraformaldehyde solution in 0.1 M PBS (5 min × 2) then rinsed in 0.1 M PBS. Mounted sections were then dehydrated in a standard alcohol series from 70% to 100% ethanol and left to air dry away. Samples were then incubated at room temperature (RT) in an acetylation solution (1.35% triethanolamine and 0.25% acetic anhydride in water) for a total of 10 mins. Next we rinsed the preparation in 2x SSPE, and then repeated the dehydration in alcohol (series) and air dried. Sections were then incubated separately in 16 μl of a hybridization solution that consisted of 50% formamide, 2x SSPE, 2 μg/μl tRNA, 1 μlg/μl BSA, 1 μg/μl poly(A) in DEPC-treated water, and 1 ng/μl of our DIG-labeled probe of interest (either sense or antisense). Slides were then coverslipped, sealed by immersion in mineral oil bath, and incubated overnight at 62°C. The next day, sections were rinsed in chloroform, decoverslipped in 2x saline-sodium phosphate-EDTA (SSPE), and sequentially washed in 2x SSPE for 1 h at RT, followed by 2x SSPE containing 50% formamide for 1.5 h at 62°C. Subsequently, sections were rinsed in 0.1x SSPE twice for 30 min at 62°C. Samples were then incubated in 0.3% hydrogen peroxide in TNT buffer (0.1 M Tris-HCl, pH 7.4, 5 M NaCl, and 0.05% Triton X-100 in DEPC-treated water) for 10 min, washed in TNT buffer (three times for 5 min), and incubated for 30 min in TNB buffer (TNT plus 2 mg/ml bovine serum albumin). As a next step, sections were incubated in a solution containing a peroxidase-coupled anti-DIG antibody for 2 h at RT (1:200 dilution in TNB; Roche Diagnostics), washed in TNT buffer (three times for 5 min), and incubated in a solution containing tyramide-linked Alexa-488 in amplification buffer provided by the manufacturer (Invitrogen). Sections were again washed in TNT buffer, three times for 5 minutes each. Sections were counterstained with Hoechst, washed in TNT, and coverslipped with Vectashield (Vector Laboratories) for histological analyses. Control experiments using the sense strand riboprobes for all genes studied did not result in signal.
Expression of α1, β2/3 and δ GABA-A receptor subunits in cortical layers of the primary auditory cortex, across key post-natal developmental stages.

FISH quantification and figure preparation
Histological sections were viewed with a Nikon microscope equipped with appropriate filters and a motorized stage, and coupled to a personal computer. To estimate the number of GABA subunit-positive neurons per unit area, neurons were counted as profiles within a grid of 100 × 100 μm squares that was superimposed over the target area of FISH-labeled sections. A minimum of 10 randomly selected, non-overlapping squares were quantifed per hemisphere, for each animal. The values obtained for each square were separately averaged for each hemisphere of individual animals and, subsequently, within groups. Comparisons across groups were performed using a standard ANOVA with a criterion of significance set at
Results
Subunit specific-positive neurons in the supragranular (II/III), granular (IV) and infragranular (V/VI) layer of primary auditory cortex (A1) were quantified at four developmental time points P0, P15, P30 and P90. Expression was assessed via FISH, as described above and detailed previously. 10
α1 subunit expression
The α1 subunit was expressed from early in development. We found that 8.5 ± 0.6 (mean ± SE), 9.2 ± 1.1 and 10.0 ± 0.7 α1-positive cells could be detected in the supragranular, granular and infragranular layers, respectively. By p15, α1 expression could be detected in 16.2 ± 0.8, 15.3 ± 1.5 and 18.0 ± 1.2 cells/area, a density that was not significantly different from p0 animals (
β2/3 subunit expression
The β2/3 subunit houses the actual binding site for GABA and modulators of the channel function. At p0, we found that 15.9 ± 1.6, 16.3 ± 1.4 and 18.3 ± 1.8 neurons expressed β2/3 in the supragranular, granular and infragranular layers of A1, respectively. At p15, we detected 17.1 ± 1.9, 15.7 ± 1.6 and 16.1 ± 1.7 neurons/area that were positive for β2/3, a value that was not significantly different from that observed at p0 (

In situ hybridization directed against the α1 subunit of the GABA-A receptor. Shown are high power images depicting α1 expression at p0 (
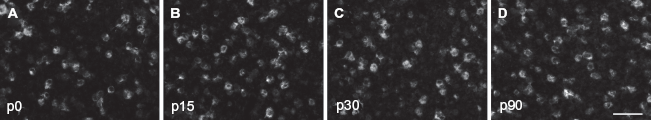
In situ hybridization directed against the β2/3 subunit of the GABA-A receptor. Shown are high power images depicting β2/3 expression at p0 (
δ subunit expression
The expression pattern for the δ subunit showed strong post-natal developmental regulation. At p0, we found that 3.1 ± 1.4, 3.7 ± 1.2 and 4.2 ± 1.7 cells were δ-positive in supragranular, granular and infragranular layers, respectively. These numbers significantly increased in the p15 animals. On average, we found 14.2 ± 1.6, 15.8 ± 1.4 and 16.1 ± 1.7 cells per area across cortical layers (
Discussion
Our findings show that whereas α1 and β2/3 subunit expression patterns are stable after birth, the expression of the δ subunit is highly increased throughout post-natal development of the auditory cortex. With respect to expression of the α1 subunit mRNA, we found it expressed immediately after birth. This finding is perhaps not surprising as it has recently been shown that co-tuned excitatory and inhibitory inputs converge upon A1 neurons by hearing onset, and that strong feed-forward inhibitory circuitry from the thalamus is established through genomic mechanisms and not dependent on auditory experience. 11 It has, however, been argued that hearing onset may be required for the maturation of synaptic properties of inhibitory transmission. 12
In its most prevalent form, the GABA-A receptor will have two α1 subunits per functional channel. 13 We hypothesized that as the circuit became functionally refined there would be a corresponding drop in α1 expression, as new alternative α subunits were added. This hypothesis was not supported by our results; instead, any modest decline observed for the p90 age group most likely refects the impact of age. 14 One consideration is that in the current study, expression profiles were examined at the mRNA level only. It is possible that mRNA for α1 is constitutively expressed in A1 neurons whereas alternate α subunits may only be called upon following sensory experience and/or in relationship to learning or memory. 15 These ideas, while speculative, warrant further inspection in future work.
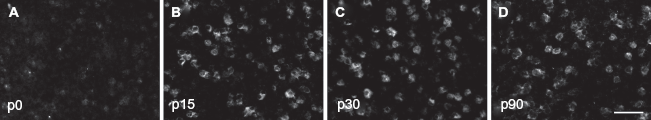
In situ hybridization directed against the δ subunit of the GABA-A receptor. Show are high power images depicting A1 expression at p0 (
Investigation into the impact of development on the expression of the β2/3 subunits was initiated to provide a measure that would reflect the total density of GABA-A receptors for any given developmental stage. This approach was intended to also serve as a reference against which the expression of other subunits could be evaluated. As the binding site for GABA, the β2/3 subunit must be expressed in all functional GABA-A receptors. Changes in the expression of this subunit might suggest places where greater or lesser inhibitory inputs are required to promote functionality of auditory circuits. 16 Analysis of transcriptional activity and its changes as a direct result of auditory experience was beyond the scope of this study. In general, one might expect attenuated β2/3 expression with advancing age as work from other groups has already shown loss of inhibitory neurons in mice approaching senescence. Such a decrease, however, was not detectable in this study as a reduced number of neurons expressing β2/3 mRNA. It is possible that β2/3 expression decreases at later (>p90) developmental stages.
Expression patterns of the δ subunit showed the most changes of all the subunits examined in the present work. In particular, δ subunit expression was relatively low at birth and significantly and progressively increased until p30. No changes were detectable between the p30 and p90 time points. Previous research has shown that expression of this subunit is associated with extra-synaptic GABA-A receptors that have been implicated as a substrate for shunt inhibition and other means of regulating neuronal excitability.17,18 Future research will be focused at addressing the extent to which changes in expression of this subunit are driven by changes in neuronal activation related to the processing of sound or auditory learning. Elucidating this relationship will be an important advance in understanding how inhibitory mechanisms are changed throughout life to shape or tailor auditory processing to a particular developmental stage.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conflicts of interest. The author confirms that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
Work supported by University of Rochester and the University of Oklahoma Health Science Center start-up funds and NIH.
