Abstract
We here report on the immunolocalization of Dynamin I (Dyn I) in neurons of the visual system of the cat. The lateral geniculate nucleus (LGN) complex displayed abundant Dyn I immunoreactivity in typical relay cells of the X-, Y- and W-pathway. The superficial and deep layers of the superior colliculus were also populated by Dyn I-immunoreactive projection neurons of the W- and Y-cell system. In primary visual areas 17 and 18, many densely packed layer VI neurons were intensely stained. A clear Dyn I signal was also demonstrated in pyramidal neurons of supragranular layers II and III, while layer IV displayed low Dyn I immunoreactivity. Additionally, area 18 displayed larger border pyramidal neurons in layer III compared to area 17. Generally, Dyn I was localized to the cell body and dendrites of neurons, to the neuropil and sometimes also to axon bundles. Typically, the Dyn I signal was not always uniformly distributed within the somatodendritic compartment. Based on its widespread distribution mainly in projection neurons Dyn I may play a fundamental role in mature neurons of different cortical and subcortical structures of the adult mammalian brain.
Introduction
In mammals, Dynamin (Dyn) proteins form a family of large GTPases in which the neuron-specific isoform is called Dyn I, the ubiquitously expressed form is called Dyn II and the isoform exclusively expressed in testis, lung and brain is referred to as Dyn III. 1 Almost two decades ago, Dyn was first isolated from calf brain and identified as a putative microtubule-associated motor protein. 2 Soon thereafter, it became clear that Dyn functions as a key molecule in synaptic vesicle endocytosis and recycling,3,4 an essential process for the continuity of cell-to-cell communication in the nervous system. From then on, the knowledge on Dyn expanded significantly by explorations of its developmentally regulated expression, 5 7 its sequence and structure 8 10 and by investigations of the mechanism by which this molecule pinches off vesicles. 11 13
Next to a role in stimulus-dependent synaptic vesicle endocytosis 14 16 Dyn I was attributed a role in preserving the cell morphology of mature neurons. Reduction of Dyn I expression levels through antisense oligonucleotide treatment in cultured neurons indeed impaired neurite formation, whereas neurite growth was accompanied by increased Dyn I expression.1,5,6
A recent large-scale proteomics screen for developmentally regulated proteins in cat visual cortex identified low expression levels for Dyn I shortly after birth, that increased significantly towards adulthood. 17 19 Given the fact that proteins with a developmentally regulated expression might play a role in neuronal plasticity and based on the presumed role for Dyn I in neurite growth, we initiated investigations on the effect of visual deprivation on the expression level of Dyn I19,20 as well as a parallel immunocytochemical investigation to define the cellular expression of Dyn I throughout the cat visual system in order to define possible restrictions to one or more functional classes of neurons. Cellular Dyn I distribution has so far been determined for rat cerebellum and hippocampus.5,21,22 We here report for the first time the neuron-specific immunolocalization of Dyn I in the neocortex, midbrain and thalamus of a mammal. Our results indicate that Dyn I exhibits a widespread distribution particularly in projection neurons of the visual system of the adult cat.
Materials and Methods
Animal care and tissue preparation
All experiments were conducted following the European Communities Council Directive of 24 November 1986 (86/609/EEC), and carried out in accordance with institutional animal welfare guidelines (K.U. Leuven, Belgium). These rules were followed strictly in all experiments. All efforts were made to minimize the animals’ discomfort and to reduce the number of animals.
Brains of six adult cats were used. All animals were deeply anaesthetised with ketamine hydrochloride (Ketalar, 10 mg/kg i.m.) prior to sacrifice with an overdose of sodium pentobarbital (60 mg/kg, i.v). For immunocytochemistry, three animals were perfused transcardially with 0.9% saline, immediately followed by cold 4% paraformaldehyde (Sigma Aldrich, St. Louis, MO) in 0.15 M sodium phosphate buffer (PBS, pH 7.4). Brains were removed from the skull, postfixed for 24 h, rinsed for 24 h, and stored at 4 °C in PBS. Coronal sections (50 μm) of cerebrum and cerebellum were cut on a vibratome (Leica VT 1000E, Van Hopplynus, Heidelberg, Germany) and collected in serial order. Series of free-floating sections were processed for immunocytochemistry. For Western analysis, three brains were immediately collected upon sacrifice, instantly frozen by immersion in dryice cooled 2-methylbutane (Merck Eurolab, Leuven, Belgium) and stored at –70 °C until use. Coronal sections (200 μm) were cut on a cryostat (Microm HM 500 OM, Waldorf, Germany), thaw-mounted onto baked slides and stored at –70 °C.
Antibody
The Dyn I (C-16) antibody (sc-6402; Santa Cruz Biotechnology, Heidelberg, Germany) is an affinity purified goat polyclonal antibody raised against a peptide mapping at the carboxy terminus of human Dyn I. As provided by the supplier, this antibody reacts with Dyn I of mouse, rat and human origin and does not cross-react with Dyn II.
Western blotting
To prove antibody specificity for cat brain tissue, Western blotting was performed as described earlier, with minor changes. 23 Briefly, grey matter containing all six layers of cat visual cortex (area 17) was isolated from 200 μm cryosections and homogenized in lysisbuffer (2% SDS, 50 mM Tris-HCl, 10% glycerol, pH 6.8). After sonication, the sample was heated for 5 min at 70 °C and centrifuged for 30 min at 10,000 rpm. Protein concentration was determined using the Micro BCA Protein Assay Reagent Kit (Perbio, Erembodegem, Belgium). Protein samples (a ½ dilution series starting from 15 μg) were loaded onto a 4%–12% NuPage gel (Invitrogen, Groningen, The Netherlands). After electrophoresis, proteins were blotted onto a PVDF membrane (Bio-Rad, Nazareth, Belgium) and blocked in 5% ECL blocking solution (GE Healthcare, Roosendaal, The Netherlands). Overnight incubation at 4 °C with anti-Dyn I (1/1,000) together with 1% blocking solution, was followed by incubation with HRP-conjugated donkey-anti-goat (1/100,000; Jackson ImmunoResearch, Cambridgeshire, UK). Immunoreactive proteins were visualized by chemiluminescent detection (ECL-plus kit, GE Healthcare).
Immunocytochemistry
Series of free-floating vibratome sections of three animals were first pretreated with 0.1% trypsine (Sigma Aldrich). This pretreatment was a prerequisite to reveal a high-quality Dyn I signal in the neocortex, and was therefore applied in all our experiments, resulting in an increased membrane permeability and thus a better exposure of the Dyn I epitope.24,25
This was followed by 3% H2O2 incubation for 20 min to block endogenous peroxidase activity. Non-specific binding was prevented by incubating the sections with 10% normal donkey serum (Chemicon, Biognost, Belgium) together with 5% bovine serum albumin for 45 min. Subsequently, overnight incubation with primary antibody was performed with goat anti-Dyn I (1/100, Santa Cruz Biotechnology) together with 1% BSA at 4 °C. The next day, sections were placed in biotinylated donkey-anti-goat (1/500, Santa Cruz Biotechnology) serum for 30 min. After incubation with streptavidin-HRP (1/500, Dako, Glostrup, Denmark), the immunoreactive signal was visualized using the glucose oxidase-diaminobenzidine (DAB)-nickel method. 26 All rinses were carried out in PBS (0.01M, 0.9% NaCl, pH 7.5), while all incubations were done in the same PBS buffer with 0.1% Triton-X-100 under gentle agitation at RT. Finally, all sections were mounted on gelatin-coated slides, air-dried, dehydrated, cleared, cover-slipped and viewed through a Leitz DM RBE microscope (Leica, Leitz Instrument, Heidelberg, Germany). The retinotopic maps of Rosenquist 27 and the stereotaxic atlas of the cat from Snider and Niemer 28 were respectively used for the localization of visual cortical areas and subcortical structures.
Immunocytochemical controls included incubation of the sections (1) omitting primary or secondary antibody, (2) substituting generic donkey IgG protein at an equivalent concentration to the primary antibody, and (3) testing the effect of serial dilutions of the primary antibody.
Neuronal specificity of the Dyn I signal in cat brain was verified
32
by performing single and double immunocytochemcial stainings for neuronal and glial cell markers (Figs. 1E–H). Upon pretreatment with 0.3% H2O2 (20 min) and preincubation with normal goat serum (NGS, 1/10, 45 min) the control sections were incubated with one of the primary cell type marker antibodies: anti-SMI-31 antiserum (1/1000; Covance) or anti-NF160 (1/1000; Boehringer Ingelheim) for neurons and anti-GFAP (1/2000; Prosan) for glial cells. The next day these antibodies were detected with biotinylated goat anti-rabbit IgGs (1/500, 30 min, Dako, Glostrup, Denmark) or biotinylated goat anti-mouse IgGs (1/200, 30 min, Dako), and peroxidase-conjugated streptavidin (1/600, 30 min, Dako). The peroxidase labeling was visualised using 3,3′-diaminobenzidine, which produced a brown reaction product in the cytoplasm and processes of immunoreactive cells. The sections were dehydrated, coverslipped and viewed through a DM RBE microscope. For GFAP and Dyn I additional double stains were performed with consecutive detection of Dyn I in gray and GFAP in brown (Fig. 1H). All the tests support neuron-specific Dyn I expression.
Results
Specificity of Dyn I antibody
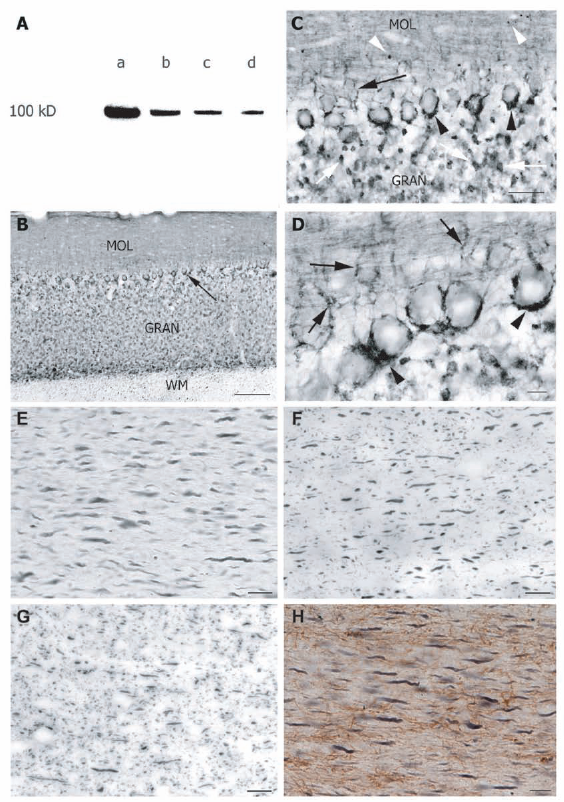
The specificity of the Dyn I labeling for cat brain tissue has not been demonstrated. We therefore performed Western analysis on cat visual cortex samples. Four protein concentrations were loaded and showed a decreasing signal according to the dilution gradient (Fig. 1A). The Dyn I antibody reacted with a single protein band of 100 kDa consistent with the molecular weight of Dyn I protein as observed for mouse, rat and human brain. 29 32 This was also the observed size of Dyn I in cat visual cortex as detected in our previous 2–D DIGE studies. 17
In addition, to further control the specificity of the Dyn I signal, stainings on the cat cerebellum were undertaken and resulted in a pattern comparable to the Dyn I immunoreactivity as found in rat cerebellum.21,22 In cat, Dyn I expression was intense in the granular and Purkinje cell layer of the cerebellar cortex (Figs. 1B–D). Much less Dyn I immunoreactivity was seen in the white matter where only short axonal segments were labeled (Fig. 1B). In the granular layer, mossy fiber glomeruli were Dyn I-positive, while the molecular layer demonstrated Dyn I-immunoreactive neuropil as well as some signal in cross-cut parallel fibers (Figs. 1B, C). The faintly stained cell bodies of the Purkinje cells were nicely outlined by the intensely stained, surrounding basket cell axons from the molecular cell layer (Figs. 1C, D), as previously demonstrated by Powell and Robinson 22 in rat cerebellum. These cells from the molecular layer make axo-somatic and axo-axonal synaptic contacts onto Purkinje cells like a basket, called a pinceau.33,34
The presence of Dyn I in neuronal but not in glial fibers was also assessed via single and double staining for the axonal marker SMI-31, the neuronal marker NFP160 and the astrocyte-specific protein GFAP (Figs. 1E–H). The morphology, size and orientation of Dyn I immunoreactive fibers resembled those of SMI-31 and NFP160 positive fibers (Figs. 1E–G). The GFAP expressing fibers clearly had a smaller diameter. Together with the lack of double staining for GFAP and Dyn I these observations confirmed that Dyn I is only expressed by neurons in cat brain.
We used this neuron-specific Dyn I antibody to perform immunostaining in three representative visual system structures of the cat, namely the LGN (Fig. 2), the SC (Fig. 3) and the primary visual cortex (area 17, area 18) in which it labeled a variety of neurons (Fig. 4).

Dyn I expression in the LGN
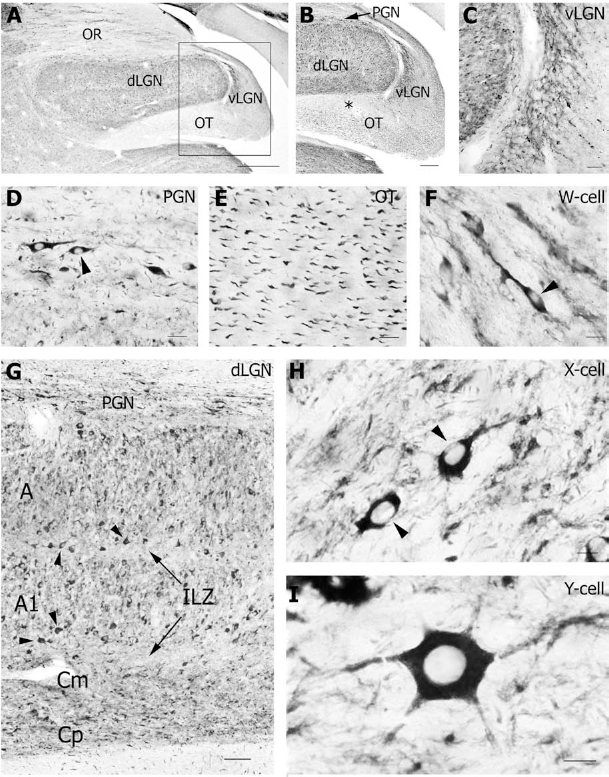
The Dyn I antibody stained numerous neurons in the LGN complex of the thalamus (Fig. 2A). All laminae of the dLGN contained many Dyn I-positive neurons and showed an obvious neuropil staining (Figs. 2A, B, G). The magnocellular laminae (A, A1 and Cm) contained medium-sized to large Dyn I-positive neurons, while Dyn I was also prominent in smaller neurons of the densely packed parvocellular C laminae (Cp) (Fig. 2G). The medium-sized neurons demonstrated an oval cell body with bipolar, elongated dendritic arbors (Fig. 2H). The large Dyn I-immunoreactive neurons were characterized by a voluminous polygonal cell body with six or more radially disposed primary dendrites (Fig. 2I). The latter neurons were often positioned near the border of the interlaminar zone (ILZ) (Fig. 2G). The ILZ mainly contained Dyn I-positive neuropil.
Also the ventral part of the LGN (vLGN) was clearly immunoreactive for Dyn I (Figs. 2B, C). Immunopositive signal for Dyn I was seen as a dense fibrillary network (Fig. 2C) together with some small cell bodies (Fig. 2F). The perigeniculate nucleus (PGN) that covers the dLGN dorsolaterally, displayed Dyn I labeling in the cell soma and the proximal part of horizontal dendrites of PGN interneurons (Fig. 2D). Axonal Dyn I expression was observed in axon bundles in the optic radiation (OR) (Fig. 2A) and optic tract (OT) (Fig. 2E). In the OT, axons were only very weakly stained just below the dLGN, while the more medial part of the OT was more strongly labeled (Fig. 2B).
A remarkable feature that we observed in neurons of the geniculate nuclei, but also in many other Dyn I-positive neurons (see below) was the unequal dispersion of the immunosignal over the soma of the neurons (Figs. 2D, F, H). Although all neurons were clearly outlined by the immunostaining, small parts of the cytoplasmic compartment of neurons definitely remained immunonegative for Dyn I. In addition, the nucleus and nucleolus were always devoid of Dyn I signal.
Dyn I distribution in the SC
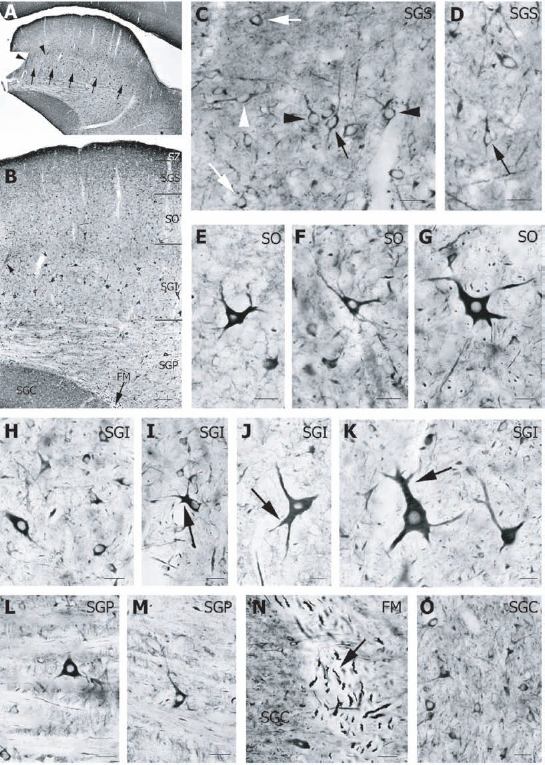
Immunocytochemistry of sections at the level of the midbrain allowed us to investigate Dyn I expression in a second visual structure that receives direct retinal input, the SC (Fig. 3). Dyn I antibody reacted in the superficial layers (stratum zonale (SZ), stratum griseum superficiale (SGS), stratum opticum (SO)), in the intermediate layer (stratum griseum intermediale (SGI)) and in the deeper layer (stratum griseum profundum (SGP)) of the SC (Figs. 3A, B). Besides a lamination pattern, small intensity differences in Dyn I neuropil staining was observed within the SGI layer (Figs. 3A, B).
Neuropil staining was obvious over all SC layers but most apparent in SZ, SGS (Figs. 3C, D) and the upper part of SGI (Figs. 3A, B). In the SGS, Dyn I labeled many small neurons with round or oval cell body and some stellate cells. Dendrites of most oval-shaped neurons projected horizontally or vertically (Figs. 3C, D). In the SO (Figs. 3E–G), triangular and multipolar neurons of irregular morphology were strongly Dyn I-positive. The signal was present over the soma and the proximal part of the cell processes. Neurons were medium-sized to large, and the latter were located at the border with the SGI layer (Figs. 3B, G). Small, medium (Figs. 3H, I) and large (Figs. 3J, K) Dyn I-immunoreactive neurons were distributed all over the SGI (Figs. 3B, H–K). In medium and large neurons, dendrites branched closely to the cell body into two or three thick processes (Figs. 3I–K). In the striate deeper layer (SGP), medium-sized neurons with triangular cell soma expressed Dyn I (Figs. 3L, M). In the cat midbrain, we also detected abundant Dyn I immunoreactivity in the substantia griseum centralis (SGC) (Figs. 3A, B, N). The signal in the SGC consisted of intense neuropil staining and many small neurons (Fig. 3O). In addition, Dyn I was found in axon bundles, as illustrated by its presence in the medial longitudinal fasciculus (FM) at the ventral medial margin of the SGC (Figs. 3B, N).
Dyn I in the visual cortex
The localization of Dyn I in the neocortex was investigated in visual area 17 and 18 (Figs. 4A, B). Dyn I stained neuropil in all layers, but most intensely in layers I and II/III. Dyn I expression was apparent in neurons of layers II/III and layers V and VI while less immunoreactivity was present in neurons of layer IV. Neurons of layer VI displayed the strongest Dyn I labeling (Figs. 4A, B). As already mentioned above, the Dyn I signal was not always equally spread out over the entire cell body. This is true for the rather small neurons of layers II/III and VI with a round to oval cell body shape (Figs. 4C, D, E), but less for the larger pyramidal neurons of layer III (Fig. 4H) and layer V (Figs. 4J–L). Also the apical dendrites of the former neurons exhibited an interrupted Dyn I pattern (Figs. 4D, E).
Layers II/III were characterized by Dyn I staining in many small pyramidal neurons, in cell processes and in neuropil (Figs. 4F–H). The cell body and the apical dendrites of these neurons were Dyn I immunopositive (Figs. 4F, G). In comparison to area 17 (Figs. 4A, G), Dyn I labeled larger pyramidal neurons at the border of layer III in area 18 (Figs. 4B, H). In addition to the soma and apical dendrites, Dyn I was also present in the basal dendrites of these neurons (Fig. 4H). Layer IV had low immunoreactivity in the neuropil and weak staining in some neurons. From these granular neurons, only the somata, but not the dendrites, were positive. Sometimes, the apical dendrites from pyramidal neurons of layer V or VI ran through layer IV (Fig. 4I). Dyn I antiserum marked medium to large pyramidal neurons of layer V in area 17 and area 18 (Figs. 4J–L). Next to a high signal in the soma and apical dendrite of these neurons, the axon hillock was also Dyn I-positive (Figs. 4J–L). Finally, Dyn I expression was most abundant in neurons of layer VI where it labeled the somatodendritic compartment of many densely packed neurons (Fig. 4M).
Discussion
Despite the well-known distribution of Dyn I in rat cerebellum and hippocampus,5,21,22 until now no detailed immunocytochemical information for the mammalian forebrain was available. The present study revealed Dyn I expression mainly in the cell body and dendrites of neurons, but also in the neuropil and in some axon bundles in vision-related cortical and subcortical structures of the cat.
LGN complex
Dyn I was expressed in afferent axon bundles of the medial part of the optic tract that penetrate the dLGN ventrally. In the dLGN itself, Dyn I was localized in relay cells with the characteristic morphology of X- and Y-cells in the magnocellular A and C laminae,35,36 but also of typically smaller W-type neurons of the parvocellular Cp layers of the dLGN 36 as well as the vLGN. 37 39 In the optic radiation the efferent axon bundles connecting the dLGN with the visual cortex, weakly stained for Dyn I.
SC
Bergmann et al 31 describe the presence of Dyn I immunoreactivity in the developing optic tectum of chicken, the non-mammalian homologue of the SC. In cat we also observed Dyn I-positive neurons in all layers of the SC, including the visually-driven SZ, SGS and SO. These layers are exclusively innervated by Y- and W-input 40 42 with the upper part of SGS receiving direct W-input, and the lower SGS and SO being innervated by direct and indirect Y-input.41,42 Histology 41 and cell recording 43 studies have demonstrated that the deeper collicular layers participate in the Y-pathway of visual processing. The large Dyn I-immunoreactive neurons in these deeper layers are again Y-type projection neurons. The superficial layers of the SC process visual information from the retina and visual cortex while the deeper layers typically receive mixed input from auditory, somatic as well as visual cortical and subcortical structures to generate a motor response. 44 46 The fact that Dyn I is present in small to large neurons over all collicular layers, favors a general function in both the sensory and the motor neurons. In addition, we observed differences in the intensity of the neuropil staining that demarcate the modular arrangements in the SC. Those ‘patches’ are probably formed by the segregation of input and output projections into one cluster. This compartmentalization is also apparent through the patchy-like expression of several other neurotransmitters, peptides and enzymes. 44 47
Visual cortex
Dyn I is mainly expressed in pyramidal neurons of the primary visual areas 17 and 18. Major Dyn I expression was found in layer VI, followed by layers II/III and V, while a clearly lower expression was detected in layer IV. Besides, minor differences were seen for Dyn I expression between the two analyzed primary visual areas. Area 18 is characterized by larger border pyramidal neurons in layer III and a thinner layer VI compared to area 17. 48 Both area 17 and area 18 receive primary retinal input but they differ in retinotopic organization and visual response properties.49,50 The observed Dyn I expression difference in area 17 and 18 could be a consequence of distinct thalamic input. Indeed, the base of layer III of area 17 and 18 receives input from Y axons from A and A1 layers and W projections from Cp neurons of the LGN while only the lower part of layer III in area 18 is innervated by Y terminals that arise from neurons located in the Cm layer of the LGN. 48
All together the distribution, density, size and morphology of the Dyn I immunoreactive neurons as detected in the cat visual system point out that Dyn I is expressed in projection neurons. This is however not absolute since certain GABAergic neuron populations, like the one of the PGN are also found Dyn I immunoreactive. Furthermore our data argue against the presence of Dyn I in a specific class of projection neurons. Instead both the magnocellular and parvocellular pathway of the mammalian visual system seem to express Dyn I.
Neuron-specific expression
In all analyzed forebrain structures, Dyn I was exclusively expressed in neurons, and not in glia, conform to the previously reported neuron-specific nature of the Dyn I isoform.8,21,29 We essentially stained the somatodendritic compartment of neurons. But, the signal was not always equally distributed over the whole somatodendritic compartment. Interestingly, this might be due to the association of Dyn I with microtubules.1,5,6 Consistent with this, it is indeed demonstrated that Dyn I is associated with distinct subcellular membrane fractions.21,29 On the other hand, we mainly detected the proximal part of dendrites as demonstrated for the large neurons of the thalamus and midbrain while in the neocortex the distal part of dendrites was stained, especially in pyramidal neurons from supra- and infragranular layers of area 17 and 18. In addition, neuropil staining was also obvious in the neocortex and other structures of the cat brain. This indicates Dyn I expression in many synaptic contacts and cell processes, since neuropil is the region where most synaptic connectivity occurs. 51 We could for instance detect a contact between two layer V neurons in the visual cortex. Other synaptic contacts were seen in the staining of cerebellar glomeruli and large basket cell synapses, as already indicated by Powell and Robinson. 22 Finally, Dyn I was also present in axonal segments in the white matter and in axon tracts like the OT, OR and FM. This is in agreement with previous observations of Dyn I expression in axons of the white matter in rat brain.21,22
Possible functions for Dyn I
The presence of Dyn I in presynaptic axons and synaptic contacts correlates well with a function at the neuronal synapse. But, Dyn I is also particularly expressed in the postsynaptic compartment of neurons. Therefore, we suggest that the function of Dyn I is not restricted to endocytotic processes at the presynaptic membrane and to synaptic membrane recycling. The expression of Dyn I in perikarya and dendrites may be explained in two ways. Firstly, Dyn I is involved in receptor-mediated endocytosis that is known to occur at postsynaptic membranes.52,53 Secondly, Dyn I participates in intracellular vesicular traffic systems. McNiven and his collaborators
12
pointed out already multiple functions for Dyn I in several cytoplasmatic compartments of neurons, like mitochondria, Golgi-complex and endosomes. In addition,
In summary, this study presents the first description of the immunolocalization of Dyn I in the visual system of the adult cat including a detailed analysis of the LGN complex, superior colliculus and primary visual cortex. We suggest that the characteristic expression of Dyn I by projection neurons reflects a fundamental role in the central nervous system and implies that Dyn I participates in several processes in many mature neurons at the subcortical and cortical level.
Disclosure
The authors report no conflicts of interest.
Footnotes
Acknowledgements
This work was supported by grants of the Fund for Scientific Research—Flanders, Belgium (FWO) and the Research Fund (OT 05/33) of the Katholieke Universiteit Leuven, Belgium. We gratefully thank Tim Leest, Ria Vanlaer and Inge Van der Auwera for technical assistance.
