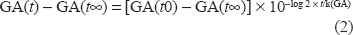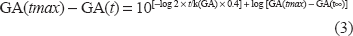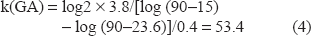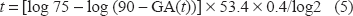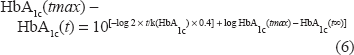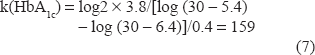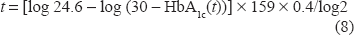Abstract
Fulminant type 1 diabetes mellitus (FT1DM) develops as a result of very rapid and almost complete destruction of pancreatic β cells. Because of an abrupt increase in plasma glucose, HbA1c and glycated albumin (GA) might increase along with duration of symptoms in FT1DM patients. We attempted to devise a formula to estimate duration of symptoms based on the increased levels in HbA1c or GA. Four patients who developed FT1DM during the course of type 2 diabetes mellitus and in whom HbA1c was measured before onset were investigated in this study. The percents of the estimated duration of symptoms calculated from HbA1c (four patients) and GA (two patients) to the actual duration were 137 ± 88% and 122%, respectively. In FT1DM patients in whom HbA1c and/or GA before onset and at the time of ketoacidosis are measured, duration of symptoms might be estimated with using the increased levels in HbA1c or GA.
Introduction
Fulminant type 1 diabetes mellitus (FT1DM) is a subtype of type 1 diabetes mellitus (T1DM) in which diabetic ketoacidosis occurs shortly after onset because of very rapid and almost complete destruction of pancreatic β cells. 1 In Japan, FT1DM is an important subtype, accounting for about 20% of all cases of acute onset T1DM. 2 The etiology of FT1DM is not fully understood, but in addition to hereditary factors such as specific human leukocyte antigen (HLA), environmental factors such as viral infection are thought to cause pancreatic β cell dysfunction. 3 In autoimmune T1DM patients, the natural course is destruction of pancreatic β cells, followed by increased plasma glucose, onset of diabetes, appearance of diabetic symptoms associated with hyperglycemia, and ultimately depletion of insulin secretion, which leads to ketoacidosis. 4 In FT1DM patients, the stages of pancreatic β cell destruction, diabetes onset, appearance of diabetic symptoms and ketoacidosis, are thought to progress over a much shorter time period as compared to autoimmune T1DM.
In diabetic patients, glycation of various proteins is known to be increased, and some of these glycated proteins are thought to be involved in the onset and progression of chronic diabetic complications. 5 Of these proteins, HbA1c is widely used clinically as a marker of glycemic control.6,7 Since the lifespan of erythrocytes is approximately 120 days, HbA1c reflects the glycemic control status of the previous 2–3 months. As well as HbA1c, glycated albumin (GA) is also used as an indicator of glycemic control. 8 Since the half-life of serum albumin is shorter than that of erythrocytes, GA reflects plasma glucose levels over a shorter period (about 2 weeks). Therefore, in cases of acute changes in glycemic control, GA is more useful than HbA1c as a glycemic control marker. 9
In FT1DM patients, because of an abrupt increase in plasma glucose after onset, HbA1c and GA also increase along with duration of symptoms. Therefore, based on the increased levels in HbA1c or GA, duration of symptoms of FT1DM could be estimated. However, when FT1DM develops from non-diabetic conditions, HbA1c and/or GA before onset are rarely measured. In non-diabetic subjects, if HbA1c or GA before onset of FT1DM are unknown, large errors in estimation of the increased levels in HbA1c or GA might occur because the normal range of both HbA1c and/or GA are broad. This represents a problem in terms of low accuracy when estimating duration of symptoms.
Recently, during the course of treatment for type 2 diabetes mellitus (T2DM), onset of FT1DM has occasionally been reported. In these patients, HbA1c has often been measured prior to onset of FT1DM. In this study, in 4 patients in whom FT1DM developed during the course of T2DM, using HbA measured before onset, we attempted to estimate duration of symptoms of FT1DM based on the degree of increases in HbA1c or GA.
Materials and Methods
Calculation of duration of symptoms
Based on data from 35 FT1DM patients previously reported, in whom HbA and GA were simultaneously measured at the time of FT1DM ketoacidosis (duration of symptoms, 3.8 ± 2.3 days; HbA1c, 6.4 ± 0.8%; GA, 23.6 ± 4.3%), 10 we devised a formula to estimate duration of symptoms using the increased levels of HbA1c or GA. Duration from onset was defined as the time from onset of diabetes to ketoacidosis and duration of symptoms was defined as the time from the appearance of diabetic symptoms to ketoacidosis. The natural course of T1DM is typically onset of diabetes with a decrease in pancreatic (3 cell function to 50%, symptomatic diabetes with hyperglycemia at 20% function and the development of ketoacidosis with depletion of pancreatic (3 cell function. 4 In FT1DM, the same phenomenon is presumed to occur. When duration of symptoms is t (days) and duration from onset is T (days), the following formula was obtained.
Shi et al 11 assumed that if glycated protein synthesis occurs irreversibly, then when hyperglycemia is treated and plasma glucose decrease, GA levels change exponentially. They showed that the predicted values using this function are almost the same as the actual values. Using this relationship, we reported that when GA before treatment (t0) is GA(t0), duration from the start treatment is t (days), GA after diabetes treatment (t) is GA(t), the treatment target value of GA is GA(t∞), and the constant is k(GA), GA(t) can be determined using the following formula. 12
When a patient with normal glucose tolerance develops FT1DM, GA is thought to increase logarithmically. When GA before onset of FT1DM (t0) is GA(t0), GA after onset (t) is GA(t), the final value of GA without treatment is GA(tmax), and the constant is k(GA), GA(t) can be determined using the following formula:
In the 35 patients with FT1DM, GA(t) was 23.6 ± 4.3% at the time of ketoacidosis and duration of symptoms (t) was 3.8 ± 2.3 days. With these values, and assuming GA(tmax) and GA(t∞) are 90% and 15%, respectively, k(GA) can be calculated by the following formula:
Duration of symptoms (t) can be calculated by the following formula using GA(t) at the time of ketoacidosis:
With regard to HbA1c duration of symptoms can be estimated by using a similar formula. Namely, when HbA1c before treatment (t0) is HbA1c(t0), duration from the start treatment is t (days), A1c after diabetes treatment (t) is HbA1c(t), the final value of HbA1c without treatment is HbA1c(tmax), and the constant is k(HbA1c), HbA1c(t) can be determined using the following formula:
In the 35 patients with FT1DM, HbA1c(t) was 6.4 ± 0.8% at the time of ketoacidosis and duration of symptoms (t) was 3.8 ± 2.3 days. With these values, and assuming HbA1c(tmax) and HbA1c(t∞) are 30% and 5.4%, respectively, k(HbA1c) can be calculated by the following formula:
Duration of symptoms (t) can be calculated using HbA1c(t) at the time of ketoacidosis, by the following formula:
Study patients
Four patients who developed FT1DM during the course of T2DM were investigated in this study. Three of 4 FT1DM patients are reported previously.13,15 HbA1c was measured before and at ketoacidosis in all patients, and GA was measured at ketoacidosis in two patients (Case 1 and 2). The clinical characteristics of the study patients are shown in Table 1.
Clinical characteristics of the FT1DM patients in this study

n.d., not determined.
Case 1 was a 66-year-old man with T2DM who was being treated with oral hypoglycemic agents. Near the time of onset of FT1DM, HbA1c was 7.1%. Four days later following symptoms of gastroenteritis and thirst developed, urinary ketones were positive, metabolic acidosis was found, and diabetic ketoacidosis was diagnosed. At the time of hospital arrival, HbA1c was 7.8%, GA was 29.5%, casual plasma glucose was 794 mg/dL, and pancreatic exocrine enzymes were increased. Urinary C-peptide was 1.1 Ng/day. On glucagon loading test, serum C-peptide was both <0.1 ng/mL before and at 6 min after loading. Thus, endogenous insulin secretion was depleted. Anti-glutamic acid decarboxylase (GAD) antibody and anti-insulinoma-associated protein-2 antibody (IA-2) antibody were both negative. Based on the above findings, FT1DM was diagnosed.
In Case 1, the GA value before onset was calculated to 19.2% (= 7.1% x 2.71) based on the HbA1c value (7.1%) before onset, and 2.87 ± 0.36 [HbA1c (Japan Diabetes Society: JDS value); by HbA1c (NGSP value): 2.71 ± 0.34] using the GA/HbA1c ratio in T2DM. 16 This study was approved by the Ethics Committee at Kinki Central Hospital and the study complied with the ethical guidelines of the Helsinki Declaration as revised in 2000.
Laboratory methods
HbA1c was measured by HPLC. The value for HbA1c (%) was estimated as a National Glycohemoglobin Standadization Program (NGSP) equivalent value (%) calculated using the formula HbA1c (%) = HbA1c (JDS) (%) + 0.4%, considering the relational expression of HbA1c (JDS) (%), as measured by the previous Japanese standard substance and measurement methods, and HbA1c (NGSP). 17 Serum GA was determined using a Hitachi 7600 autoanalyzer (Hitachi Instruments Service Co., Tokyo, Japan) by the enzymatic method with albumin-specific proteinase, ketoamine oxidase and albumin assay reagent (Lucica GA-L; Asahi Kasei Pharma Co., Tokyo, Japan). 18
Results
In Case 1, the difference in HbA1c before and after ketoacidosis (ΔHbA1c) was 0.7%, and the difference in GA (ΔGA) was 10.3% using a pre-onset GA of 19.2% calculated from the HbA1c (see section in Study Patients). In this case, assuming the onset of FT1DM from normal glucose tolerance, by adding ΔHbA1c and ΔGA to a pre-onset HbA1c of 5.4% and GA of 15.0%, respectively, after ketoacidosis HbA1c of 6.1% and GA of 25.3% were calculated. With these values, according to Formulas 5 and 8, duration of symptoms was calculated. In Cases 2–4, the methods for calculation were similar.
Figure 1 shows the results for estimated duration of symptoms using HbA1c or GA compared to the actual duration of symptoms in the study patients. In Case 1, the estimated duration of symptoms calculated from ΔHbA1c is 2.6 days and the estimated duration of symptoms calculated from ΔGA is 4.6 days. From the clinical course, the actual duration of symptoms was 4 days. The ratio of estimated duration of symptoms calculated from ΔHbA1c and ΔGA, to actual duration of symptoms 65% and 115%, respectively. Thus, the estimated duration of symptoms calculated from ΔGA was closer to the actual duration of symptoms. In Case 2, the estimated duration of symptoms calculated from ΔHbA1c is 5.3 days, it from ΔGA is 7.1 days and the actual duration of symptoms of 5 days. In Case 3, the estimated duration of symptoms calculated from ΔHbA1c is 4.6 days, which was close to the actual duration of symptoms of 4 days. On the other hand, in Case 4, the estimated duration of symptoms calculated from ΔHbA1c is 5.4 days, thus showing a discrepancy with the actual duration of symptoms of 2 days. The estimated duration of symptoms calculated from ΔHbA1c was 4.4 ± 1.3 days compared to the actual duration of symptoms of 3.8 ± 1.3 days, and the ratio of the estimated duration of symptoms to the actual duration of symptoms was 137 ± 88%. The estimated duration of symptoms calculated from GA was 5.6 days compared to the actual duration of symptoms of 4.5 days, and the ratio of the estimated duration of symptoms to the actual duration of symptoms was 122%.
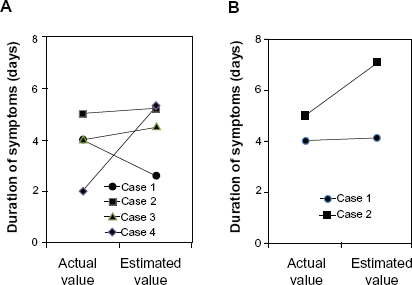
Comparison of the estimated duration of symptoms and the actual duration of symptoms, in the patients who developed fulminant type 1 diabetes mellitus (FT1DM) after type 2 diabetes mellitus (T2DM).
Discussion
The present study showed that in the patients with FT1DM in whom glycemic control marker before onset and at ketoacidosis were measured, duration of symptoms could be estimated using HbA1c or GA. Because a half-life of albumin is about 14 days, GA is used as a marker for plasma glucose over the previous 2 weeks to 1 month.9,19 In FT1DM patients with an abrupt increase in plasma glucose for a short time, HbA1c shows a discrepancy with plasma glucose and is normal or only mildly increased at ketoacidosis. On the other hand, GA reflects acute glycemic changes compared to HbA1c. GA or GA/HbA1c ratio is useful as a marker for short-term glycemic changes in FT1DM. 10 Therefore, GA might be superior to HbA1c to estimate FT1DM duration of symptoms.
Because islet-associated autoantibodies such as anti-GAD antibody are not detected in FT1DM, a mechanism of β cell dysfunction different from that in autoimmune T1DM is thought to exist. Although the specific mechanism of β cell dysfunction in FT1DM is unclear, β cell dysfunction is presumed to involve some type of viral infection as a trigger, with direct β cell damage due to the virus, and in addition, the activation of immunocompetent cells, which occurs after viral infection. 3 In FT1DM, similar to the natural course of autoimmune T1DM, β cell dysfunction occurs before ketoacidosis develops, but the mechanism and timing are unclear. To elucidate these factors, it is important to pinpoint the onset period or period when pancreatic β cell destruction begins.
Episode of ketoacidosis is always accompanied in FT1DM as shown in the diagnostic criteria. The acute appearance of hyperglycemic symptoms, including thirst, polydipsia and polyuria, may be characteristic before ketoacidosis. However, patients with FT1DM often do not present with typical hyperglycemic symptoms, and instead complains of nonspecific symptoms such as generalized fatigue, nausea or epigastralgia. Therefore, clinicians may have a difficulty in accurately determining duration of symptoms from the patient's symptoms. At the time of FT1DM diagnosis, a significant positive correlation between GA and urinary CPR has been reported. 20 Therefore, in FT1DM patients with a marked decrease in endogenous insulin secretion, the increased levels in GA is small because ketoacidosis develops for a very short time. Based on these results, the increased levels in GA could be used to estimate duration of symptoms in FT1DM patients. To the best our knowledge, this is the first study to estimate duration of symptoms based on laboratory values in FT1DM patients. Our attempt to estimate FT1DM duration of symptoms, in addition to leading to an understanding of the clinical course of FT1DM, is also hoped to help elucidate the pathophysiology of FT1DM.
As a precaution when using HbA1c and GA to estimate duration of symptoms, if there are factors that affect lifespan of erythrocyte or half-life of albumin, the accuracy of estimated duration of symptoms might be low. For example, HbA1c may be apparently low in patients with bleeding, hemolytic anemia, hypersplenism, during iron treatment for iron deficiency anemia, and during treatment with erythropoietin for renal anemia. GA may be apparently low in conditions with increased albumin catabolism, such as patients with nephrotic syndrome and hyperthyroidism. In patients with liver cirrhosis, in which albumin catabolism is prolonged, GA is apparently high. 8 When performing studies using HbA1c or GA, the above conditions should be excluded. Therefore, patients with any of the above conditions were excluded in this study. A negative correlation between GA and BMI has been reported, 16 but with very rapid glycemic changes as in FT1DM, this relationship with BMI could be ignored. In this study, duration of symptoms was estimated using our estimation formula in a small number of FT1DM patients. In the future, in a larger number of FT1DM patients, accuracy of the estimation formula and superiority of using GA should be investigated.
Conflicts of Interest
There are no conflicts of interest.
Author Contributions
Conceived and designed the experiments: MK. Analysed the data: AK, MK. Wrote the first draft of the manuscript: AK. Contributed to the writing of the manuscript: AK, MK. Agree with manuscript results and conclusions: AK, TS, SF, MY, SO, JM, HS, MK. Jointly developed the structure and arguments for the paper: AK, TS, SF, MY, SO, JM, HS, MK. Made critical revisions and approved final version: AK, TS, SF, MY, SO, JM, HS, MK. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.