Abstract
Safety and efficacy of combination therapy of pitavastatin and fenofibrate were examined in consecutive case series with fasting serum triglycerides ≥ 150 mg/dL despite receiving pitavastatin 1 or 2 mg daily for over 2 months and additionally administered micronized fenofibrate 67 mg daily for another 4 to 16 weeks. Such low doses were selected in consideration of safety, and normal liver and renal functions were incorporated in inclusion criteria. In result, a total of 56 cases were examined. The addition of fenofibrate 67 mg to pitavastatin 1 mg/2 mg yielded a 36.8%/35.6% reduction in triglycerides and 6.4%/12.4% elevation in high-density lipoprotein cholesterol, respectively. Almost 70% of the patients achieved triglycerides <150 mg/dL. Statistically significant elevation and decrease were observed in high-density lipoprotein cholesterol level and low-density lipoprotein cholesterol, respectively. Laboratory tests for liver, renal and muscle function statistically significantly elevated after starting fenofibrate co-administration, which were considered comparable to the effect of fenofibrate alone. No myopathy or serious adverse events were reported. In conclusion, while the safety and tolerability need to be further examined over the longer term, and careful monitoring is still needed, this regimen could be considered as one of the treatment option for hypercholesterolemia associated with hypertriglyceridemia.
Introduction
Recently, the efficacy and safety of concomitant use of statins and fibrates in patients with dyslipidemia has been accumulated by several clinical studies in Western populations.1–3 However, the tolerability of anti-dyslipidemic drugs is considered to differ between Western and Asian populations; for instance, the recommended doses of statins in Europeans and Americans are generally 2- to 4-fold greater than that in Japanese. Therefore, the efficacy and safety confirmed in Europeans and Americans may not be directly extrapolatable to Japanese.
In this study, to examine the safety and efficacy of such combination therapy in Japanese hypercholesterolemic patients with elevated fasting serum triglycerides (TG), we collected consecutive case series of patients who had suboptimal level of TG despite statin therapy for treatment of hypercholesterolemia and normal liver and renal function. To simplify the patient characteristics and analytical method due to limited sample size which had been considered feasible to be accomplished, we selected pitavastatin and fenofibrate to research for the following reasons.
Currently, atorvastatin, pitavastatin and rosuvastatin are clinically available in Japan as statins with efficient TG-lowering effect when administered alone in addition to potent low-density lipoprotein cholesterol (LDL-C)-lowering effect at approved regular doses. Pitavastatin and rosuvastatin are scarcely metabolized through cytochrome P450 and show fewer drug-drug interactions. 4 However, rosuvastatin had been reported to be associated with a higher risk of proteinuria or renal failure as compared with other statins. 5 Therefore, we selected pitavastatin which had established the safety profile in the Japanese population by LIVES study 6 involving approximately 20,000 patients over 2 years with no particular concerns about the safety of pitavastatin as compared with that of other statins.
Concerning fibrates, bezafibrate and fenofibrate are available for clinical use as potent TG-lowering agents in Japan. Although a trial of combined therapy with pitavastatin and bezafibrate has already been conducted in Japanese, 7 there are no reports of concomitant therapy with pitavastatin and fenofibrate. The FIELD study 3 had provided data to suggest the safety of fenofibrate administered in combination with statins. Moreover, fenofibrate has been reported to unlikely affect the blood levels of pitavastatin when administered concomitantly. 8 Based on the above, we selected fenofibrate to research.
To further ensure the safety of this study, we selected low doses of those agents. Intended subjects were those who were receiving pitavastatin 1 mg or 2 mg daily, which are the recommended initial doses in Japan, and to be co-administered micronized fenofibrate 67 mg daily. The daily dose of micronized fenofibrate indicated for cholesterol-lowering therapy is 201 mg in Europe and the US and 134 mg to 201 mg in Japan, whereas that for TG-lowering therapy is only 67 mg to 201 mg in those areas. Although a single use of micronized fenofibrate 67 mg is approved only for TG-lowering therapy, and there have been reported few studies on the clinical results of micronized fenofibrate 67 mg, it was considered efficacious enough to additionally lower the TG levels that have not been sufficiently controlled by a statin alone.
The main objective of the study was to evaluate whether micronized fenofibrate 67 mg efficiently and safely reduce the fasting serum TG level that had been suboptimal level despite pitavastatin treatment 1 mg or 2 mg.
Methods
This study was conducted in accordance with the ethical principles in the Declaration of Helsinki and the Ethical Guideline for Clinical Research by the Ministry of Health, Labor and Welfare of Japan. The protocol was approved by the institutional review board of the Nonprofit Organization Chubu Clinical Research Network (Aichi, Japan) and all patients provided written informed consent. This study was conducted in an exploratory fashion with a consecutive case series collected in 20 medical facilities belonging to the Nonprofit Organization Chubu Clinical Research Network (Aichi, Japan) between November 22, 2008 and September 30, 2009. This study is registered in University Hospital Medical Information Network Clinical Trials Registry (UMIN-CTR) as the ID of 000001828. Subjects were selected among those who were considered to need additional TG-lowering medication by micronized fenofibrate 67 mg due to suboptimal fasting TG level as of $150 mg/dL despite receiving pitavastatin 1 mg or 2 mg for at least 2 months for treatment of hypercholesterolemia. Prospectively setting inclusion and exclusion criteria, we collected the safety and efficacy information of the additional fenofibrate treatment for 12 weeks in principle or by the time fasting blood sample was obtained between week 8 and 16. For each of those who were receiving pitavastatin 1 mg or 2 mg at the baseline, thirty patients were considered adequate to assess the efficacy and safety of this treatment. Patients were excluded if they had followings: the age of <20 years; estimated glomerular filtration rate of <60 mL/min/1.73 m2, which was evaluated by the equation issued by the Japanese Society of Nephrology in 2008 as of 194 x serum creatinine1.094 x age−0.287 (for females, x0.739 in addition); 9 serum creatinine of $1.0 mg/dL; creatinine kinase (CK), aspartate aminotransferase (AST) or alanine aminotransferase (ALT) of higher than the upper limit of normal (ULN) range. Patients were also excluded if they were already treated with fibrates or any other lipid-modifying agents or were judged to be unsuitable for any reason by their physician. Physicians discontinued co-administration of fenofibrate and pitavastatin for the patients as usual clinical practice if they develop myalgia with concurrent elevation of CK to higher than 3 times the ULN or had serum creatinine of $1.5 mg/dL, and the patients stopped participating in the study. The end of the study was determined basically as week 12. Lipid and safety profiles were collected at week 0, 4 and 12. If patients visited without fasting or did not visit at week 12, the end of the study determined as the day on which the fasting blood sample was obtained between week 8 and 16. When the patients were discontinued or withdrew from the study before completion, data up to that day were collected.
Lipid profile included TG, total cholesterol, high-density lipoprotein cholesterol (HDL-C), non-HDL-C, remnant-like particle cholesterol (RLP-C) and LDL-C. LDL-C was calculated by the Friedewald equation when TG was of <400 mg/dL. Safety profile included AST, ALT, CK, serum creatinine, gamma-glutamyl transpeptidase (γ-GTP), blood urea nitrogen, uric acid, fasting plasma glucose and HbA1c.
The primary efficacy variable was the percentage change in TG from the baseline to the end of the study. The secondary efficacy variables were the percentage of patients achieving TG < 150 mg/dL at the end of the study, the percentage changes in other lipid measures from the baseline to the end of the study and the changes in lipid measures at week 4 and 12. The safety variables included the changes in blood laboratory parameters mentioned above from the baseline to the end of the study. Physician-reported adverse events were also collected. An adverse event was defined as any untoward medical occurrence in a patient after the start of the study, which did not necessarily have a causal relationship with the treatment. Of the discontinuation criteria determined for an individual patient, elevations of CK to higher than 3 times the ULN with myalgia and serum creatinine of $1.5 mg/dL were prespecified as adverse events.
Statistical analysis was conducted by the third party CLINICAL STUDY SUPPORT, Inc, Aichi, Japan. with the use of SAS software, version 9.1.3 (SAS Institute Inc., Cary, NC). The results were examined in an exploratory manner. All analyses were conducted in a full analysis-set approach, which included all the patients who received at least 1 dose of fenofibrate and had at least one post-treatment information in addition to the baseline. The categorical data of the baseline patient characteristics were expressed as the number of patients (percentage). The continuous variables were expressed as mean ± standard deviation. The median, minimum and maximum were also expressed for a skewed distribution. Differences in patient demographics between two groups were analyzed with the use of one-sample t-test or Fisher's exact test. The change and percentage change from the baseline in the efficacy variables were analyzed with the use of one-sample t-test. Wilcoxon's signedrank test was used to analyze the change and percentage change for a skewed distribution. The changes in the clinical laboratory values for the safety were expressed as mean and 95% confidence interval. All statistical tests were two-sided and P values less than 0.05 were interpreted as statistically significant.
Results
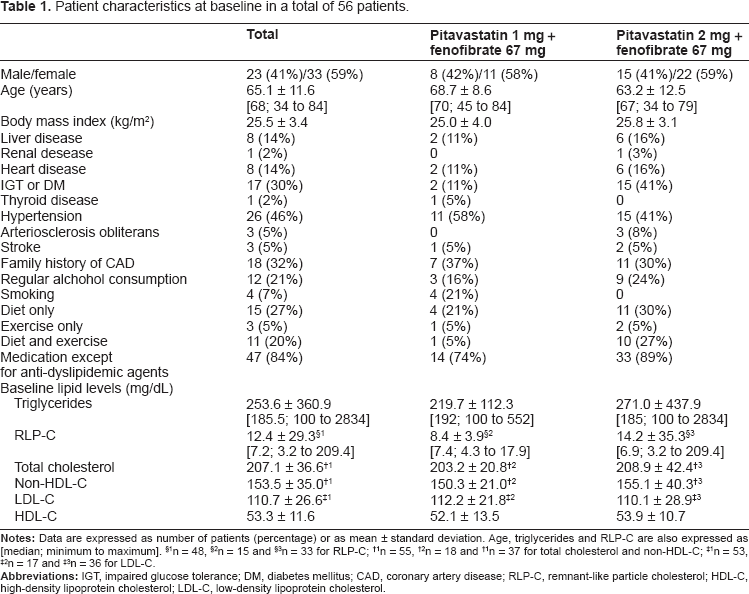
Of the 58 patients enrolled, 20 were receiving pitavastatin 1 mg, and another 38 were 2 mg. One patient for each group, two patients in total were excluded because of the lack of data after the initial visit. Protocol violation was found in some patients. At baseline, TG levels of 4 patients in pitavastatin 2 mg group and 1 in pitavastatin 1 mg group were less than 150 mg/dL; ALT levels of 10 patients in pitavastatin 2 mg group and 3 in pitavastatin 1 mg group, AST levels of 2 patients in pitavastatin 2 mg group, CK levels of 2 patients in pitavastatin 2 mg group were higher than the ULN; serum creatinine levels of 2 patients were of or higher than 1.0 mg/dL; eGFR levels of 3 patients in pitavastatin 2 mg group and 2 in pitavastatin 1 mg group were less than 60 mL/min/1.73 m2. Those were included in the analyses in accordance with a full analysis-set approach, and a total of 56 patients were therefore analyzed. Table 1 shows the patient characteristics at the baseline. Males accounted for 41% of the analyzed population. The patients were relatively elderly with the mean age of 65.1 ± 11.6 years, and had an average physique of the Japanese population with the mean body mass index of 25.5 ± 3.4 kg/m2. The main complications were impaired glucose tolerance including diabetes mellitus and hypertension (30% and 46%, respectively). Of all the patients, 32% had a family history of coronary artery disease, 21% gave a history of habitual alcohol consumption, 52% were receiving diet or exercise therapy, or both, and 84% were receiving some other medications concomitantly with anti-dyslipidemic treatment. In regard to the baseline lipid levels, LDL-C were controlled at 110.7 ± 26.6 mg/dL by administration of pitavastatin 1 mg or 2 mg for at least 2 months, while TG were controlled at 253.6 ± 360.9 mg/dL (median, 185.5 mg/dL; range, 100 to 2834). More patients had impaired glucose tolerance or diabetes mellitus (P = 0.0307) and received both of diet and exercise therapy in pitavastatin 2 mg group (P = 0.0233). Smoking were seen only in pitavastatin 1 mg group (P = 0.0114).
Patient characteristics at baseline in a total of 56 patients.
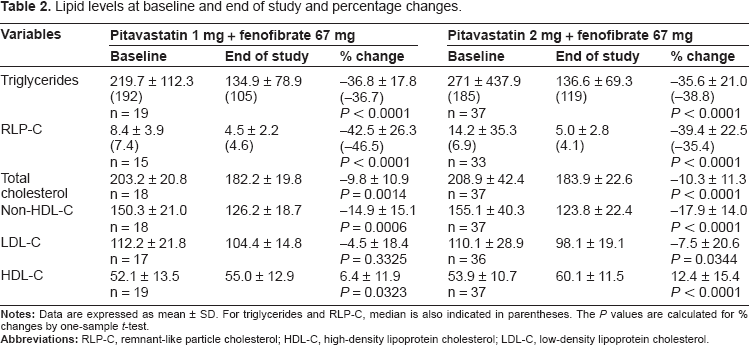
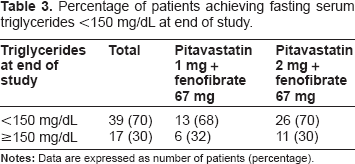
As the primary efficacy variable, the mean percentage change in TG from the baseline to the end of the study was -36.8% (P < 0.0001) in patients who received micronized fenofibrate 67 mg in addition to pitavastatin 1 mg, and -35.6% (P < 0.0001) in those the fenofibrate was added on to pitavastatin 2 mg (Table 2). In relation to the secondary efficacy variables, the proportion of patients achieving TG <150 mg/dL at the end of the study was 68% (13 in 19 patients) in the pitavastatin 1 mg group, and 70% (26 in 37) in the pitavastatin 2 mg group (Table 3). In total patients, TG was well controlled after 4-week additional co-administration of micronized fenofibrate on pitavastatin (Fig. 1). The percentage change in RLP-C was -42.5% (P <0.0001) in the pitavastatin 1 mg group and -39.4% (P < 0.0001) in the pitavastatin 2 mg group, indicating excellent improvement similar to the TG reduction (Table 2). The levels in RLP-C in total patients changed in parallel with that in TG (Fig. 1). As shown in Table 2, statistically significant improvement in all lipid parameters except for LDL-C in the pitavastatin 1 mg group were observed. Increase in HDL-C in total patients was statistically significant at week 4 and 12, and decrease in LDL-C was significant at week 12 (Fig. 1).
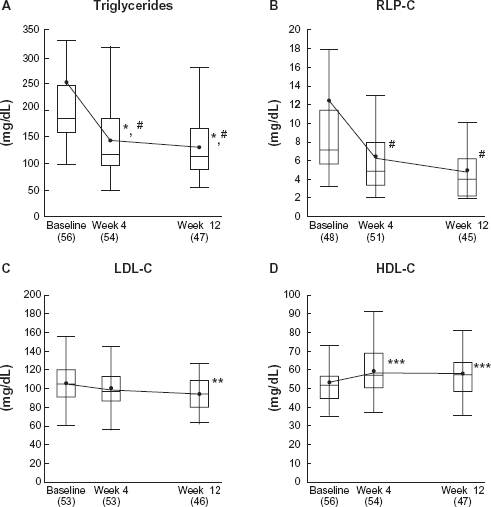
Lipid levels at baseline, week 4 and week 12 in total patients.
Lipid levels at baseline and end of study and percentage changes.
Percentage of patients achieving fasting serum triglycerides <150 mg/dL at end of study.
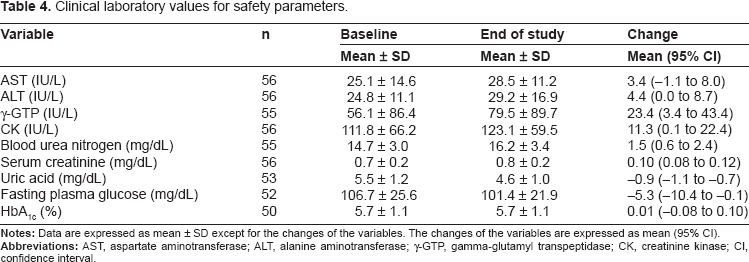
As for the safety outcomes, the clinical laboratory values at baseline and the end of the study and the mean changes are presented in Table 4. Statistically significant elevation in ALT, γ-GTP, CK, serum creatinine and blood urea nitrogen and reduction in uric acid and fasting plasma glucose were observed. However, no clinically significant changes of these parameters were found except for one patient who had the γ-GTP values higher than 3 times the ULN, which was reported as an adverse event. HbA1c changed neither in total patients nor in patients with diabetes or glucose intolerance. The change in HbA1c in patients with diabetes or glucose intolerance was -0.08 [95% CI, -0.31 to 0.16] (n = 17), and that in non-diabetic patients or patients with normal glucose tolerance was 0.05 [95% CI, -0.02 to 0.13] (n = 33).
Clinical laboratory values for safety parameters.
No serious adverse events, including rhabdomyolysis, were reported. No prespecified adverse events fulfilling the discontinuation criteria from the study were noted. A total of 8 patients were recorded as experiencing adverse events self-reported by the investigators. All of them were mild to moderate. Among those, 4 patients were withdrawn from the study in accordance with the judgment by the investigators, and one discontinued the study drug on her own will. All the 5 patients recovered after the treatment discontinuation. Of those, 4 patients were recorded to have adverse events in terms of signs and symptoms were reported as paroxysmal atrial fibrillation, dizziness, left shoulder pain and a strange feeling in the mouth for each. Paroxysmal atrial fibrillation and dizziness occurred in the patients who had the same episodes before, and was not considered to be related to fenofibrate and pitavastatin. In the patient who experienced the left shoulder pain and stopped taking the treatment on her own will, the symptom disappeared. The patient with the strange feeling in the mouth developed the feeling after the start of fenofibrate administration, and the feeling disappeared after discontinuation of fenofibrate. Another 4 patients were reported to have adverse events in terms of clinical laboratory abnormalities. A 75-year-old male who received pitavastatin 2 mg + fenofibrate 67 mg was reported to have γ-GTP elevation, whose values at week 0, 4 and 12 were 30, 22 and 250 IU/L, respectively, where the ULN was 86, and the treatment was continued until study completion. A 68-year-old female who received pitavastatin 2 mg + fenofibrate 67 mg was reported to have elevation in AST, ALT, γ-GTP and CK, whose values at week 0, 4 and 12 were 22, 46 and 54 IU/L for AST where the ULN was 40 IU/L, 15, 36 and 56 IU/L for ALT where the ULN was 40 IU/L, 12, 31 and 107 IU/L for γ-GTP where the ULN was 30 IU/L and 96, 117 and 169 IU/L for CK where the ULN was 162 IU/L, respectively, and the treatment was continued until study completion. Of the 2 patients who were reported to have elevation in AST, ALT and γ-GTP a 64-year-old female who received pitavastatin 2 mg + fenofibrate 67 mg was discontinued at week 4 because of the elevation in AST from 21 IU/L to 52 IU/L where the ULN was 33 IU/L, ALT from 20 to 56 IU/L where the ULN was 30 IU/L and γ-GTP from 22 to 110 IU/L where the ULN was 47 IU/L, respectively, and a 64-year-old male who received pitavastatin 1 mg + fenofibrate 67 mg was discontinued at week 8 because of the elevation in AST from 18 IU/L at week 0 to 32 IU/L at week 4 and 34 IU/L at week 8 where the ULN was 33 IU/L, ALT from 18 IU/L to 24 and 41 IU/L where the ULN was 30 IU/L and γ-GTP from 36 IU/L to 60 and 392 IU/L where the ULN was 47 IU/L, respectively. These abnormalities disappeared or improved after treatment discontinuation without other specific treatment. Follow-up data of the last patient described above were available; the values improved for AST 39 IU/L at week 12 to 23 IU/L at week 16, for ALT 53 IU/L to 24 IU/L and for γ-GTP to 282 IU/L at week 12 and 136 IU/L at week 16, respectively. This patient had previously experienced worsening of the liver function following administration of terbinafine, which was recorded as a drug allergy.
Discussion
Several studies in Japan, Korea and Europe10–18 have demonstrated the consistent LDL-C-lowering effect of pitavastatin 1 mg and 2 mg with the mean percentage change in LDL-C in the range of 31% to 34% and 38% to 44%, respectively. Pitavastatin monotherapy demonstrates a TG reduction in the range of 23% to 30% in patients with baseline TG ≥ 150 mg/dL,11,14 an HDL-C elevation by 12% in patients with baseline HDL-C <40 mg/dL 19 and RLP-C reduction by 31% to 38%.20,21 In such patients who had received pitavastatin 1 mg or 2 mg over 2 months but with suboptimal TG levels, the lipid profiles were improved to more cardioprotective levels by additional treatment with fenofibrate 67 mg.
Durrington et al 2 reported a TG reduction of approximately 25% in 49 type 2 diabetic patients with hyperlipidemia following 6-week administration of micronized fenofibrate 67 mg. Hamamoto et al 22 reported a TG reduction of 28% and HDL-C elevation of 11%, respectively, following 1- to 5-month administration (mean, 3.2 ± 1.1 months) of fenofibrate 67 mg in 68 consecutive case series of outpatients with hypertriglyceridemia in a department of cardiovascular medicine. Tokuno et al 23 reported a TG reduction of 38% and HDL-C elevation of 12%, respectively, after 3-month administration of micronized fenofibrate 100 mg in patients with type 2 diabetes. In this study, the extents of TG reduction and HDL-C elevation were almost similar to those studies. RLP-C, non-HDL-C, TC and LDL-C were also improved.
As for the safety of combination therapy of statin and fenofibrate, it has been confirmed in subpopulation of the FIELD study 3 involving about 890 patients treated with the combination of fenofibrate and a statin and the ACCORD study 24 in which the long-term efficacy and safety of combined therapy with simvastatin and fenofibrate was prospectively examined. Although the evidence obtained from those studies conducted in Europe and the US can not be directly extrapolated to Asians, the information obtained from the present study is considered to be reasonably consistent with those. In this study, the mean levels in ALT, γ-GTP, CK, blood urea nitrogen and serum creatinine were statistically significantly elevated. Fenofibrate are known to increase AST and ALT, as a result of activation of PPARα. 25 Blood urea nitrogen and serum creatinine have also been reported to increase following fenofibrate monotherapy.1,22 Although there still remain needs for careful monitoring on co-administration of a statin and fenofibrate case by case, the statistically significant elevation in ALT, γ-GTP, CK, blood urea nitrogen and serum creatinine observed in this study were considered comparable to those in existing reports.1,22,26 The elevation in γ-GTP which a 64-year-old male experienced and resulted in discontinuation at week 8 was attributed to the administration of fenofibrate, but a cause of it was unknown in detail. It was suspected to be attributed to his background, ie, he had the same episode of liver function abnormality after administration of terbinafine, by the investigator. In fact, it was improved after discontinuation of fenofibrate. Given those, the safety of co-administration of micronized fenofibrate 67 mg daily on pitavastatin 1 mg or 2 mg daily was confirmed at least for up to 16 weeks in this study population.
Unfortunately, primary outcome was not prevented by fenofibrate alone and fenofibrate add-on to statin therapy in the FIELD study 3 and the ACCORD study, 24 respectively. However, especially in patients with type 2 diabetes who have both of elevated TG and reduced HDL-C, fenofibrate might have the further cardioprotective effect even when added on to statin therapy.24,27 Further examination should be needed to confirm the long-term benefit of TG-lowering and/or HDL-C-elevating therapy by fenofibrate.
There were several potential limitations in this study. At first, sample size is too small to evaluate the factors that might affect the effectiveness and safety of this treatment. The second limitation was lack of therapeutic information before starting pitavastatin treatment because this study was observational in nature, collecting consecutive case series meeting the study criteria, under daily clinical practice conditions. For that limitation, it was unable to further evaluate heterogeneity of this study population in terms of dyslipidemia, response to pitavastatin treatment and the effect on lipid profile of co-administration of pitavastatin and fenofibrate. Moreover, the long-term safety and tolerability needs to be further verified, because the duration of this study was for up to only 16 weeks.
The effectiveness and safety of this combined therapy was satisfactory as a whole.
Conclusion
Co-administration of pitavastatin and fenofibrate should be considered as one of the options for the treatment of hypercholesterolemic patients with hypertriglyceridemia, in whom control of the lipid profile sometimes becomes problematic in routine clinical practice.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Acknowledgements
Financial support for this trial was obtained from Nonprofit Organization Chubu Clinical Research Network (Aichi, Japan) with the unrestricted grant from Kowa Company, Ltd (Aichi, Japan). We thank Tatsuya Isomura as for the statistical analyses and the APPROACH-J study investigators in Appendix for conducting this study.
Appendix. APPROACH-J study investigators

| Investigator | Affiliation |
|---|---|
| Kiwami Chikada | Chikada Clinic |
| Satomi Hayakawa | Hayakawa Clinic |
| Shigeki Hotta | Hotta Otona Kodomo Clinic |
| Masashi Kakihana | Yamate Clinic |
| Kengo Kanemaki | Kanemaki Clinic |
| Akihisa Kimura | Kimura heart Clinic |
| Atsushi Kinoshita | Kinoshita Clinic |
| Takenori Mase | Mase Clinic |
| Mitsuharu Mori | Jodo Clinic |
| Junichi Nakagawa | Nakagawa Naika Clinic |
| Hirohiko Nomura | Mitsuba Clinic |
| Hideo Okabe | Okabe Naika |
| Masatoshi Otake | Shimoyama Clinic |
| Yukihiko Sato | Sato Medical Clinic |
| Hiroyuki Shinjo | Wakaba-dori Clinic |
| Satoshi Suzuki | Owariasahi Clinic |
| Mitsuru Yamaguchi | Komura Clinic |
| Hideko Yamamoto | Yamamoto Medical Clinic |
| Yumiko Yano | Fujimoto Geka Naika |
| Tatsuya Yasukawa | Yasukawa Clinic |
