Abstract
Introduction:
The long half-life of atorvastatin and fenofibrate makes them suitable for alternate day therapy. Hence, we aimed to study the efficacy, safety, and cost-effectiveness of alternate day therapy with atorvastatin and fenofibrate combination in mixed dyslipidemia.
Methods:
Eligible patients with mixed dyslipidemia were randomly allotted into 2 equal parallel groups—alternate day therapy group (group 1) and daily therapy group (group 2). Patients in groups 1 and 2 received fixed dose combination of atorvastatin 10 mg and fenofibrate 160 mg on alternate days and daily, respectively, for 12 weeks. Mean percentage change from baseline in triglycerides (TGLs), non-high-density lipoprotein cholesterol (non-HDL-C), HDL-C, low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), and TC–HDL ratio, incidence of adverse effects, and cost-effectiveness were compared in both the groups.
Results:
Among 110 patients randomized, 99 completed the study till 12 weeks treatment duration. The TGLs, non-HDL-C, TC, and LDL-C decreased by 56.4%, 49.7%, 36.5%, and 39.2%, respectively, in alternate day therapy group and by 57.5%, 51.2%, 37.5%, and 39.4%, respectively, in daily therapy group. The HDL-C levels increased by 20.1% in alternate day therapy group compared to 21.8% in daily therapy group. No statistically significant difference was seen between both the groups in mean percentage change in lipid parameters from baseline to end of 12 weeks. Incidence of adverse events was reasonably less in alternate day therapy group.
Conclusion:
Alternate day therapy with atorvastatin–fenofibrate combination is an effective and safe alternative to daily therapy in mixed dyslipidemia. Apart from significant cost savings, reasonable reduction in the incidence of adverse events is seen with alternate day regimen. However, larger studies are needed to more reliably confirm our interesting but preliminary results.
Keywords
Introduction
Cardiovascular diseases are the leading cause of mortality worldwide. 1 Atherosclerosis is a major contributor to cardiovascular diseases and the most important risk factor for the development of atherosclerosis is dyslipidemia. It has been reported that increased cholesterol level is the sixth leading global risk factor for death worldwide. 2 According to World Health Organization, the estimated global prevalence of raised cholesterol levels among adults is 39% (37% for males and 40% for females). 3 Recent estimates also depict that 53% of adults in the United States have dyslipidemia. 4
Mixed dyslipidemia is characterized by increased triglycerides (TGLs) and low-density lipoprotein cholesterol (LDL-C) along with decreased high-density lipoprotein cholesterol (HDL-C). Presence of small-dense LDL particles increases the risk of atherogenesis in patients with mixed dyslipidemia. 5 Mixed dyslipidemia is the commonest type of dyslipidemia in patients with diabetes and metabolic syndrome and is strongly associated with the development of atherosclerosis and cardiovascular events. Due to increasing diabetic population, the prevalence of mixed dyslipidemia is also high. 6,7 According to the recent estimates, mixed dyslipidemia is prevalent in 21% of the adults in the United States. 4
Statins primarily reduce LDL-C but have only modest effects on HDL-C and TGL. On the other hand, fibrates primarily reduce TGL and increase HDL-C but have only modest effects on LDL-C. Hence, combination of statins and fibrates is frequently used for the treatment of mixed dyslipidemia. 8 –12
One of the major limitations of long-term pharmacological treatment of dyslipidemia is suboptimal adherence to drug therapy. 13,14 Recent studies have shown that noncompliance with statins is around 30% to 50% thereby decreasing the therapeutic effects. Further around 50% of patients who begin treatment with hypolipidemic drugs discontinue the treatment at the end of first year. 13 –15 Common reasons for poor patient compliance with these drugs are the adverse effects produced by them like myopathy and high cost of drug therapy. 14,16,17 One of the ways to minimize adverse effects, to reduce cost of drug therapy, and to improve patient compliance is to give these drugs on alternate days instead of daily therapy. Both atorvastatin and fenofibrate have long half-life (t½), which makes them suitable for alternate day therapy. Hence, this pilot study was designed to evaluate the efficacy and safety of alternate day therapy with fixed dose combination (FDC) of atorvastatin 10 mg and fenofibrate 160 mg in comparison with daily therapy in the treatment of mixed dyslipidemia.
Methods
Study Design
The present pilot study is a randomized, active-controlled, 2-group, parallel arm, open-labeled, single-center study. It was conducted in Sri Ramachandra Medical College Hospital, Chennai, a tertiary care teaching hospital in South India, from September 2010 to December 2011. The study was performed in accordance with declaration of Helsinki and ICH-GCP guidelines. The study protocol was reviewed and approved by Institutional Ethics Committee (CSP/10/AUG/11/26). The study was registered in Clinical Trials Registry—India before enrollment of the first study participant (CTRI/2010/091/001328). Written informed consent was obtained from all study participants before performing any study-related procedure.
Eligibility Criteria
Patients of both gender aged >18 years who were diagnosed to have mixed dyslipidemia (TGLs ≥ 200 mg/dL and non-HDL-C ≥ 130mg/dL) and met the criteria for pharmacological treatment per updated National Cholesterol Education Program—Adult Treatment Panel III (NCEP-ATP III) guidelines were included in the study 18 (Non-HDL-C = Total cholesterol − HDL-C).
Patients already taking statins, fibrates, or other hypolipidemic drugs, patients with active hepatic disease and elevated liver enzymes (serum glutamic oxalo-acetic transaminase (SGOT), serum glutamic pyruvic transaminase (SGPT), and alkaline phosphatase >3 times the upper normal limit), uncontrolled diabetes mellitus in spite of drug therapy (hemoglobin A1c > 7%), serum creatinine >1.2 mg/dL, elevated creatine phosphokinase (CPK) levels, and muscular disorders, patients who had myocardial infarction, coronary artery bypass grafting, percutaneous transluminal coronary angioplasty, or cerebrovascular accident within 6 months preceding the study, patients receiving concomitant drugs that might interfere with metabolism of study drugs, postmenopausal women on hormone replacement therapy, pregnant and lactating women, women of child-bearing age not using acceptable methods of contraception, and patients unwilling to give informed consent were excluded from the study.
Study Procedure
Patients who met the eligibility criteria were randomized to 2 parallel groups (alternate day therapy group and daily therapy group) in 1:1 ratio using computer-generated simple randomization in blocks of 10. Allocation concealment was maintained using serially numbered opaque-sealed envelopes. Study drug was the FDC of atorvastatin 10 mg and fenofibrate 160 mg. Patients on alternate day therapy group and daily therapy group received the study drug on alternate days and daily, respectively, for 12 weeks.
There were 3 scheduled visits during the study; baseline visit, after 6 weeks, and after 12 weeks (end of study visit). Serum lipid profile (total cholesterol [TC], HDL-C, LDL-C, TGL, and TC–HDL ratio), creatinine, SGOT, and SGPT were analyzed in the baseline and after 12 weeks of therapy. Lipid profile estimations were done after overnight fasting using venous blood samples. Serum TC was measured using cholesterol oxidase method, TGLs using enzymatic assay, HDL-C using polymer–polyanion, and LDL-C using direct measure. Non-HDL-C was calculated using the formula (Non-HDL-C = Total cholesterol − HDL-C). The CPK levels were estimated only for patients with myalgia.
The study drug was supplied as 6-week calendar pack to all study participants along with compliance card and patient diary. All the study participants were instructed to take the study drug orally with a glass of water after food at bedtime. All the study participants were advised to continue low-fat diet and physical exercise (as per NCEP-ATP III guidelines) throughout the treatment period.
Study End Points
The primary efficacy end points were percentage change in TGL and non-HDL levels from baseline after 12 weeks of therapy. Secondary efficacy end points include percentage change in LDL-C, HDL-C, TC levels, and TC–HDL ratio from baseline after 12 weeks of therapy, cost-effectiveness, and patient compliance in both the groups. Safety end points were incidence of adverse events in both groups and percentage change in liver enzymes SGOT and SGPT from baseline after 12 weeks of therapy.
Assessment of Patient Compliance
Patient compliance was assessed by counting the number of empty blister packs and unused study medications, if any, returned by the study participants and by looking at the patient compliance card. Noncompliance was defined as less than 80% or more than 120% of the predicted number of empty blister packs being returned during the scheduled visits.
Cost-Effectiveness Analysis
Since non-HDL-C represents the atherogenic form of lipoproteins, 6 the drug treatment cost incurred for 1% reduction in non-HDL-C level during treatment period was compared in both the groups to analyze cost-effectiveness.
Adverse Event Reporting During the Study
All the adverse events observed/complained by the study participants were reported in the case report form along with the information about severity (mild, moderate, or severe) and causal relationship to the study medication. Biochemical parameters assessed for safety profile were serum creatinine, SGOT, and SGPT.
Statistical Analysis
Since this is a pilot study, no formal sample size calculation was done. A convenient sample of 110 patients was chosen for this study. All statistical analyses were performed using SPSS for windows version 16. Demographic data (age, sex, etc) and baseline patient characteristics were summarized by treatment groups using descriptive statistics to assess difference if any between them. Mean and standard deviation (SD) were provided for continuous data, while absolute frequencies and percentage were provided for categorical data. The TGL, LDL-C, TC, HDL-C, and non-HDL levels in each group before and after intervention were compared using paired “t” test. The mean percentage change in the above-mentioned lipid parameters from baseline to 12 weeks between both the groups was compared using student “t” test. Nonparametric tests were used for the data that did not follow normal distribution. P value <.05 was considered statistically significant. All the analyses were based on modified intention-to-treat (ITT) principle. Modified ITT population included all the study participants who had completed at least 1 follow-up visit and had posttreatment lipid parameter values.
Results
Of the 160 patients screened, 50 patients were excluded due to screening failure. The remaining 110 patients were randomly assigned to 2 groups—alternate day therapy group and daily therapy group, each consisting of 55 participants. Baseline patient characteristics were similar in both the groups as shown in Table 1.
Baseline Patient Characteristics (Randomized Population).a
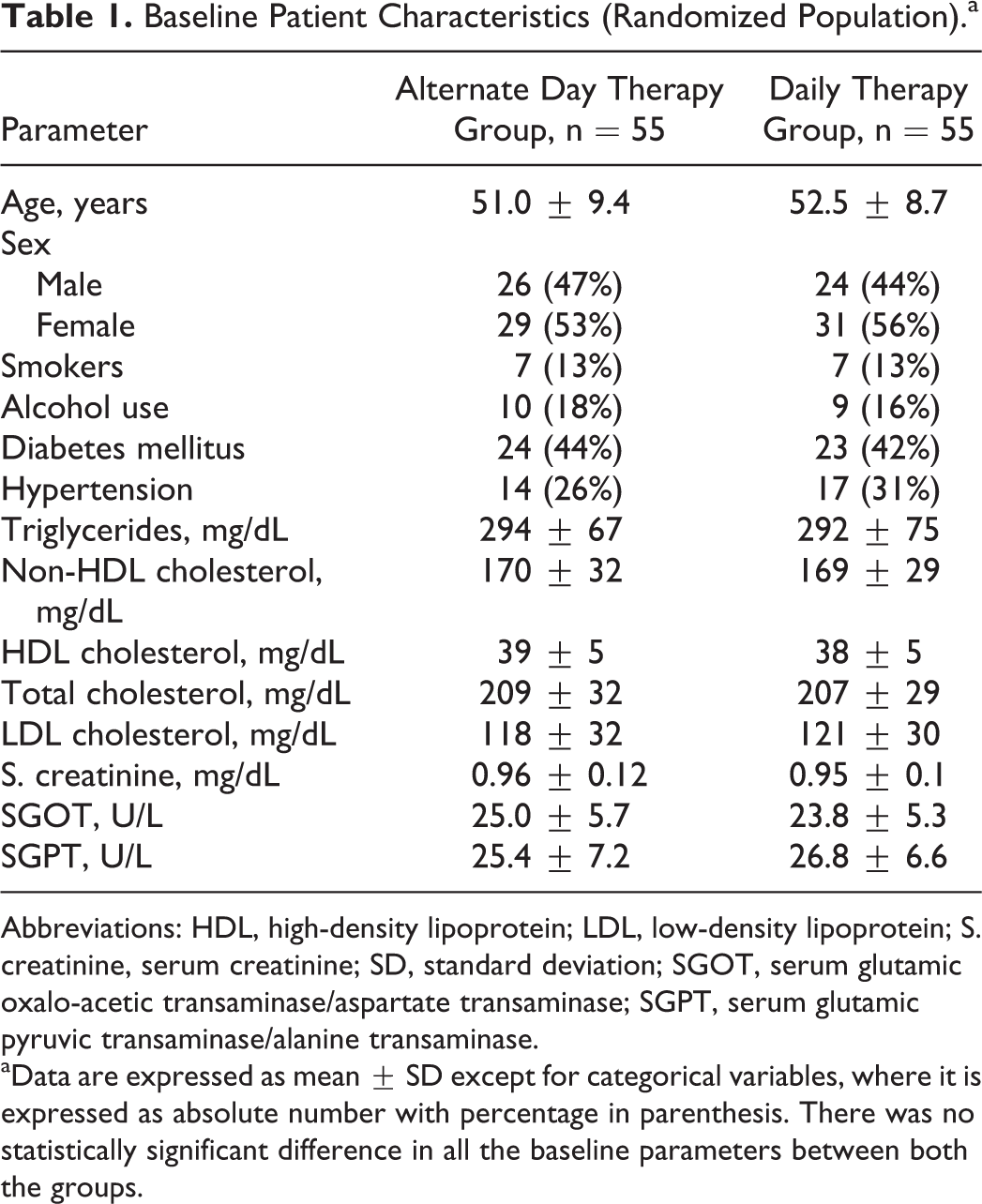
Abbreviations: HDL, high-density lipoprotein; LDL, low-density lipoprotein; S. creatinine, serum creatinine; SD, standard deviation; SGOT, serum glutamic oxalo-acetic transaminase/aspartate transaminase; SGPT, serum glutamic pyruvic transaminase/alanine transaminase.
aData are expressed as mean ± SD except for categorical variables, where it is expressed as absolute number with percentage in parenthesis. There was no statistically significant difference in all the baseline parameters between both the groups.
Among 110 patients randomized, 99 completed the study till 12 weeks treatment duration, while 11 patients discontinued. In all, 4 patients in alternate day therapy arm and 5 patients in daily therapy arm were lost to follow-up before the first follow-up visit. One patient in both arms withdrew consent due to personal reasons (relocation to different place). In all, 51 patients in alternate day therapy arm and 50 patients in daily therapy arm were included in modified ITT population. Remaining 9 patients were excluded from the analysis due to lack of posttreatment lipid values, since they didn’t even turn up for a single follow-up visit. The flow of study participants is shown in Figure 1.
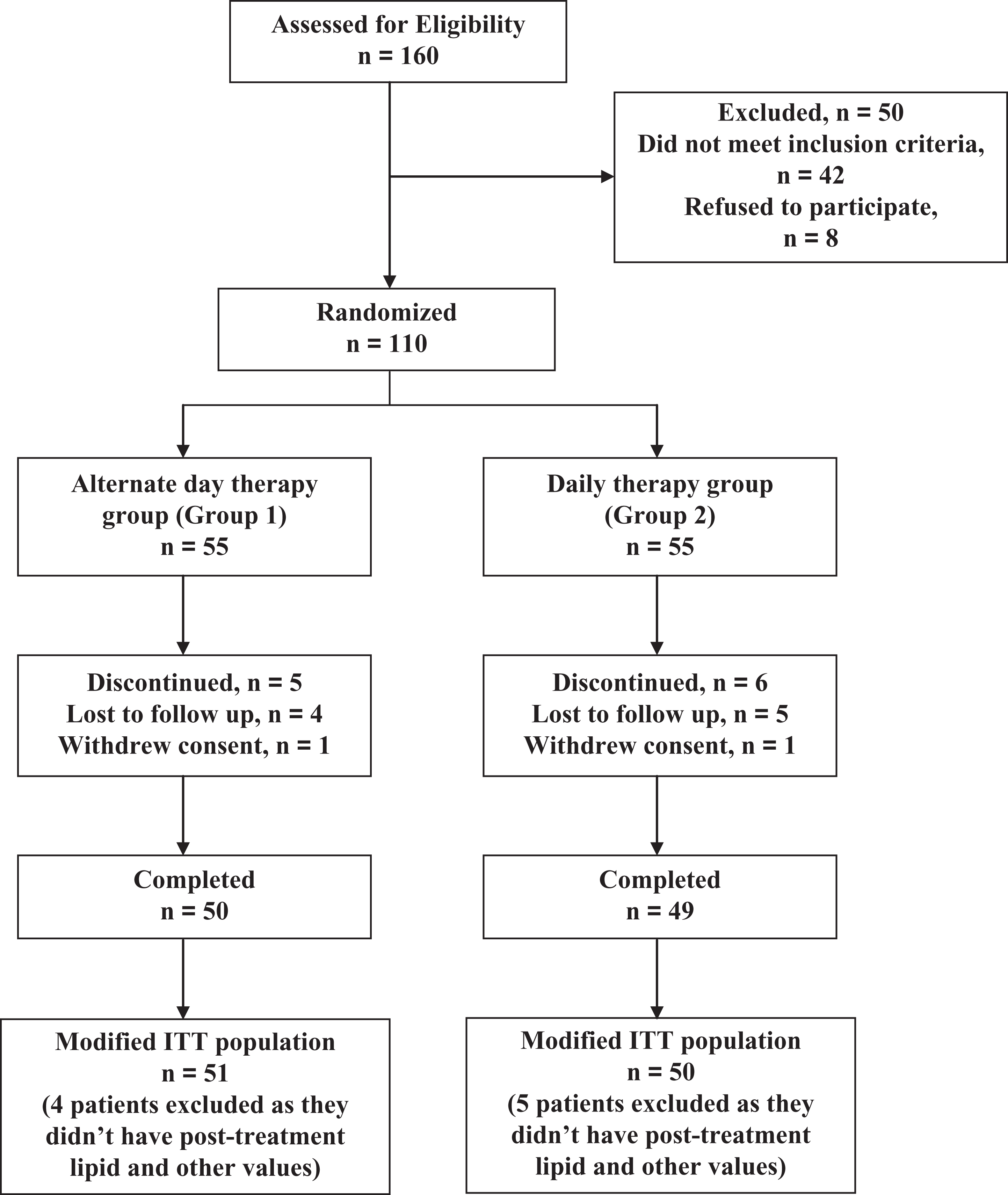
Flow of study participants.
Efficacy Profile
The reduction in TGLs, non-HDL-C, TC, LDL-C, and TC–HDL ratio and increase in HDL-C after 12 weeks of therapy were highly significant (P < .001) when compared to baseline measurements in both the groups (Table 2).
Pre- and Posttreatment Lipid Profile Values in Both Groups.a
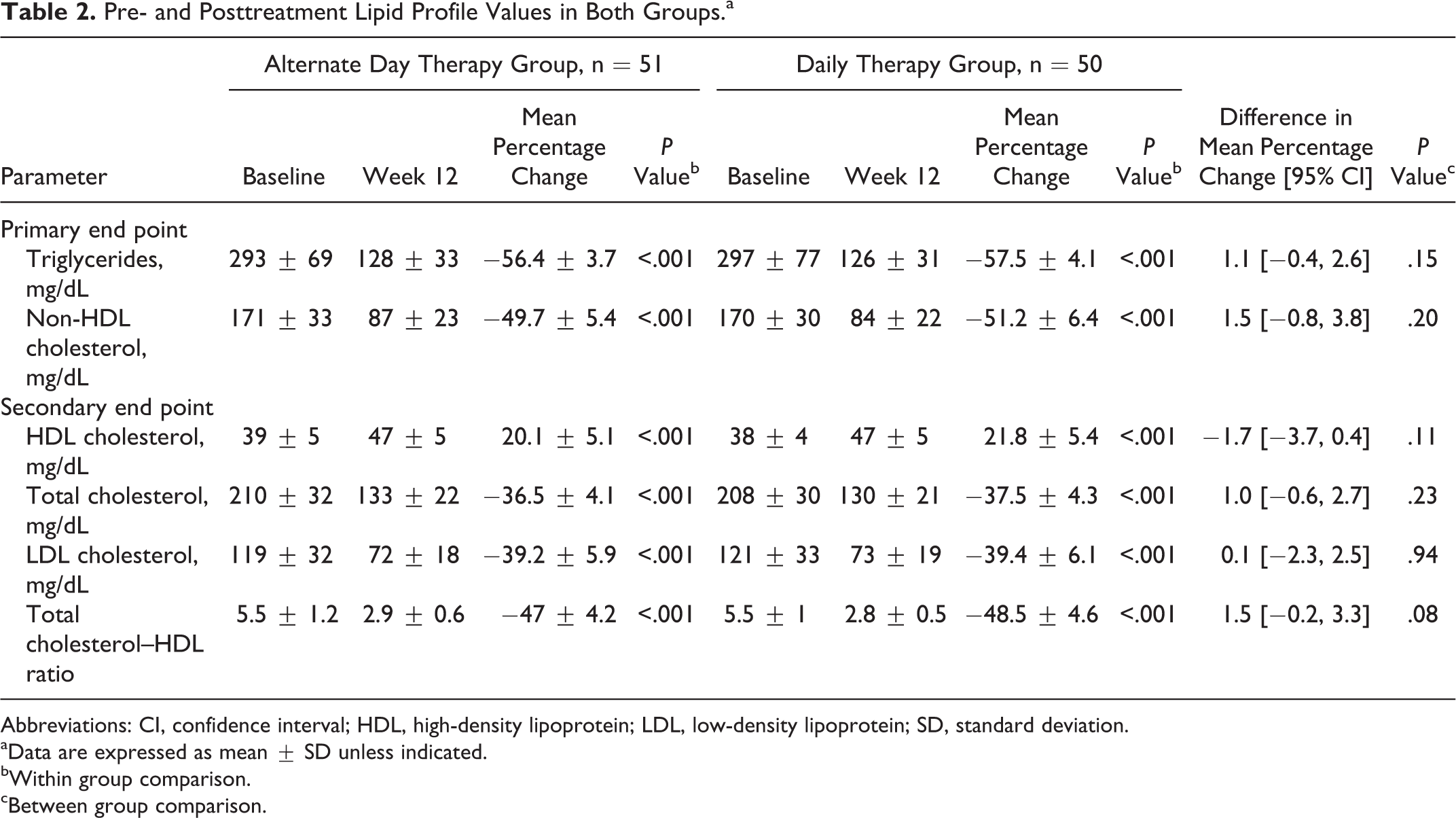
Abbreviations: CI, confidence interval; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SD, standard deviation.
aData are expressed as mean ± SD unless indicated.
bWithin group comparison.
cBetween group comparison.
Triglycerides, non-HDL-C, TC, and LDL-C decreased by 56.4%, 49.7%, 36.5%, and 39.2%, respectively, in alternate day therapy group and by 57.5%, 51.2%, 37.5%, and 39.4%, respectively, in daily therapy group. The HDL-C levels increased by 20.1% in alternate day therapy group compared to 21.8% in daily therapy group (Table 2 and Figure 2).

Mean percentage change from baseline in lipid profile values after 12 weeks of therapy in both the groups. Error bars in the chart represent standard deviation (SD).
The decrease in TGLs, non-HDL-C, TC, and LDL-C and the increase in HDL-C were comparable in both the groups. No statistically significant difference was observed between both the groups in the mean percentage change in all the lipid parameters from baseline after 12 weeks of therapy (Table 2 and Figure 2).
All patients in both the groups were compliant with the drug treatment. The mean drug treatment cost (±SD) incurred for 1% reduction in non-HDL level was 9.2 (±1) Indian Rupee (INR) and 17.9 (±2.5) INR in alternate day and daily therapy groups, respectively (P < .001).
Tolerability Profile
The study drug was well tolerated in both the groups. No statistically significant difference was observed between the baseline and the posttreatment mean serum creatinine, SGOT, and SGPT levels in alternate day therapy group. But a mild (but not clinically meaningful) increase in posttreatment serum creatinine (P = .03) and SGOT levels (P < .01) compared to baseline was noted in the daily therapy group (Table 3). However, no statistically significant difference was noted between the 2 groups in the mean percentage change in creatinine, SGOT, and SGPT levels from baseline after 12 weeks of therapy.
Pre- and Posttreatment Serum Creatinine, SGOT, and SGPT Levels in Both Groups.a

Abbreviations: S. creatinine, serum creatinine; SD, standard deviation; SGOT, serum glutamic oxalo-acetic transaminase; SGPT, serum glutamic pyruvic transaminase.
aData are expressed as mean ± SD unless indicated.
bMann-Whitney U test.
Two patients in the alternate day therapy group and 3 patients in the daily therapy group had headache that was mild to moderate in intensity and was controlled by analgesics (paracetamol). Three patients in the alternate day therapy group and 6 patients in the daily therapy group had mild to moderate myalgia, but none of them had increased serum CPK levels and hence were continued on the same treatment without any interruption. One patient in the daily therapy group had moderate itching that was controlled by antihistamines (cetirizine). The adverse events seen in both the groups are summarized in Table 4. No discontinuation of treatment due to adverse effects was seen in either of the study groups. There was no incidence of rhabdomyolysis or serious adverse events in either of the groups.
Incidence of Adverse Events in Both Groups During 12-Week Study Period.a

aData denote absolute number of patients with percentage in parenthesis.
Discussion
The present study evaluated the efficacy and safety of alternate day therapy with FDC of atorvastatin 10 mg and fenofibrate 160 mg in comparison with daily therapy in reducing TGLs, non-HDL-C, TC, and LDL-C and increasing HDL-C in patients with mixed dyslipidemia.
The study results demonstrate a comparable efficacy of alternate day therapy with daily therapy in controlling all the lipid parameters. The difference in mean percentage change in lipid parameters between both the groups was not statistically significant. This might be due to the long half-life of both atorvastatin and fenofibrate and synergistic effect of both drugs in reducing the lipid levels. The half-life of atorvastatin is 19.5 ± 9.6 hours, which increases with age. 19 But the half-life of inhibitory activity for 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase is still longer, that is, 20 to 30 hours due to the contribution of active metabolites. 20 Atorvastatin is a statin whose metabolite has HMG-CoA reductase enzyme inhibitory activity similar to that of nonmetabolized atorvastatin. The t ½ of fenofibrate is 20 to 27 hours. 11,19
No statistically significant difference was observed in the primary and secondary outcomes in either of the groups with respect to gender, diabetic status, smoking, alcoholism, and different age groups.
Two patients in the alternate day therapy group and 1 patient in the daily therapy group failed to attain the target TGLs goal. This can be attributed to high baseline triglycerides level (>400 mg/dL) in all 3 patients and noncompliance to diet and exercise despite being compliant with drug therapy. One patient in the daily therapy group did not attain the target HDL-C goal, which might be due to chronic smoking, alcoholism, and very low baseline HDL-C value of 28 mg/dL in that patient. Similarly, 2 patients in both the groups failed to attain LDL-C target goals, which might also be due to very high baseline LDL-C levels (>190 mg/dL) in these patients and failure to follow dietary advice. However, all patients in both the groups attained the target TC goals at the end of 12 weeks.
The dose of atorvastatin in the FDC used in the present study is 10 mg in contrast to high doses of the same drug used in the previous studies. Past studies evaluating atorvastatin–fenofibrate combination have used 20 to 40 mg of atorvastatin along with 100 to 200 mg of fenofibrate. Athyros et al evaluated FDC of 20 mg atorvastatin and 200 mg of fenofibrate, in comparison with monotherapy of both the individual drugs. 21 They reported a reduction of 37%, 46%, and 50% in TC, LDL-C, and TGLs, respectively, with 22% increase in HDL-C levels in the FDC group, which is comparable with the results of our present study in both alternate day therapy and daily therapy groups.
Similarly previous studies evaluating alternate day regimen with atorvastatin monotherapy have used various doses ranging from 10 to 40 mg of atorvastatin. Matalka et al used 10 mg atorvastatin initially followed by doubling the dose (20 mg) after 6 weeks. 22 The mean daily dose of atorvastatin used by them was 18 mg in alternate day therapy group and 12 mg in daily therapy group. Keles et al evaluated the lipid-lowering efficacy of 20 mg atorvastatin when administered on alternate days compared with daily administration. 23 Both the above-mentioned studies reported that lipid-lowering efficacy of alternate day atorvastatin therapy was comparable to that of daily therapy along with reasonable cost savings.
Our study is the first of its kind to evaluate the combination of atorvastatin and fenofibrate when given on alternate day basis for the treatment of dyslipidemia. So far only 1 study has been reported in the literature regarding alternate day therapy with high-dose fenofibrate (250 mg). In that study, Kayikcioglu et al evaluated the efficacy of alternate day simvastatin and fenofibrate in patients with mixed dyslipidemia. 24 They used simvastatin 10 mg and fenofibrate 250 mg as alternate day regimen in such a way that the patients received fenofibrate 250 mg on the days when simvastatin 10 mg was not taken. They concluded that alternate day regimen was as effective as daily regimen in reducing the lipid levels but with a lower cost and less adverse effects.
Despite low doses of both atorvastatin and fenofibrate used in our present study, mean percentage change in lipid parameters were comparable to that observed in previous studies. This can be attributed to the fact that Asians require a lower dose of lipid-lowering drugs like statins when compared to the Western population, which in turn might be due to genetic variations in hepatic drug transporters and metabolism of statins. 25,26
The study drug was well tolerated in both the groups. However, the incidence of drug-related adverse events were significantly and meaningfully less frequent among patients receiving alternate day therapy, thus reflecting a better tolerability and safety of alternate day lipid lowering therapy. This can be attributed to frequent drug-free intervals in the alternate day therapy group. Although a mild increase in posttreatment serum creatinine and SGOT levels were seen in daily therapy group, it was not clinically meaningful.
Patients in both groups were fully compliant to drug therapy, and no difference in treatment compliance was observed between the groups. This might be due to weekly reminders given to all study participants during the treatment period and short duration of the study treatment period (12 weeks).
Both atorvastatin and fenofibrate are expensive drugs. The minimum cost of generic drug that contains a FDC of atorvastatin 10 mg and fenofibrate 160 mg in Indian market is around 10 INR per tablet. 27 The mean cost of drug therapy required for 1% reduction in non-HDL-C during 12 weeks treatment period in the present study was 9.2 INR in alternate day therapy group while the same for daily therapy group was 17.9 INR. This shows that alternate day therapy provides a reduction in the treatment cost of around 48% when compared to daily therapy that amounts to minimum cost savings of 1752 INR (approximately 32 US$) per year for a patient. This cost savings could be extremely useful in developing countries like India, where the cost of drug therapy highly influences the treatment option.
The main reason for decreased patient compliance with statins, as reported in the literature, is due to its adverse effects and high treatment costs. Since alternate day therapy is associated with fewer incidences of adverse events and less treatment cost when compared to daily therapy, patients on alternate day therapy are expected to have better compliance with lesser discontinuation rates.
However, there are a few limitations of our study. Small sample size of the current study may limit extrapolation and generalization of our study results. It is possible that a type II error inherently attributed to the small sample size may have inadvertently resulted in the inability to show a significant difference in lipid levels between our 2 treatment groups. Thus our results are at best preliminary and hypothesis generating. A much larger study with a better statistical power will be needed to more reliably confirm our results. Because of the shorter duration of our study (12 weeks), we could not measure the effect of therapy on cardiovascular end points. The open label nature of this study could be a source of bias, especially with regard to the adverse events. A double-blind study of similar kind can solve this issue. Inflammatory markers for atherosclerosis like high-sensitivity C reactive protein levels were not measured in the present study due to financial constraints since it was an investigator-funded study.
Conclusion
This pilot study has revealed that alternate day therapy with atorvastatin and fenofibrate combination is as good as daily therapy in controlling the lipid parameters. At the same time, alternate day therapy is associated with fewer incidences of adverse events and significant reduction in drug treatment cost when compared to daily therapy. Therefore, better patient compliance and less treatment discontinuation rates are expected with alternate day regimen.
To conclude, alternate day regimen is as effective as daily regimen in controlling lipid parameters during short-term therapy. But the effect of alternate day therapy on long-term cardiovascular outcomes is not known. Hence, larger studies with long follow-up are needed to more reliably confirm our interesting but preliminary results and conclusions.
Footnotes
Authors’ Note
Name of the institution where the work reported was done Sri Ramachandra Medical College and Research Institute, Chennai 600116, India.
Acknowledgments
The authors wish to acknowledge Dr Ramesh, Professor of Cardiology, SRMC for his encouragement and support in conducting the study. We acknowledge Mr David, Biostatistician for helping in statistical analysis and Dr S. Sandhiya, Department of Clinical Pharmacology, JIPMER for critically reviewing the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
