Abstract
It still remains unknown whether angiotensin-receptor blockers (ARBs) are cardioprotective in patients with type 2 diabetes. The recent two clinical trials, the ROADMAP and the ORIENT, have suggested that fatal cardiovascular events or cardiovascular deaths were unexpectedly higher in olmesartan group. These results suggest that aggressive blood pressure lowering may cause a higher risk in some high-risk patients, especially in those with preexisting coronary heart disease, indicating a possibility that the J-curve phenomenon may exist in some group of patients.
Angiotensin-receptor blockers (ARBs) have recently been demonstrated to be renoprotective in patients with type 2 diabetes and overt nephropathy or microalbuminuria,1,2 as shown in Figure 1. However, it still remains unknown whether ARBs are cardioprotective in patients with type 2 diabetes. All of the recent studies have indicated that ARBs are equal to or not inferior to calcium channel blocker (CCB), amlodipine,3–5 except for one study (Jikei Heart Study), claiming that valsartan was superior to amlodipine in the cardiovascular outcome, 6 which is very controversial because of its equivocal primary endpoint setting.
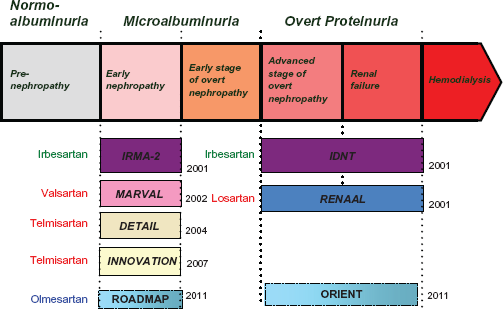
Clinical large trials of angiotensin-receptor blockers (ARBs) in patients with hypertension and chronic kidney disease.
Recently, the results of the two clinical trials, the ROADMAP (Randomized Olmesartan and Diabetes Microalbuminuria Prevention Study) and the ORIENT (Olmesartan Reducing Incidence of Endstage Renal Disease in Diabetic Nephropathy Trial), which investigated the renoprotective and cardioprotective effects of one of ARBs, olmesartan, have been published.7,8 Several studies have reported that the antihypertensive and renoprotective effects of olmesartan might be more pronounced than other ARBs, such as candesartan,9,10 and thus the studies were expected to show better results than the studies which had been done with other ARBs. Unexpectedly, both studies showed a higher rate of cardiovascular deaths in patients taking olmesartan compared to placebo.
The ROADMAP was a randomized, double-blind, placebo-controlled, multicenter trial conducted in Europe. The trial included 4,447 patients with type 2 diabetes and at least one additional cardiovascular risk factor, but without overt evidence of nephropathy. Patients were randomized to receive either 40 mg of olmesartan or placebo daily. Patients were permitted to receive other antihypertensive medications, but not ACEIs or ARBs. The ROADMAP Study indicated that olmesartan was associated with a delayed onset of microalbuminuria, even though blood-pressure control in both groups was excellent according to current standards. However, it also showed that the rate of fatal cardiovascular events was higher with olmesartan among patients with preexisting coronary heart disease. 7
Another study, the ORIENT, was a randomized, double-blind, placebo-controlled, multicenter trial conducted in Japan and Hong Kong. Patients (n = 566) with type 2 diabetes and overt nephropathy were randomized to receive olmesartan 10 mg to 40 mg or placebo daily. The ORIENT did not only demonstrate significant difference of the primary renal endpoint, but it also indicated that there were 10 cardiovascular deaths in the olmesartan group versus 3 in the placebo group. 8
Although the ORIENT has not been published yet except for an abstract form, 8 the ROADMAP trial has been published, 7 which showed that a greater number had fatal cardiovascular events was attributable in part to a higher rate of death from cardiovascular causes in the olmesartan group than in the placebo group among patients with preexisting coronary heart disease (11 of 564 patients [2.0%] vs. 1 of 540 [0.2%], P = 0.02). Furthermore, it was suggested that the death was higher in the subjects either in the lowest quartile of blood pressure or in the highest quartile of blood-pressure reduction during follow-up. 7 Therefore, excessive reduction of blood pressure in some high-risk patients may confer a predisposition to an increased risk of a death, suggesting a possibility that J-curve effect of antihypertensive drugs may really exist. However, the conclusion about causal effect relationship between blood pressure and cardiovascular death cannot be drawn from the two studies. Many clinical findings hitherto have excluded a possibility that J-curve may exist in the range of blood pressure target, such as 130/80 mmHg in type 2 diabetes, whereas a recent observational subanalysis of the International Verapamil SR-Tandolapril Study (INVEST) suggested that tight control of systolic blood pressure among patients with diabetes was not associated with improved cardiovascular outcomes. 11

Effects of angiotensin-receptor blockers (ARBs) or angiotensin-converting enzyme inhibitors (ACEIs) and calcium channel blockers (CCBs) on cardiovascular events in large clinical trials.
These findings remind us of the recent controversy about the aggressive blood glucose-lowering therapy might increase the risk of death in subjects with type 2 diabetes, suggested by the ACCORD study. 12 The American Heart Association states that the group at the bottom-left part of the curve (with very low blood pressure and low cholesterol levels) tends to be different from the general population in other ways. Those differences may contribute to the apparent increase in mortality, and that the notion that effectively lowering blood pressure or cholesterol levels may be harmful is unfounded. 13 Further studies are required to investigate whether the increased cardiovascular death found in the two trials with olmesartan might be a general phenomenon with ARBs or all antihypertensive drugs, or alternatively an incidental phenomenon.
The FDA said that it has not concluded that Benicar (olmesartan) increases the risk of death, and that patients taking the drug for hypertension should not stop taking it unless told to do so by a healthcare professional.
Disclosure
The author (Y.H.) received speaker's fees from MSD Japan, Novartis Pharma, Novo Nordisk, Daichi-Sankyo Co., and Eli Lilly Co. The author (H.K.) received research grants from MSD, Daichi-Sankyo Co. and lecture fees or consultation fees from MSD Japan, Daichi-Sankyo Co. Pfizer Co., Novartis Co. Novo Nordisk, Eli Lilly Co. Bohringer-Ingelheim Co., and Astellas Co.
The authors confirm that they have permission to reproduce any copyrighted material.
