Abstract
Asthma is characterized by the accumulation of eosinophils in the airways in most phenotypes. Eosinophils are inflammatory cells that require an external survival-prolonging stimulus such as granulocyte macrophage-colony-stimulating factor (GM-CSF), interleukin (IL)-5, or IL-3 for survival. In their absence, eosinophils are programmed to die by spontaneous apoptosis in a few days. Eosinophil apoptosis can be accelerated by Fas ligation or by pharmacological agents such as glucocorticoids. Evidence exists for the relevance of these survival-prolonging and pro-apoptotic agents in the regulation of eosinophilic inflammation in inflamed airways. Much less is known about the physiological significance and mechanisms of spontaneous eosinophil apoptosis even though it forms the basis of regulation of eosinophil longevity by pathophysiological factors and pharmacological agents. This review concentrates on discussing the mechanisms of spontaneous eosinophil apoptosis compared to those of glucocorticoid- and Fas-induced apoptosis. We aim to answer the question whether the external apoptotic stimuli only augment the ongoing pathway of spontaneous apoptosis or truly activate a specific pathway.
Introduction
Eosinophilic granulocytes account only for approximately 3% of blood leukocytes in healthy individuals. Similar to neutrophilic granulocytes, they are cells specialized to kill pathogens by the secretion of toxic mediators but also able to regulate function of other immune cells. Although evolutionary, the function of eosinophils is thought to be the innate immune response against parasitic helminthes; 1 they are also critically involved in the pathogenesis of allergic, gastrointestinal, and hypereosinophilic disorders and in tumor immunity.2–5 Allergic asthma is characterized by the accumulation of eosinophils in the airways. The current evidence suggests that eosinophils are critical mediators of asthma exacerbations and airway remodelling.6–9
The biology of eosinophils differs from many other immune cells or malignant cell lines in their requirement for an external stimulus for continuation of survival. In the absence of any such stimulant (eg granulocyte macrophage-colony stimulating factor (GM-CSF), interleukin (IL)-5 or IL-3), they die by spontaneous (also termed as “passive”) apoptosis in a few days. 10 Apoptosis is characterized by cell shrinkage, nuclear coalescence, chromatin condensation, and DNA fragmentation leading to the formation of apoptotic bodies and in vivo, to their ingestion by macrophages or other phagocytes. Generally, apoptosis can be induced via two different pathways, extrinsic (receptor mediated) or intrinsic (mitochondrial centered) (Fig. 1). 11
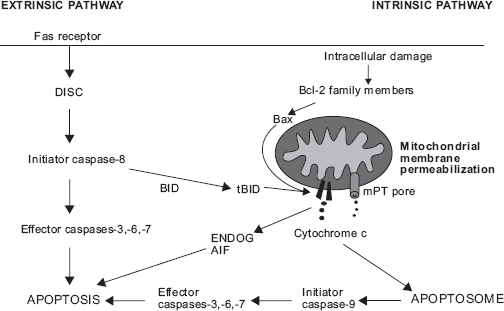
The main features of extrinsic and intrinsic apoptosis pathways. Fas receptor-mediated pathway is shown as an example of extrinsic apoptotic pathway. Extrinsic apoptosis is initiated by ligation of death receptor Fas leading to the formation of DISC and activation of caspases. Sometimes, BID cleavage into truncated BID (tBID) and mitochondrial route is required for caspase activation in extrinsic apoptosis. Intracellular stress conditions initiate intrinsic pathway of apoptosis, where Bcl-2 family members and MMP play major roles. MMP can be mediated by pore-forming activity of Bax and/or tBID or by mPT. If caspases are inhibited, apoptosis may be executed by apoptosis-inducing factor (AIF) and endonuclease G (ENDOG).
A majority of the studies on the regulation of eosinophil apoptosis have used allergic asthma as a starting point, ie they have focused on the significance and mechanisms of survival-prolonging cytokines.10,12,13 Given the importance of eosinophils in certain phenotypes of asthma,14,15 this approach is very sensible. However, eosinophils, albeit in low numbers, are also present in the blood and tissues in healthy individuals. It has been estimated that the mean turnover of eosinophils is approximately 2.2 × 108 cells/kg/day. Once in the circulation, eosinophils have a half-life of approximately 8-18 h and a mean blood transit time of approximately 25-26 h.10,16,17 In two recent studies, migration of radiolabeled eosinophils was followed in healthy individuals in real time. Eosinophils, after leaving the blood circulation, transited through the lungs and accumulated in the liver and spleen.16,18 Evidence was also attained regarding the re-entrance of eosinophils from the liver to the circulation. 16 However, 48 h after injection of the radiolabeled eosinophils, approximately 50% of them resided in the liver, 30% in the spleen, and the rest in other organs. It is reasonable to hypothesize that the liver may be the primary site of eosinophil clearance through apoptosis, possibly spontaneous apoptosis. No direct evidence, however, exists about the occurrence of spontaneous (or passive) apoptosis in vivo. As studying the mechanisms of eosinophil survival and apoptosis in tissue samples is largely impossible, a vast majority of eosinophil studies have used eosinophils isolated from the peripheral blood by the CD16-negative selection. Indirect evidence of the appearance of spontaneous eosinophil apoptosis in blood was obtained recently in a study evaluating the aging of cells in blood samples, where eosinophils showed a decrease in forward side scatter (FSC) values indicative of smaller cell size typical to apoptosis. 19 Furthermore, the occurrence of blood eosinophil apoptosis, even though not spontaneous, was demonstrated in mice in vivo after the administration of Siglec-F antibody. 20
Eosinophil apoptosis can be induced by several agents facilitating clearance of eosinophilic inflammation. Eosinophil apoptosis can be induced eg by ligation of Fas or by ligation of tumor necrosis factor receptor (TNFR) family member CD30 and by many pharmacological agents, such as glucocorticoids, theophylline, and leukotriene modifiers.21–27 Plenty of evidence exists supporting the occurrence of steroid- induced eosinophil apoptosis in the airways of steroid-treated asthmatics.28–31 Many cell types of the airways, such as bronchial epithelial cells, bronchial smooth muscle cells, fibro- blasts, T cells, and eosinophils express Fas ligand (FasL),29,32–34 and in T cells, the expression is reduced by Th2 cytokines GM-CSF, IL-5, and IL-4.
35
Neutralization of FasL enhanced airway eosinophilia in a mouse model of allergic asthma providing evidence that FasL is a relevant pro-apoptotic agent for eosinophils
To combat the eosinophilia associated with several disease conditions, understanding the signaling patterns related to eosinophil survival and apoptosis is extremely important. Ideally, a novel pharmacological agent aimed specifically to deplete eosinophils by inducing eosinophil apoptosis could be targeted to cover all the following options: (1) inhibit the action or signaling of survival-prolonging factors, (2) mimic the action and/or signaling of known external inducers of apoptosis such as FasL or glucocorticoids, and (3) enhance the intrinsic pro-apoptotic signaling pathway during spontaneous (passive) eosinophil death. This review focuses on the signaling of spontaneous eosinophil death, a phenomenon that is largely neglected, and compares it with the mechanisms of known inducers of eosinophil apoptosis, FasL, and glucocorticoids.
Inhibitory Signals for Spontaneous Apoptosis
Many eosinophil survival-prolonging inflammatory agents, such as GM-CSF, IL-5 and IL-3, are present in the lungs of asthmatics, and eosinophil apoptosis has been shown to be reduced in the airway submucosa of patients with steroid-untreated asthma when compared to healthy controls.29,38–40 Eosinophil survival may be prolonged up to 1-2 weeks in response to these cytokines, and IL-5 is the most potent. 41 GM-CSF, however, seems to be the main eosinophil survival-prolonging cytokine in asthmatic airways.39,40 Pathways activated by IL-5/GM-CSF include Lyn/Syk-Ras-Raf-1-extracellular signal-regulated kinases (ERK) 1/2, Jak2-STAT1, and PI3K-Akt in eosinophils.42–49 Of these, the ERK pathway does not seem to be involved in the survival-prolonging action.50,51 In addition, inhibition of Bax translocation to mitochondria by IL-5 and GM-CSF has been shown in eosinophils.49,52 TGF-β, interestingly, abrogated IL-5/GM-CSF-induced eosinophil survival, and this mechanism involved inhibition of tyrosine phopshorylation of Jak2, Lyn, and ERK 1/2 as well as inhibition of phosphorylation of STAT1 and Akt.53–55
In addition, other significant survival-prolonging factors seem to exist because delayed apoptosis of blood and nasal polyp tissue eosinophils was only partly prevented by anti-GM-CSF, anti-IL-5, and/or anti-IL-3 antibodies.56,57 Many pathogenic components, cytokines (eg TNF-α, leptin, interferons (IFNs)) and allergens also prolong eosinophil survival.10,58–65 Generally, NF-kB may be the most important transcription factor mediating eosinophil survival, as its inhibition turns eosinophils into the apoptotic cascade.64,66
TNF- α, which can be produced locally by mast cells, was demonstrated to be an anti-apoptotic factor for eosinophils if NF-kB was not inhibited. This effect was proposed to be mediated via TNF receptors, NF-kB, and induction of GM-CSF production.60,67 IFN-γ is produced by T helper 1 cells, and its effects on eosinophils seem to be complex. IFN-γ inhibited IL-3- or IL-5-induced differentiation of eosinophils from cord blood mononuclear cells 68 but prolonged eosinophil survival in vitro. 69 Leptin is a cytokine that is mainly produced by adipocytes of white adipose tissue with the main function related to inhibition of appetite. Leptin has been shown to increase eosinophil survival even though it remains unclear whether the survival-prolonging concentrations may be reached in vivo. 70
In addition, CD40-CD40L interaction has been shown to prolong eosinophil survival. Freshly isolated blood eosinophils did not express CD40, but the expression was strong after 48 h of culture. The mechanism of CD40L-induced survival prolongation involved induced expression of cellular inhibitor of apoptosis proteins (cIAPs). 71 CD40L-deficient mice showed decreased eosinophilic lung inflammation 72 h but not 24 h after allergen challenge 72 suggesting that CD40-CD40L interaction affects maintenance of eosinophilic airway inflammation. In the absence of any of these or other survival-prolonging factors, eosinophils proceed into spontaneous apoptosis.
Progression of Spontaneous Apoptosis
When eosinophils isolated from human blood are cultured in the absence of any inducers or inhibitors of apoptosis, approximately half of them undergo spontaneous apoptosis in 2 days.64,73 We have applied several methods for the determination of eosinophil apoptosis providing basic information on the progression of apoptosis and the cascade of apoptotic events in spontaneously dying eosinophils. According to our combined data over time, apoptotic values obtained with Annexin-V/ propidium iodide double-staining are continuously higher when compared to those obtained with other standard methods of apoptosis determination (DNA fragmentation assay, morphological examination, ΔΨm dissipation) (Fig. 2, unpublished observation). This suggests that cell surface expression of phosphatidylserine (PS) precedes many well-known manifestations of apoptosis in eosinophils. Early occurrence of PS exposure has been previously demonstrated in eosinophils, 74 and evidence exists also of PS exposure as a caspase-dependent event.52,74,75 Mitochondrial events such as cytochrome c release and ΔΨm dissipation were shown to occur after PS exposure in eosinophils, which was in contrast to lymphocytes. 74 It seems that the order of events is stimulus dependent as well as cell- type dependent.74,76,77 To further support early time-course of PS exposure, it was shown that PS exposure preceded cell shrinkage and DNA fragmentation in a lymphoma cell line by using three different stimulants to induce apoptosis. 78 During the process of apoptosis, early appearance of PS is logical since PS functions as a cell surface signal for phagocytes to ingest the apoptotic cells and attraction of phagocytes and phagocytosis may be considered as one of the most important events for occurrence of apoptosis in a non-inflammatory fashion. 37 Indeed, inhibition of PS exposure during apoptosis led to more than 50% reduction in engulfment of apoptotic cells. 79 However, from the methodological point of view, it is recommended to measure apoptosis using a combination of different methods, not solely Annexin-V-assay despite the early appearance of PS in apoptotic cells. All methodologies to analyze apoptosis have their drawbacks.80,81
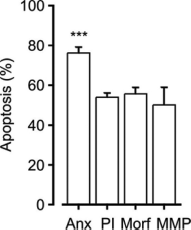
Comparison of percentages of spontaneous eosinophil apoptosis obtained by different apoptosis determination methods. Apoptosis was determined by Annexin-V FITC/propidium iodide double-staining (Anx,
Mediators of Spontaneous Eosinophil Apoptosis
Bcl-2 members and mitochondrial events during spontaneous eosinophil apoptosis
Members of Bcl-2 family are critical in monitoring intracellular damage and important for mitochondrial membrane permeabilization (MMP) to occur, especially in the intrinsic pathway of apoptosis. The Bcl-2 family consists of a group of anti-apoptotic proteins and two groups of pro-apoptotic proteins.82,83 Because eosinophils undergo apoptosis quite rapidly, the expression of proteins regulating longevity is balanced toward pro-apoptotic members. Generally, pro-apoptotic Bcl-2 family members Bax and Bid are strongly expressed in untreated human eosinophils.49,84,85 Cleavage of Bax and Bid into pore-forming fragments enables permeabilization of the outer mitochondrial membrane and release of cytochrome c. It was shown that during spontaneous eosinophil apoptosis, Bax is clustered and re-localized into mitochondria, independent from caspases, and this leads to the release of cytochrome c to the cytosol and activation of caspases.52,86 An accelerated Bax translocation is observed in dexamethasone- treated eosinophils. 86 Also Bid is processed during spontaneous apoptosis and at a faster rate during Fas- and glucocorticoid- induced apoptosis. 84 Spontaneous, FasL-, and dexamethasone- mediated eosinophil apoptosis were reduced by 30, 50, and 25%, respectively, in cultured bronchoalveolar lavage (BAL) eosinophils from Bid-deficient mice, suggesting that Bid has a lesser role in spontaneous and glucocorticoid-induced apoptosis and is a more critical mediator in FasL-induced apoptosis. 85 It seems clear that extrinsic (FasL-induced) apoptosis requires an additional mitochondrial loop in eosinophils.
As expected in cells prone to undergo apoptosis, the expression of anti-apoptotic Bcl-2 members Bcl-2, Bcl-xL, and Mcl-1L is generally low in eosinophils.87–90 However, the level of Bcl-2 expression seems to depend on the status of the patient and origin of eosinophils, because higher expression of Bcl-2 was found in the lung eosinophils of patients with asthma and children with severe exacerbations when compared to eosinophils of healthy individuals or children with mild- to-moderate exacerbations, respectively.91–93 Anti-apoptotic Mcl-1L is degraded during spontaneous apoptosis and in an accelerated manner during glucocorticoid-induced apoptosis. 90
In addition to the pore-forming activity of cleaved Bax or Bid, MMP can be mediated via mitochondrial permeability transition (mPT) pore.11,94 It is a channel formed to the merging point of inner and outer mitochondrial membranes in response to Ca2+, oxidants, or pro-apoptotic Bcl-2 family members leading to free passage of solutes and molecules up to 1.5 kDa.94,95 mPT does not seem to be important for spontaneous apoptosis or Fas-induced apoptosis but is a critical mediator of eosinophil apoptosis induced by glucocorticoids.73,96,97 As discussed above, pores formed in the outer mitochondrial membrane by the cleaved Bax and/or Bid are probably responsible for the MMP in the pathways of spontaneous and Fas-induced eosinophil apoptosis.84,90
Caspases and calpains
Caspases are
Eosinophils have been shown to express caspases-3, -6, -7, -8, and -9.52,75,87 Many apoptotic events during spontaneous or induced eosinophil apoptosis are reduced or prevented by pan-caspase inhibitors suggesting that eosinophil apoptosis is mediated by the activation of the caspase cascade.52,75,96,100 Caspase-9, accounted as the initiator caspase activated in response to mitochondrial apoptotic pathway, has been shown to be processed during spontaneous and induced apoptosis.52,84,101–103 However, its inhibition by Z-LEHD-FMK did not prevent spontaneous apoptosis or FasL-mediated apoptosis, suggesting that it may not function as a critical initiator caspase in these pathways.84,101 However, a possibility exists that the inhibitor used was inefficient, because according to our data it inhibited only 65% of caspase-9 activity in eosinophils. It can be suspected that the residual 35% of caspase-9 activity was enough to activate effector caspases. 101 Also caspase-8 activity has been detected in spontaneously dying eosinophils in some but not all studies. But similar to caspase-9, its inhibition did not prevent apoptotic events during spontaneous apoptosis.52,84,101–103 Altogether, the initiator caspase responsible for the proceeding of spontaneous apoptosis is not clear. In neutrophils, activation of caspase-8 was shown to be dependent on initiator caspase-9 104 and may be actually activated by effector caspase-3, as previously described. 105 In eosinophils, activation of both initiator caspases (8 and 9) has been detected during Fas- and glucocorticoid-induced apoptosis.84,106,107 The evidence indicates that caspase-8 functions as the critical initiator caspase in FasL-mediated eosinophil apoptosis as its inhibition was reported to prevent Bid-cleavage and reduce apoptosis. 84
Activation of effector caspases-3 and -6 seems to be a general feature of eosinophil apoptosis. Involvement of these caspases has been found in spontaneous eosinophil apoptosis and apoptosis induced by various stimuli.52,75,87,96,103 Lamin degradation and DNA fragmentation are caspase-6-dependent events in eosinophils, and inhibition of caspase-6 delayed or halted apoptosis at the level of chromatin condensation but did not prevent apoptosis. 75 This is consistent with the results in other cell types.108–111 Also PS externalization was shown to be partly dependent on caspase-6. 75 Inhibition of caspase-3 partially prevented DNA fragmentation in eosinophils.75,96
Calpains are
Reactive oxygen species (ROS)
ROS induce apoptosis of human eosinophils and are often involved in the mitochondrial pathway of apoptosis.114,115 Thiol-antioxidant glutathione is considered to form the most important antioxidant defence in mitochondria. 116 Spontaneous eosinophil apoptosis was reduced by antioxidants that elevate intracellular levels of glutathione and by hypoxia86,115,117,118 suggesting a role of ROS in mediating spontaneous apoptosis and importance of glutathione in the regulation of intracellular oxidant levels in eosinophils. In a similar manner, the antioxidants increasing glutathione reduced Fas-induced apoptosis. 115 Also, glucocorticoid-induced apoptosis was prevented by mimetic of superoxide dismutase (SOD) and hypoxia, indicating the involvement of ROS. 86 ROS seem to mediate also eosinophil apoptosis induced by many other stimulants.73,115,119 However, the exact mechanism of increased oxidant levels remains unclear. Decrease in the levels of an important mitochondrial antioxidant MnSOD was demonstrated during spontaneous apoptosis as well as in glucocorticoid-treated cells. 86 Levels of a cytosolic antioxidant were not similarly decreased. It is possible that loss of mitochondrial antioxidant defence at least partly explains the increased ROS during spontaneous and glucocorticoid-induced eosinophil apoptosis.
Mitogen-activated protein kinases (MAPKS) and mammalian sterile 20-like kinase (MST)
MAPKs are serine/threonine kinases mainly activated by proinflammatory cytokines, growth factors, and environmental stress. MAPK family consists of c-Jun N-terminal kinases (JNK) 1-3, ERK 1/2, 3, 5 and 7, and p38 family members and a serial of phosphorylation cascades leads to activation of MAPK. MAPKs phosphorylate transcription factors resulting in transcription of genes involved in apoptosis, survival, proliferation, and differentiation. Additionally, MAPKs affect function of numerous other proteins via phosphorylation.120,121 JNK has been previously shown to mediate apoptosis through several pathways: AP-1-mediated transcription of FasL and TRAIL- receptor 1,122,123 phosphorylation of Bcl-2 family protein members,120,124,125 mPT induction,126,127 and phosphorylation of histone H2AX required for DNA fragmentation. 128
Some evidence has been gathered regarding the role of JNK as a mediator of spontaneous eosinophil apoptosis, even though results are contradictory. Spontaneous eosinophil apoptosis was decreased by a peptide inhibitor of JNK but not by the other JNK inhibitors tested. Furthermore, modest or no activation of JNK and lack of activation of c-Jun has been demonstrated in spontaneously dying eosinophils.73,129 Instead, JNK was involved in glucocorticoid-induced eosinophil apoptosis and its activation was dependent on oxidants. 86 Indeed, increased level of ROS is one possible general activation mechanism for JNK in eosinophils proceeding toward apoptosis. Additionally, activation of JNK pathway has been previously demonstrated to occur in response to FasL in lymphocytes,130,131 and some evidence points to the role of JNK in FasL-induced eosinophil apoptosis. 132 The other MAP kinases ERK 1/2 and p38 seem to mediate eosinophil survival, not apoptosis.49,50 Interestingly, p38 MAP kinase seems to be active in isolated eosinophils, and its inhibition by a pharmacological inhibitor induces apoptosis. 50
Mst1 belongs to a group of germinal center kinases (GSKs) that is involved in many functions of immune cells such as trafficking, proliferation, and apoptosis. 133 Mst1 has been shown to be involved in the activation of MAPKs such as JNK. 134 Caspase-mediated cleavage and release of 36 kDa fragment of Mst1 was demonstrated to correlate with eosinophil apoptosis but not with neutrophil apoptosis. Cleavage of Mst1 was increased by FasL and decreased by IL-5, suggesting an important role of this kinase in mediating eosinophil apoptosis. 135
Summary and Conclusions
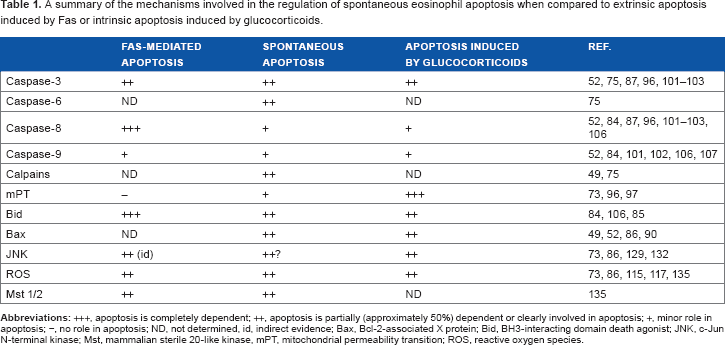
Eosinophil apoptosis induced by FasL or glucocorticoids is a physiologically or clinically relevant mechanism of eosinophil clearance. Plenty of evidence exist about the clinical relevance of steroid-induced eosinophil apoptosis in the airways of steroid-treated asthmatics.28–31 Most likely, spontaneous eosinophil apoptosis occurs in a physiological situation, even though direct evidence is difficult to obtain. The signaling pathway of spontaneous apoptosis seems to overlap with the pathways of FasL or glucocorticoid-stimulated apoptosis. A summary of different pathways is shown in Table 1. Premitochondrial phases of FasL-stimulated apoptosis have unique features such as Fas-associated protein with death domain (FADD) phosphorylation and activation of initiator caspase-8. However, mitochondrion has an important role in all of these pathways of apoptosis as suggested by common dependence on mitochondrial ROS and processing of pro- apoptotic Bcl-2 family members. The mechanism of MMP may differ between these pathways of apoptosis. Bax and Bid seem to be important mediators of MMP in eosinophils undergoing spontaneous apoptosis, while FasL-stimulated apoptosis was dependent on Bid. mPT is emphasized during glucocorticoid-induced apoptosis. Effector caspases seem to be similarly activated in all three apoptotic routes. Activation of JNK may also be common for these pathways of apoptosis, even though additional evidence is required to address it. In a physiological situation, some level of mitochondrial disruption may already be ongoing at the time the cell encounters a pro-apoptotic stimulant, and the stimulant probably adds pro-apoptotic signals that merge at the level of mitochondria to augment, amplify, and finalize the ongoing process of (spontaneous) apoptosis. Thereby, enhancement of the ongoing spontaneous apoptosis is a relevant therapeutic strategy to treat diseases with eosinophilic inflammation.
A summary of the mechanisms involved in the regulation of spontaneous eosinophil apoptosis when compared to extrinsic apoptosis induced by Fas or intrinsic apoptosis induced by glucocorticoids.
Footnotes
Author Contributions
PI wrote the first draft of the manuscript. HK contributed to the writing of the manuscript. HK and EM made critical revisions. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
