Abstract
The aim of this article is to report the organ-specific correlation with tryptophan (Trp) metabolism obtained by analyses of tryptophan 2,3-dioxygenase knockout (TDO-KO) and quinolinic acid phosphoribosyltransferase knockout (QPRT-KO) mice models. We found that TDO-KO mice could biosynthesize the necessary amount of nicotinamide (Nam) from Trp, resulting in the production of key intermediate, 3-hydroxyanthranilic acid. Upstream metabolites, such as kynurenic acid and xanthurenic acid, in the urine were originated from nonhepatic tissues, and not from the liver. In QPRT-KO mice, the Trp to quinolinic acid conversion ratio was 6%; this value was higher than expected. Furthermore, we found that QPRT activity in hetero mice was half of that in wild-type (WT) mice. Urine quinolinic acid levels remain unchanged in both hetero and WT mice, and the conversion ratio of Trp to Nam was also unaffected. Collectively, these findings show that QPRT was not the rate-limiting enzyme in the conversion. In conclusion, the limiting factors in the conversion of Trp to Nam are the substrate amounts of 3-hydroxyanthranilic acid and activity of 3-hydroxyanthranilic acid 3,4-dioxygenase in the liver.
Introduction
The tryptophan (Trp) degradation pathways can be divided into four pathways as follows: serotonin, kynurenine, glutarate, and niacin pathways (Fig. 1). Many biologically important compounds such as serotonin, melatonin, kynurenine, kynurenic acid (KA), quinolinic acid (QA), and niacin are synthesized from Trp. Of these pathways, we are interested in the conversion pathway of Trp to niacin. This is because a deficiency of Trp and a metabolic disorder of Trp is pellagra, which is cured by administration of niacin.

Trp degradation pathway in the liver.
We have previously reported that the conversion percentage of niacin from Trp is around 2% in molar ratio, the rate-limiting enzyme is quinolinic acid phosphoribosyltransferase (QPRT), and this pathway mainly exists in the liver, while nonhepatic tissues are not associated with this conversion pathway.1–20 However, our recent findings21,22 have revealed that the latter is incorrect.
An enzyme that governs the biosynthesis of niacin from Trp
The urine total amount of nicotinamide (Nam) and its catabolites, such as N1-methylnicotinamide (MNA), N1-methyl-2-pyridone-5-carboxamide (2-Py), and N1-methyl-4-pyridone-3-carboxamide (4-Py), reflects the formation of niacin-active compounds. We investigated the relationships between the urine total amount of Nam and its catabolites and other urine Trp catabolites such as anthranilic acid, KA, xanthurenic acid (XA), 3-hydroxyanthranilic acid (3-HA), and QA.1–20,23 We found a significant correlation between the urine total amount of Nam and its catabolites and urine QA.
Next, we examined the enzyme that controls the formation of niacin from Trp in the liver.2–5,10–12,15,16,18,19,23 The relationship between tryptophan 2,3-dioxygenase (TDO) and urine Nam and its catabolites was not significant. The relationship between 3-hydroxyanthranilic acid 3,4-dioxygenase (3-HADO) and urine total amount of Nam and its catabolites was significant (P = 0.040). The relationship between kynureninase and urine Nam and its catabolites was not significant. The relationship between QPRT and urine Nam and its catabolites was not significant. From these results, we concluded that the enzyme that governs the biosynthesis of Nam from Trp is 3-HADO.
Organ correlation with Trp metabolism in wild-type mice
More than 90% of the Trp intake is said to be metabolized by the Trp-kynurenine-glutarate pathway (Fig. 1) in the liver.21,24,25 2-Amino-3-carboxymuconate-6-semialde-hyde (ACMS) is a very unstable compound; therefore, it non-enzymatically cyclizes to form QA. QA is a key intermediate of the conversion. QA then enters the nicotinamide adenine dinucleotide (NAD) pathway.
The NAD metabolism in the liver is shown in Figure 2. QA is converted to nicotinic acid mononucleotide (NaMN), nicotinic acid adenine dinucleotide (NaAD), and then to NAD. NAD is then hydrolyzed to form Nam. Nam is distributed to nonhepatic tissues. Also, in the liver, Nam is converted to nicotinamide mononucleotide (NMN) and NAD. When excessive Nam is present in the body, it is catabolized to form MNA, 2-Py, and 4-Py, which are eliminated into the urine.

NAD metabolism in the liver.
The Trp degradation pathway in nonhepatic tissues is shown in Figure 3. The first enzyme is not TDO, but indoleamine 2,3-dioxygenase (IDO). The activity of kynurenine 3-monooxygenase and kynureninase in nonhepatic tissues is weak compared with that of the liver enzymes. Therefore, some parts of kynurenine and 3-hydroxykynurenine (3-HK) are converted to side intermediates such as KA and XA. Furthermore, 3-HA is an end product, because 3-HADO does not exist in nonhepatic tissues. 21 Namely, Trp is not a precursor of NAD in nonhepatic tissues.

Trp degradation pathway in nonhepatic tissues.
Te NAD biosynthetic pathway in nonhepatic tissues is shown in Figure 4. In nonhepatic tissues, Nam is only a precursor of NAD. Nam generated from Trp in the liver is transported to nonhepatic tissues via the blood. Nam incorporated into nonhepatic tissues is converted to NMN and NAD. Nonhepatic tissues cannot catabolize Nam. Thus, surplus Nam in nonhepatic tissues returns back to the liver where it is catabolized.

NAD biosynthetic pathway in nonhepatic tissues.
The organ correlation with Trp metabolism in wild-type (WT) mice is summarized in Figure 5. The first enzyme in liver Trp degradation is TDO. A small amount of Trp is also degraded in nonhepatic tissues. In this case, the first enzyme is IDO. We investigated the distribution of the enzymes involved in the Trp–niacin metabolism.21,26,27 We found that 3-HADO was only in the liver, and not in nonhepatic tissues. 21 Therefore, 3-HA is an end product of the Trp degradation pathway in nonhepatic tissues. Namely, nonhepatic tissues cannot biosynthesize niacin from Trp. Also, a certain amount of 3-HA is taken up by the liver, and 3-HA is used as a niacin precursor. In nonhepatic tissues, NAD is synthesized only from Nam. The upper metabolites in the Trp– niacin pathway such as 3-HA, KA, and XA are detected in the urine. Their origins are thought to be nonhepatic tissues. The lower metabolites in the Trp–niacin pathway such as QA, MNA, 2-Py, and 4-Py are detected in the urine. Their origin is thought to be the liver.

Organ correlation with Trp metabolism in WT mice.
TDO-KO Mice
The necessary amount of niacin can be synthesized only by IDO
We found that mice can biosynthesize the necessary amount of niacin from Trp by catalysis of IDO (exists in nonhepatic tissues) even in the absence of TDO (mainly exists in the liver). 21 A retardation of body weight gain was not observed in tryptophan 2,3-dioxygenase knockout (TDO-KO) mice. Namely, we found that the necessary amount of Nam was synthesized from Trp by IDO, which exists in nonhepatic tissues. In nonhepatic tissues, 3-HADO does not exist. 21 Consequently, 3-HA is sent out to the blood stream and some of it is then taken and metabolized into Nam in the liver, which is then distributed to nonhepatic tissues by the blood stream.
Side-intermediates of the Trp–niacin pathway originate from nonhepatic tissues
We obtained interesting findings in the experiment with TDO-KO mice (Table 1). The urinary excretion amounts of the upper intermediates of the Trp–niacin pathway such as kynurenine, KA, XA, and 3-HA were higher in the TDO-KO mice than in the WT mice. On the other hand, the amounts of the lower intermediates such as QA, Nam, MNA, 2-Py, and 4-Py were lower in the TDO-KO mice than in the WT mice. These findings indicate that the urinary excretion amounts of the upper metabolites reflect the conversion ability of Trp to 3-HA in nonhepatic tissues, but do not reflect that ability in the liver. The conversion ability of Nam from Trp was very low in TDO-KO mice, because TDO was absent.
Urinary excretion of Trp-Nam metabolites in WT and TDO-KO mice fed a niacin-free diet. 22
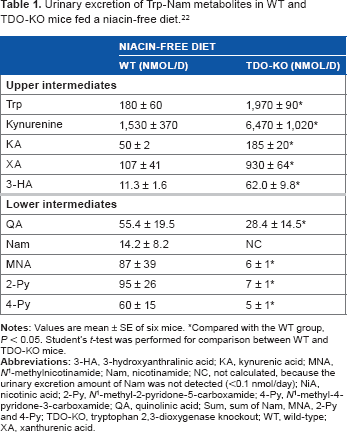
Compared with the WT group, P < 0.05. Student's t-test was performed for comparison between WT and TDO-KO mice.
Table 2 shows the relative niacin conversion activity from exogenous Trp, kynurenine, 3-HK, and 3-HA.28–30 Each compound was orally fed to niacin-deficient animals. Trp and 3-HA showed niacin conversion activity, but kynurenine and 3-HK did not. These findings indicate that liver cells can incorporate Trp and 3-HA, but cannot incorporate kynurenine and 3-HK. From these findings, we summarized the Trp to Nam metabolism in TDO-KO mice (Fig. 6). 3-HADO does not exist in nonhepatic tissues. 21 Hence, 3-HA in nonhepatic tissues, which is synthesized from Trp by IDO, is sent out to the blood stream, and some of the 3-HA is then taken up by the liver, where the 3-HA is metabolized into Nam. The synthesized Nam is distributed to nonhepatic tissues via the blood stream, and then, the Nam is converted to NAD.
Relative niacin conversion activity to Nam (molar ratio).

Ref. 30 ,

Organ correlation with Trp metabolism in TDO-KO mice.
QPRT-KO Mice
True niacin-deficient animals cannot be created using nutritional techniques, because the vitamin niacin can be synthesized from Trp. 22 We wanted to establish a truly niacin-deficient model animal using a protocol that does not involve manipulating the dietary Trp. We generated mice that do not have the qprt gene. 21 The body weight of QPRT-KO mice decreased when they were fed a diet without preformed niacin; however, it did not decrease when they were fed a diet with preformed niacin.
QPRT activity was not affected by the kind of diet, ie, with or without preformed niacin. Its activity was 1.0 μmol/hour/g of liver in WT mice, 0.5 μmol/hour/g of liver in hetero type mice, and 0 μmol/hour/g of liver in QPRT-KO mice.
In QPRT-KO mice, QA is not metabolized. Therefore, QA is completely eliminated into the urine in QPRT-KO mice. The amount of urine QA excreted was not affected by the kind of diet. The excretion amount percentage of QA was around 6% of the intake of dietary Trp. These values were higher than expected.
The urine QA was almost the same between the groups of WT and hetero mice. This was unexpected, because we thought that QPRT is the rate-limiting enzyme of the conversion pathway of Trp to Nam. Thus, these results indicate that QPRT is not the rate-limiting enzyme. The Trp to Nam conversion percentages in WT and hetero mice were 1.4% and 1.2%, respectively. This finding also indicates that QPRT is not the limiting enzyme in the Trp to Nam conversion.
The degradation pathway of Trp in QPRT-KO mice, which does not have QPRT, is shown in Figure 7. Therefore, QA does not convert into niacin-active compounds. All of the QA formed is eliminated into the urine. Therefore, the vitamin niacin is an essential nutrient for QPRT-KO mice.

Niacin (= dietary Nam) is an essential nutrient for QPRT-KO mice.
In conclusion, these findings21,22 show that the limiting factors in the conversion of Trp to niacin are based on the amounts of 3-HA and the conversion ability of 3-HADO in the liver.
Future Directions
The formation of QA is catalyzed by 3-HADO, whose activity is much higher than that of other enzymes involved in the Trp degradation pathway. Thus, generally speaking, 3-HADO is not the limiting enzyme. Nevertheless, our findings suggest that the limiting step may be the conversion reaction of 3-HA to QA. The supply of 3-HA from nonhepatic tissues to the liver may have an important role. We thought that the supply of 3-HA was significantly smaller in nonhepatic tissues than in the liver, but it turned out to be much higher than expected. Thus, IDO in nonhepatic tissues plays an important role in the supply of 3-HA. A 3-HA uptake transporter in liver membranes may have a critical role. The formation of QA from 3-HA is catalyzed by 3-HADO.
Further studies on organ–organ relationships are required to elucidate the entire Trp metabolism. In addition, we will investigate 3-HADO.
Author Contributions
Designed the study and drafted the manuscript: KS. Reviewed the manuscript and helped in the study design: TF. Both authors approved the final manuscript.
Footnotes
Acknowledgment
This is a manuscript of the Proceeding for ISTRY 2015, which was held in September 15–18, 2015, in Grand Rapid, MI, USA.
