Abstract
The aryl hydrocarbon receptor (AHR) is a cytosolic receptor for low molecular weight molecules, of which the most widely recognized ligand is 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), and the most widely recognized effect, chloracne. Adverse effects of manipulation were most recently and graphically demonstrated by the poisoning of Viktor Yushchenko during the Ukrainian presidential elections of 2004. However, recent research has revealed a receptor with wide-ranging, and at times, paradoxical actions. It was arguably among the first biological receptors to be utilized by dermatologists, dating from the time of topical tar preparations as a therapeutic agent. I provide a review outlining the role AHR plays in the development, cellular oxidation/antioxidation, responses to ultraviolet light, melanogenesis, epidermal barrier function, and immune regulation and its relationship to tryptophan metabolism. Finally, I will review the role of AHR in diseases of the integument.
Keywords
Background
The AHR is a cytosolic receptor for low molecular weight molecules, binding and becoming activated by sterically planar ligands approximately three benzene rings in size. 1 It is maximally expressed in interface tissues including the liver, lungs, skin, 2 and the gastrointestinal tract. 3
In the cytosol, AHR exists in a latent state as part of a multiprotein complex (Fig. 1). Chaperoning proteins include the heat shock protein 90 (hsp90), hsp23, and hepatitis B virus X-associated protein 2 (XAP2). 4 Pp60 src is an associated signaling partner that is released into the cytosol on ligand binding. 4 This binds to the epidermal growth factor receptor (EGFR) and initiates mitogen-activated protein kinase (MAPK) signaling.5,6

In the cytosol, AHR exists in a latent state as part of a multiprotein complex. Chaperoning proteins includes heat shock protein 90 (hsp90), hsp23, and hepatitis B virus X-associated protein 2 (XAP2). On ligand binding, pp60src is released and binds to the epidermal growth factor receptor (EGFR), initiating MAPK signaling. The remainder of the complex translocates to the nucleus where it binds to the aryl hydrocarbon receptor nuclear transporter (ARNT), promoting the transcription of genes with xenobiotic response elements (XRE) in their promoters. Crosstalk occurs with the estrogen receptor (ER), retinoblastoma protein (Rb), retinoic acid (Reti), and NF-κB pathways. Control is provided by two loops, exportation of the AHR to the cytoplasm with subsequent degradation and transcription of the aryl hydrocarbon receptor repressor (AHRR).
On ligand binding, the receptor complex translocates to the nucleus. 7 Here it binds to the aryl hydrocarbon receptor nuclear transporter (ARNT). The AHR-ARNT heterodimer interacts with several histone acetyltransferases and chromatin remodeling factors. 8 This promotes the transcription of genes containing xenobiotic response elements (XRE) in their promoters.
Two feedback loops regulate AHR activity. First, AHR is released to the cytosol and degraded by the 26 S proteasome pathway. 9 Second, binding to the XREs results in the transcription of the aryl hydrocarbon receptor repressor (AHRR), which regulates the activity of the AHR via a negative feedback loop. 10
There is also crosstalk between the AHR and other pathways. These include the estrogen receptor, 11 the retinoblastoma protein (Rb), thus inhibiting cell cycle progression 12 and the retinoic signaling pathways. 13 AHR can also bind the p65 subunit of nuclear factor kappa light chain enhancer of activated β cells (NF-κB), thereby either suppressing or activating (depending on cellular context) the expression of NF-κB-dependent genes.14,15
Tryptophan and the Aryl Hydrocarbon receptor
Many tryptophan-based molecules are AHR ligands. These include 6-formylindolo [3,2-b] carbazole (FICZ) 16 and 2-(1′Hindole-3′-carbonyl)-thiazole-4-carboxylic acid methyl ester (ITE), a tryptophan-cysteine dimer. 17
The kynurenine pathway (Fig. 2) is the metabolic pathway via which L-tryptophan is metabolized to nicotinamide adenine dinucleotide (NAD). In addition to generating endogenous supplies of nicotinamide, it has important immune-regulatory roles. 18 Several metabolites known to act as endogenous ligands of the AHR include kynurenine, 19 kynurenic acid, 20 and cinnabarinic acid. 21
Relationship with tryptophan metabolism
Tryptophan is metabolized by four primary pathways.
The kynurenine pathway
The kynurenine pathway (Fig. 2) is the metabolic pathway via which L-tryptophan is metabolized to NAD. Indoleamine 2,3-dioxygenase (IDO) is the rate-limiting enzyme of the kynurenine pathway (KP). It is induced by several proinflammatory molecules, especially interferon gamma (IFN-γ). 22 It is essential that the inflammatory response be controlled and not surprising that one of the downstream KP metabolites, kynurenine, 19 provides a negative feedback loop through AHR to promote the generation of Treg cells. 19 Other downstream AHR ligands include kynurenic, 20 cinnabarinic, 21 and xanthurenic acid. 23
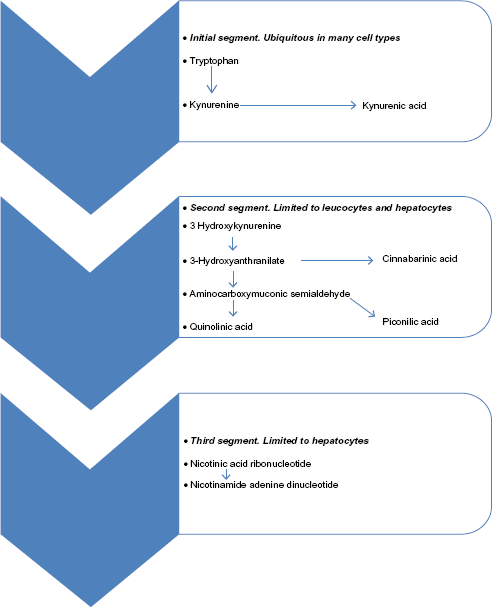
The kynurenine pathway (Reproduced from Noakes, RR, Int. J. Tryptophan Res. 2013;6:67–71).
Reflecting the complexity and multilayered nature of the control mechanisms, it has been reported that kynurenic acid promotes the generation of inflammatory cytokines. 20 In addition, IDO is upregulated by AHR activation, suggesting that positive feedback loops operate within the microenvironment. 24
More recently, it has been demonstrated that the AHR chaperoning protein pp60src can phosphorylate IDO-1, promoting transforming growth factor beta 1 (TGFβ1) production by dendritic cells, leading to disease tolerance. 25 TGFβ1 production in dendritic cells in response to the downstream KP metabolite quinolinic acid has also been reported. 26
Pleiotropic responses are seen with AHR ligands, with some promoting the generation of Treg cells (regulatory ligands), and others, TH17 expansion (effector ligands). 27 The former include TCDD and kynurenine, and the latter, FICZ. 28 Effector ligands such as FICZ are rapidly metabolized and produce only transitory stimulation 29 of AHR, whereas regulatory ligands such as TCDD are long-lived.
Much research on the kynurenine pathway has focused on its neuro-inflammatory roles. The nervous system, however, expresses only low levels of the AHR compared to the integument, 2 and it is in the skin that the relationship is likely to be of greatest significance.
AHR is important in the development of the KP. Langerhans cells (LCs) from AHR-null mice display significantly reduced IDO activity 30 compared to the wild phenotype. In addition, the archetypical AHR ligand, TCDD has been shown to induce IDO in dendritic cells, which promotes Treg generation.20,31
Several pharmacological agents known to modulate the KP are also ligands of AHR. These include tranilast32,33 and leflunomide.34,35
The tryptamine pathway
The tryptamine pathway, catalyzed by tryptophan hydroxylase and dopamine decarboxylase, has been implicated in AHR activation. Tryptamine is a potent AHR activator acting either as a direct ligand 29 or as a precursor for downstream ligands such as indole acetic acid. 29 In addition, shunting through this pathway via carcinoid tumors can result in nicotinamide deficiency (pellagra).
The serotonin pathway
5-Hydroxy-tryptophan, a proximal metabolite, is an AHR agonist. 36 .
Tryptophan photoproducts
Tryptophan is readily photoxidized by UV light. The products include kynurenine, 1 tryptamine, 37 FICZ, and 6,12-di-formylindolo[3,2-b]carbazole (dFICZ), the latter two representing high affinity compounds. 38
The skin
The structure of the skin is shown in Figure 3. The epidermis is composed principally of keratinocytes and continuously regenerated from the basal layer. It serves as a barrier against the environment. As an interface with the external environment, it contains specialized antigen-processing cells known as LCs. The cells responsible for melanin production, melanocytes, are found in the basal layer of the epidermis and transfer pigment granules containing melanin to neighboring keratinocytes via dendritic processes. In keratinocytes, these localize over the cell nucleus and provide protection for the genetic material against environmental ultraviolet radiation.

Stylized image of the skin.
The dermis is divided into a finer papillary and a coarser reticular dermis and provides mechanical strength. Fibroblasts produce collagen, elastin, and glycosaminoglycans that act as an intercellular ground substance imparting viscoelastic properties.
The dermis is also home to a population of cells essential in immunological function including mast cells, dermal dendrocytes, and a resident population of CD4, CD8, CD45, memory, and FOXP3-regulatory T cells.
All cells in the skin express AHR. The highest levels of expression are found in the outer layers of the epidermis (stratum spinosum and granulosum), melanocytes, fibroblasts, and LCs. 39
Function of the Aryl Hydrocarbon Receptor
In addition to the traditional role of metabolizing environmental toxins, AHR plays a role in the development, cellular oxidation/antioxidation, responses to ultraviolet light, melanogenesis, epidermal barrier function, and immune regulation.
Developmental roles
AHR-null mice display growth retardation, reduced liver size, abnormalities in vascular structure, portal tract fibrosis, dermal fibrosis, and decreased fertility,40–43 supportive of a role for the AHR in embryogenesis. Patent ductus venosus is the most consistent abnormality, and the resulting porto-systemic shunt is likely responsible for reduced liver size and portal tract fibrosis.
Oxidant/antioxidant responses
The best known function of AHR is the induction of the cytochrome P450 genes, 44 which are involved in the metabolism of drugs and the detoxification of environmental toxins. The phase I enzymes, cytochrome P450 (CYP) 1A1, CYP1A2, and CYP1B1, contain XRE in their promoters and respond to AHR signaling. 45 These are monoxygenases that introduce functional groups prior to conjugation with water-soluble molecules by phase 2 enzymes. Phase 2 enzymes are under the control of the nuclear factor erythroid derived 2, like 2 transcription factor (Nrf2), which regulates cellular antioxidant responses. Nrf2 promoter sequences are known to contain XRE, 46 allowing coordination of phase 1 and 2 responses.
Ultraviolet light
Tryptophan is a chromophore for ultraviolet (UV) light in the cellular cytoplasm. The photoproducts include FICZ, a potent AHR ligand. 16 On ligation and subsequent dissociation of the AHR complex, the chaperoning protein pp60src binds to the EGFR and initiates MAPK signaling. This increases transcription of cyclooxygenase 2 (COX-2) 47 and matrix metalloproteinase-1. 48 The former plays a role in cutaneous carcinogenesis 49 and the latter in photoaging. 50 Thus, AHR-mediated cytoplasmic events influence carcinogenesis and aging independent of DNA damage.
Melanogenesis
The tanning response is vital to protect the skin from harmful UV light. Keratinocytes produce α-MSH in response to UV light, which promotes melanin synthesis in local melanocytes. Melanin is then transferred back to the keratinocytes in small packages (melanosomes) via the dendritic processes. The melanosomes localize over the cell nucleus, providing protection against UV radiation.
Poisoning with toxins known to be powerful AHR ligands has been reported to result in hyperpigmentation. Mass poisoning with cooking oils contaminated with polychlorinated biphenyl (PCB) was associated with cutaneous hyperpigmentation. 51 Likewise, exposure to TCDD has been reported to result in hyperpigmentation. 52
Melanogenesis is also inducible in cultured melanocytes by FICZ. 53 In addition, c-kit/stem cell factor system is important in melanocyte hemostasis, 54 and c-kit has XREs in its promoter. 55 Thus the AHR plays a role in the tanning response and protection against environmental ultraviolet light.
Epidermal barrier function
An intact epidermal barrier is essential in providing protection against environmental insults and limiting transepidermal water loss. It is most commonly conceptualized as a bricks-and-mortar model. The bricks are compacted keratinocytes containing keratin 1 and 10 filaments. The protein, filaggrin, is responsible for mediating the assembly of the keratin filaments. A cornified cell envelope is synthesized below the plasma membrane comprising cross-linked molecules of envoplakin, periplakin, and involucrin. 56 Cross-linking is mediated by tissue transglutaminase. The mortar is provided by free fatty acids, cholesterol, and ceramides.
Atopic dermatitis is due to defects in skin barrier function secondary to loss-of-function mutations in the filaggrin gene. 57 A mouse model with constitutive expression of AHR was reported as displaying itching, skin inflammation, and immunological imbalances resembling atopic dermatitis. 58 Interestingly, but somewhat counterintuitively, EGFR expression has been reported to be reduced in patients with atopic dermatitis 59 and EGFR signaling attenuates the development and relapse of atopic dermatitis. 60 This appears to be at odds with the anticipated increased availability of the chaperoning pp60 src protein and associated increased EGFR signaling expected with constitutive expression of AHR. These apparent contraindications may represent species specificity, although it has been reported that EGFR signaling inhibits the AHR-mediated differentiation of human keratinocytes, 61 suggesting that feedback loops are present and that constitutive expression of a receptor may produce different features to one under feedback control.
Immune regulation
Keratinocytes
Keratinocytes express an impressive array of cytokines. 62 Cytokines of keratinocyte origin known to contain XRE in their promoters include IL-1β63 and IL-8. 64
Langerhans cells
LCs are epidermal dendritic cells involved in antigen presentation. More recently, their role in immune regulation and telerogenesis has been recognized. 65 AHR is expressed in LCs, and AHR-null LCs display evidence of impaired function. 30
Interleukins
TH17 T cells are a subgroup of T cells and are important in mediating responses to bacteria and yeast. They play a pivotal role in psoriasis 66 and are the target for several biological agents used in the management of psoriasis. They also play a role in autoimmune disease. 67 They produce IL-17 and IL-22. 68 AHR promotes the expansion of TH17 lymphocytes and is obligatory for IL-22 production. 68 Reflecting the complexity of the interplay between control systems, AHR has also been found to promote IL-21 and IL-23 production and may display inhibitory effects on TH17 depending on the ligand. 19 In human skin, LCs induce a special subset of T cells, TH-22, which produce IL-22 (but not IL-17, which is under control of AHR). 69
AHR is also known to crosstalk with NF-κB 15 and thus is involved in the regulation of inflammatory and immune responses, cell survival, and proliferation.
Dermatopathological Correlations
Archetypical correlations
Chloracne
Chloracne (Fig. 4) is the archetypical toxic response of the skin to TCDD. The name is a misnomer, as it is characterized by atrophy of the sebaceous glands, keratinocyte hyperpigmentation, and epidermal hyperplasia. Recently, the skin lesions were recognized as hamartomas 70 and, in the case of TCDD poisoning, represent a TCDD metabolizing compartment. 71 Curiously, mice (with the exception of the hairless mouse) do not develop chloracne-like lesions on exposure to TCDD, 72 and there are likely to be significant species differences in the action of the AHR.

Proposed pathogenesis of chloracne. Ligand binding to the AHR leads to nrf-2-mediated expression of Epgn, Slpi, and Sprr2d. This causes infundibular hyperkeratosis and obstruction of the pilosebaceous unit. With the progress of time, the sebaceous gland undergoes atrophy and a MADISH is formed.
TCDD promotes terminal differentiation of keratinocytes, 73 possibly mediated by changes in the expression of transforming growth factor alpha (TGFα) 74 and epidermal growth factor (EGF). 75
It has been proposed that these altered cytokine profiles and abnormalities in lipid metabolism, which have also been identified, 76 may play a role in pathogenesis.
Recently, it has been reported that transgenic mice with constitutional activation of nrf-2, which contains XRE in its promoter, develop chloracne-like lesions. 77 This is reportedly related to the upregulation of the growth factor epigen (Epgn), secretory leukocyte peptidase inhibitor (Slpi), and small proline-rich protein 2d (Sprr2d). In hair follicles, the latter three were identified as the likely causes of infundibular acanthosis, hyperkeratosis, and cyst formation. 77
Cutaneous Carcinogenesis
AHR is involved in cutaneous carcinogenesis
Exposure to tar has long been recognized by dermatologists to cause pre-malignant lesions, known as tar keratosis. They are considered to be due to polycyclic aromatic hydrocarbons (PAHs), which are components of tar. 78 PAHs are known ligands of AHR.
Human subjects are less susceptible to the carcinogenic effects of tars compared to mice, as evidenced by the long historical use of tar preparations with few reports of carcinogenesis. Nonetheless, scrotal carcinomas were historically reported amongst London chimney sweepers.
Carcinogenicity may be dependent upon on whether the induction of cytochrome p 450 enzymes results in the detoxification or generation of potential carcinogens. 79
Stimulation of AHR leads to MAPK signaling.5,6 This can be mediated by tryptophan photoproducts such as FICZ 16 generated on exposure to UV radiation. MAPK signaling leads to the expression of COX-2, 47 which plays a role in cutaneous carcinogenesis. 49
Disorders Involving TH17 Lymphocytes
Psoriasis is the archetypical TH17-mediated disease. TH17 responses are also important in autoimmune diseases.
Psoriasis
Psoriasis (Fig. 5) is a common cutaneous disorder affecting 3% of the population. It is believed to be due to overactivity in the TH17 limb of the immune response and is characterized by well-defined erythrosquamous plaques with a distinctive silvery scale found predominantly on extensor surfaces. Several variants are recognized, including flexural variants, a sebo-psoriatic variant, which overlaps with seborrheic dermatitis, and less common pustular variants. Flexural and sebopsoriatic variants are likely to be related to TH17 hyper-reactivity to commensal Candida sp. and Malassezia sp.

Proposed involvement of the AHR in the pathogenesis of psoriasis. Enhanced TH 17 responsiveness to bacterial antigens likely explains the guttate variant seen after streptococcal infections. The sebo-psoriatic and flexural variants are probably due to enhanced responsiveness to Malassezia and Candida sp., respectively.
The AHR is known to be involved in development of the TH17 subset of T cells and their cytokines, IL-17 and IL-22. 68 IL-17 is known to be involved in the pathogenesis of psoriasis 66 and autoimmunity. 67 Deficiency of the AHR is known to exacerbate psoriasis. 80
Dermatologists, if unknowingly, have been using AHR modulation in the management of psoriasis since the introduction of coal tar. PAHs, and possibly other components, are known to be active at AHR. 81
Phototherapy is a highly effective treatment for psoriasis. It is notable that one of the known endogenous ligands of AHR, FICZ, is a photoproduct of cutaneous tryptophan.
Scleroderma
Scleroderma (Fig. 6) is a fibrotic disease. It is divided into localized (morphea) and generalized (progressive systemic sclerosis) variants. The latter is distinguished by the presence of Raynaud's phenomenon (transient digital vasospasm usually in response to cold) and pulmonary, esophageal, and renal involvement. The cause is unknown, but it is considered an autoimmune disorder. A congenital form of scleroderma with a poor prognosis, known as infantile stiff skin syndrome, is due to mutations in the fibrillin-1 gene. 82 Fibrillin-1 is a component of the microfibrils of the extracellular matrix and a binding site for cellular integrins, allowing resident cells to bind to the extracellular matrix. Antibodies to fibrillin-1 have been detected in patients with both morphea 83 and scleroderma. 84

Proposed involvement of the AHR in scleroderma. A complex interplay involving anti-endothelial and fibrillin-1 antibodies, enhanced TH 17 responsiveness, and kynurenine metabolites are likely to be involved. AHR is involved in the mediation of several of these processes.
TGFβ is a major fibrotic cytokine and displays altered expression in scleroderma. 85 TGFβ is secreted from the cell in a large latent complex (LLC) that includes the active cytokine, a dimer of its processed N-terminal pro-peptide (latency associated peptide or LAP), and one of three latent TGFβ binding proteins (LTBP-1, -3, or -4). As implied by mouse models and confirmed biochemically, fibrillin-1-rich microfibrils contribute to targeting of the LLC to the extracellular matrix by direct interaction with LTBPs. Failed matrix sequestration of the LLC in fibrillin-1-deficient patients and mice promotes increased availability of the TGFβ family of cytokines.
The sclerodermoid disorders include eosinophilic fasciitis and eosinophilia-myalgia syndrome, both of which86,87 are associated with L-tryptophan ingestion. The majority of L-tryptophan is converted by the KP to NAD, several intermediates of which have been identified as AHR ligands.19–21 In addition, agents used in the management of Parkinson's disease, which are inhibitors of kynureninase, 88 have been reported to have cutaneous sclerosis as a side effect. 89 This would be consistent with prolonged activation of AHR by the KP metabolite, kynurenine, mediating cutaneous sclerosis.
Noakes et al. 26 reported TGFβ1 production in dermal dendritic cells and endothelium in response to quinolinic acid, a KP metabolite, in a human subject. In patients with morphea, KP activation has been identified in infiltrating leucocytes, endothelium, the basal layer of the epidermis, and eccrine units, 90 suggesting that an ample supply of AHR ligands is present in this condition.
The pro-fibrotic cytokines are TH17-driven in systemic sclerosis, 91 supportive of AHR involvement in view of the central role it plays in TH17 expansion. In addition, pp60 src released by AHR on ligand binding has been demonstrated to play a significant role in fibroblast activation. 92 Fibroblasts from AHR-deficient mice display increased TGFβ production 93 and marked dermal fibrosis. 94 Once again, the concept of effector and regulatory ligands is relevant. Long-acting ligands such as TCDD 95 have been reported to produce cutaneous sclerosis. In these circumstances, long-acting ligands appear to produce an AHR-null effect. Phototherapy is an evidenced-based therapeutic modality used in the management of scleroderma. 96 It is tempting to postulate that the effects are mediated via AHR. FICZ is a photoproduct of tryptophan, which acts as an effector ligand at AHR and thus may be expected to reduce dermal fibrosis.
Tranilast is an agent marketed in Japan and Korea for the management of both allergies and keloids. It has reported activity at AHR 33 and a modulatory effect on the KP. 32 As an antifibrotic agent, tranilast is anticipated to act as an effector ligand at AHR.
Scleroderma, both generalized and localized, is currently considered to be an autoimmune disorder. AHR is important in regulating immune responses. TCDD is potently immunosuppressive, 97 promoting the development of Treg cells. The endogenous ligand, kynurenine, has also been reported to have the same action. 19 Thus, modulation of AHR may be a means of suppressing autoimmune disorders and promoting Treg cell differentiation. Nonetheless, the effects of AHR are complicated by the regulatory nature of some ligands and the effector nature of others. FICZ as an effector ligand has been reported to promote TH17 expansion and worsen experimental allergic encephalomyelitis. 98 The AHR agonist ITE has an inhibitory effect on scarring by inhibition of TGFβ1 myofibroblast differentiation, although this appears to be in a manner independent of the AHR receptor. 99
The initial precipitating event is thought to be endothelial damage. 100 This may be precipitated by viruses. 101 Anti-endothelial antibodies have been found in scleroderma. 101 This results in elevated levels of vascular endothelial growth factor (VEGF). 102 VEGF is an inducer of IDO, 103 allowing the generation of downstream metabolites and activation of the AHR. In addition, TGFβ sustains IDO expression, 104 thereby establishing an autocrine loop, which may account for the delayed response traditionally seen with immunosuppressive therapy.
Disorders of Cutaneous Barrier Function
Atopic dermatitis
Atopic dermatitis is a common skin condition affecting up to 20% of children and 3% of adults. It is characterized by a poorly defined pruritic eruption, which histologically displays spongiosis. As previously noted, loss of function in the filaggrin gene is thought to be responsible.
EGFR signaling is known to attenuate the development and relapse of atopic dermatitis. 60 EGF/EGFR signals through the MAPK pathway, which is activated by pp60src released on AHR ligand binding. 4 The use of EGFR inhibitors in cancer patients is associated with cutaneous eruptions, which share 105 many features with atopic dermatitis.
Coal tar is a traditional treatment for atopic dermatitis. Van den Bogaard et al. 81 demonstrated AHR-mediated skin barrier repair in atopic dermatitis patients with tar preparations. Counterintuitively, however, TCDD has been reported to exacerbate atopic dermatitis, 106 representative of the pleotropic regulatory and effector responses seen with AHR.
Phototherapy is the therapeutic modality used in atopic dermatitis. 107 It would be tempting to postulate that its effects are mediated by FICZ.
Disorders of Pigmentation
Vitiligo
Vitiligo (Fig. 7) is a de-pigmenting disease characterized by an autoimmune attack on, and subsequent loss of, melanocytes. Vitiligo is a partly understood condition. Current models would suggest that a primary abnormality is present in melanocytes. 108 Oxidative stress leads to melanocyte death, 109 and the subsequent autoimmune response, which develops against liberated melanocyte antigens, results in the perpetuation of the condition.

Proposed involvement of AHR in the pathogenesis of vitiligo. TH 17-mediated autoimmunity and a reduced capacity of melanocytes to manage oxidative stress are AHR mediated.
AHR plays a central role in cellular antioxidant responses via Nrf2 signaling. In addition, TH17 responses, in which AHR is known to play a role, are involved in the autoimmune responses in vitiligo. 110 Importantly, polymorphisms in AHR have been reported as a susceptibility factor for vitiligo. 111
In addition, AHR is involved in the tanning response. It has also been demonstrated that hyperpigmentation induced by cigarette smoking is mediated via AHR. 112 The c-kit/stem cell factor system is involved in melanocyte homeostasis, 113 and both contain XREs that respond to the AHR.
Malassezia-Associated Diseases
Malassezia species, especially M. furfur and M. globosa, generate a number of potent AHR ligands including indirubin, FICZ, indolo[3,2-b]carbazole (ICZ), malassezin, and pityriacitrin. 114 These organisms are normal commensals of the human integument and are known to play a role in a number of dermatological conditions. Tryptophan is excreted in human sweat 115 and is the source of these metabolites. 116
Pityriasis versicolor
Pityriasis versicolor is a pityriasiform eruption predominantly involving sebaceous areas of the skin. Its presentation varies from hypopigmented macules in tanned skin to erythematous or brown macules depending on the species of Malassezia sp. involved.
Pityriasis versicolor is due to the overgrowth of Malassezia species, most commonly M. furfur and M. globosa. Impairment of the tanning response is characteristic of colonization, and depigmentation persists long after mycological clearance. Many species produce malassezin, an AHR agonist that has been reported to cause apoptosis of melanocytes. 117
Seborrhoeic dermatitis
Seborrhoeic dermatitis is a pruritic eruption involving the seborrheic regions of the skin classically displaying a greasy scale. It is currently considered to be mediated by Malasezzia sp. based on the known response to agents directed against Malassezia. The AHR ligands Malassezin and ICZ are selectively produced by Malassezia yeast isolated from the scalps of patients with seborrhoeic dermatitis 118 compared to healthy individuals.
Cutaneous Disease known to Have an Association with Smoking
Tobacco smoke is a source of PAH, 119 a known AHR ligand. Smoking has a reported association with palmoplantar pustulosis, a localized version pustular psoriasis. 120 AHR is known to be involved in the development of the TH17 subset of T cells and their cytokines IL-17 and IL 22. 68 Increased levels of IL-17 have been reported in both the serum and lesions of patients with palmoplantar pustulosis. 121
Hidradenitis suppurativa, a chronic noninfectious suppurative eruption arising in apocrine-bearing skin, has also been reported to be precipitated by smoking. 122 Interestingly, reduced numbers of sebaceous glands have been reported in patients with hidradenitis suppurativa. 123 This may be relevant considering that chloracne is characterized by sebaceous gland atrophy.
Conclusion
AHR is potentially involved in a range of cutaneous disorders, and successful manipulation of this receptor is likely to offer significant therapeutic benefits. Yet, the recognition that both effector and regulatory ligands exist means the results of manipulation can be both paradoxical and unpredictable. This paradox, however, is well known in clinical practice. Psoriasis, a TH17-mediated disorder, may occasionally destabilize into the more dangerous pustular variants in response to treatment with tar preparations or phototherapy, which are active at AHR. A better understanding of the role of AHR would likely reduce these adverse reactions.
It would be tempting to postulate that the effects of phototherapy are mediated via AHR, thus raising the possibility that appropriate agonists may achieve the same therapeutic response without patient inconvenience, cost of equipment, and use of potentially carcinogenic ultraviolet light. This would represent a significant advance in the management.
The sclerodermoid disorders remain poorly understood, and treatments are suboptimal. Involvement of the kynurenine metabolites and their interactions with AHR has long been suspected based on the documented activity of this pathway in several of these disorders. A better understanding of these interactions would allow the development of improved management strategies.
Malassezia sp. have long remained a quandary. The normal skin commensal produces no disease in the majority of the population, yet is responsible for a range of common problematic disorders in a significant minority. Why it produces different disease patterns in different individuals is also unknown. An understanding of the interactions it has with the AHR may help to clarify this and lead to improvements in treatment.
There remain many unwritten chapters in the saga of AHR, which is part of a complex and multilayered regulatory system within the skin. Its manipulation in the management of cutaneous disorders dates from the earliest days of dermatology; yet its potential remains to be fully realized. Over the last several decades, it has held a prominent place in the field of toxicology, yet this may be a receptor whose time has come.
Footnotes
Author Contributions
Conceived the concepts: RN. Wrote the first draft of the manuscript: RN. Made critical revisions: RN. The author reviewed and approved of the final manuscript.
