Abstract
Aryl hydrocarbon receptor (AhR), a ligand-activated transcription factor, is crucial in maintaining the skeletal system. Our study focuses on encapsulating the role of AhR in bone biology and identifying novel signaling pathways in musculoskeletal pathologies using the GEO dataset. The GEO2R analysis identified 8 genes (CYP1C1, SULT6B1, CYB5A, EDN1, CXCR4B, CTGFA, TIPARP, and CXXC5A) involved in the AhR pathway, which play a pivotal role in bone remodeling. The AhR knockout in hematopoietic stem cells showed alteration in several novel bone-related transcriptomes (eg, Defb14, ZNF 51, and Chrm5). Gene Ontology Enrichment Analysis demonstrated 54 different biological processes associated with bone homeostasis. Mainly, these processes include bone morphogenesis, bone development, bone trabeculae formation, bone resorption, bone maturation, bone mineralization, and bone marrow development. Employing Functional Annotation and Clustering through DAVID, we further uncovered the involvement of the xenobiotic metabolic process, p450 pathway, oxidation-reduction, and nitric oxide biosynthesis process in the AhR signaling pathway. The conflicting evidence of current research of AhR signaling on bone (positive and negative effects) homeostasis may be due to variations in ligand binding affinity, binding sites, half-life, chemical structure, and other unknown factors. In summary, our study provides a comprehensive understanding of the underlying mechanisms of the AhR pathway in bone biology.
Introduction
Bone undergoes continuous remodeling through the sequential activity of osteoblast (bone-forming) and osteoclast (bone-resorbing) cells. The dynamic equilibrium between osteoblasts and osteoclasts is crucial to maintain the integrity of the musculoskeletal system.1,2 It is a highly complex process governed by several endogenous and exogenous factors, such as nutrients and environmental factors. 3 Nutrient metabolites and environmental factors (pollutants) can activate transcription factors (eg, aryl hydrocarbon receptor) to regulate gene expression and alter physiological processes. The aryl hydrocarbon receptor (AhR) is one of the well-known receptors that mediate various biological actions through different endogenous, natural, and synthetic molecules and environmental pollutants. 4 Recently, several studies have focused on the above-mentioned factors, AhR signaling, and age-related diseases. The build-up of damage to intracellular macromolecules is the hallmark of aging, which is influenced by genetic, environmental, and dietary variables. 5 These elements impact mitochondrial function and induce premature senescence, accelerating aging. It’s interesting to note that many environmental and dietary variables, such as flavonoids and carotenoids, alter AhR signaling and mitochondrial function.
Aryl hydrocarbon Receptor, a highly conserved transcription factor (AhR), plays an important role in aging.6 -10 Aryl hydrocarbon receptor is a ligand-activated transcription factor, and it belongs to the Per-Arnt-Sim superfamily of proteins. 11 AhR is composed of 848 residues and consists of three functional domains, namely basic helix-loop-helix (bHLH domain), Per-ARNT-Sim (PAS) domains (A and B), and the transactivation domain (TAD). 12 The amino-acid sequence is highly conserved among these domains. 13 The bHLH domain is located at N-terminal, and it can divide into the basic domain and HLH domain that determines the dimerization of protein molecules and the combination of AhR with DNA. 14 PAS A and PAS B domains are mainly involved in binding to ligands along with the release of heat shock protein 90 (HSP-90), and these domains enhance the stability of heterodimer AhR-ARNT (aromatic hydrocarbon receptor nuclear transfer protein), which causes conformational alterations in DNA. 15 AhR combines with Hsp90, p23, ARA9, and several other proteins in an inactive form. The binding of AhR to its ligand causes a conformational change, facilitating AhR translocation into the nucleus, where it undergoes heterodimerization with ARNT. Once inside the nucleus, AhR-ARNT dimer activates xenobiotic response elements (XRE), which promotes transcription and mediates various biological processes such as immune response, toxicity, and bone remodeling. 2 The aryl hydrocarbon receptor is unique in itself because of its omnipresent nature as it is expressed in the retina, liver, spleen, pancreas, kidney, lungs, esophagus, testis, epithelial cells, placenta, heart, brain, and skeletal muscles.16,17
Several inflammatory genes have varying frequencies of xenobiotic response elements (XREs) in their promoter regions, suggesting that AhR modulates inflammatory responses.18 -21 Through interactions with other signaling pathways like the NF-κB system, AhR controls inflammatory signals. 22 It has long been understood that this route regulates the expression of the inflammatory genes IL-1β, IL-6, IL-8, TNF-α, and others. Even though there is substantial evidence about AhR and its activation in various physiological processes, its role in bone remodeling is not well known. Considering this aspect, our study explicitly summarizes AhR role in osteoblast and osteoclast biology and identifies the novel signaling pathways through GEO dataset analysis in musculoskeletal pathobiology.
Bone Remodeling
Bone remodeling is a perpetual and essential process that persists throughout an individual’s life. This cyclic activity primarily encompasses the removal of mineralized bone facilitated by osteoclasts, succeeded by the generation of bone matrix through the action of osteoblasts. 23 During the resorption phase, osteoclasts break down old bones, and in the reversal phase, bone formation occurs as osteoblasts deposit new bone matrix until the resorbed bone is entirely replaced. 24 This dynamic process consistently adapts the microarchitecture of the bone to maintain its integrity.
Bone remodeling is influenced by major systemic regulators, including parathyroid hormone (PTH), growth hormones, sex hormones, glucocorticoids, and notably calcitriol. 25 Various growth factors such as insulin-like growth factors (IGFs), tumor growth factor-beta (TGF-beta), and cytokines like interleukin-1 (IL-1) and interleukin-6 (IL-6), along with prostaglandins and bone morphogenic protein (BMP), intricately modulate the activity of osteoblasts. 26 Additionally, key proteins like type-1 collagen, osteocalcin, osteopontin, osteoprotegerin, and sialoprotein play crucial roles in regulating bone remodeling. 27 The receptor activator of nuclear factor kappa-B ligand (RANKL) assumes a pivotal role in bone remodeling, while osteoprotegerin (OPG), secreted by osteoblasts, serves a protective function for skeletal integrity against excessive bone resorption. OPG achieves this by binding to RANKL, thereby preventing the activation of RANKL signaling with the Receptor activator of nuclear factor-κB (RANK). 28 Maintaining the integrity of the skeletal system necessitates a delicate balance between the processes of bone formation and bone resorption. 29 It is crucial to uphold equilibrium between the rates of bone formation and bone resorption. If the bone resorption rate is higher than bone formation, then it will lead to bone loss resulting in osteoporosis and increase fracture rates.30,31 The underlying mechanism driving heightened bone resorption involves an upregulation in the expression of inflammatory cytokines and RANKL. 32
AhR Signaling Pathway
The AhR exerts its downstream change in gene expression through canonical and non-canonical signal transduction mechanisms. 33 In the canonical signaling pathway, AhR forms a complex comprising AhR, heat shock proteins 90, X-associated protein 2 (XAP-2), and p23 in the cytosol. 34 After activation through ligands, AhR is translocated from the cytoplasm to the nucleus and dissociates itself from the complex. Further, the ligand-AhR complex combines with the AhR nuclear translocator (ARNT) and binds to a specific DNA promoter sequence called a xenobiotic responsive element (XRE), which leads to the activation of genes. 35 In the non-canonical pathway, AhR forms a complex with various other transcription factors in the nucleus. This leads to the binding of AhR to non-XRE elements, which further regulates the expression of target genes. 36 For instance, evidence has shown crosstalk between AhR and estrogen receptor (ER), which subsequently binds to DNA elements in the absence of ER ligand.37 -40 AhR can also bind to Kruppel-like factor 6 (KLF-6), a transcription factor that forms the AhR-KLF-6 complex and regulates the cell cycle. 41 Following exposure to a diverse range of exogenous and endogenous ligands, AhR collaborates with numerous nuclear factors to regulate gene expression. This intricate molecular interplay allows AhR to modulate the expression of specific genes in response to various environmental and internal cues (Figure 1). Some of the important pathways regulated by AhR signaling are summarized below.
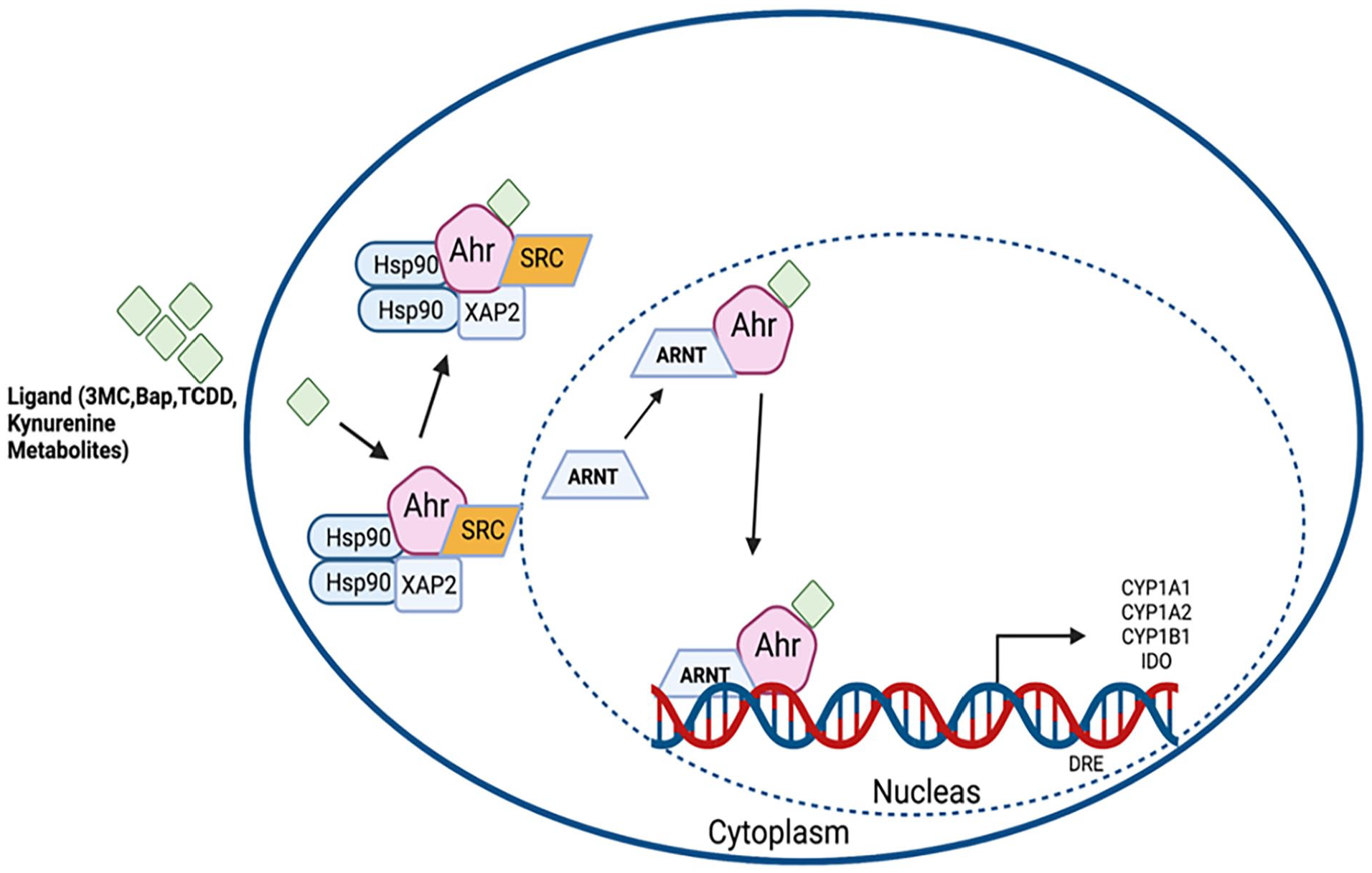
Schematic representation of AhR canonical signaling pathway. AhR forms a complex comprising of AhR, 2 heat shock protein 90, and X-associated protein 2 (XAP-2) in the cytosol. After activation through ligands, AhR is translocated from the cytoplasm to the nucleus, where it dissociates itself from the complex. Further, the ligand-AhR complex combines with the AhR nuclear translocator (ARNT) and binds to a specific DNA promoter sequence called a xenobiotic responsive element (XRE), which leads to the activation of genes (CYP1A1, CYP1A2, CYP1B1, IDO).
AhR and NF-κB Signaling Pathway
The transcriptional factor NF-κB is essential for cell survival, cytokine production, and transcription regulation. The association of AhR and NF-κB is complicated and still not clear. Although, recent studies demonstrated that the binding site of AhR overlaps with the NF-κB binding site. AhR can bind to various subunits of NF-κB and activate the AhR- NF-κB signaling pathway. 42 RelB, a subunit of NF-κB, combines with AhR and acts as a coordinator of the inflammatory response.43,44 A substantial amount of evidence suggests that AhR can regulate NF-κB activity directly via the association with RelA or RelB and indirectly by suppressing SOX2.42,45-47 It has also been reported that different ligands of AhR, such as 3 -MC, BaP, TCDD, and β-NF, can directly interact with AhR and regulate the NF-κB signaling pathway. 33 Furthermore, various studies have pointed out that AhR directly influences NF- κB pathway through AhR ligands such as 3 -MC, BaP, and β-NF, which bind to NF-κB membrane receptors and further inhibit osteoclast formation48 -50 (Figure 2). Naruse et al showed that 3 -MC inhibited the formation of mono and multi-nuclear osteoclast-like cells in a dose-dependent manner. The 3 -MC decreased the levels of RANKL mRNA in ST2 cells but did not affect the mRNA levels of osteoprotegerin (OPG), M-CSF, and the receptor of 1α,25(OH)2 Vitamin D3. 48 Voronov et al suggested that inhibition of osteoclasts by BaP could be due to crosstalk between the receptor activator of NF-κB ligand (RANKL) and AhR signaling cascades competing for NF-κB, a common transcription factor for both pathways. RAW264.7 cells were exposed to various concentrations of RANKL and BaP. The effect on NF-κB activation, nuclear translocation and the effect of NF-κB inhibitors on BaP-mediated CYP1B1 gene expression was assessed. Their results demonstrated that BaP inhibited RANKL-induced NF-κB activation and nuclear translocation. Furthermore, BaP-induced CYP1B1 gene expression was inhibited by 2 NF-κB inhibitors in a dose-dependent manner, exhibiting that NF-κB is involved in a BaP-mediated signaling pathway. 49 The study performed by Hsu et al evaluated the anti-oxidative and anti-inflammatory potential of β-NF in human endothelial cells treated with TNF-α. β-NF pretreatment significantly reduced TNF-α-induced intracellular ROS and TNF-α-induced monocyte binding and transmigration. The suppression of nuclear translocation of NF-κB, p65, and Akt, as well as the phosphorylation of ERK1/2 and p38, was associated with the inhibition of adhesion molecules. The study’s findings revealed that β-NF (β-naphthoflavone), a potent agonist of AhR, suppresses TNF-α-induced NF-kB and ERK1/2 activation and ROS generation, thereby suppressing the expression of adhesion molecules. 50

Schematic representation of AhR-NF-κB signaling pathway. AhR binds to different NF-κB subunits, and the AhR-NF-κB signaling pathway is activated. RelB, an NF-κB component, interacts with AhR to coordinate the AhR pathway. AhR ligands, including 3 -MC, BaP, and β-NF, bind to NF-κB membrane receptors and further restrict the development of osteoclasts by direct effect on the NF-κB pathway.
AhR and Wnt Signaling Pathway
Similarly, as AhR signaling, Wnt signaling is activated by ligand-receptor binding, but instead of chemical ligands, Wnt signaling is activated by Wnt and R-spondin families. 51 Wnt signaling is crucial for cell growth, proliferation, and differentiation and has emerged as a key regulator for bone formation. 52 Wnt proteins are highly conserved across all the species, ranging from mice, humans, Zebrafish, drosophila, and Xenopus. 53 Well-established evidence suggests that AhR regulates Wnt signaling pathways through canonical Wnt signaling.54 -57 AhR ligand TCDD binds to aryl hydrocarbon receptor 2 (AhR2) and up-regulates R Spondin 1 (Rspo1) expression, further causing activation of the Wnt signaling pathway.58 -60 Wnt signaling activates in an AhR-dependent manner, directly influencing osteoblast proliferation and differentiation. 61 Wnt proteins bind to lipoprotein receptor protein 5 (LRP5) and lipoprotein receptor protein 6 (LRP6) ligands. After binding, it stabilizes and enriches β-catenin in the cytoplasm. Furthermore, this activated β-catenin enters the nucleus and interacts with the osteoblast transcription factor, Runx2, and engages in bone formation (Figure 3). Suppression of Wnt signaling leads to decreased osteogenic differentiation, resulting in various bone-related complications such as osteoporosis, osteoarthritis, bone tumors, and inflammatory arthritis. 62

Schematic representation of AhR-Wnt signaling pathway. The Wnt signaling pathway is activated when the AhR ligand TCDD binds to the aryl hydrocarbon receptor (AhR) and increases the expression of R Spondin 1 (Rspo1). Wnt signaling activation happens in an AhR-dependent way and directly affects osteoblast differentiation and proliferation. Lipoprotein receptor protein 5 (LRP5) and lipoprotein receptor protein 6 (LRP6) ligands are recognized by Wnt proteins. After binding, it causes β-catenin to be stabilized and enriched in the cytoplasm. Additionally, this activated β-catenin moves into the nucleus, where it interacts with the Runx2 transcription factor for osteoblasts to promote bone production.
AhR and MAPK Signaling Pathway
Mitogen-activated protein kinases (MAPKs) are responsible for cell differentiation, proliferation, and survival. 63 MAPK family is mainly composed of ERK, p38, and c-JUN-N. There is a direct relation between AhR and MAPK pathway. AhR activates the MAPK pathway by binding to its ligand (TCDD). 64 Upon ligand binding, the inactivated AhR, which is localized in cytoplasm as a complex comprising of HSP90 dimer, XAP-2, p23, and SRC (steroid receptor coactivator) protein kinase, dissociates itself from the complex and translocates into the nucleus by ARNT which leads to conformational changes. Further, the dissociated components of the complex, such as SRC, activate the MAPK pathway through Ras-Raf signaling. 63 Once SRC is activated, it leads to a series of cascade events and further initiates ERK, p-38, and C-JUN-N signaling, which induces phosphorylation and activation of Runx2. Runx2 is the main transcription factor for bone development, leading to osteoblast proliferation, differentiation, and bone matrix formation 65 (Figure 4).

Schematic representation of AhR-MAPK signaling pathway. AhR binds to its ligand (TCDD) to activate the MAPK pathway. When a ligand binds to the inactivated AhR, which is present in the cytoplasm as part of a complex with HSP90 dimer, XAP-2, p23, and SRC (steroid receptor coactivator) protein kinase, the complex dissociates, and the inactivated AhR is transported into the nucleus by ARNT, causing conformational changes. Additionally, the complex’s dissociated components, like SRC, further stimulate the MAPK pathway through Ras-Raf signaling. Once SRC is activated, it triggers cascade events and starts ERK, p-38, and C-JUN-N signaling, which causes Runx2 to be phosphorylated and activated. Runx2 is thought to be the primary transcription factor to produce bone matrix, osteoblast differentiation, and proliferation.
A substantial amount of evidence shows the regulation of MAPKs through AhR ligands, such as TCDD, and bone development. 66 MAPK’s can regulate bone formation and act directly on the osteoblasts, which indicates the importance of MAPKs in bone homeostasis.67,68 Thompson et al reported that activation of ERK1 and ERK2 is essential for bone growth and development. 69 Studies conducted by Bozec et al and Stevenson et al demonstrated that ERK, p38, and c-JUN-n promote osteoclast differentiation.70,71 Induction of ERK1/ERK2 signaling increases eNOS production and decreases RANKL, which results in augmentation of bone formation and thus reduces bone loss.72,73 In vitro findings indicate that p38 and c-JUN-N positively regulate osteoblast proliferation and differentiation.74,75 Wang et al demonstrated that another AhR agonist, Napthoflavone, activated ERK/MAPK signal pathways in an AhR-dependent manner. 66 Furthermore, Yu et al showed that activated AhR inhibited the proliferation and differentiation of osteoblast by enhancing ERK/MAPK phosphorylation. 76 Based on the literature review, most studies have pointed out the beneficial effects of MAPKs on bone formation through an AhR-dependent manner, suggesting the crucial role of AhR in shaping overall bone homeostasis. Several endogenous and exogenous metabolites can act as AhR ligands and affect the above-mentioned signaling pathways and biological processes.
AhR Role in Bone Homeostasis
Bone-related disorders such as osteoporosis and osteoarthritis cause significant health and socioeconomic burdens. With age, osteogenic differentiation of mesenchymal stem cell (MSC) potential decreases, and elevated adipogenic differentiation in the bone marrow.77,78 The build-up of fat in the bone marrow speeds up the disease process.
The defining characteristic of mesenchymal stem cells (MSCs) is their capacity to maintain the ability to differentiate into multiple cell types, including osteoblasts. The 2 key pathways that regulate this differentiation are bone morphogenic protein (BMP) and Wnt pathways. Several transcription factors, such as Runt-related transcription factor 2 (RUNX2), govern these processes. RUNX2 stimulates differentiation into osteoblasts and inhibits adipogenesis.79,80 Some reports indicate that TCDD-activated AhR influences the variety of these downstream effects that alter the course of osteogenic differentiation.81,82 Several groups have shown that activation of AhR by its ligands (eg, TCDD, FICZ) inhibits in-vitro differentiation of bone marrow-derived stem cells into osteoblasts.83 -85 The well-known AhR ligand, 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) activates AhR and negatively affects osteoblast differentiation.81,86 Monnouchi et al reported that treatment of human periodontal ligament cells with benzo[a]pyrene (BaP) reduced mRNA expression of osteogenic genes, alkaline phosphatase activity, mineralization, and collagen synthesis. 87 The study by Liu et al demonstrated that TCDD treatment in human fetal palate MSCs reduced cell proliferation through AhR-induced inhibition of BMP-2/transforming growth factor β 1 (TGFβ 1)/SMAD pathway. Their findings highlighted that TCDD reduced the expression of RUNX2 and BMP-2, which is required for osteogenic differentiation, suggesting that the crosstalk between BMP-2/TGFβ1/SMAD and AhR is crucial for osteogenesis. 88 It is well known that TCDD is a component of cigarette smoke and has a high affinity for AhR. Therefore, keeping this aspect in mind, Yun et al performed a study to analyze the effect of TCDD on bone regeneration and explored AhR antagonism as a potential therapeutic approach to alleviate the effects of TCDD on Bone. Their results demonstrated that TCDD inhibited ALP activity, migratory capacity, and matrix mineralization.
In contrast, treatment with the AhR antagonists (α-Naphthoflavone, resveratrol, 3,3′-Diindolylmethane, and luteolin) mitigated these effects, suggesting that AhR should be investigated as a therapeutic target to counter the adverse effects of cigarette smoke on bone healing. 83 Another study conducted by Watson et al demonstrated the impact of TCDD on osteogenic differentiation in human bone-derived MSCs (hBMSCs). Exposure of hBMSCs to 10 nm TCDD resulted in decreased ALP activity and a reduced matrix mineralization at the terminal stages of osteogenic differentiation. They found attenuated expression in DLX5 and reduced expression of osteogenic markers such as ALP, OPN, and IBSP. Moreover, they used GNF351 AhR antagonist, which blocked TCDD enhancing matrix mineralization and rescued expression of genes associated with osteogenic regulation, extracellular matrix, and/or maintenance of multipotency. 85 Yun et al used TCDD to activate AhR and study its effects on osteogenic markers using a human osteoblast-like cell line. They found that TCDD inhibits osteogenic differentiation, whereas co-treatment with an AhR antagonist showed a protective effect against TCDD. Treatment with AhR antagonist restored the migratory capacity. Their findings suggested that AhR may be a potential therapeutic target for patients who smoke cigarettes and those exposed to secondhand smoke or other environmental sources of aryl hydrocarbons. 84
In the last decade, several studies demonstrated the important role of AhR in bone homeostasis. Jämsä et al reported that TCDD affects bone remodeling by altering the structure of the AhR transactivation domain and further reduces overall bone strength in Han/Wistar (H/W) and Long-Evans (L-E) rat strains. 89 Yoshikawa et al investigated the roles of AhR ligands, l6-formylindolo[3,2-b] carbazole (FICZ), in subchondral bone metabolism and temporomandibular joint osteoarthritis. They studied the mechanisms behind smoking-induced bone loss in temporomandibular joints using a temporomandibular joint-osteoarthritis (TMJ-OA) mouse model. Yoshikawa et al showed the protective role of FICZ-an endogenous AhR ligand, which prevented mandibular subchondral bone loss by repressing osteoclast activity. 90 Our group showed that kynurenine (KYN) elevates miRNAR-29b-1-5p levels in BMSCs, which negatively affects osteogenic differentiation. We also reported KYN mediates the anti-osteogenic effect regulated through AhR signaling. 8 Our group also demonstrated that AhR plays a vital role in the induction of osteoclastogenesis. We showed that blocking AhR signaling using an AhR antagonist, or AhR siRNA, downregulates the KYN/RANKL-mediated increase in c-fos and NFATc1 and inhibits the formation of multinucleated osteoclasts. 9
Few studies demonstrated an important role of AhR in osteoclast differentiation.49,76,91 -93 Liu et al showed that a low dose of indoxyl-sulfate for a short period (3 days) stimulated osteoclast precursor cell differentiation. On the other hand, longer exposures (5 days) resulted in suppressed osteoclast differentiation. 91 Iqbal et al demonstrated that Bap and TCDD interact with AhR and induce osteoclastic bone resorption by activating cytochrome P450 1a/1b (Cyp1) enzymes. Bap and TCDD augmented osteoclast formation in bone marrow cell cultures, and oral gavage with Bap and TCDD stimulated bone resorption and osteoclastogenesis in vivo. 92 Voronov et al demonstrated that Bap at higher concentrations (10−5M) reduced osteoclastogenesis in the RAW264.7 cell line. 49 Ilvesaro et al reported that a short duration of exposure to TCDD does not cause any alterations in the function of osteoclasts. 94 The study performed by Fu et al demonstrated that the treatment of bone marrow cells with 6-formylindolo[3,2-b] carbazole (FICZ) and TCDD promoted osteoclastogenesis. 93 In a separate study, we reported elevated levels of KYN involved in muscle loss with age. 10 We demonstrated a 2-fold increase in reactive oxygen species levels (ROS) and a reduction in muscle size and strength followed by an increase in muscle lipid peroxidation in young mice after kynurenine treatment. Moreover, we reported that inhibition of Ahr in vitro and Ahr knockout in vivo did not prevent a KYN-induced increase in ROS, indicating that kynurenine can directly enhance ROS independent of Ahr activation. 10 Recently, our group demonstrated the involvement of male sex hormones in AhR signaling. 6 We reported that the Kyn-activated AhR signaling and testosterone prevent transcriptional activity in mesenchymal linage cells, suggesting the protective function of male sex hormones in reducing Kyn’s negative effects on cortical bone. Testosterone may be crucial for regulating Kyn/AhR signaling in musculoskeletal tissues, indicating that the interaction between Kyn signaling and male sex hormones may affect age-related musculoskeletal fragility. 6
The findings are inconsistent, and this might be because of different ligands that bind to AhR and play diverse roles. This might be due to several reasons, such as the affinity of ligands, combining efficiency, duration, and pathways that these ligands not only activate but also play a crucial role in regulating them.
Recent studies in vitro studies showed role of AhR in the pathogenesis of osteosarcoma, a malignant bone tumor prevalent among children, adolescents, and young adults.95,96 Vorontsova et al investigated AhR functional activity in 3 primary cell cultures derived from osteosarcoma patients. Their findings demonstrated functional AhR activity in all cell lines but with variable activation patterns of target genes in response to ligand stimulation. 97 Yang et al demonstrated that treatment of the MG-63 osteosarcoma cell line led to the degradation of AhR expression via activation of the AhR signaling pathway 98 . They observed elevated levels of cyclooxygenase-2 and receptor activator of nuclear factor-κB ligand at both protein and mRNA levels after 24 and 72 hours of TCDD treatment. Furthermore, TCDD-treated MG-63 cells exhibited increased prostaglandin E2 production and upregulation of the chemokine receptor CXCR4. These TCDD-induced alterations in RANKL, COX-2, PGE2, and CXCR4 expression were mitigated by CH223191, an AhR inhibitor 98 . These findings underscore the significance of AhR signaling in osteosarcoma and advocate for further exploration of AhR-targeted therapies for malignancy.
Role of Endogenous Metabolites in AhR Signaling
Aging is an accumulation of changes, leading to slow physiological, metabolic, and physical functions. 5 Aging is accelerated by several factors, such as genetic, environmental, and nutritional factors. 99 With age, a change in cellular metabolism affects AhR signaling. It is known that several endogenous cellular metabolites act as AhR ligands, such as tryptophan metabolites. Tryptophan is degraded into several metabolites, such as kynurenine (KYN), kynurenic acid (KYNA), and quinolinic acid (QUIN). Kynurenine and its metabolites act as an AhR ligand. 100 Alterations in the kynurenine pathway accelerate age-related diseases. 101 Previously, our group demonstrated that kynurenine reduces the proliferation of bone marrow mesenchymal stem cells (BMSCs), inhibiting osteoblast proliferation and differentiation. 102 Our group recently reported that increased KYN levels are detrimental and induce bone loss in mouse models 103 suggesting a role for AhR. We also demonstrated that activation of the AhR pathway through kynurenine increases senescence-associated β-galactosidase activity and p21, suggesting an important role in premature senescence. 104
Another endogenous metabolite of the Kyn pathway, the 3-hydroxy anthranilic acid (3-HAA), possesses pro-oxidant properties. 105 Forrest et al measured the concentrations of various kynurenine pathway metabolites in the plasma of osteoporosis patients before, during, and after 2 years of treatment with the raloxifene or etidronate drugs. They discovered that patients with osteoporosis had lower baseline levels of 3-hydroxyanthranilic acid than healthy controls but higher levels of anthranilic acid and lipid peroxidation products. In parallel, both drug treatments studied restored the levels of 3-hydroxyanthranilic acid, anthranilic acid, and lipid peroxidation products to control levels, while tryptophan levels increased significantly when compared to baseline values. 105 It has been demonstrated that 3-hydroxykynurenine can reduce the viability of osteoblast-like cells. 106 A recent study conducted by Wu et al identified 2 novel AhR endogenous ligands 3α,5α-tetrahydrocorticosterone and 3α,5β-tetrahydrocorticosterone (5α- and 5β-THB), which can modulate neural development and differentiation of neuroblastoma. They treated zebrafish embryos with 5α- and 5β-THB and found an increase in HuC, a neurogenesis marker. 107 Furthermore, they found that 5α- and 5β-THB augmented the expression of myelinating glial cell markers, sex-determining region Y-box 10, and myelin-associated proteins myelin basic protein and improved the mobility of zebrafish larvae via the AhR2 pathway. The findings of their study indicate that AhR regulates zebrafish neurogenesis and gliogenes and the differentiation of oligodendrocyte or Schwann cells. In a nutshell, 5α- and 5β-THB are endogenous ligands of AhR and have therapeutic potential for neuroblastoma treatment. 107 Besides, tryptophan metabolites, other endogenous AhR ligands regulate various metabolic pathways. For instance, Lumichrome is a riboflavin metabolite identified as an AhR ligand in rats as early as the 1980s. 108 Heme degradation products such as bilirubin and its metabolic precursor biliverdin are recognized as AhR endogenous ligands that directly activate AhR transformation and CYP isozymes transcription. 109 Yao et al reported that in the hyperhomocysteinemia (HHcy) mouse model, lipoxin A4, a metabolite of arachidonic acid, markedly increases AhR activity and upregulates CD36 expression. 110 Further studies are required to elucidate and provide deeper insight into these complex mechanisms of bone metabolism, which will help design the therapeutic strategy for treating various bone diseases.
Role of Exogenous Metabolites in AhR Signaling
Many agonists of AhR (Table 1), such as 6-formylindolo2, 3carbazole (FICZ), TCDD, and Bap are known to play an important role in musculoskeletal diseases such as rheumatoid arthritis, osteoporosis, inflammatory arthritis. 111 In rheumatoid arthritis (RA), AhR activation by TCDD and FICZ can increase the severity and disease progression, followed by bone destruction and osteoclast differentiation. 112 The study performed by Kazantseva et al demonstrated the effects of smoking on inflamed rheumatoid synovial tissue. Various carcinogenic chemicals present in cigarette causes AhR activation. Higher expression of CYP1A1 and AHRR was reported in the patients who smoke compared to healthy subjects. 112 Altogether, these findings highlight that AhR plays a vital role in deciding the severity and progression of RA. A variety of AhR agonists play a critical role in regulating osteogenic differentiation. It has been reported that bone-derived MSCs treated with TCDD amend the transcriptomic profile of various osteogenic biomarkers, Extracellular matrix synthesis (ECM) markers and lead to suppressing osteogenic differentiation. 85 Benzo[a]pyrene (BAP), another activator of AhR, is a carcinogen found in cigarette smoke and grilled meats. Zhou et al reported that BAP altered the TGFβ1/SMAD4 and TGFβ 1/ERK/AKT pathways, reducing the proliferation rate and self-renewal of MSCs. 113
A list of common Exogenous and Endogenous AhR ligands with their abbreviations and their source (Table adapted from Neavin et al 111 ).

Studies conducted in mouse models of arthritis revealed that AhR nuclear translocation and expression are upregulated.114,115 Study performed by Tong et al demonstrated that the AhR suppresses the osteogenesis of stem cells in collagen-induced arthritic mice through the inhibition of β-catenin. 114 Procházková et al showed that levels of β-catenin were reduced in both TCDD-induced AhR activation and arthritic mice, suggesting the Wnt pathway is affected. 115 Furthermore, documented evidence highlights that long-term use of certain AhR ligands (FICZ, TCDD, Bap) might cause severe side effects such as liver toxicity, carcinogenicity, and high embryo mortality.116,117
Therapeutic Utility of Natural AhR Ligands for Musculoskeletal Health
Many natural AhR ligands are being studied extensively by the scientific community worldwide (Table 2) . These natural AhR ligands can directly or indirectly activate AhR through different mechanisms. For instance, Quercetin, a natural plant-derived AhR ligand, demonstrated that life span could be extended in Caenorhabditis elegans by directly activating AhR through a ligand binding mechanism. 118 Similar findings were reported, indicating the positive effect of quercetin on aging in an AhR-dependent manner in Drosophila melanogaster. 119 Another natural AhR ligand, curcumin (Polyphenol compound) can bind to AhR directly and block downstream signaling by inhibiting AhR phosphorylation by protein kinase C 120 or by binding with endogenous AhR ligand-FICZ thereby activating AhR indirectly. 121 Curcumin is reported to have anti-inflammatory properties, making it an attractive drug against arthritis. The study conducted by Zheng et al demonstrated that curcumin was effective in alleviating the pain caused due to arthritis in Lewis rats. 122 Green tea is a product of the dried leaves of Camellia sinensis, has anti-inflammatory properties. 123 Fukuda et al reported that catechins in green tea suppress the activity of cytochrome P450 1A1 through the AhR activation pathway. 124 The polyphenolic compounds isolated from green tea (aqueous extract) reduced the progression of arthritis in mice. 125 The study carried out by Hasan et al demonstrated that Indole-3-carbinol (I3C) alleviated arthritis by decreasing the level of inflammatory markers in adjuvant-induced arthritis rats. 126 Resveratrol, another natural AhR ligand, is a polyphenol compound in red wine and grape skins. 127 Resveratrol acts as an AhR inhibitor and inhibits the binding of AhR to TCDD, further leading to a reduction in CYP1A1 expression. 127
Natural Ahr ligands and their mechanism of action in Ahr dependent manner (Table adapted from Hui and Dai). 128

Berberine, another natural alkaloid, is reported to have beneficial effects against rheumatoid arthritis. Oral administration of berberine in rats reduced the progression of collagen-induced arthritis by encouraging the differentiation of Treg cells due to the activation of AhR signaling. 131 Tetrandrine is a bis benzylisoquinoline alkaloid discovered in the roots of Stephania tetrandra S. Moore and is currently being utilized in Chinese medicine to treat arthralgia and rheumatalgia. 132 Interacting with AhR encourages AhR localization in the nucleus of T cells, increases the expression of AhR-related reporter genes, and improves the transcription of target genes. 133 The study performed by Yuan et al showed that tetrandrine relieved collagen-induced arthritis in mice by restoring the balance between Th17 and Treg cells via AhR signaling. 133 Since the 1960s, Sinomenine, an alkaloid derived from the roots of Sinomenium acutum, has been utilized in China as an anti-rheumatoid arthritis treatment. By boosting the expression of AhR target genes, encouraging AhR/Hsp90 dissociation, and encouraging AhR nuclear translocation, it attenuates arthritis by stimulating the development and function of Treg cells in an AhR-dependent way.134,135 Numerous cruciferous vegetables contain the compound indole-3-carbinol (I3C). Studies have shown that I3C attenuated arthritis by reducing inflammatory markers and ameliorates the histopathological changes of the tibiotarsal joints in adjuvant-induced arthritis rats by promoting the generation of Tregs cells and down-regulating the percentages of Th17 cells in vivo and in vitro in an AhR-dependent manner.126,136 3,3′-diindolylmethane has been linked to having anti-tumorigenic and anti-inflammatory activities through the regulation of AhR, making it one of the best-characterized and most bioactive components in the widely utilized cruciferous plants. 137 Dong et al reported that 3,3′-diindolylmethane lowered the levels of inflammatory cytokines such as IL-1, NO, and TNF-alpha, as well as the expression of RANKL in osteoclasts and fibroblast-like synoviocytes (FLS) to prevent osteoclastogenesis, it also attenuated the clinical and histologic indicators of inflammation and tissue damage. 138
The Natural AhR agonists with fewer side effects will be promising candidates for the therapeutic approach against rheumatoid arthritis. Future studies are needed to decipher and understand the exact mechanisms regarding the functioning of these natural ligands and identify new targets for treating various bone diseases. AhR plays a crucial role as a contributing factor in many bone diseases; therefore, AhR can be used as the target for novel pharmacotherapeutic agents. Nevertheless, more studies still need to be carried out to understand the clinical aspects and molecular mechanism of AhR to consider its use in clinical trials in the near future.
Identification of Novel Genes Regulated by AhR Signaling in Bone Homeostasis by GEO2R Analysis
We performed GEO data analysis to understand the molecular mechanism involved in bone biology. The primary purpose of using the Gene Expression Omnibus (GEO) dataset was to find the studies that align with our notion suggesting that AhR is essential for bone biology and homeostasis. We searched the published literature through the GEO dataset for studies demonstrating AhR’s importance in bone formation and development. We used the search terms “AhR-bone development”; “AhR-bone formation” and found 2 studies aligned with our notion. The GEO dataset archived by Xiong et al was analyzed to determine differentially expressed genes, highlighting the importance of AhR in bone biology (GEO accession GSE11893). 139 Briefly, the study investigated transcriptional response in zebrafish larvae exposed to TCDD (1,2,4 and 12 hours). The study’s findings suggested that TCDD activation of AhR downregulated numerous chondrogenic transcripts, leading to jaw malformation in zebrafish embryos. We performed the GEO2R analysis for 4 groups (Jaw Vehicle Control- 1 Hour vs Jaw TCDD- 1 Hour; Jaw Vehicle Control- 2 Hours vs Jaw TCDD- 2 Hours; Jaw Vehicle Control- 4 Hours vs Jaw TCDD- 4 Hours; Jaw Vehicle Control- 12 Hours vs Jaw TCDD- 12 Hours). An expression density plot was generated using GEO2R to check data normalization to complement boxplot analysis. The GEO2R interactive tool was used to determine common genes involved in the AhR pathway in both treatment and control groups by calculating log base 2 of fold change (log2(FC)). No alterations were made in Benjamini & Hocheberg false discovery rate method for p-value adjustment as it is widely used to perform GEO2R analysis. The rest of the parameters were set to default settings. The GEO2R analysis identified 8 genes (CYP1C1, SULT6B1, CYB5A, EDN1, CXCR4B, CTGFA, TIPARP, and CXXC5A) involved in the AhR pathway, which play a crucial role in bone remodeling. These genes were found in all treatment conditions: Jaw Vehicle Control- 1 Hour versus Jaw TCDD- 1 Hour; Jaw Vehicle Control- 2 Hours versus Jaw TCDD- 2 Hours; Jaw Vehicle Control- 4 Hours versus Jaw TCDD- 4 Hours and Jaw Vehicle Control- 12 Hours versus Jaw TCDD- 12 Hours. AhR was significantly expressed after 4 and 12 hours; the expression was not detected during the first 1 to 2 hours. The AhR was expressed from 4 to 12 hours, which correlated with significant upregulation of CYP1C1, SULT6B1, and CYB5A. Therefore, this indicated that, in turn, these 3 genes are AhR-dependent/AhR-associated. It signifies that AhR is involved in bone morphogenesis, as these 3 genes are critical for normal bone biology. However, after 12 hours of TCDD exposure, the expression of osteogenic genes like EDN1, CXCR4B, CXC5A, TIPARP, and CTGFA was downregulated (Figure 5). Another GEO dataset archived by Bennett et al (GEO accession GSE76276) was analyzed to emphasize the importance of AhR in maintaining the integrity of hematopoietic stem cells and how it affects overall bone biology. 140 Briefly, the study investigated the effect of AhR gene knockout on expression profiles of hematopoietic stem cells from young and old mice. The study’s findings indicated that conditional deletion of AhR resulted in the alteration of several gene and signaling networks crucial for the normal function of hematopoietic stem cells. We performed GEO2R analysis for 2 groups (Young AhR KO vs Young Floxed AhR and Old AhR KO vs Old Floxed AhR). Initially, we performed GEO2R analysis for Old AhR KO versus Old Floxed AhR. We identified the top 250 differentially expressed genes. Based on the fold change, out of the top 250, the top 20 most upregulated (Table S1) and most downregulated genes (Table S2) were selected. Moreover, based on the GEO2R analysis and literature review, out of the top 20 most upregulated genes, we found 3 novel genes, namely, Defb14, ZNF 51, and Chrm5, involved in bone physiology and development (Figure 6a-c). It has been reported that Defb14 is involved in controlling bone mineral density. 141 ZNF 51 mainly regulates skeletogenesis and plays an important role in tooth, cartilage, and bone development. It is responsible for maintaining the structural integrity of the skeletal system. 142 A study by Kauschke et al demonstrated that Chrm5 is crucial for bone homeostasis as it enhances bone formation and decreases bone resorption. 143 Furthermore, out of the top 20 most downregulated genes, based on the GEO2R analysis and literature review, we found 7 novel genes, namely Cdh2, Oosp1, Ttc30b, Fabp4, Cldn5, H28, and Epcam, which are involved in bone homeostasis. Out of 7 genes, we selected the top 3 genes (Cdh2, Oosp1, and Ttc30b) for further analysis based on their fold change (Figure 6d-f). A study reported by Marie et al showed that Cdh2 is essential for cell-cell adhesion and signaling in bone remodeling. 144

AhR activation regulates bone remodeling genes in Zebrafish. GEO2R data analysis showing 8 bone homeostasis-related genes (CYP1C1, SULT6B1, CYB5A, EDN1, CXCR4B, CTGFA, TIPARP, and CXXC5A) differentially regulated after activation of AhR signaling in jaw of Zebrafish. The data were retrieved from the GEO dataset uploaded by Xiong et al (GEO accession GSE11893); significance was determined by GEO2R adjusted *P-value < .05. **P-value < .001 (n = 3/group).
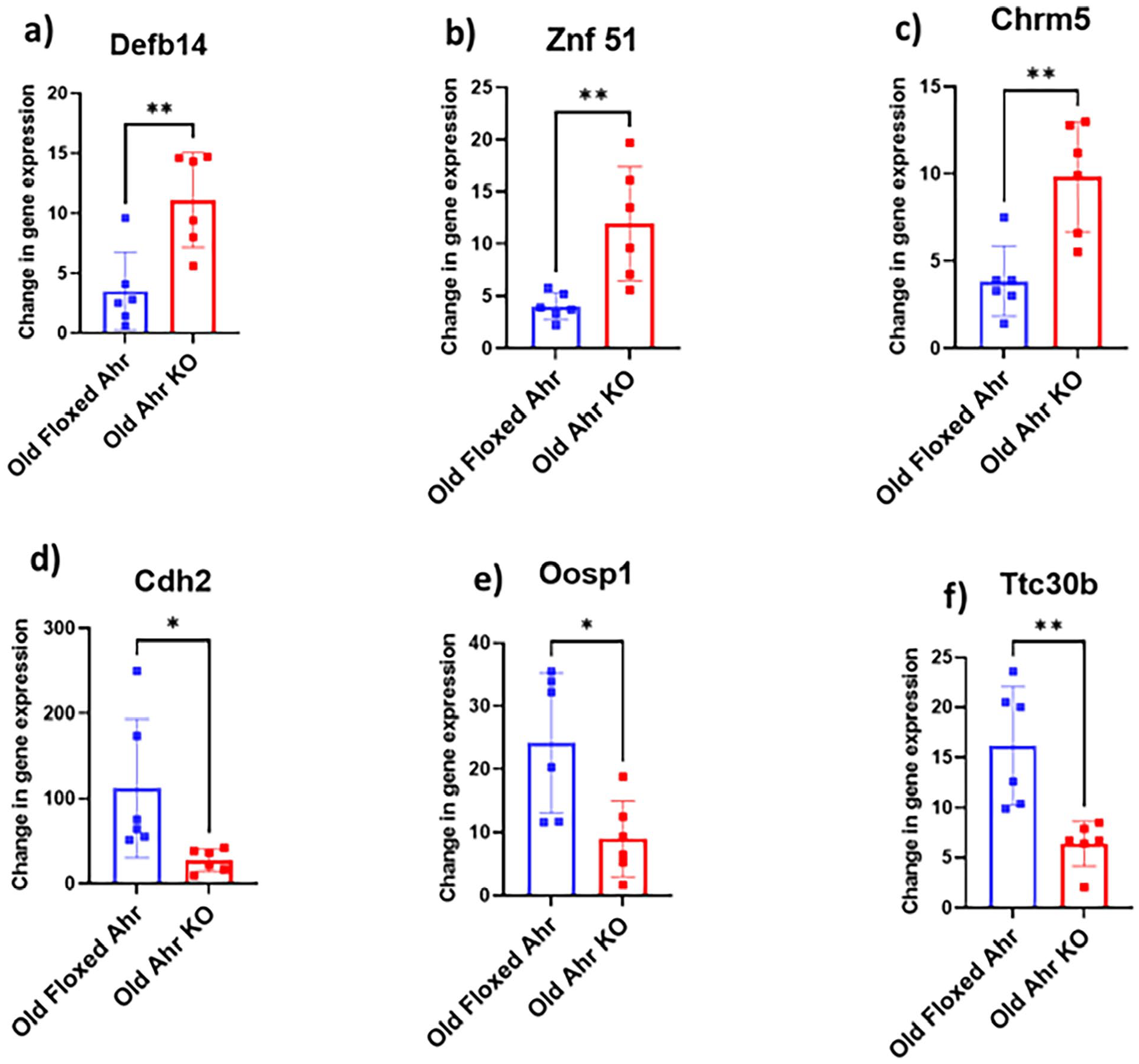
Identification of novel bone-related genes involved in AhR signaling with age: GEO2R study (Young AhR KO vs Young Floxed AhR) showing elevated levels of (a) Defb14, (b) ZNF 51, and (c) Chrm5 genes and top 3 genes (d) Cdh2, (e) Oosp1, and (f) Tth30b downregulated in AhR knock out mice. The data were retrieved from the GEO dataset uploaded by Welle et al (GEO accession 76276). The significance was determined by GEO2R adjusted *P-value < .05. **P-value < .001 (n = 5-6).
Furthermore, we performed GEO2R analysis for Young AhR KO versus Young AhR-FLoxed. We identified the top 250 differentially expressed genes. Out of the top 250, 20 most upregulated (Table S3) and the top 20 most downregulated (Table S4) were selected based on their fold change. Based on the literature review and GEO2R analysis, out of the top 20 most upregulated genes, we found 15 genes involved in bone physiology. Out of those 15 genes, we discussed here the top 3 (Cdk7, Eed, and Raasf5) most upregulated genes involved in bone biology based on their fold change as well as literature review and their ability to influence bone remodeling for further analysis (Figure 7a-c). Cyclin-dependent kinase-7 (Cdk-7) is the master regulator of all the cyclin-dependent kinases through the phosphorylation of T-loops. Cdk-7 is crucial for bone homeostasis because it is vital in maintaining hematopoietic stem cells. Studies have pointed out that deleting Cdk-7 leads to abnormalities in bone marrow, such as adipose tissue deposition and reduction in bone mineral content. 145 Rassf5 is essential for osteoblastogenesis. It facilitates the proliferation and differentiation of osteoblasts and suppresses osteoclasts. A study by Song et al demonstrated the significance of Rassf5 in bone remodeling. 146 The embryonic ectoderm development gene (Eed) is responsible for chondrocytes’ proliferation and differentiation. It plays a crucial role in preserving the overall structure of the growth plate. A study conducted by Mirzamohammadi et al showed that Eed is necessary for the proliferation and differentiation of chondrocytes and plays a key role in preserving the overall integrity of growth plates. 147 In the top 20 most downregulated genes, we selected Haptoglobin (Hp), Rnf114, and Rab27b based on the literature review and their ability to impact bone remodeling (Figure 7d-f). Haptoglobin is responsible for maintaining bone microarchitecture and bone volume. Haptoglobin prevents excessive osteoclastogenesis. 148 Ring finger protein 114 (Rnf114) regulates the RANKL signaling pathway and mediates osteoclastogenesis. Rnf114 suppresses osteoclast proliferation and differentiation, thereby augmenting bone formation. 149 Rab27b is a small GTPase that regulates the RANKL release pathway. It decreases bone resorption and enhances bone volume. 150

Identification of novel bone-related genes involved in AhR signaling: GEO2R study (Old AhR KO vs Old Floxed AhR) showing elevated levels of (a) Cdk7, (b) Rassf5, and (c) Eed genes and top 3 genes (d) Hp, (e) Rnf114, and (f) Rab27b downregulated in AhR knock out mice. The data were retrieved from the GEO dataset uploaded by Welle et al (GEO accession 76276). The significance was determined by GEO2R adjusted *P-value < .05. **P-value < .001 (n = 5-6).
AhR Pathway Affected Gene Ontology Enrichment Analysis of Biological Processes Involved in Bone Formation
We performed gene ontology enrichment analysis and pathway annotation using DAVID (Database for Annotation, Visualization, and Integrated Discovery) for those genes involved in the AhR pathway to gain a deeper understanding of the biological process and functions regulated by these genes (Table S5). Our analysis revealed that the annotation cluster mainly consisted of the xenobiotic metabolic process, p450 pathway, oxidation-reduction, and nitric oxide biosynthesis process, which were significantly enriched as compared to other functions. Moreover, we also performed Gene Ontology Enrichment Analysis using the GO consortium for the genes involved in the AhR pathway to check their role specifically in bone biological processes using the Gene Ontology Resource software tool. All the parameters were set at default settings (Analysis Type: Panther, Annotation Version, and Release Date: GO Ontology database DOI: 10.5281/zenodo.4735677 Released 2021-05-01, Analyzed List: Homo Sapiens, Reference List: Homo Sapiens-all genes in the database, Annotation Set: GO biological process, Test Type: Fisher, Correction: False Discovery Rate). Gene Ontology Enrichment Analysis for the genes involved in the AhR pathway identified 54 biological processes associated with bone homeostasis. Mainly, these processes include bone morphogenesis, bone development, bone trabeculae formation, bone resorption, bone maturation, bone mineralization, and bone marrow development (Table S6). Overall, our analysis showed a strong correlation between AhR signaling pathways and their role in bone development and homeostasis.
Conclusion
With an enhanced comprehension of the relationship between AhR signaling and bone homeostasis, it is tempting to conclude that AhR signaling indeed influences bone biology. However, conflicting evidence also highlights the positive and adverse effects of AhR signaling on bone. Some studies indicate positive effects, while others demonstrate detrimental effects on bone homeostasis. These conflicting findings challenge the scientific community to reach a consensus regarding the utilization of AhR as a therapeutic option for bone disorders and diseases. Nevertheless, evidence suggests that AhR plays a significant role in osteogenic differentiation, bone formation, and remodeling, indicating its potential as a target for designing diverse treatment strategies against various bone ailments. The effects of AhR agonists and antagonists on bone remain unclear, hindering the clinical application of AhR-related therapies for bone disorders. The inconsistencies in findings may be attributed to several factors, including differences in ligand binding affinity to AhR, binding sites, half-life, chemical structure, and various unknown factors. However, further research is still required to investigate the underlying mechanisms of AhR action and its influence on bone remodeling.
Supplemental Material
sj-docx-1-try-10.1177_11786469241246674 – Supplemental material for The Role of Aryl Hydrocarbon Receptor in Bone Biology
Supplemental material, sj-docx-1-try-10.1177_11786469241246674 for The Role of Aryl Hydrocarbon Receptor in Bone Biology by Sagar Vyavahare, Pankaj Ahluwalia, Sonu Kumar Gupta, Ravindra Kolhe, William D Hill, Mark Hamrick, Carlos M Isales and Sadanand Fulzele in International Journal of Tryptophan Research
Footnotes
Author Contributions
SV and SF conceived the study. SF reviewed the literature and identified suitable publications. SV wrote the original draft of the manuscript with the help of SF. PA, SKG, SV, and RK participated in generation of data, figure, and tables of the manuscript. WDH, MH, CMI and SF edited the manuscript. All authors read and approved the final manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is based upon work supported in part by the National Institutes of Health NIA00059 (SF), and AG036675 (National Institute on Aging-AG036675 S.F, MH, WH, C.I). The above-mentioned funding did not lead to any conflict of interest regarding the publication of this manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting this study’s findings will be available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
