Abstract
Cutaneous sclerosis, resembling that seen in subcutaneous morphea, is a feature of eosinophilic fasciitis and eosinophilia-myalgia syndrome, two conditions in which the kynurenine pathway is known to be activated. To investigate the possibility of activation of the kynurenine pathway in morphea, skin biopsies were taken from involved and non-involved sites in a series of three patients with morphea. Immunohistochemical stains for quinolinic acid and indoleamine 2,3-dioxygenase (IDO) were performed.
Introduction
The ingestion of L-tryptophan is associated with eosinophilic fasciitis1,2 and the eosinophilia myalgia syndrome. 3 Although initially attributed to the contaminant 1,1-ethylidenebis tryptophan detected in the product manufactured by Showa Denko, 4 other authors have reported the same adverse reaction from L-tryptophan supplied by other manufacturers. 5 Activation of the kynurenine pathway was reported in these patients 6 as it was in patients suffering from the so called toxic oil syndrome. 7 The latter was reported in Spain in 1981 as a result of adulterated rapeseed (canola) oil and shared many features with eosinophilia-myalgia syndrome and eosinophilic fasciitis.
Further evidence supporting the role of quinolinic acid in eosinophilic fasciitis was published in 2006 when a human volunteer underwent a series of subcutaneous injections of quinolinic acid. 8 A rise in the peripheral eosinophil count, cutaneous eosinophilic infiltration, and production of the fibrotic cytokine transforming growth factor (TGF) beta 1 by dermal dendrocytes and endothelial cells was reported.
Little is known about the normal role of the kynurenine pathway in skin. N-methyl-D-aspartate (NMDA) receptors have been found to be present in non-neuronal tissue and have been demonstrated to have functional roles in keratinocytes. 9 Fibroblasts express the kynurenine pathway with significant polymorphisms. 10 The kynurenine pathway is active in macrophages. 11 On the basis of current knowledge, L-tryptophan is metabolized to quinolinic acid in leucocytes and keratinocytes, diffuses into dermal connective tissue, after which it enters the vascular and lymphatic plexuses before being transported to the liver for conversion to nicotinamide adenine dinucleotide (NAD). Indoleamine 2,3-dioxygenase (IDO), the rate limiting enzyme, 12 is ubiquitous and found in many cell types. 13 Activation occurs by both an increase in transcript number and enzyme activation.14,15 IDO activity increases with age. 16 Overall the results favor increased activity of the kynurenine pathway in morphea with evidence of background activity in control specimens. The first two segments of the kynurenine pathways are expressed in the basal layer of the epidermis, eccrine units, and endothelium. The bulge area of the hair follicle has previously been reported as a site of increased IDO activity and may be a site of relative immune privilege. 17
Materials and Methods
The study was undertaken at the Queensland Institute of Dermatology (P.O Box277 Holland Park, QLD 4121 Australia www.qldid.com.au.). Ethics committee approval was obtained from the Ethics Committee of Greenslopes Private Hospital (Newdegate Street Greenslopes QLD 4120 Australia. www.greenslopesprivate.com.au.). Immunohistochemical stains (IHC) were performed at the Queensland Institute of Medical Research (300 Herston Road Herston QLD 4006. Australia. www.qimr.edu.au.).
Patients
Summary of patient's clinical features.
Punch biopsies were taken from involved and non-involved skin. Hematoxylin and Eosin stains and immunohistochemistry stains for IDO were fixed in formalin. Quinolinic acid stains were prepared as frozen sections in OTC medium and liquid nitrogen. Immunohistochemical stains were performed at the Queensland Institute of Medical Research histotechnology facility. The protocols were as follows:
Quinolinic acid
Sections were fixed in -20°C acetone for 10 min before being rehydrated in phosphate-buffered saline (PBS), pH 7.4. Endogenous peroxidase activity was blocked by incubating the sections 3.0% H2O2 in PBS for 10 min.
Sections were washed in three changes of PBS for 5 min each and non-specific antibody binding inhibited by incubating the sections in 10% normal goat serum for 30 min.
Abcam rabbit anti-quinolinic acid antibody (ab 37106) was used. Antibody was diluted 1:200 in PBS and applied for 90 min at room temperature, sections washed in three changes of PBS for 2 min each. They were than incubated with Dako Envision Plus anti-rabbit for 30 minutes at room temperature.
Sections were washed in three changes of Tris buffered saline (TBS) for 5 min each and color developed in Vector Nova Red for 5 min. They were then washed in gently running tap water for 5–10 min to remove excess chromogen, then lightly counterstained in Mayers’ hematoxylin and dehydrated through ascending graded alcohols, cleared in xylene, and mounted.
Negative controls were performed using antibody diluent without antibody. No positive control is available.
Indoleamine 2,3-dioxygenase
Sections were de-waxed and rehydrated through xylol and descending graded alcohols to water.
Endogenous peroxidase was blocked by incubating slides in 1.0% hydrogen peroxide plus 0.1% sodium azide in TBS for 10 min before being washed in water three times.
Sections were transferred to Dako Antigen retrieval Solution pH 6.0 and heated for 20 min at 100 °C in a Biocare Medical Decloaking Chamber. Slides were removed and allowed to cool for 20 min at room temperature then washed in TBS 3 times for 5 min each.
Non-specific antibody binding was inhibited by the application of 10% goat serum and 1.0% bovine serum albumin (BSA) in TBS for 30 min.
Abcam ab 55305 mouse anti-IDO antibody (0.5 mg/mL) was used. Anti-IDO was diluted 1:150 and applied overnight at room temperature before being washed in three changes of TBS for 5 min each.
Dako Envision Plus anti-mouse Monoclonal ANTI-FLAG® M2-Peroxidase (HRP) was applied for 30 min before being washed in three changes of TBS for 5 min each.
Color was developed in 3,3′-diaminobenzidine (DAB) with H2O2 as substrate for 5 min and excess chromogen removed by washing in water.
Sections were lightly counterstained in Mayer's hematoxylin then dehydrated through ascending graded alcohols, cleared in xylene, and mounted.
Positive control was performed on human tonsil non germinal center tissue looking for cytoplasmic and nuclear labeling in lymphoid cells outside the germinal center. Negative controls were performed using antibody diluent without antibody.
Results
The kynurenine pathway (Noakes R. Effects of tranilast on the urinary excretion of kynurenic and quinolinic acid under conditions of I tryptophan loading. Int J Tryptophan Res. 2013;6:67–71).


Patient 1, 2 and 3: Involved hematoxylin and Eosin × 20.
Cells of hematopoietic origin
As expected, an infiltrate of inflammatory cells producing quinolinic acid is seen in involved tissue; this is not evident in non-involved tissue (Figs. 2–4). These cells also express IDO (Figs. 5, 6).
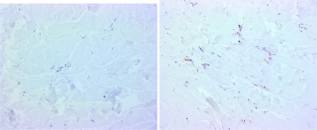
Patient 1: Quinolinic acid, non-involved (left) and involved (right) × 200. Patient 2: Quinolinic acid, non-involved (left) and involved (right). Patient 3: Quinolinic acid, non-involved (left) and involved (right) × 200. Patient 2: Indoleamine 2,3-dioxygenase, non-involved (left) and involved (right). Patient 3: Indoleamine 2,3-dioxygenase, non-involved (left) and involved (right) × 200. Patient 1: Quinolinic acid, Non-involved. Eccrine unit × 100.




Eccrine units
There are no published reports on kynurenine activity in eccrine units, yet the findings suggest it is an active site. The eccrine units at the control site in patient one showed background activity (Fig. 7). In patient two, the involved site displayed strong expression in the eccrine unit with little or no expression at the control site (Fig. 8). In patient three, there is strong expression in the eccrine unit at the involved site (Fig. 9). This data supports activity of the kynurenine pathway in the eccrine unit. It is of interest that the basement membrane zone of the epidermis, eccrine units, and pilosebaceous units are sites of involvement in cutaneous lupus erythematosus. Whether loss of IDO mediated immunological tolerance plays a role in this disorder is speculative but plausible.
Patient 2: Quinolinic acid, Eccrine unit non-involved (left) and involved (right) × 200. Patient 3: Eccrine unit, Quinolinic acid, involved × 100.

Epidermis
Immunohistochemistry stains support activity of the first two segments of the kynurenine pathway in the basal layer with little evidence of activity in the spinous and granular layers (Fig. 10). It is of interest that one of the described dermatopathological reaction patterns, the lichenoid pattern, is characterized by immunological attack on this zone. Although speculative, it is plausible that kynurenine mediated loss of immunological tolerance may play a role in this reaction pattern.
Patient 3: non-involved, Epidermis × 100.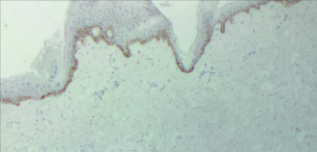
Vascular and lymphatics
IHC stains show evidence of activation of the kynurenine pathway with quinolinic acid synthesis in the endothelium (Fig. 11). IDO activity has previously been reported in endothelium
18
and this data would suggest the second segment of the pathway is also active in endothelium.
Patient 1: Endothelial cells, Quinolinic acid, involved × 200.
Discussion
The kynurenine pathway is the metabolic pathway via which L-tryptophan is converted to NAD. L-tryptophan is the least common of the essential amino acids and depletion of it within the microenvironment not only starves cells of an essential amino acid,19,20 but can lead to antiproliferative and apoptotic effects on T cells 21 mediated through the GCN2 kinase. 22 The kynurenine metabolites quinolinic acid and 3-hydroxyanthranilic acid suppress T cell proliferation. 23
In the kynurenine pathway, tryptophan is initially oxidized to formylkynurenine by IDO-1, IDO-2, 24 or tryptophan 2,3-dioxygenase (TDO). The latter is a hepatic enzyme and is induced by tryptophan or corticosteroids. 25 IDO-1 is found extrahepatically and is potently induced by interferon gamma. 6 The kynurenine metabolites themselves have immunoregulatory roles promoting the apoptosis of Th1, but not Th2 cells. 26 The kynurenine metabolite 3-hydroxyanthranilic acid promotes a cytokine shift in favor of TH2 cells. 27
Most interest has focused on the role of kynurenine metabolites in neuroinflammation. Kynurenic acid 28 and picolinic acid 29 are neuroprotective whilst quinolinic acid 30 and 3-hydroxyanthranilic acid 20 are neurotoxic.
There are reasons to suggest the kynurenine pathway is active in morphea.
Firstly, morphea is associated with eosinophilia 31 and the kynurenine metabolites 3-hydroxyanthranilic acid and quinolinic acid promote a TH2 shift.16,17
Secondly, morphea is often associated with neurological findings. 21 The latter occur most commonly, but not exclusively, in frontoparietal disease. The kynurenine metabolites with the exception of kynurenic acid 18 and picolinic acid 19 are neurotoxic 20 with quinolinic acid mediating its effect through NMDA receptors. 32 Alterations in nerve conduction studies have been reported in morphea. 33
Finally, generalized scleroderma is associated with Raynaud's phenomenon or chronic episodic digital ischemia. Although the etiology of this remains controversial, nitric oxide has been reported to play a role and 3-hydroxyanthranilic acid is known to inhibit the expression and activity of inducible nitric oxide synthase. 34
Although the causes of most cases are unknown, morphea has been reported to follow vaccination, radiotherapy, and infections, 35 all known inducers of interferon gamma production, the main inducer of IDO-1. 15 Histologically early lesions of morphea display a superficial and deep inflammatory lymphocytic infiltrate and it is possible that the infiltrating lymphocytes are responsible for the production of interferon gamma.
We have previously published on the results of quinolinic acid injections in a human volunteer. 8 Both a peripheral eosinophilia and an interstitial infiltrate of eosinophils were reported. TGF-beta production by dermal dendrocytes and endothelial cells was noted. Elevated levels of the fibrotic cytokine TGF-beta is found in morphea. TGF-beta 1, 2 and 3 are secreted by most cell types, generally in a latent form, requiring activation before they can exert biological activity. This activation of latent TGF-beta, which may involve plasmin, thrombospondin, and possibly acidic microenvironments, appears to be a crucial regulatory step in controlling their effects. 36 Regrettably, there exists little published data on associations between kynurenine metabolites and possible activation of the TGF-betas, although it is likely both are entwined due to their immunosuppressive properties. 37 There are reported alteration in the kynurenine pathway in the hyper-fibrinolytic states seen in chronic renal failure patients on dialysis, 38 which could potentially suggest that they play a role in plasminogen conversion to plasmin and activation of latent TGFs. This is, however, speculative.
Our results indicate both IDO and quinolinic acid are expressed in the basal layer of the epidermis, eccrine units, and vascular endothelium in addition to infiltrating inflammatory cells. The concentration of metabolites in the microenvironment is likely to be critical in disease states as the kynurenine pathway is a physiological metabolic pathway and pathological consequences should not be seen at physiological concentrations. The importance of concentration has already been reported in relation to the neurotoxic effects of quinolinic acid. 39 It is of interest that the adnexal structures, which are lost in morphea, correspond to the site of maximal activity of the kynurenine pathway supporting an etiological role.
Thus we provide data to support evidence of activation of the kynurenine pathway in morphea.
The question as to whether the kynurenine pathway is responsible for the increased synthesis and activation of TGFs beta in morphea is more difficult to answer. It is suggested, however, by the overlap between eosinophilia myalgia syndrome and eosinophilic fasciitis, which is now included in the spectrum of localized scleroderma.
The issue would be best answered by demonstrating that manipulation of this pathway produced clinical improvement. There is already some data, albeit mixed, available on this. Tranilast, a 3 hydroxyanthranilic acid analogue, has been reported to be of benefit in anecdotal reports 40 and has marketing approval for treatment of keloid scars in Japan. None the less, definitive answers require further investigation.
The principal limitations of this study are the small number of patients and the dynamic nature of the system under investigation with IHC studies providing a snapshot only. This may not be representative of the behavior of the system over a longer time frame.
Author Contributions
Conceived and designed the experiments: RN, NM. Analyzed the data: RN, NM. Wrote the first draft of the manuscript: RN, NM. Contributed to the writing of the manuscript: RN, NM. Agree with manuscript results and conclusions: RN, NM. Jointly developed the structure and arguments for the paper: RN, NM. Made critical revisions and approved final version: RN, NM. All authors reviewed and approved of the final manuscript.
Disclosures and Eethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests.
