Abstract
B-1 cells are a peculiar subpopulation of B cells found in the peritoneal and pleural cavities in mice. These cells are typically IgM+ and CD11b+. B-1 cells are able to migrate from the peritoneal cavity to non-specific inflammatory sites in mice. In addition, they can differentiate into mononuclear phagocyte-like cells in vitro; however, it is still unknown whether B-1 cells are capable of performing phagocytosis in vivo. Here we further characterized B-1 cells as phagocytes in vitro, and we investigated their ability to phagocytose apoptotic cells and bacteria in vivo. Our results demonstrate that B-1 phagocytes are able to uptake apoptotic thymocytes and Escherichia coli bacteria, both in vitro and in vivo. These findings indicate that along with macrophages, B-1 phagocytic cells might play a role in fundamental processes such as tissue remodeling, resolution of inflammation and pathogen clearance.
Keywords
Introduction
Phagocytosis is accomplished predominantly by professional phagocytes such as polymorphonuclear cells, monocytes and macrophages. 1 Other cell types, such as fibroblasts and epithelial cells, are also capable of engulfing particles but to a much lesser degree than professional phagocytes. These so-called nonprofessional phagocytes uptake a more limited range of particles mainly because of their restricted repertoire of phagocytic receptors; 1 therefore, they can not play a critical role in processes such as tissue remodeling and immunity.
Unlike professional phagocytes, most B cell subsets are not able to perform phagocytosis. 2 B-1 cells, however, constitute a peculiar fraction of the B cell population in this respect. This population of cells is found mainly in the peritoneal and pleural cavities in mice, and they are rarely found in the spleen and are not detected in lymph nodes. They are typically IgMhigh, IgDlow, CD23–, CD43+, CD19+ and CD11b+, and they are subdivided into B-1a (CD5+) and B-1b (CD5–) subsets.3,4 It is known that B-1 cells are able to migrate from the peritoneal cavity to non-specific inflammatory sites in mice;5,6 in addition, these cells differentiate into phagocyte-like cells in vitro. These phagocyte-like cells are capable of ingesting mannose-coated particles, 6 and they are also able to phagocytose and kill bacteria in culture. 7 Moreover, malignant CD5+ B cells can assume a macrophage-like phenotype capable of ingesting particles. 8
Previous studies have discussed the mechanisms underlying the ability of B-1 cells to differentiate into phagocytes, as well as the ontogenic and physiological implications of this phenomenon. Several investigators have demonstrated that CD5+ pre-B cells can differentiate into macrophage-like cells.9,10 A recent report has shown that undifferentiated B-1 cells express both myeloid and lymphoid lineage-specific transcription factors, whereas after undergoing differentiation into phagocytes, they predominantly display a myeloid profile. 7 In addition, the CD5 and IgM antigens, which are markers classically associated with the lymphoid lineage, are lost when B-1 cells migrate from the peritoneal cavity in response inflammatory stimuli. 11 Altogether, these “biphenotypic” features displayed by B-1 cells suggest that certain B lymphocytes and macrophages share a closer lineage relationship than is predicted by current models of hematopoietic differentiation. 12
Despite the phagocytic and microbicidal properties of B-1 cells, it remains to be determined to what extent they would be effective in performing these features in vivo, ie, the physiological relevance of the previous in vitro observations. Here, we investigated the ability of B-1 cells to phagocytose apoptotic cells and bacteria in an effort to elucidate the possible influence that these cells have on apoptotic cell clearance and infection.
Material and Methods
Animals
Male BALB/c mice, 6–12 weeks old, were provided by the animal facility of the Federal University of São Paulo, Brazil. Animal handling and housing were performed in accordance with the NIH Guide for Care and Use of Laboratory Animals.
Apoptotic Thymocytes
Thymic cells from BALB/c mice were harvested and maintained in RPMI 1640 media. A single-cell suspension of thymocytes was prepared by thymic disruption in a nylon filter chamber. Cells were washed once with phosphate-buffer saline (PBS) and submitted to 500 cGy of gamma irradiation using a Gamma-cell 3000 Elan 137Cs source (Nordion International Inc., Kanata, Ontario, Canada).
Bacteria
Escherichia coli strain k91, kindly provided by Renata Pasqualini (MD Anderson Cancer Center, USA), was maintained as previously described. 6 Before each experiment, E. coli were cultured in Luria-Bertani media (LB) (Difco) at 37 °C, harvested at mid-log phase, and washed twice before inoculation into the peritoneal cavities of animals or in vitro culture.
B-1 Cell cultures
Mouse B-1 cells were cultured as previously described. 6 Briefly, cells were collected by washing the peritoneal cavity with ice-cold RPMI 1640 media (Sigma-Aldrich, St. Louis, MO, USA). Non-adherent cells were discarded after the peritoneal cells were incubated on glass dishes for 1 h at 37 °C in 0.5% CO2. Adherent cells were detached by scraping and transferred to culture flasks with RPMI 1640 media containing 10% heat-inactivated fetal calf serum (Gibco/BRL, Carlsbad, CA, USA). After 6 days without media replacement, floating cells, which mostly consisted of B-1 cells, were collected and analyzed by flow cytometry or used to obtain B-1-derived mononuclear phagocytes.
B-1-Derived Mononuclear Phagocytes(Bdmps)
BDMP cells were obtained as previously described. 13 Briefly, after six days of culture, the non-adherent cells (1 x 10 6 cells) obtained as described above were harvested, washed, and subcultured in fresh media (RPMI 1640 containing 10% heat-inactivated fetal calf serum) on 6-well culture plates containing glass coverslips. After 24 h of culture, these cells acquired a typical fusiform shape with pseudopodia projecting from both cell poles.
Bone Marrow Macrophages(Bmms)
Bone marrow-derived macrophages were generated from marrow cells obtained from BALB/c mice. Approximately 10 6 cells were cultivated in 6-well tissue culture plates containing square glass coverslips in each well for 7 days in RPMI 1640 media plus 15 mM of HEPES, 2 g/L sodium bicarbonate, and 1 mM L-glutamine and supplemented with 20% fetal bovine serum and 30% L929 cell-conditioned media, which contains macrophage colony-stimulating factor. 14 After 72 hours, the media was replaced by RPMI 1640 supplemented with 10% fetal bovine serum and 5% of the conditioned media derived from cultures of L929 cells. Cultures were kept at 36 °C in an incubator with 5% CO2.
Flow Cytometry
To identify the B-1 cells in our in vitro cultures, the cells were double-labeled with allophycocyanin (APC)-conjugated anti-Mac-11 (CD11b) and phycoerythrin (PE)-conjugated anti-IgM. All antibodies were obtained from BD Biosciences Pharmingen (San Diego, CA, USA). After staining, the cells were fixed with 1% paraformaldehyde. Analysis was performed with a FACSCalibur flow cytometer (Becton Dickinson, Mountain View, CA, USA). A total of 10,000 events were counted and the frequency of each cell surface marker was determined using Cell Quest software (Becton Dickinson).
Immunofluorescence and Confocal Laser Scanning microscopy
Cells were submitted to immunofluorescence with antibodies that allowed for identification of macrophages and B-1 cells. For in vitro experiments, adherent cells were maintained in culture on glass coverslips for one day, after which they were fixed with 3.7% formaldehyde for 30 min, washed three times with PBS and permeabilized with 0.5% Triton X-100 for 10 min. The cells were then treated with RNase for 30 min and incubated with anti-IgM-APC and anti-CD11b-FITC or anti-IgM-FITC and anti-CD11b-APC (BD Biosciences Pharmingen). After 2 h, the cells were washed three times with PBS and the nuclei, apoptotic bodies or E. coli were stained with propidium iodide. The cells were mounted onto glass slides with Vecta-Shield (Vector Laboratories, Burlingame, CA, USA). For in vivo experiments, cells were removed from the peritoneal cavity after the treatments and fixed with 3.7% formaldehyde for 30 min, washed with PBS, and spun down on poly-L-lysine-coated glass slides in a cytological centrifuge (3 x 10 4 cells/slide, 1,000 rpm for 2 min). The cells were then submitted to immunofluorescence as described above. The cell preparations were analyzed by confocal laser scanning microscopy (Zeiss LSM 510) with a 40X objective. Optical sections of 0.5–1 µm were made and orthogonal projections were used for analysis of phagocytosis.
Purification of B-1 Cells and macrophages
Murine B-1 cells were isolated from peritoneal cavity cell suspensions by magnetic cell sorting. The cells first underwent negative selection with anti-CD23 microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany), according to the manufacturer's instructions. Following the first selection, CD23” cells were positively selected with anti-CD19 microbeads (Miltenyi Biotec). A 97% pure B-1 cell suspension was obtained, as verified by flow cytometry. For purification of macrophages, the same method was used but with the anti-F4/80 antibody for positive selection (BD Biosciences Pharmingen).
Assessment of in Vitro phagocytosis
BDMP cells or macrophages were obtained as described above. The cells were plated in 6-well plates containing glass coverslips with 1 ml complete RPMI 1640 media in the presence of apoptotic thymocytes (1.0 x 10 7 ) or E. coli (1.5 x 10 7 colony-forming units). Phagocytic cells plus apoptotic thymocytes or bacteria were incubated for 1 h at 37 °C in an incubator with 5% CO2. The cells on the glass coverslips were then washed with PBS, fixed with 3.7% formaldehyde for 30 min and washed three times with PBS. The cells were then treated with Triton X-100 and RNase as described above, and actin filaments were stained with phalloidin-FITC. The cell nuclei, apoptotic bodies or bacteria were stained with propidium iodide. The preparations were observed by fluorescence microscopy (X81 inverted with a DSU spinning disk, Olympus). An average of 100 cells was counted on several microscopic fields to determine the percentage of BDMP cells or macrophages that internalized apoptotic thymocytes or bacteria, as well as the average number of particles per cell. The phagocytic index (PI) was calculated as previously described. 3 All experiments were done three times, in triplicate.
Assessment of in Vivo phagocytosis
Apoptotic thymocytes (1.0 x 10 7 ) or bacteria (1.5 x 10 7 colony-forming units) in 0.5 mL of PBS were injected into the peritoneal cavities of BALB/c mice. After 1 h, the peritoneal cells were collected by washing and submitted to purification by magnetic cell sorting as described above. Macrophage purification was performed by positive selection with anti-F4/80. Purified cells were labeled for confocal microscopy using the protocol described above. These experiments were performed twice, in triplicate.
Bactericidal Activity
To assess the intracellular killing capacity of B-1 lymphocytes, the E. coli strain k91 was injected into the peritoneal cavity as described above. After one hour, B-1 cells were purified by magnetic cell sorting, and 1 x 10 6 cells were lysed with 0.9 ml sterilized water. The lysates were seeded on LB agar plates and incubated at 37 °C for 24 h. Live bacteria were counted and the results were expressed as the number of colony-forming units (CFU). In each set of experiments, macrophages were also purified, and bacterial content was determined as described above. All experiments were done three times, in triplicate.
Statistics
Results are expressed as the mean ± SD. Data were analyzed by Student's t-test (INSTAT software, GraphPad, San Diego, CA). P < 0.05 indicated statistical significance.
Results
Phagocyte Characterization
B-1 cells are preferentially restricted to the pleural and peritoneal cavities in mice. 11 They correspond to the main B lymphocyte inhabitants of the peritoneal cavity (20%–30% of all cells and 80% of lymphocytes, CD19+CD11b+IgM+CD23–). Almeida and colleagues established in 2001 that non-adherent B-1 cells that exist in adherent peritoneal cell cultures are spherical, small and refringent cells. 6 When transferred to fresh culture media, those cells differentiate into fusiform mononuclear cells with phagocytic capability. When that occurs, the cells are named “B-1-derived mononuclear phagocytes” (BDMPs), as described by Ghosn and colleagues. 13 Here, free suspended B-1 cells were obtained from the supernatants of mouse peritoneal cell cultures. Concurrently to the expression of CD11b and IgM (Fig. 1A), these cells are also CD19+ and B220+ (data not shown). Figure 1A shows the flow cytometry analysis of B-1 cells generated from our in vitro experiments, with the great majority of cells expressing the CD11b and IgM markers. Twenty-four hours after collection and transfer to a new culture medium, a large proportion of these cells (>80%) adhered to the glass coverslips and spread as fusiform cells; we will refer to these cells as B-1-derived mononuclear phagocytes (BDMP) (Fig. 1B). The adhesion of B-1 cells occurred 24 hours after subculture, when there was reduction in the expression of B lymphocyte markers (IgM, CD19, B220), as shown in flow cytometry by Popi and et al 7 while myeloid marker (CD11b) remained unaltered. 7 Figure 1C shows the differentiation of B-1 cells into phagocytes that display fusiform and spread morphology and simultaneously express IgM and CD11b.
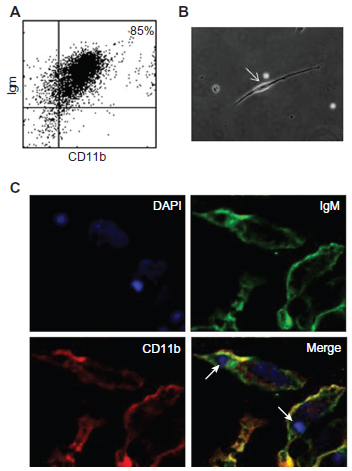
Characterization of the B-1-derived mononuclear phagocyte (BDMP) cells.
Phagocytosis of Apoptotic Cells and E. Coli by Bdmp Cells in vitro
As described above, B-1 cells were collected, subcultured, and induced to differentiate into phagocytes in vitro (creating BDMPs). Apoptotic thymocytes or Escherichia coli were added to these cultures or to macrophage cultures and the cells were allowed to interact for 1 h. Confocal microscopy analysis showed that the BDMP cells were able to ingest both apoptotic cells (Fig. 2A) and bacteria (Fig. 2B).
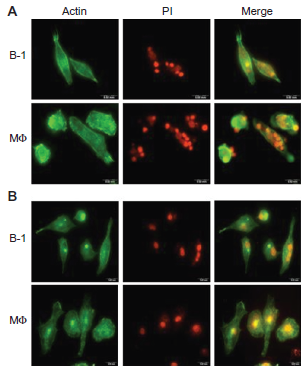
In vitro phagocytosis of apoptotic thymocytes and bacteria by BDMP cells. Images of BDMP cells and macrophages exposed to apoptotic thymocytes (
Phagocytosis of Apoptotic Cells and E. Coli in Vitro and in vivo
BDMP cells and macrophages were incubated with apoptotic thymocytes (1.0 x 10 7 ) or E. coli (1.5 x 10 7 colony-forming units) for 1 h. Figure 3 shows that both BDMP cells and macrophages have the ability to phagocytose apoptotic bodies and bacteria in vitro. The in vitro phagocytic index showed that B-1 cells and macrophages have similar phagocytic capacities for apoptotic thymocytes (Fig. 3A) and bacteria (Fig. 3B). There was no statistically significant difference in phagocytosis ability between B-1 cells and macrophages. The confocal analysis image in Figure 2 corroborates these data because it shows that the particles are within the cells. To assess in vivo phagocytosis, mice received an intraperitoneal (i.p.) injection of 1.5 x 10 7 murine apoptotic thymocytes or 1.5 x 10 7 colony-forming units of bacteria. After 1 h, peritoneal cells were harvested and confocal microscopy analyses were performed. Figure 4 shows that the B-1 cells were able to ingest apoptotic cells (Fig. 4A) and bacteria (Fig. 4C) in vivo. An orthogonal projection of the same image shows apoptotic bodies inside the B-1 cells (Fig. 4B). B-1 cells were identified by the concomitant expression of IgM (FITC) and CD11b (APC). Macrophages also performed phagocytosis after the injection of apoptotic bodies (data not shown). The in vivo phagocytic activity of B-1 cells was also measured by labeling the apoptotic bodies with propidium iodide prior to injection, and the cells from the peritoneal wash were analyzed by flow cytometry after staining with B-1 markers. Analysis by flow cytometry showed that nearly 30% of B-1 cells recovered from peritoneal cavity of mice had internalized propidium iodide-labeled apoptotic thymocytes after 1 h (data not shown). This indicates that B-1 cells might also play a role in the clearance of apoptotic bodies in vivo, along with other cells such as macrophages.
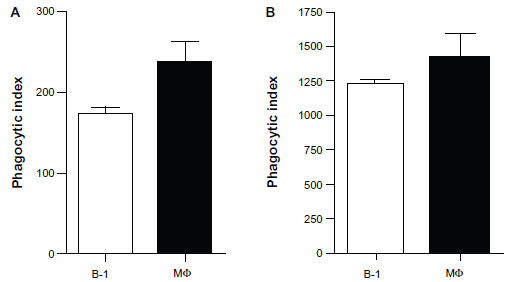
In vitro phagocytic indices of apoptotic thymocytes and bacteria.
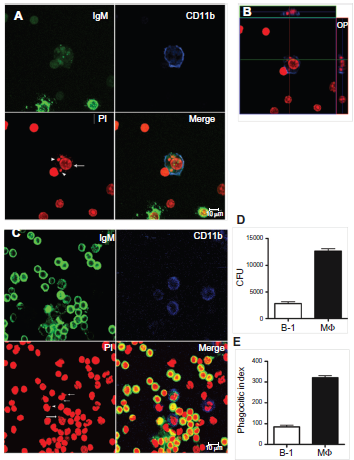
In vivo phagocytosis of apoptotic thymocytes by mouse B-1 and resident macrophage peritoneal cells.
We next investigated whether B-1 cells could internalize E. coli bacteria in vivo. For these experiments, mice were inoculated i.p. with 1.5 x 10 6 E. coli, and the cells from the peritoneal wash were analyzed by confocal microscopy, as described above. We found that B-1 cells and macrophages phagocytose bacteria in vivo 1 h after injection. In Figure. 4C, the uptake of bacteria by the B-1 cells is indicated by an arrow, while bacteria phagocytosed by macrophages are indicated by an arrowhead. B-1 cells displayed approximately one-fourth of the in vivo phagocytic capacity of macrophages, as shown by the data obtained in the CFU analysis (Fig. 4D) and the phagocytic index (Fig. 4E).
Discussion
Van Furth and Kohn in the 1968 demonstrated that monocytes are generated in the bone marrow of mice. 15 From there, monocytes differentiate from stem cells to pro-monocytes, then mature into monocytes in the medulla, migrate to the blood stream as monocytes and travel to different organs to become macrophages. 15 The demonstration of monocyte differentiation into several types of cellular macrophages, including Kupfer cells, microglia, osteoclasts, and peritoneal macrophages, supported the concept of a “Mononuclear Phagocyte System”. 16 This concept, however, is currently under debate. For example, it has been demonstrated in mouse embryos (7 dpc) that there exists a “primitive” macrophage population derived from the yolk sac that is not related to monocytes. These cells appear in the embryonic liver on day 11 (pc), when definitive hematopoiesis occurs.17,18 Studies have clearly demonstrated that PU.1 null mice have a lack of monocyte-derived macrophages, but they still have yolk sac-derived macrophages. Considering the fundamental role that the PU.1 transcription factor plays in macrophage differentiation, these data support the theory that fetal phagocytes have a separate lineage from macrophages that arise during definitive hematopoiesis.
In addition to the demonstration that two lineages of macrophages exist in the mouse during ontogeny, 19 cells with both myeloid and lymphoid characteristics and phagocytic abilities have also been described. For example, malignant CD5+ B cells can transform into cells with macrophage-like morphology that are capable of ingesting large particles by phagocytosis. 20 In addition, a developmental relationship between B-1 cells and macrophages has been established with the identification of biphenotypic B cell/macrophage progenitors in the fetal liver 9 and adult bone marrow. 10 The presence of a biphenotypic B/macrophage lineage precursor in mammals suggests a close evolutionary relationship between B cells and monocyte-derived macrophages, which indicates a possibility of a common phylogenetic predecessor with attributes of both cell types; in fact, this has been demonstrated in teleost fish. 20
Borrello and Phipps demonstrated that splenic B-1a cells became phagocytes when co-cultivated with fibroblasts, and they termed these cells “B/macrophages”. 12 These authors proposed that macrophages, defined by the concept of the mononuclear phagocyte system as cells derived from blood monocytes, could also originate from B-1 cells. Conversely, Almeida and colleagues clearly showed that B-1b cells proliferate when cultured with adherent mouse peritoneal cells and can become new mononuclear phagocytic cells with no relation to blood-derived monocytes. 6 Notably, whereas monocytes migrate from the bone marrow, B-1b cells migrate from the peritoneal cavity to non-specific inflammatory lesion sites. These findings, however, have been criticized based on the possibility that BDMP cells might be a contaminant in the cultures with monocyte-derived macrophages. This hypothesis has been refuted by a study by Popi and colleagues. 7 With the use of single-cell PCR, they showed that during differentiation, BDMP transcription factors related to the lymphoid lineage are decreased, but there is a concomitant persistence of myeloid gene expression; thus, BDMP cells continue their differentiation towards a myeloid commitment despite preserving immunoglobulin gene rearrangement.
In spite of the great volume of information regarding the origin,21,22 properties,23,24 and participation of B-1 cells in normal22,25,26 or pathologic conditions,27–29 the phagocytic ability of B-1 cells in vivo remains to be further established. Here we showed that B-1 cells can phagocytose both E. coli and thymocytes in vitro and in vivo. In addition to their phagocytic ability in vitro, the capacity of BDMP cells to kill microorganisms and the roles these cells play in the dynamic of inflammation and in the mechanisms of innate and acquired immunity remain largely unknown.
In this study, we demonstrated that B-1 cells also have the ability to phagocytose thymocyte apoptotic bodies in vivo, even though they are less effective than macrophages in this respect. It is intriguing that B-1 cells, after differentiation into BDMP cells in vitro, behave like macrophages, while the same ability to phagocytose is not seen in vivo. These findings merit further investigation. The phagocytic indices show that there are no statistically significant differences between BDMP cells and macrophages (Fig. 3A). Confocal microscopy analyses corroborate these data and indicate that engulfed particles are inside the cells and not adhering to the cell surface.
We further demonstrated that B-1 cells are capable of ingesting apoptotic bodies and E. coli bacteria, and our results show that BDMP cells have this ability in vitro, in a manner that is comparable to that of macrophages. Moreover, we further demonstrated that B-1 cells are capable of ingesting apoptotic bodies and E. coli bacteria. Results show that BDMP have this ability in vitro comparable to that of macrophages. However, the phagocytic ability of B-1 cells in vitro is increased regarding BDMP. The explanation for this phenomenon is still not clear but a hypothesis that may explains this fact is that B-1 cells in the peritoneal cavity are not yet differentiated into professional phagocytes. Popi and colleagues 7 demonstrated that the expression of lymphoid restricted transcription factors was decreased or abolished during B-1 cell differentiation. In that study their established that B-1 cells obtained in vitro express myeloid and lymphoid characteristics, although they have a predominant myeloid committed profile after differentiation into phagocytes. 7 Therefore, we believe the B-1 cells in the peritoneal cavity are not able to advance their differentiation status, posing the question as to whether B-1 cells are fully mature cells. Thereby, when B-1 cells interacts with antigens, such as apoptotic bodies or bacteria, in the peritoneal cavity we suggest that B-1 cells have no enough time to undergo complete differentiation in phagocytes in vivo. For this reason, the performance of B-1 cells in vivo turn these cells less phagocytic compared to in vitro assay. Moreover, Plytycz and Seljelid 30 proposed the hypothesis that B-1 cells are ‘living fossils’, suggesting that they are primordial cells in the phylogenesis of the haematopoietic system that resisted full differentiation into the distinct lineages of T, B and macrophage cells. Additionally, Alugupalli and colleagues demonstrated that mice deficient in both T and B cells, but not in B-1 and marginal zone B cells, cleared B. hermsii efficiently, suggesting that certain B cell subclasses such B-1 cells are sufficient to eliminate B. hermsii. 31 In contrast, Xid mice, which are severely deficient in B-1 cells, suffered significantly longer and more severe episodes of bacteremia compared with wild-type mice, suggesting that B-1 cells are likely to be the most critical immune cells for the clearance of B. hermsii. 31 In this study, we demonstrated that B-1 cells also phagocytose thymocyte apoptotic bodies in vivo, even though they are less efficient than macrophages in this respect. Our data suggest that B-1 cells may have evolved from phagocytic predecessors that are involved in the innate phase of immune responses in higher vertebrates. Evidence for this hypothesis lies in a study on teleost fish, one of the earliest groups to demonstrate adaptive immunity. 32 This study revealed a transitional period in B lymphocyte evolution, during which a subset of cells important for innate immunity and phagocytosis evolved into a highly specialized component of the adaptive arm of the immune response in higher vertebrates. In addition, it has been shown that a substantial proportion of Xenopus laevis B cells are phagocytic; 20 therefore, these studies also support the hypothesis that B cells evolved from a phagocytic predecessor cell type, an event that could help explain the functional and developmental relationships between B-1 cells and macrophages in mammals.
Overall, the data presented here validate the hypothesis that vertebrates have at least two professional mononuclear phagocyte systems–-one derived from monocytes and another derived from B-1 cells. These B-1 cells are reminiscent of the preceding evolutionary layer of the immune system, as proposed by Herzenberg. 11
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
We are grateful to Dr. Luiz C. Godoy of Department of Biological Engineering, Massachusetts Institute of Technology, Cambridge, MA, USA, for his insightful contributions for the manuscript. This work was supported by Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP).
