Abstract
Fungal diseases are major causes of morbidity and mortality among the immunocompromised, including HIV-infected individuals and patients with cancer. Individuals without a weakened immune system can also suffer from these infections. Not surprisingly, fungi are a major target for the immune system, rendered visible to it by expression of pathogen-associated molecular patterns/signatures. We now appreciate the roles of both innate and adaptive immunity in eliminating fungal infections, and how a disproportionate or inadequate immune response can diminish the host's capacity to eliminate fungi. This review focuses on our current understanding of the roles of innate and adaptive immunity in clearing common and emergent fungal pathogens. A clearer understanding of how the host's immune response tackles fungal infection may provide useful clues as to how we might develop new agents to treat those diseases in the future.
Introduction
Emerging evidence indicates that fungi are an increasingly important class of pathogens in plants and mammals. 1 The correlate between the incidence of fungal infection and clinical fungal-related disease has risen dramatically in the last two decades, which would suggest an increasing pool of susceptible, immunocompromised individuals. These could conceivably include individuals infected with human immunodeficiency virus (HIV), patients with a hematologic or solid cancer, and transplant recipients. 2
Multiple species of fungal pathogen have come to be associated with various human diseases, 3 against which the innate and adaptive immune responses are considered to be the principal defenses. 4 The mechanistic aspects of these immune responses (innate or adaptive) vary depending on the fungal species encountered, 5 the target organism, and the site of infection. Countering these, pathogenic fungi have developed multiple mechanisms to evade host immune defenses. Survival within phagocytes from where fungi can later disperse throughout their host is one particular elegant strategy.1,6 To maintain a stable host–fungi interaction, the immune response is segregated into an innate first-line defense, which is latterly strengthened by a second tier, adaptive response.
This review summarizes the two major types of host immune response to fungi, inclusive of the major cellular players including macrophages, neutrophils, dendritic cells (DCs), and T- and B-cells. It also addresses our current knowledge of the mechanistic aspects of each cell type's antifungal action. Finally, I briefly discuss insights gained from studies of immune cell function and how these may inform future vaccine strategies.
Innate Immunity and Fungal Infection
Neutrophils, macrophages, and DCs are all critical to the antifungal response. 7 Upon infection, these innate immune cells are rapidly recruited to sites of infection by virtue of their production of inflammatory cytokines, chemokines, and/or complement units.8–10 The release of inflammatory cytokines, as well as reactive oxygen intermediates and antimicrobial peptides, can then clear the fungi in target organs.
Neutrophils, a class of professional phagocyte, can also engulf and/or kill invading fungi. 11 One of its more recently discovered antifungal mechanisms is the neutrophil extracellular trap, which, together with an oxidative burst and the release of antimicrobials, constitutes a potent antifungal response. 12 Activated neutrophils release the cytokines IL-12 and IL-18 (Fig. 1) and mediate antifungal responses via their expression of receptors such as Toll-like receptor 4 (TLR4) and/or the complement receptors (eg, CR1). 13 For example, Candida albicans can induce reactive oxidative intermediates via their cell wall components although the necessity of the oxidative burst for Aspergillus conidia killing remains controversial.14,15 Clearly, there are gaps in our knowledge of how neutrophils and fungal pathogens interact; filling these may require new investigative methods.
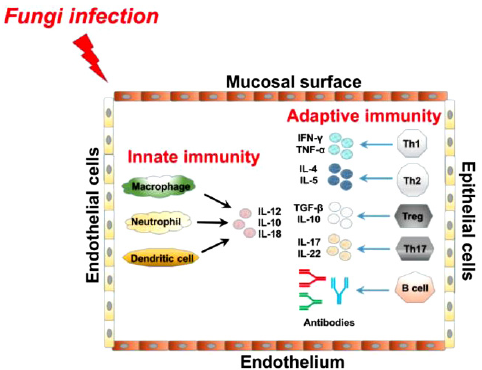
Immunity against fungal pathogens. Fungi usually infect their host via epithelial or endothelial cells, invading both the mucosal and endothelial surfaces. Cells in both the innate and adaptive wings of the immune system are activated by fungal infection, which subsequently generate different antifungal effectors. Upon initial fungal infection, innate immune cells (including macrophages, neutrophils, and DCs) release cytokines such as IL-12, IL-10, and IL-18. Cellular players in the adaptive immune response then secrete various cytokines against fungal infection: Th1 cells produce IFN-γ and TNF-α; Th2 cells produce IL-4 and IL-5; Th17 cells generate IL-17 and IL-22; and TReg cells produce TGF-β and IL-10. B-cells also secrete antibodies to target fungal pathogens.
Macrophages have long been appreciated for their role in balancing the effector cytokine species required for neutrophil recruitment and activation, as well as enhancing or inhibiting innate immunity. 16 For example, following infection by A. conidia, macrophages can produce pro-inflammatory cytokines, including TNF-α and IL-1β. 17 However, they can also stimulate IL-10 production, which is a typical anti-inflammatory cytokine (Fig. 1). 18 The strength of either response may be dialed up by TLR signaling, inclusive of TLR4 and TLR2, 19 which would suggest that different TLR signaling pathways contribute to dissimilar biological responses upon fungal infection. A. conidia can also induce NF-kB translocation in both the TLR2 and TLR4 signaling cascades. 20 Additionally, it was reported that human macrophage anti-C. albicans activity and the inhibition of phagolysosomal fusion in macrophages are both nonoxidative mechanisms. 21 It is worth noting that C. albicans may attempt to mitigate these effects by preventing macrophage proliferation. 22 The administration of corticosteroids suppresses the production of IL-1α, TNF-α, and MIP-1α in macrophages, all of which are protective against aspergillosis. 23
DCs are particularly potent players in the immune response as they initiate both innate and adaptive immune responses to various fungi including Cryptococcus neoformans, Aspergillus fumigatus, and C. albicans. 24 The signaling pathways triggered by DCs largely depend on the infectious agent. 25 DCs also capture and process antigens, express lymphocyte costimulatory molecules, and migrate to lymphoid organs and secrete cytokines to initiate immune responses including IL-12 and IL-10 (Fig. 1). Similar to macrophages, DCs can access the TLR system for antifungal host defense. 26 However, DCs largely mediate antifungal immunity by initiating and buffering T-cell responses. 27 Future research on DC targeting may prove to be fruitful in terms of generating potent vaccines against fungal pathogens.
NK cells show capacity against multiple types of fungi, including C. albicans and A. fumigatus in vitro.28,29 NK cells produce interferon gamma (IFN-γ) and play a fungicidal role in fighting against C. neoformans. 30 Furthermore, other innate immune cells including mast cells, basophils, and eosinophils contribute to the fungal protection as well.
Epithelial cells are also very important to the antifungal role. For instance, mucosal epithelial surface is the initial site of C. albicans for contacting with host, and epithelial cells upregulate TLR4 and subsequently protect against tissue damage caused by C. albicans. 31 Furthermore, endothelial cells can also interact with fungi, and the interaction between C. albicans and endothelial cells has been reported, 32 such as in blood vessels; however, its mechanism might involve complex processes, which would be potentially great for both bench work and clinical research in future.
Clearly, the innate immune response to fungi is important, but remains poorly understood. In particular, the regulatory mechanism at play may prove to be useful in improving our understanding of treatment options.
Adaptive Immunity and Fungal Infection
The role of adaptive immunity in the antifungal immune response is also well appreciated. 33 Immune-regulatory CD4+ T helper cells are of key importance, which can be functionally categorized as one of the five groups: Th1, Th2, Th9, Th17, and TReg cells.
Th1 cells can be activated by DCs via TLR signaling, activated in response to the recognition of immutable fungal molecules. 34 Th1 cells can then help to optimize the activation of phagocytes at sites of infection. 35 Th1 cells can also secrete signature pro-inflammatory cytokines such as IFN-γ and TNF-α (Fig. 1). Any diminished ability of Th1 cells to mediate inflammatory signaling to phagocytes (such as macrophages) may lead to the decline of the infected patient. Thus, modulating Th1 cells can boost the therapeutic efficacy of antifungal agents.
Th2 cells, activated by IL-4 and IL-13, generate cytokines including IL-5 (Fig. 1) that can limit the Th1 response, as well as activating M2 macrophages, which are harmful to patients with severe fungal infections and fungal-related allergic responses. 36 Hyperactivated Th2 cells have also been linked to cystic fibrosis. 37 As an indication of the complexity of these issues, Th2-associated antibody responses can also partially increase the Th1 cell response. 38
Th17 cells act principally at mucosal surfaces, including the lungs, where these cells play important roles in protective antifungal immunity. 39 Th17 cells produce IL-17 and IL-22 (Fig. 1) largely following their activation by signals transduced by the myeloid differentiation primary response 88 (MYD88) pathway (including the key signaling effector SYK-CARD9) and the mannose receptor pathways in DCs and macrophages. 40 Defective Th17 cells render patients susceptible to mucosal fungal infections, such as hyperimmunoglobulin E syndrome, which results from mutations in the stat1 gene in autosomal dominant mucocutaneous candidiasis. 41 Moreover, TReg cells generating anti-inflammatory cytokines including TGF-β and IL-10 (Fig. 1) have been described in fungal infections of both mice and humans. 42 In experimental fungal infections, TReg cells have been shown to regulate both inflammation and immune tolerance in the respiratory and/or gastrointestinal mucosa. Collectively, Th1, Th2, Th17, and ThReg cells are essential to the host's susceptibility or resistance to invasive fungal infections. Future research efforts will be required to determine whether other T-cell subsets (eg, Th9) also operate within this framework.
CD8+ T-cells can also block the fungal growth such as C. albicans in vitro. The observation that CD8+ T-cells can provide protection even without the presence of CD4+ T-cells against fungi infection such as Histoplasma capsulatum. Furthermore, CD8+ T-cells can produce IFNγ against Pneumocystis carinii infection and induce the clearance of the fungal infection. 43
Antibody generation by B-cells is critical for fungal clearance, such as clearing pneumocystis from the lung (Fig. 1). Moreover, titers for antibodies against pneumocystis have been measured in young individuals, similar to other pathogenic fungi such as C. albicans. 44 Emerging evidence also indicates that this organism is widely experienced in nature with antibody production, particularly IgGs and IgM, constituting part of the natural host response. 45 Many different analytic techniques have been used to examine pneumocystis preparations or antigens and identify pneumocystis-specific antibodies. The fine-tuning of adaptive immune responses during fungal infections would also be a useful tool for treating fungal-related human diseases.
Conclusion and Outlook
Given that the pool of immunocompromised individuals is rapidly expanding, there would appear to be an urgent need to develop novel, more potent antifungal drugs. Aspects of the innate and acquired immune response to fungi have been intensely investigated. Furthermore, novel analytic techniques capable of detecting immune responses elicited by fungi have proven to be a promising area of scientific research.
Future clinical therapies for invasive fungal infections may include drugs that enhance the antifungal activity of immune effectors. This option would be especially relevant for immunocompromised hosts in clinical trials, although the safety and efficacy of novel antifungals remains questionable. Another potential approach is the modulation of key signaling regulators. For example, findings that the innate and adaptive immune response to common fungal pathogens can be mediated by TLRs, MYD88, and/or noncoding RNAs, such as microRNAs, suggest that their manipulation might be a useful tool for the enhancement of antifungal resistance.
Clearly an improved understanding of the roles and mechanisms of the host–pathogen interaction may pay dividends in terms of our development of novel antifungal therapies, with more investment in this research area now needed to stimulate interest in solving current and future challenges posed by fungal disease.
Author Contributions
The author conceived, organized, drafted, reviewed, and approved the manuscript.
