Abstract
Since the introduction of a cytology-based screening program for cervical cancer in developed countries a dramatic reduction of specific morbidity and mortality has been reported. In addition to this in 1972 zur Hausen proposed HPV as a causal factor of cervical cancer. About 15 viral subtypes have been identified: the so called high-risk subtypes for cancer development. Despite this the clinical use of subtype identification in a prevention strategy has not substantially been reported. Simple identification of HPV carrier status cannot reliably be used as a prognostic factor for cervical cancer development nor as a useful alternative to cytological examination. Therefore we are establishing a link between the classical cytological examination and the techniques of molecular biology. Our strategy detects HPV in a semi-quantitative way as well as the E7-m-RNA activity and T-lymphocyte-transformation in order to better calculate how serious an infection is, how active the virus and how active the defense reaction is.
Background
In a conventional PCR diagnostic a single-strand DNA consisting of 15 to 25 base pairs is usually used as a primer. This gives an extremely high degree of specificity of the primer to the DNA under identification. 1 Only this DNA is discovered leaving others with even slightly differing DNA sections unidentified.
Using a so-called “degenerated primer”, this extreme specificity can be eased and other similar DNA sequences can be identified alongside the specific DNA under examination. Using the “degenerated primers” a single analysis is capable of identifying all HPV sub-types currently regarded as carcinogenic. If the identification of all carcinogenic sub-types in a single analysis is defined as a specific search, then the use of primers has the maximum possible level of specificity.2,3 Our approach does not differ from the current “gold standard” HPV analysis techniques.
What is new and in the view of the authors significant for the effectiveness of any HPV diagnostic is the parallel detection of human DNA in the same test. To make a general distinction between a “banal superficial contamination” and a “massive output of pathogenic substances”, the nowaday method uses a simple type of numerical analysis. An exact determination of the number of pathogens or a substance concentration is not so appropriate when screening for cancer using this diagnostic procedure.4,5 In order to have the same sort of information with the HPV infection6,7 we propose the amount of viral DNA in relation to the amount of human cellular DNA (derived from the amount of haemoglobin-DNA) as an indicator of the infection severity.
The density of human papilloma viruses depends heavily on the location from which the sample is taken. Regions with a relatively high quantity of the virus are directly adjacent to virus-free regions. For this reason, a semi-quantitative analysis (HPV-DNA quantity in proportion to the quantity of the human haemoglobin gene) is appropriate as a simple means of orientation.
The currently available HPV diagnostic methods do guarantee general evidence of HPV at the place of removal, but they cannot give reliable information on the true quantity of HPV. Thus evidence of HPV alone does not provide information on the actual degree of the infection.
In this way, a relatively small amount of material is required which would remain undetected in conventional analysis for HPV-DNA. In the case of a similarly negative human DNA analysis result, it is obvious that the sample contained very little or no material. An apparently inconspicuous, negative result in conventional HPV diagnostics is in fact only due to the material being unsuitable for analysis.
In this point, the strategy is superior to any other strategy currently known. With the additional detection of human DNA, the seriousness of the infection can be better ascertained than with simple HPV-DNA detection.
In the case of hepatitis B specific investigation of IgG and IgM antibodies can provide reliable information on the stage and activity of the viral infection (acute, chronic). Unfortunately, IgM and IgG antibodies proved to be of no value in establishing the stage and severity of HPV infection. The viral activity can be derived from the degree of expression of E6 and E7 genes. In order to ensure that a detected HPV infection is not a coincidental and irrelevant finding but that the virus is in fact engaged in cell degeneration, we also involved determination of the quantity of m-RNA of the oncogenes E6 and E78,9—in addition to the semi-quantitative determination of the amount of HPV-DNA—as an expression of an anticipated, incipient degeneration.10–12 No other contemporary diagnostic method addresses the key clinical question “How active is the virus?” using molecular biological methods.
Illustrates the importance of detecting both HPV-DNA and human DNA. The scheme shows cells with their nucleus (the blue lines indicate human DNA). The middle cell contains HPV-DNA in its upper section, packed in virus capsid, in the lower section of the cell there is unpacked HPV-DNA in the cytoplasma and shown in the cell nucleus. Only the simultaneous detection of HPV-DNA and human DNA can provide a clear image of the seriousness of the infection. Illustrates the importance of detecting E7-m-RNA. With the help of the cell, the virus produces its oncogenous m-RNA. Our working group was able to detect a region of the oncogene Onkogen E7 which is central to the malignant development of cells (shown in purple).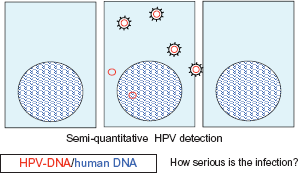
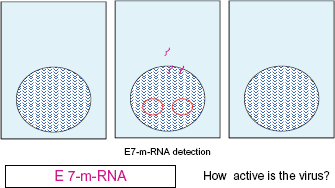
According to the current state of knowledge, the activation of the oncogenes E6 and E7 stands in direct causal connection with the degeneration of a cell. At a molecular biological level, it represents the initiating stage of the cancerous development of a cell. The detection of the activation of the gene is thus the first and most reliable indication of an anticipated cancerous development.
A number of contemporary institutions accept the importance of the body's defense situation, but none of them give the physician a parameter for determining the strength of the body's defense system.
For the selection of a simply determined parameter, there was a choice between the parameters from the interferon cascade, a gene from the tumor necrosis factor-cascade (TNF) or a gene which has been well known and studied for approximately 10 years and which was initially detected in degenerative and inflammatory brain diseases (Brain → Brn). 13 For reasons of practicability, the authors opted for the Brn 3a gene to determine the extent of the body defense system in order to reduce the number of unknown factors.
In 1998 it was observed that the gene Brn-3a is over-expressed in cells which show cervical intraepithelial neoplasia (CIN) lesions and it has recently been generally accepted that the gene Brn-3a plays a central role in the cellular defense mechanisms against malignancy.
14
Based on these facts we took the expression of gene Brn-3a as a measurement of the body defense mechanisms.
Illustrates the importance of detecting Brn 3a. The infected cell in the middle reacts to the infection and the expression of oncogenous m-RNA by producing m-RNA which serves the purpose of cell defense. An example of the activation of cell defense is the expression of the gene Brn 3a, which was known to the working group (shown in green). (In principle, the detection of TNF or interferon-m-RNA would also have been conceivable). This is the only way to adequately address the question: “How active is the defense?”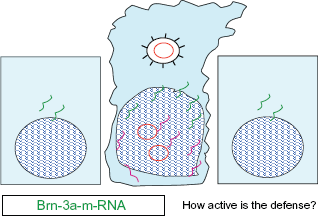
Conclusion
The most common way to obtain a biological sample for HPV tests is brushing the cervical (and the vaginal) surface tissue. It is known that regions of cells with no HPV particles can be found in the close neighborhood of focused epithelia with high concentration of HPV (similarly to skin warts caused with relative HPVs). Two subsequent sample obtaining actions can provide two samples with different numbers of cells with and without HPV infection.
Our diagnostic method described does not allow a more precise type of diagnosis and also only provides a distinction between positive (high-risk type) and negative (non-high-risk) type, with no further therapeutic use.
Therefore our criteria for a current diagnostic is:
The diagnostic method should also enable semi-quantitative detection. The activity of the oncogenous virus should be identified. The defense situation of the body should also be identified.
In our opinion a modern diagnostic has to cover:
15
The concentration of HPV-DNA in correlation to cellular genomic DNA in smear, in which HPV-DNA is detected. A semi-quantitative detection of E2, E6/E7 m-RNA in cervical smears, in which HPV-DNA is detected. Detection of cellular response activity. To look at the immune-defense status either semi-quantitative detections of the relation of BRN 3a/BRN 3b mRNA in cervical smears or (better) T-lymphocyte-transformation activity has to be examined.
Prospective Use
Attaching importance to both the cytological and the molecular biological findings could have consequences in clinical terms. The resulting examination strategy might take the following form, for example:
Patients with Pap I and Pap II with negative HPV screening should be examined at longer intervals, but especially when other sexual contacts have occurred within an existing partnership. Here, the focus should be on semi-quantitative HPV-DNA identification in addition to cytological diagnosis.
Patients in whom a Pap IIw or Pap IIID is detected for the first time should be routinely subjected to a Brn 3a E6/E7 diagnosis. If the E6/E7 diagnosis reveals high levels and the Brn 3a low levels, the HPV infection can be expected to be severe, even if the Pap IIw or Pap IIID findings themselves did not provide a basis for this expectation.
Patients with a Pap IVa or possibly a Pap IVb finding who show a high Brn 3a titer and a low E6/E7 finding could certainly remain under close monitoring or under antiviral therapy without having to take on an unnecessarily high risk.
Patients who have never had a Pap finding higher than Pap II and who have then been inoculated with an HPV vaccine would show inoculation success with the laboratory constellation “high HPV-DNA titer, high Brn 3a titer and low E6/E7 titer”. Patients who have been treated operatively or conservatively by means of an antiviral therapy could have their therapy monitored using EMBIVUD, thus enabling the success of the therapy to be shown.
A patient with a false low grade lesion can be identified in time to undertake the necessary treatment.
A patient with a high grade lesion but with good defending mechanisms may not need treatment.
HPV vaccines can make our proposed way extremely useful and reasonable.
Further Investigations
For a period we saw a changing in interpretation of the Brn-3a activity. It was not clear whether Brn3a is directly correlating with the cellular defense or it directly stimulates E6 and E7 oncogene expression. In 2006 it was reported that Brn-3a activates the expression of the human papilloma virus oncogenes. Brn-3a is expressed at high levels in high-grade cervical intra-epithelial neoplasia (CIN 3) compared to normal cervical biopsies.16,17 Brn-3a may contribute to tissue formation by binding to regulatory regions of HPV-16 and HPV18. The expression of their oncogenic E6 and E7 is regulated this way which can be seen in the T cell response.18–23 Therefore we decided to develop a T-Lymphocyte-transformation-test instead of Brn-3a-test to better assess cellular defense.
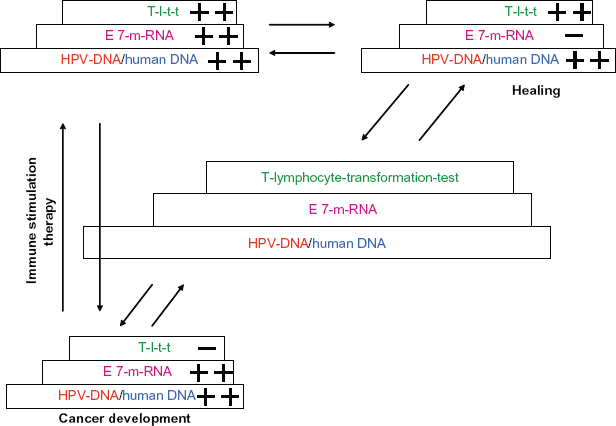
Illustrates our fundamental concept. Only the joint assessment of the individual examinations described in these diagnostic steps (proportion of HPV-DNA to human DNA, quantity of E7 m-RNA, and quantity of m-RNA of Brn-3a) allow a clear image of the seriousness of the HPV-infection. Explains the assessment of the severity of an HPV-induced cell change. Due to the varying parameters, the examination can provide clear information on the status of the HPV infection. The detection of a high quantity of HPV-DNA (with a comparatively low quantity of human DNA), with a high quantity of E7 m-RNA and a low quantity of T-lymphocyte-transformation-test indicates cancer development. The detection of equally high quantities of HPV-DNA (with a comparatively low quantity of human DNA), with a small quantity of E7 m-RNA and a high quantity of T-lymphocyte-transformation-test indicates a tissue which has gained control over the cancer development. Only this diagnostic method allows a clear therapy strategy to be observed using immune stimulation.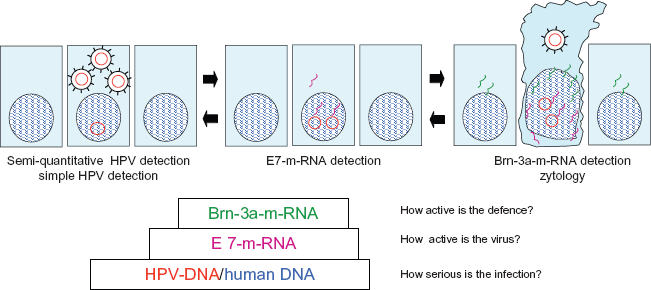
We hope our strategy will serve clinicians, particularly oncologic gynaecologists, in making determinations.
Footnotes
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
