Abstract
The vagina is a dynamic and finely tuned ecosystem in which homeostasis depends on mutually beneficial interactions between a human female and her resident microorganisms, an ecosystem that can be thrown off balance by a wide variety of both intrinsic and extrinsic factors. Although a functional equilibrium provides stability to the ecosystem considered crucial to maintaining vaginal health, “normal flora” is a concept currently being redefined. New methodologies enable molecular analyses of the vaginal microbiota which have widened the definition of “normal” from a single specific microbiological profile to a range of functional microbial equilibria dependent upon pertinent host and microbial factors. One of the strongest influences on the vaginal microbiota is the hormonal changes that define the reproductive phases of a woman's life. The vaginal environment is particularly responsive to estrogen, a hormone that creates distinctive changes in the vaginal microbiota. This review summarizes the components of a healthy vaginal ecosystem during the reproductive years, including the characteristics of a healthy equilibrium and factors that can disturb a functional balance. It also summarizes what is known about the vaginal microbiota in childhood and after menopause. Healthful ecosystems at any stage of a female's reproductive life will be characterized by a microbiota that both maintains physiological function and though changeable, adapts to normal perturbation without succumbing to disease.
Keywords
Introduction
The vaginal vault is colonized within 24 hours of a female child's birth and remains colonized until death, 1 comprising an ever-changing yet fine-tuned ecosystem with numerous factors (both internal and external) that have the potential to disrupt the ecosystem's fragile balance. 2 Over a woman's lifespan—with variations in endogenous hormone levels, sexual activity, hygiene practices, and other potential influences—the community of bacteria that inhabit the vagina will be in constant flux. 2 The concept of “normal flora” as a static and well-defined microbial population therefore is in need of revision, particularly as better information (including that gained through recently developed molecular methodologies that are not dependent on culture) is changing the current paradigm. It is now recognized that a spectrum of microbial profiles can produce a stable vaginal ecosystem with the ability to maintain vaginal health without succumbing to disease.
This review will follow the changes in the “normal flora” over a woman's lifespan from birth through menopause, including factors that may influence the microbiota, the role that the vaginal ecosystem plays in the maintenance of vaginal health, and how recently developed molecular methodologies contribute to an updated definition of a healthy vaginal vault.
The Vaginal Ecosystem in the Reproductive Years
The vaginal ecosystem is primarily driven by the dramatic hormonal changes which mark the reproductive stages of a woman's life—puberty, menarche, fertility, pregnancy, and menopause. 3 During a woman's reproductive years, the fluctuating levels of hormones which regulate the menstrual cycle are an important influence on the vaginal microbiota. 3 There are multiple other potential influences in this stage of her life as well, such as choice of contraception, 2 use of personal hygiene products or medications, 4 the presence of sexually transmitted diseases, 5 as well as numerous factors related to sexual behaviors (age at which sexual activity begins, frequency of sex, number of sexual partners, specific sexual practices engaged in, and even the introduction of semen). 4
The consensus of prior culture-based studies is that lactobacilli form the numerically dominant group of organisms in the “normal flora” of the adult women in their reproductive years.3,6 Colonization of the infant vagina with lactobacilli begins during the birth process, with organisms transferred from the mother's own vagina and flourishing in the infant as a result of residual maternal estrogens. 7 At puberty the baseline level of estrogen rises, and cyclic changes in circulating estrogen levels begin. 8
Rising estrogen levels stimulate the proliferation of vaginal epithelial cells, with a mid-cycle peak in intracellular glycogen levels in the vaginal mucosa and a subsequent increase in lactic-acid producing microbes in the vaginal milieu. 3 Glycogen is metabolized by the lactobacilli present, its breakdown resulting in an increase in lactic acids and thereby lowering vaginal pH to 4.0–4.5. This acidity level appears to inhibit colonization by numerous other bacterial species. 9
By late adolescence the morphology of the vulva is mature and the menstrual cycle has become well established. Lactobacillus spp have been observed at higher levels at the stages of life when estrogen levels are highest, 10 and lactic-acid producing microbes are numerically dominant in the reproductive woman 6 at approximately 10 7 lactobacilli per gram of vaginal secretion. 10 A dominance of lactobacilli in the vaginal microbiota has been considered the defining factor of a healthy vaginal equilibrium, so much so that the Nugent system used to diagnosis bacterial vaginosis is based on the quantitation of lactobacilli in a vaginal smear. 11
Microbial Studies in Reproductive Women
Although lactobacilli are the dominant strain in most healthy women of reproductive age, only a single strain of Lactobacillus is typically cultured from any one individual; 12 Antonio reported that 92% of cultured individuals had a single strain. 13 The most common Lactobacillus species reported by traditional culture techniques are L. crispatus, L. acidophilus, and L. fermentum, with L. brevis, L. jensenii, L. casei, L. delbrueckii, and L. salivarius also isolated. 10
Lactobacillus species have been observed to inhibit in vitro growth of numerous other bacterial species. 10 The Gram-stain method of Nugent has traditionally specified Lactobacillus-dominant microbiota as a healthy vaginal ecosystem. 11
The mechanism by which lactobacilli stabilize the vaginal microbiota and keep pathogenic microorganisms in check has been theorized to be either maintenance of a low pH or the production of hydrogen peroxide.10,14 Hillier et al 15 demonstrated a significant correlation between the presence of hydrogen-peroxide producing lactobacilli and inhibition of colonization of the vagina by potentially pathogenic competitors. 15 H2O2-producing lactobacilli were also observed to have viricidal activity. 14 It has been observed, however, that up to 42% of women who do not demonstrate a lactobacilli-dominant vaginal population are nonetheless able to maintain functional vaginal ecosystems. 2
Other bacteria commonly identified in the microbiota by traditional methods include Staphylococcus species, Ureaplasma, Corynebacterium, Streptococcus, Peptostreptococcus, Gardnerella, Bacteroides, Mycoplasma, Enterococcus, Escherichia, Veillonella, Bifidobacterium, and Candida.10,16 Prevalence rates for common organisms are displayed in Table 1.
A comparison of prevalence rates for common organisms reported in normal flora in the literature: traditional (gram-stain and culture-based) methods vs. molecular in adult premenopausal women.
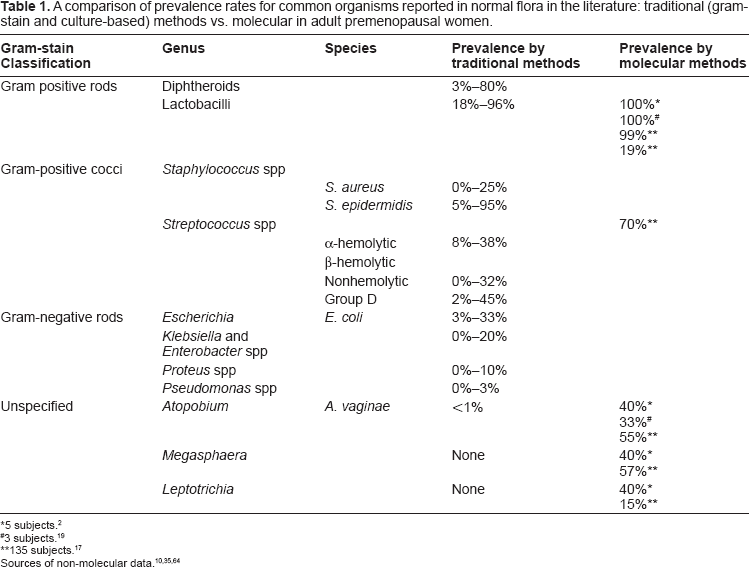
5 subjects. 2
3 subjects. 19
135 subjects. 17
Recent application of molecular techniques in the evaluation of the vaginal microbiota in healthy women of reproductive age has added depth to the existing body of literature. Using polymerase chain reaction (PCR) amplification of the 16 subunit of ribosomal ribonucleic acid (16S rRNA), deoxyribonucleic acid (DNA) segments were sequenced from vaginal samples of five healthy women. Data obtained were consistent with previous studies in the finding, that four of the five (80%) women had lactobacilli-dominated ecosystems. 2 Although two of these women were colonized solely with L. crispatus, formerly recognized as one of the most common species, 13 the other two were found to be colonized only with L. iners, an organism that does not grow on standard media used for Lactobacillus isolation. 2 The fifth woman, through molecular analysis, was demonstrated to have a microbial community dominated by Atopobium, found in one woman with lactobacilli-dominated flora as well. Two of the women also had high levels of both Megasphaera species and Leptotrichia species. None of the novel organisms identified are readily culturable and had not been previously identified as numerically significant organisms; in fact, Megasphaera had not previously been identified in the vaginal environment. 2
Maintenance of vaginal acidity via production of lactic acid and other organic acids by the microbiota is widely believed to be critical to the maintenance of vaginal health. Interestingly, Atopobium, Megasphaera, and Leptotrichia are all known to be producers of lactic acid. 2 Thus, the finding that other lactic-producing organisms apparently anchor functional ecosystems indicates that while maintaining a community of lactic-acid producers remains critical to vaginal homeostasis, the composition of that community can vary within a bacterial population. 2
A second expanded molecular study in which Zhou et al 17 used 16s rRNA techniques to evaluate the vaginal microbiota in 144 women of reproductive age confirmed their earlier findings. 17 The investigators found eight distinct patterns among the study populations. Five of the eight groups were dominated by Lactobacillus species, with L. iners, recovered in 66% of all women sampled, the most common species identified. Also recovered were L. crispatus, L. jensenii, and L. gasseri.
One of the eight groups was dominated by Atopobium with high numbers of various members of the order Clostridiales (including Megasphaera), one group was mostly comprised of Streptococcus species with a low level of Lactobacillus, and the last was predominantly Clostridiales (particularly Lachnospiraceae, not related to any previously described organism) with low levels of Atopobium, Micromonas, Mobiluncus mulieris, and Peptoniphilus. Six of the groups were, again, dominated by lactic-acid producing bacteria; the other two groups were dominated by organisms whose physiologies are not yet well understood. Maintenance of an acidic environment in the vaginal vault, however, was well-conserved. 17 This study confirmed that Atopobium is common in healthy, fertile women. In addition, it demonstrated that a high percentage of healthy women host fastidious anaerobes of the order Clostridiales which have not been recovered through traditional cultivation. 17
An additional 16s rRNA molecular study by Burton et al 18 using PCR followed by denaturing gradient gel electrophoresis also found L. iners to be the most common species of Lactobacillus identified in healthy young women, found in 42% of the 19 subjects analyzed. 18 Organisms not typically found in the vagina were identified in three subjects (Arthrobacter, Caulobacter, and Butyrivibrio), suggesting that environmental exposures such as swimming pools and soil may influence the vaginal population. 18
In yet another study using 16S rRNA to identify cultured isolates, Verhelst et al 19 also identified Lactobacillus as the dominate organism in healthy vaginal environments, with L. crispatus and L. gasseri the most dominant. 19 It is probable that the old specification of L. acidophilus, considered by traditional techniques to be the most common Lactobacillus species, was actually a lumping together of L. crispatus, L. gasseri and L. iners, which the newer molecular techniques now make possible to differentiate. 19
A summary of the findings on the specific organisms identified in the microbiota in women of reproductive age is also displayed in Table 1.
A healthy Vaginal Equilibrium as a Barrier to Disease
A healthy host-vaginal microbiome is an ecosystem in which a functional equilibrium is established through reciprocal and mutually beneficial interactions between the host and her resident microorganisms. This healthy equilibrium acts to provide a barrier to both new colonization by pathogenic organisms and overgrowth of organisms that are otherwise commensal. 2 It is known that disturbances in the vaginal microbiota can be a prelude to the development of bacterial vaginosis (BV) and other disease states. 2 BV, in turn, is believed to be a factor in the development of STDs, 4 pre-term birth,4,21 pelvic inflammatory disease, 4 and infertility. 20
A functional equilibrium can also inhibit the transmission of sexually transmitted diseases (STDs). 4 Research has also implicated disturbances of the vaginal microbiota with an increased risk of human immunodeficiency virus (HIV) infection; more recent research has shown, in addition, that a “normal” vaginal microbiota (as defined by Nugent score) lowers the probability of heterosexual transmission of HIV. 22 A disrupted microbiota has been associated with a variety of undesirable clinical events, including adverse pregnancy outcomes, post gynecology (GYN) surgical infections, and pelvic inflammatory disease. 4
In light of the fact that the incidence of male-to-female transmission with just a single, coital act is 50% for gonorrhea, 40% for chlamydia, and 30% for trichomoniasis, it has been considered critical for sexually active women to maintain “normal flora” in the vaginal vault. Although in gonorrhea, chlamydia, syphilis, and trichomoniasis disruption of the vaginal ecosystem has clearly occurred (a pathogenic organism is introduced into an otherwise healthy ecosystem), in general the metamorphosis of a stable equilibrium into a disease state is not yet well understood. 4 The introduction of a known pathogen does not always result in disease,4,23 while organisms which are habitual constituents of the vaginal microbiota (i.e. C. Albicans, G. vaginalis, E. coli) can become pathogenic when the equilibrium is disturbed. 10 The chain of events which tips the scales from a functional equilibrium to the development of BV is still cloudy,10,23 but apparently involves alteration of existing equilibrium that allows a particular organism (invading or commensal) to achieve replicative dominance. Functional pathogenesis depends on the relative numbers and virulence of the specific organism, in concert with the composition of the existing microbiota as well as the lower genital tract immune system of the host.4,10,24
Factors that Influence Microbiota in Women of Reproductive Age
Effect of Extrinsic Factors
Numerous studies have evaluated the potential of specific factors to upset the delicate balance of the vaginal ecosystem. Longitudinal studies have found that women experience transient changes in the microbiota over time, and that those changes are dramatic enough to shift a functional equilibrium into a disrupted and potentially pathogenic state.5,25–27 Diagnostic evaluation of the microbiota has been traditionally performed by Gram stain followed by scoring of the microbiota via the method of Nugent, which assigns scores according to the number of specific bacterial morphotypes seen per microscopic 1000X field. Scores of 0–3 have been defined as normal flora, scores of 7–10 have been defined as BV, while a score of 4–6 has been defined as a transitional intermediate state. 11
Recent research has revealed that the microbiota of the woman of reproductive age undergoes continual transformation. In a study of 51 healthy women, Schwebke et al, 26 using gram-stain and culture methodology, investigated the effect of the number of sexual partners, sexual practices, and the use of vaginal medication or spermicides on the stability of the vaginal microbiota. While many things were associated with day-to-day fluctuation in the microbiota, only the number of partners and frequency of receptive oral sex were associated with a shift of a functional equilibrium into a disrupted or diseased state. 26 Only 22% of the women in that study maintained a Nugent-defined normal flora throughout the entire study period. 26 Keane et al 25 assessed the effect of specific sexual practices; the use of spermicide, lubricant, or contraception; and the effect of personal hygiene practices. None of these factors demonstrated a significant effect on the microbiota as follows: 48% of the women in this study maintained a Nugent-defined normal flora throughout the one cycle, 19% had intermediate flora throughout, and 33% had normal flora initially that became abnormal over the course of the study, but without clinical symptoms. Ten percent had normal flora that progressed to BV. 25 A study by Eschenbach et al 14 also found no influence of oral contraceptives on the vaginal microbiota. 14
Interestingly, Zhou et al 17 concluded that race might play a genuine genetic role in the composition of the vaginal microbial population. In a study that evaluated microbial difference in 144 healthy women using molecular techniques, analysis of data by race revealed dramatic differences in the types of vaginal communities identified in whites versus blacks. 17 The incidence of BV has also been demonstrated to vary markedly across races, with a 6% prevalence in Asians, 9% in whites, 6% in Hispanics, and 23% in blacks, differences which cannot be explained by other variables such as sociodemographics or sexual behaviors alone.28–30
Effect of Menses
The strongest influence on the stability of the vaginal ecosystem has been observed to be menstruation; deviation from Nugent-defined normal flora was found to occur in the first 9 days of the cycle in 71% of subjects. 25 Earlier investigations into the role of the menstrual cycle on the microbiota had also identified cyclical changes. Barlett et al 31 found a 100-fold decrease in the numbers of aerobes in the week preceding menses as compared to numbers during the menstrual flow. 31 Sautter et al 32 found similarly that the numbers of specific species fluctuated over the cycle, while the species isolated from an individual remained constant. 32 In a more extensive study, Johnson et al 33 observed that the total number of bacteria did not change over the course of the menstrual cycle, but that a greater variety of organisms was present during menses than over the rest of the cycle. 33 Wilks and Tabaqchali also found no alterations in the total numbers of organisms over the cycle, although the mean number per species declined from 4.6 in week 1 to 2.9 in week 4. No particular organism was selectively depleted. 34
In a more recent study, Eschenbach et al 6 evaluated the influence of the phase of the menstrual cycle on the microbiota and found that although Lactobacillus prevalence did not change dramatically, growth of non-Lactobacillus species increased during the menstrual flow, with heavy growth of non-Lactobacillus species occurring in 72% of subjects during the menstrual flow as compared to 40% from days 19–24 (p = 0.002). 6 The rate of recovery of heavy growth of lactobacilli increased over the menstrual cycle. 6 Numerous studies have evaluated the influence of choice of menstrual protection on the vaginal microbiota35–37 and have determined that menstrual products have no significant effect on the normal changes that occur during menses. 38
Effect of Pregnancy
In the pregnant woman, levels of estrogen, mainly in the form of estriol, rise steadily over the course of pregnancy, with urine levels rising from a low in the first trimester of around 4 μg/24 hours to a high in the third trimester of up to 50,000 μg/24 hours. 39 Estrogen stimulates deposition of glycogen in vaginal epithelial tissue, which is metabolized to glucose within vaginal epithelium and then converted to lactic acid by cellular metabolism. Recent data on changes in the vaginal microbiota during pregnancy are sparse and are meaningless unless the time of sampling is specified. One recent study, using molecular techniques, looked at vaginal smears from a total of 200 healthy women with Nugent-defined normal flora late in the first trimester of their pregnancies and found that 57% had recoverable lactobacilli, with L. crispatus and L. gasseri the most common, followed by L. jensenii and L. rhamnosus. 40
The Vaginal Ecosystem in Childhood and after Menopause
A review of the current literature can be broken into the following phases of a woman's lifetime: birth and early childhood, puberty, reproductive years (which include menses and pregnancy), and menopause.
Birth and Early Childhood
During the birth process and shortly after, the vaginal epithelium is high in glycogen content as the result of residual maternal estrogens, providing an environment in which lactic acid-producing microbes can thrive. 1 Vaginal pH during early childhood is neutral or slightly alkaline. 3 Due to the absence of adrenal or gonadal action, as maternal estrogen depletes, the prevalence of lactic-acid producing microbes present in neonates also decreases. 3
Analyses of the resident microbiota in children have been few. Hammerschlag et al, 41 in an ambitious study, looked at organisms present in the vaginal ecosystems of 100 girls, aged 2–15, by traditional methods and found high rates of colonization of diphtheroids (78%), Staphylococcus epidermidis (73%), α-hemolytic streptococci (39%), Escherichia coli (34%), and the mycoplasma Ureaplasma urealyticum (UU). 41 Lower rates of prevalence were found with Corynebacterium vaginale (13.5%), Candida species (28%), Klebsiella (15%), Group D streptococci (8.5%), S. aureus (7%), Haemophilus influenzae (5%), Pseudomonas aeruginosa (5%), Proteus (5%), and Acinetobacter species (3%). 41
Lactobacilli were isolated most frequently from older girls (a 45% prevalence in those under 2 years had increased to 88% by age 11); while enteric organisms were isolated most frequently from younger girls (E. coli was found in 90% of infants). Corynebacterium was more prevalent in children under 2 or over 10 years of age (18% and 63% respectively). 41
Mycoplasmas tended to be found together; all six children colonized with M. hominis were also colonized with U. urealyticum. 41 M. hominis was more prevalent in those over 10 years of age (none was found in children under 2 years), while the prevalence of U. urealyticum was similar in all age groups. C. albicans and C. tropicalis were the most common yeast species identified and were more prevalent in infants and teenagers (post pubertal) than in older children (3 to 10 years). C. tropicalis was isolated only from infants. 41 Neisseria gonorrhoeae, Chlamydia trachomatis, and Trichomonas vaginalis were only rarely identified. Although the presence of N. gonorrhoeae and Chlamydia are considered conclusive evidence of abuse in children, this study did not address the issue of sexual transmission of any organisms. In addition, the analysis of the data is difficult since both premenarchal and postmenarchal girls were included, and the described lack of sexual activity in some girls was not specified as virginal. 41
A second study by Hammerschlag et al, and similarly using traditional methods, 42 looked specifically at anaerobic bacteria in the vagina of 25 girls (aged 2 months to 15 years). 42 The most prevalent organism was S. epidermidis (84%). Diphtheroids, α-hemolytic streptococci, E. coli, Klebsiella, P. aeruginosa, C. vaginale, H. influenzae, and S. aureus were also identified. All but one of the subjects were colonized by anaerobic streptococci. 42
A more recent study by Jaquiery et al 43 evaluated the microbiota in girls across income groups (Hammerschlag et al had looked only at low-income children) who underwent surgery at a children's hospital. Jaquiery (looking only at premenarchal girls) found, like Hammerschlag et al, that anaerobes, diphtheroids, coagulase-negative staphylococci, and E. coli were very common. 43 Jaquiery et al, who solicited information about prior sexual abuse and excluded those subjects, did not find Gardnerella vaginalis, T. hominis, genital Mycoplasmas, or N. gonorrhoeae in any culture. 43 C. trachomatis was also negative for all specimens. 43 A complete list of isolates found in the vaginal microbiota of children is found in Tables 2 through 4.
Aerobic and facultatively anaerobic species identified in the vaginal cultures of girls under 16 years of age.
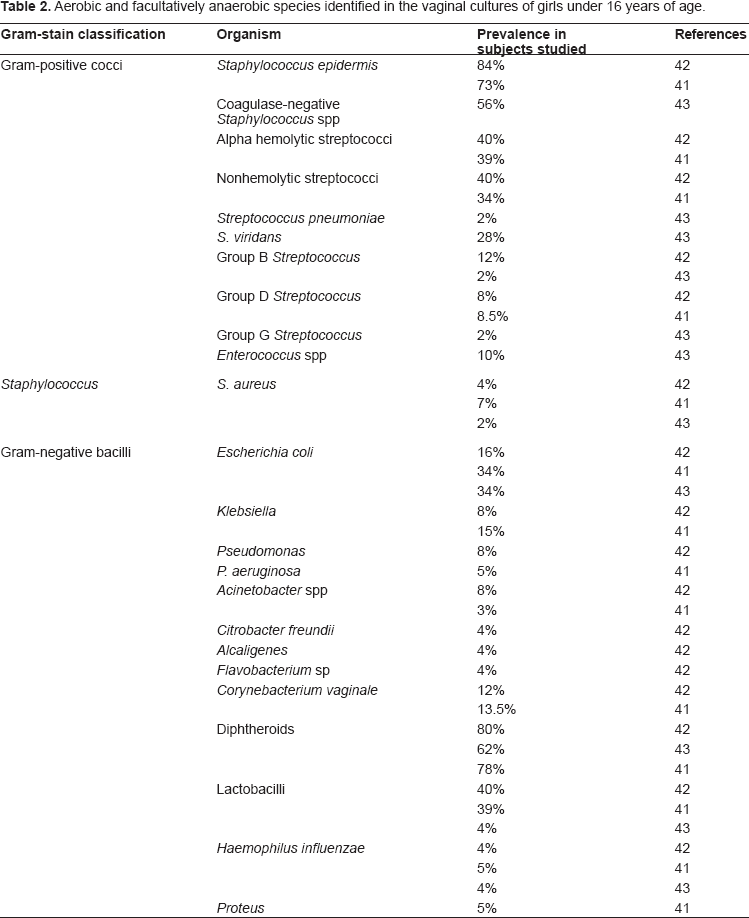
Obligate anaerobe species isolated from vaginal cultures in girls under 16 years of age.
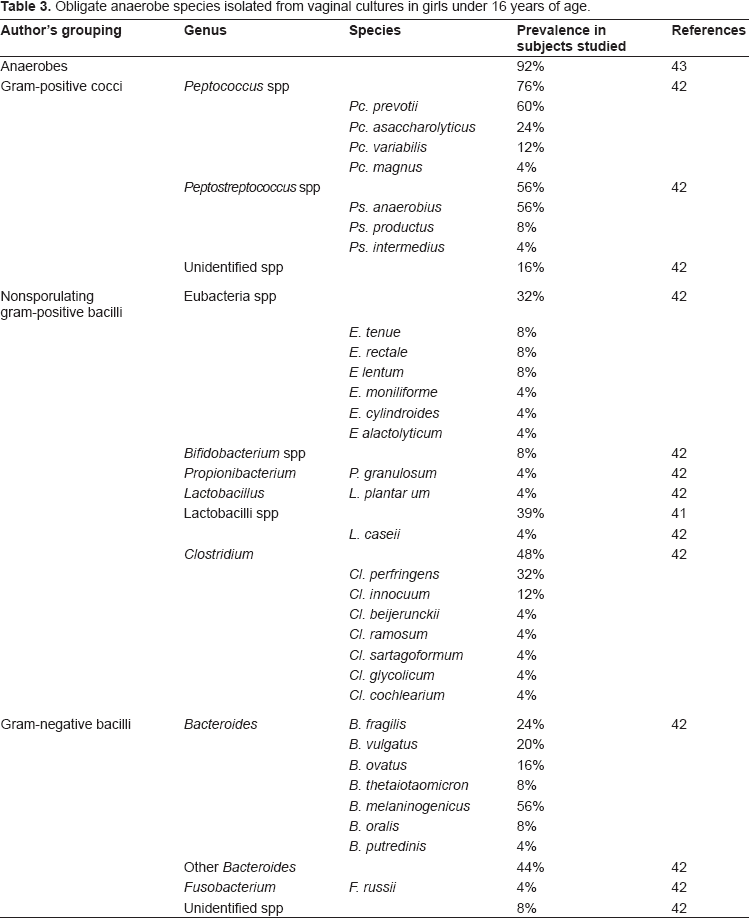
Atypical components of the vaginal microbiota in girls under 16 years of age.

Puberty Period
Both the morphology and physiology of the vulva and vagina change at puberty. 44 With adrenal and gonadal maturation, cyclic hormonal patterns are established and menstruation begins. Mid-cycle estrogen levels produce peaks in the glycogen content of the vaginal epithelium, which increases the prevalence of lactic-acid producing microbes in the microbiota. 3
The vaginal microbiota was assessed in 171 girls (aged 13 to 21) by Shafer et al, also by traditional methods. 45 Lactobacillus species were isolated from nearly 70% of all subjects, with sexually active girls almost twice as likely to carry lactobacilli than non-sexually active girls. C. trachomatis, N. gonorrhoeae, and T. vaginalis were isolated only from sexually active adolescents; sexually active subjects had a mean of 6.05 organisms/subject, while non-active subjects had an average of 3.12 organisms apiece. 45 Active subjects had higher rates of G. vaginalis, lactobacilli, Mycoplasma species, and U. urealyticum than non-active participants.
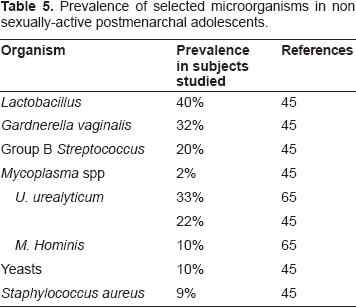
Findings for teens who were not sexually active were similar to that reported by Hammerschlag et al for female children, while those for the sexually active group were more similar to results reported for adult women. 45 Gardnerella was recovered from non-sexually active adolescents, but the prevalence nearly doubled with sexually activity. 45 Yeast was also recovered from a significant percentage (10%) of non-sexually active girls. In this study, Mycoplasmas were not found in adolescents who were not sexually active, confirming an early study which found a linear relationship between the presence of Mycoplasma in the vaginal microbiota and sexual activity. 46 Results of that analysis are displayed in Table 5.
Prevalence of selected microorganisms in non sexually-active postmenarchal adolescents.
Menopausal and Postmenopausal Years
Menopause is marked by a dramatic reduction in estrogen production, resulting in drying and atrophy of the vaginal epithelium. 47 When estrogen levels drop, glycogen content in the vaginal epithelium drops as well, leading to depletion of lactobacilli. Falling numbers of lactobacilli result in a subsequent rise in vaginal pH, since glucose is not converted to lactic acid. 48 High pH promotes growth of pathogenic bacteria, particularly colonization by enteric bacteria. 3 Overall, the makeup of the vaginal microbiota will depend upon duration, rate, and severity of estrogen deficiency.
In an assessment of vaginal health in 921 women by Gram-stained smears, only 46.3% of postmenopausal women had Lactobacillus-dominated populations. 49 Hillier et al 50 evaluated 76 healthy postmenopausal women without any hormonal intervention 50 Lactobacilli were recovered from 49% of the women. Although nearly half of the postmenopausal women retained colonization with lactobacilli, it was observed that concentrations of facultative lactobacilli were 10 to 100 times lower than those observed in women of reproductive age. Additional organisms isolated in substantial numbers were G. vaginalis (27%), U. urealyticum (13%), Prevotella (33%), and coliforms (41%). 50
Gupta et al 48 studied 80 menopausal women with and without the use of hormone replacement therapy (HRT) by traditional culture-based methods. 48 Vaginal pH was significantly lower in users as compared to nonusers as follows: 80% of nonusers had a vaginal pH higher than 4.5, while 87% of users had vaginal pH lower than or equal to 4.5. 48 Estrogen replacement was shown to restore pH levels to those found in fertile women. 48
There was also a dramatic difference between the groups in terms of the percentage of lactobacilli-dominant populations; 95% of users had dominant lactobacilli, while 80% of nonusers had microbial populations dominated by other species. 48 Lactobacillus and Gardnerella were frequently isolated among users of HRT. Lactobacilli spp, however, were found in only 20% of nonusers of HRT, while Bacteroides, and E. coli were isolated from 40% and 35%, respectively, of those subjects. 48
Pabich et al 51 also evaluated HRT influence on the vaginal microbiota in 463 community-dwelling postmenopausal women with urinary tract infection (UTI) by culture methods and found that 83% of women on HRT had lactobacilli present in the vaginal vault, with 65% of women on HRT found to have high numbers of resident lactobacilli. 51 The rate of colonization with E. coli was inversely associated with the presence of lactobacilli in postmenopausal women, with heavy growth of lactobacilli associated with a lower frequency of E. coli colonization of the vagina. 51
In a molecular study using denaturing gradient gel-electrophoresis (DGGE) analysis, Heinemann et al 52 found that lactobacilli were recovered from the vagina of at least 95% of women in both the HRT and untreated groups. 52
In 87% of women using HRT, less than three species were identified in the vaginal population. Most subjects had a single organism, usually (90% of cases) a Lactobacillus, with L. iners, L. crispatus, L. gasseri, and L. jensenii the most common. Ninety-one percent of non-users were colonized by more than one organism, often high numbers of organisms associated with the development of BV (Bacteroides, Prevotella, and Gardnerella) or UTI (E. coli and Enterococcus). Bacteroides, E. coli, and Prevotella, in fact, were found at levels ten times higher than in women who were being treated with HRT. 52
Despite the presence of potentially pathogenic organisms in HRT users, it was observed that in HRT-treated women, the same Lactobacillus species were recovered at each sampling time, conferring substantial stability on the vaginal environment. 52 In HRT-treated women lactobacilli were more often the dominant (and only) colonizer and fewer potentially pathogenic bacteria were found. The incidence of clinical bacterial vaginosis was six times higher in non-HRT treated women than in those using replacement therapy (5.6% vs 31%). 52 Only 20% of non-HRT users had Nugent-defined normal flora throughout the entire period. 52
Devillard et al 9 also using molecular methods, evaluated the vaginal microbiota of 19 women on HRT. 9 Sixty-eight percent had normal flora by Nugent scoring (a percentage that was 30% before use of estrogen began) with Lactobacillus dominance; L. iners and L. crispatus were most common. Potential urogenital pathogens were present in 44%, including Gardnerella, Prevotella, and E. coli. Despite the presence of these pathogenic organisms, 61% of women treated with HRT still had Nugent-defined normal flora after 3 months, supporting the putative lactobacilli-influenced stability of vaginal environment. 9
Interestingly, molecular methods demonstrated the presence of lactobacilli in every subject, which through traditional Nugent methodology would yield a technical prevalence rate of BV of zero, as compared to the 10.5% observed by traditional Nugent methodology at baseline in this study. This implies that asymptomatic Nugent-defined cases of BV may represent a microbiota that has numbers of lactobacilli sufficient to restrain other bacteria to an asymptomatic condition, but their presence may be only detectable by molecular methods.9,45
In a prospective study on the effects of HRT on vaginal microbiota, Galhardo et al 47 looked at 48 women greater than 5 years past menopause, over a 3-month period, by examining vaginal smears by Gram strain. No patients at baseline had lactobacilli as defined by Gram stain. After 1 month, 46% of those in the treatment group had normalized (lactobacilli > 90%); at 3 months, 74% had achieved normalization. Vaginal pH was also monitored and was 7 in both groups at baseline. At 30 days, pH had dropped to 5 in the treatment group; by 60 days, to 4.7; and by 90 days it had reached 4.5 (the upper limit of normal for reproductive women). 47 Use of HRT in women definitively in menopause was associated with normalization of vaginal pH as well as the composition of the microbiota (as normal was defined).
A summary of the results of microbial identifications in menopausal women is displayed in Table 6.
Comparison of vaginal microbiota in healthy postmenopausal women with and without hormone replacement therapy.
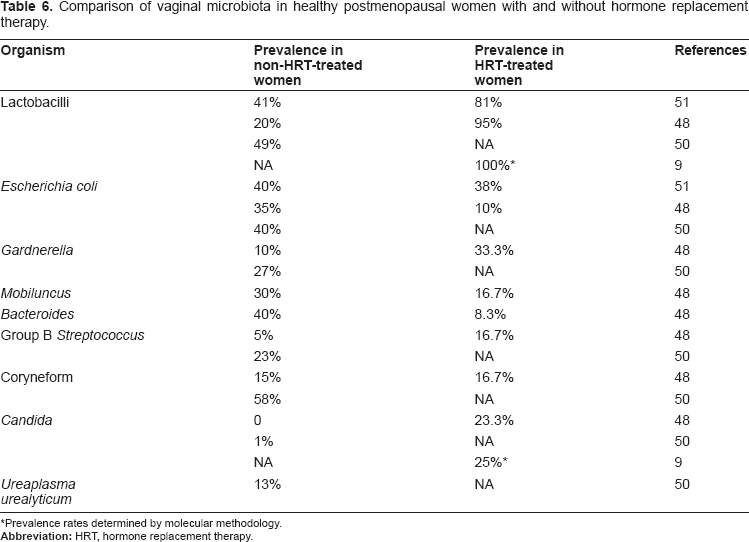
Prevalence rates determined by molecular methodology.
Methods for Measuring Vaginal Microorganisms
Traditional bacterial identification may include several distinct lines of investigation, including microscopy (both traditional light microscopy and electron microscopy), 53 culture, serology, and newer molecular techniques; nonetheless, comprehensive analysis of the vaginal microbial community is still an elusive goal. Complete description of the often complex community in the vagina has been frustrated by the available tools (traditionally culture-dependent or biochemical methods) which leave many organisms either uncultured or unidentified. 19 For example, one study used standard culture media to evaluate 522 women for the presence of Clostridium difficile and found only one patient (0.2%) who carried this organism. When cultures were repeated, however, using a media more suited to that organism, Clostridium difficile was cultured from 58 women (11%). 10 An additional limitation of many existing studies has been that single sampling events provide only a snapshot of a dynamic environment that undergoes continuous change. 23
The following are the current methods used to measure vaginal microbiota.
Culture-Specific Tests
The culture of organisms of interest, requiring recovery of the organism as well as regrowth, has been traditionally the gold standard and is significantly more sensitive than Gram staining for aerobic bacteria. 48 It continues to be the definitive measure of bacterial identification, 54 and it facilitates further study including antigenic studies, antibiotic susceptibility studies, genetic studies, and both in vitro and in vivo disease models of disease. 54
Successful culturing of a specific organism, however, is dependent on an appropriate fulfillment of the diverse and complex growth requirements of each individual bacterium.9,23 Successful culture requires the proper selection of an appropriate media; inoculation of an adequate amount of pathogen; and incubation at optimal temperature, atmosphere, and appropriate length of time. 54 Broad-spectrum media has allowed for successful culture of many new isolates of well-known species as well as the identification of new species. Many organisms, however, remain unculturable and thus unidentified. 54
Gram Staining/Microscopy
Gram staining reliably identifies common organisms 48 and staining techniques provide definitive identification for some organisms that are non-culturable, such as Mycobacterium leprae. 55 Some organisms are not susceptible to the staining methods, however, and some organisms are gram variable. 54 The isolation rate for any particular organism can vary widely depending on the detection method used, and even when the detection method is consistent, results can vary between laboratories as interpretation is fairly subjective. 54 Both sensitivity and specificity, in comparison to other techniques, is low. 54
Molecular Techniques
Although traditional identification methods based on microscopy (following differential staining) or on culture of an isolated organism remain the cornerstone of bacteriology, it is estimated that 99% of the bacteria that inhabit our environment are not identifiable by traditional culture techniques. 56 Traditional techniques are also frequently dependent on recognition of phenotypic features that are unstable and that fluctuate with changes in environmental conditions such as temperature or growth substrate. 57 Existing data about types and numbers of bacteria present in the vagina have not correlated well with clinical symptoms, an observation which may well be the result of the incomplete analysis of the vaginal ecosystem available through traditional methods. 58
Recent development of molecular techniques that can identify (without the need for laborious microscopy or prior culture) the organisms comprising the vaginal microbial community are proving useful in defining organisms that have defied identification through traditional culture means. 58 These techniques are already widely employed in industrial microbiology, providing an easy way to screen for a variety of potentially deleterious microbes in a single test 59 and confirming that many novel microorganisms remain to be identified.19,60 Molecular studies currently provide the best approach to a comprehensive analysis of all microbial species present within the complex vaginal ecosystem. 2
Although various approaches to molecular identification exist, most employ sequencing of the DNA that encodes 16S rRNA. 61 Analysis has revealed a wealth of genetic microbial diversity, increasing the number of culture-defined divisions in the bacterial kingdom from about 12 different phyla in 1987 62 to a reorganization of Monera into two separate “Domains” with multiple kingdoms in each, comprising upwards of 40 different phyla, a number which continues to grow steadily. 60 One problem fueled by rapid genetic identification of new organisms is that no guidelines exist to determine what level of genetic differences constitute either specialization or clinically relevant differentiation. 61 Molecular techniques will continue to yield new information, particularly as an adjunct to traditional culture and Gram-stain methods, while existing taxonomic difficulties are resolved. 9
No single methodology produces bacterial identification with 100% accuracy. 54 Most bacteriological research utilizes sequential application of a combination of microscopy, culturing, and molecular techniques with possible use of serology or culture in animal systems. 61 Whether or not molecular approaches will supplant traditional approaches in identification of clinical pathogens is currently a matter of debate. 61 Finally, identification of previously unculturable, original microbiota, even when present in large numbers, does not prove pathogenic complicity of these microorganisms. 61
Conclusion
A healthy vaginal ecosystem, in which a constructive equilibrium is achieved by a set of variable mutually beneficial interactions, clearly confers health on the vaginal vault. What combination of organisms may define that ecosystem is still elusive. What has become clear is that the traditional understanding of “healthy” has widened, largely through molecular data, to include a spectrum of possible combinations, depending on a variety of both host and microbial factors.
Dominance by lactic-acid producing microbes appears to be the cornerstone of microbial health. Lactobacillus, long considered a crucial component of a healthy vaginal ecosystem, is still believed to be foundation of most healthy microbiota. Recent research, based on genetic studies, however, has revealed that other lactic-acid producers, including previously unrecognized lactobacillus species, can form the foundation of a healthy microbiota as well. A healthy microbiota buffers the vaginal milieu from disruption that may make it vulnerable to the multitude of potential pathogens that may also inhabit the vagina, particularly organisms that cause STDs.
Newborns acquire their vaginal microbes initially from their mother; throughout childhood a lack of high levels of circulating estrogen keeps lactobacilli and, without sexual intrusion, microbial diversity low. Maturation at puberty establishes a cyclic fluctuation of estrogen levels; peak estrogen levels at the midpoint of the menstrual cycle encourage Lactobacillus colonization of the vaginal microbiota. Specific changes in the microbiota, associated with fluctuations in estrogen during the menstrual cycle, have been difficult to define conclusively. Disruption of the microbiota during menses may provide a vulnerable window in which other organisms may gain replicative dominance. The microbiota during pregnancy, particularly in light of the fact that the presence of BV is associated with pre-term delivery, should be a focus of further research. Use of HRT during menopause has been shown to restore vaginal pH and Lactobacillus domination in postmenopausal women, with an associated improvement in vaginal health.
Transient, intermittent perturbations in the composition of the vaginal microbiota have been demonstrated to be surprisingly common in women with healthy vaginal ecosystems, and what pushes a functional equilibrium towards disease, particularly BV, is still unclear. A clear correlation between specific microbial profiles and clinical significance has also been elusive; 40% of all BV defined by Nugent scores is asymptomatic. 63 Not surprisingly, response to common therapeutic options has been less than satisfactory. In fact, according to US Food and Drug Administration Criteria, the 30-day cure rate is less than 40%, with a full 80% of patient experiencing recurrence of symptoms within 9 months. 63
Molecular studies bring a promising new window from which to look at the composition of the vaginal microbiota, although correlation of molecular findings with those obtained by traditional culture method will continue to be a research goal. Molecular studies have identified species not previously detected by culture but have missed species that can be identified by traditional means. A lack of correlation between microbial findings and clinical significance is particularly relevant to molecular analysis of the vaginal community, which yet provides little utility for the purposes of diagnosis or choice of treatment.
Emerging molecular methodologies provide a way to more completely evaluate the dynamics of the vaginal microbiota at every stage of a woman's life. A greater understanding of the variety of microbial populations that can maintain a functional vaginal ecosystem, the culprit organism(s) of BV, and the complex interplay of both host and microbial factors that maintains a healthy vaginal microbiota will be necessary to formulate effective therapies for BV. Given the association of BV with both vaginal pathologies and complications of pregnancy, these should be important research goals.
Footnotes
Acknowledgement
The authors are grateful to Ms. Z. Schwen, Strategic Regulatory Consulting (Cincinnati, OH) for assistance in this manuscript.
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
