Abstract
Antibiotic resistance has been emerged as a major global health problem. In particular, gram-negative species pose a significant clinical challenge as bacteria develop or acquire more resistance mechanisms. Often, these bacteria possess multiple resistance mechanisms, thus nullifying most of the major classes of drugs. Novel approaches to this issue are urgently required. However, the challenges of developing new agents are immense. Introducing novel agents is fraught with hurdles, thus adapting known antibiotic classes by altering their chemical structure could be a way forward. A chemical addition to existing antibiotics known as a siderophore could be a solution to the gram-negative resistance issue. Siderophore molecules rely on the bacterial innate need for iron ions and thus can utilize a
Introduction
Marc Sprenger, Director of the WHO's secretariat for antimicrobial resistance, recently stated that “Many such infections are rapidly becoming resistant to life-saving drugs”; 1 thus, we may be on the verge of the post-antibiotic era. Indeed, it was also proposed that many of the procedures and conditions such as simple operations or cancer immunosuppression, which we take for granted today, may become impossible due to these organisms. 2
Extended-spectrum beta-lactamases (ESBL) have become a global scourge in the past 20 years. Initially thought to be only nosocomial problems have now become commonplace in community-acquired infections. It has been predicted that soon ESBL-producing
There are three definitions of resistance, which apply to more and more pathogens; these are multidrug resistant (ie, resistant to at least three different drug classes), extensively drug resistant (ie, resistant to all but one or two drug classes), and pan resistant (ie, resistant to all approved antibiotics).
4
All of these infections are very challenging from a clinical perspective, besides the escalating epidemiological issues. More recently, the spread of carbapenem-resistant enterobactericeae (CRE) has become a global issue. The carbapenem class of antibiotics has become the
In particular, it is the recent escalation of multiple mechanisms of resistance among gram-negative species, which are causing the greatest concern. These species go beyond
Overview of current-/late-stage developmental gram-negative agents.

It has been hypothesized that it may be possible to harness a vital bacterial survival mechanism to enable access to the bacterial cell. Such a process depends on the need for the essential element, iron. Bacteria secrete aggressive ironcomplexing proteins known as siderophores, which scavenge iron from their environment to survive. This process could be compared to mining by solubilizing iron ions in mineral form or biologically complexed iron ions such as iron bound to transferrins. Siderophores (from the Greek
Human interest in this
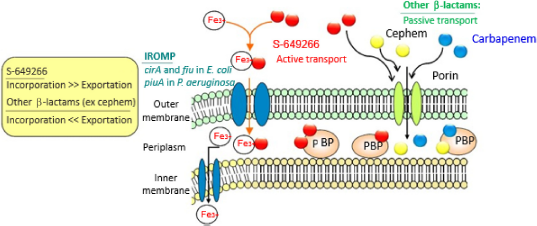
Depiction of siderophore action.
However, there are significant technical issues with susceptibility testing of these agents in that the standardized methods, eg, Committee for Laboratory Standards Institute or European Committee for Antimicrobial Susceptibility Testing, as they require specific media, such as Mueller Hinton Broth (MHB) supplemented with certain ions such as calcium and magnesium. However this medium, and possibly other test media, contains levels of iron, which are not representative of physiological concentrations of ferrous or ferric ions. Thus, in standard media, minimum inhibitory concentrations (MICs) tend to be 4–32 times higher than in iron-depleted settings. There have been two approaches to removing these iron ions by using cation-binding resins, which are added to MHB. The two tested agents are apo-T (solid media) 12 and Chelex (broth media). 13 These agents remove all ionic components but require subsequent addition of essential zinc, calcium, and magnesium ions to be supplemented to the test media after cation-binding treatment. These laboratory methods have been validated by in vivo or animal infection models, which are clearly depleted in terms of free iron ions. 14 This technical approach will need to be overcome once the drugs move into late clinical development as routine approaches will yield inaccurate and inappropriate, misleading high MICs.
Currently, there are three such
MC-1
MC-1 is a novel siderophore-conjugated monocarbam antibiotic, which has shown activity against MDR
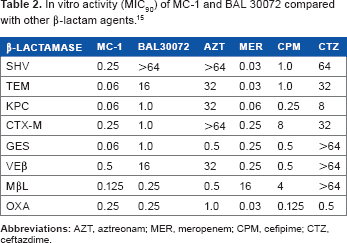
In vitro activity (MIC90) of MC-1 and BAL 30072 compared with other β-lactam agents. 15
Additionally, an isogenic panel of
BAL30072
BAL30072 is a monosulfactam conjugated with an ironchelating dihydroxypyridone moiety, which was developed by Basilea Pharmaceutica. It has been investigated in combination with a range of antibiotics commonly used against gram-negative organisms, but with the emergence of multidrug-resistant strains, they are less active. Gram-negative pathogens with β-lactam-resistant phenotypes were evaluated and compared with the activities of reference drugs, including aztreonam, ceftazidime, cefepime, meropenem, imipenem, and piperacillin/tazobactam.
BAL30072 showed potent activity against MDR
BAL30072 was bactericidal against both
In the direct comparison of BAL30072 with MC-1, the latter agent was more active in vitro against all enzyme isogenic strains, although the difference among metallo-β-lactamases and OXA strains was similar having MIC90s of 0.125 and 0.25 mg/L, respectively. 15 This drug was not listed in current clinical trials (ClinicalTrials.gov accessed August 2016); thus, no human tolerability data are available.
S-649266 (Cefiderocol)
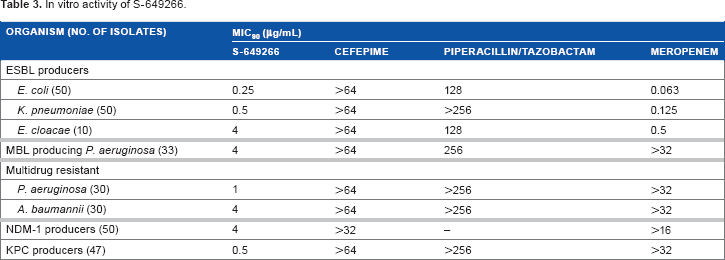
S-649266, or cefiderocol, is a novel siderophore cephalosporin antibiotic with a catechol moiety on the 3-position side chain (Fig. 2). Two sets of recent clinical isolates were used to evaluate the antimicrobial activity of S-649266 against Enterobacteriaceae. These sets included 617 global isolates collected between 2009 and 2011 and 233 β-lactamase-identified isolates, including 47 KPC, 50 NDM, 12 VIM, and 8 IMP producers.
18
The MIC90 values of S-649266 against the first set of
In vitro activity of S-649266.
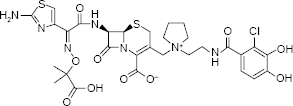
S-649266 (cefiderocol).
The antibacterial activity of S-649266 against carbapenemase producers and its stability against clinically relevant carbapenemases were also investigated. The catalytic efficiencies
It is increasingly appreciated that adaptation is a major mechanism associated with the acquisition and evolution of antibiotic resistance. Adaptive resistance is a specific type of nonmutational resistance that is characterized by its transient nature. It occurs in response to certain environmental conditions or due to epigenetic phenomena like persistence. It has been proposed that this type of resistance could be the key to understanding the failure of some antibiotic therapy programs. However, adaptive resistance mechanisms are still somewhat unclear. Equally the genetics behind some of the changes involved in adaptive resistance may explain the phenomenon of
Previous siderophore-based β-lactam compounds, such as monobactam (MB-1) and the monocarbam (SMC-3176), demonstrated inconsistent activity probably due to development of adaptive resistance.
21
,
22
Thus, Ghazi et al
23
examined S-64926, MB-1, and SMC-3167 in the neutropenic mouse model to determine the relative penetration through outer membrane via iron transporter systems as well as the stability of the three molecules against serine- and metallo-carbapenemases. Using this established thigh infection model,
A pharmacokinetic model providing probability of target attainment (PTA) data described the time courses of S-649266 concentrations in plasma and urine, which were used to predict efficacy for optimizing dosage regimens.
24
The simulations for the subjects with normal renal function suggested that S-649266 at 2 g q8h would exhibit efficacy for the target pathogens (ie, carbapenem-resistant
The clinical development of these three siderophore agents is less than clear. Only Shionogi Pharmaceuticals with S-649266 is registered on ClinicalTrials.gov with their Phase 2/3 program, which is comparison of S-649266 with imipenem/cilastatin for the treatment of complicated urinary tract infections in adults. This is a multicenter, double-blind international study with a focus on enrolling patients with multidrug-resistant, carbapenem-susceptible, gram-negative pathogens.
25
There is no clinical trial information on either MC-1 or BAL30072. A novel (Phase 3) study of S-649266 will enroll only patients with evidence of carbapenem-resistant pathogens, regardless of the primary infection site. This
Conclusions
The onslaught of multidrug-resistant bacteria is a global problem with the emergence and geographical expansion as a major clinical threat. In the past few years, we have seen several novel approaches to combatting bacterial resistance; these include synthetic peptides, cationic antimicrobial peptides (CAMPs), lantibiotics, lipophosphoxins, nosokomycins, and β-sitosterol.
The synthetic peptides have been studied as potential antibacterial agents for over 20 years, a recent focus on multidrug-resistant gram-negative species. McGrath et al have engineered a synthetic peptide that shreds and dissolves the double-layered membrane, which are considered to be a prime defensive mechanism of gram-negative species. This spiral peptide called KLAKLAKKLAKLAK acts by puncturing this unique bacterial bilayer without affecting eukaryotic cells. However, these peptides are subject to normal host enzyme destruction of some enzymes excreted by the bacterium itself. Thus, in order to combat this negative impact, increase in dosing will be needed, which may bring increased toxicity and manufacturing costs. The authors showed in vitro activity against key nosocomial pathogens with a dose-dependent killing. This synthetic peptide eliminates biofilms that may be important in settings where bacteria establish microcolonies and then seed to cause infection such as bone and joint infections. Animal models are now required to fulfill the next steps of drug development. 26
CAMPs are essential natural innate immune defense mechanisms that inhibit colonization by pathogens and aid in the clearance of infections. Gram-negative species are a major target but some evolved resistance mechanisms. Such mechanisms undoubtedly contribute to virulence and survival of pathogens. Over 1,200 natural CAMP molecules have been identified from prokaryotes and vertebrates. They possess certain constant features including cationic, amphipathic, and relatively hydrophobic. CAMPs destabilize the bilayer membrane by interacting with anionic head groups and hydrophobic fatty acid chains. This enables the CAMP to destabilize the membrane, leading to cell lysis. It has been hypothesized that CAMPs also have intracellular targets that also contribute to cell wall disruption and cell death. Two CAMPs are in clinical use, namely, polymyxin B and colistin (polymyxin E); however, bacterial resistance has recently been reported in China 27 and very recently in USA. 28 Bacterial resistance occurs via surface remodeling, usually lipopolysaccharide modification, capsule production, biofilms, efflux pumps, and proteolytic degradation. 29 Clearly with such an array of CAMP resistance mechanisms, this may limit the value of the class in the clinical setting. Moreover, the known renal toxicity of colistin is another hurdle to be overcome. It has been proposed that establishment of some of these CAMP resistance mechanisms are additions to the bacterial pathogenicity. Deeper understanding of the CAMP resistance mechanisms may help yield further gram-negative antibiotics.
In an effort to overcome some of these issues, Torcato et al 30 designed and characterized two new molecules, namely, R-BP100 and RW-BP100. These analogs have two amino acids, in which Tyr is replaced with a Trp and/or the Lys residues replaced with Arg. These analogs are active against a wide range of gram-negative species as well as, unusually for peptides, all tested gram-positive bacteria. Various studies show the target site of the bacterial membrane. These minor, but significant, structural changes may yield potential new class of antibacterial agents for a broad range of multidrug-resistant species.
Draper et al 31 examined posttranslationally modified ribosomally synthesized antimicrobial peptides, lantibiotics, with a broad-spectrum antimicrobial activity. One lantibiotic 3147 showed a wide range of anti-gram-positive activity. Draper et al tested the enhancement of 3147 with polymyxin B and E using synergism tests. Using low levels of a polymyxin, the lantibiotic 3147 activity against gram-positive and some gram-negative species. They hypothesized that use of 3147 may allow for use of lower, thus less, toxic concentrations of polymyxin.
Panova et al
32
discovered a new series of compounds termed lipophosphoxins (LPPOs), which showed specific activity toward gram-positive species. LPPOs are bactericidal in activity and localize to the plasma membrane in bacteria but not in eukaryotic cells. LPPOs create pores in the bacterial membrane. Of key concern with any new class of compound toxicity to humans is essential thus showing no genotoxicity in the Ames test, do not cross monolayer of Caco-2 cells and well tolerated by mice when given orally but not via the peritoneum. As the agents withstand low pH, it has been proposed that they may not be viable systemic antibiotics but may have a role as nonabsorbed antibiotics such as in
MRSA is still a major nosocomial pathogen, although recent additions to the armamentarium such as modified glycopeptides and oxazolidinones have recently been approved for clinical use; it is undisputed that MRSA will find a way to resist these new agents. Tomoda
33
reported on a new member of the phosphoglycolipid family, the nosokomycins from
Li et al
34
reported the use of a phytosterol, β-sitosterol, to protect against cell lysis by pneumococcal pneumolysin, a potent virulence mechanism of
There is clearly a huge amount of research into alternative methods to combat antibiotic-resistant bacterial infections. However, many of these molecules are still in their early stages with very few having been exposed to humans. Indeed, a few seem to have been tested in animal models. So although there is much excitement at these innovations, we have to turn to classes we understand and have a long-standing safety record.
Thus, we resort to the exploration of one of the oldest antibiotic classes, the β-lactams. Most recently, extension of the β-lactam/β-lactamase inhibitor combinations (eg, ceftazidime/avibactam or ceftolazane/tazobactam) and modified carbapenems are clinical options. Of particular interest is the recent announcement of the initial Phase 3 clinical study of meropenem–vaborbactam in complicated urinary tract infections, which was compared with piperacillin/tazobactam. Notably the clinical efficacy was 98.4%; yet, the microbiological eradication was 66.7% compared with 57.7% reported with piperacillin/tazobactam.
35
The second Phase 3 study comparing this new combination with
Author Contributions
Conceived and designed the experiments: GT. Analyzed the data: GT. Wrote the first draft of the manuscript: GT. Contributed to the writing of the manuscript: GT. Agree with manuscript results and conclusions: GT. Jointly developed the structure and arguments for the paper: GT. Made critical revisions and approved final version: GT. Author reviewed and approved of the final manuscript.
