Abstract
Decision to test for Clostridium difficile infection (CDI) is usually made when patients have loose stools with Bristol stool score of ≤5. We aimed to assess the relationship between bacterial load of C. difficile and Bristol stool scale, as well as stool frequency in stool samples collected from patients infected with the organism. Samples were collected at baseline, during therapy, and at the end of therapy. Spearman correlation test was used to evaluate these relationships. No correlation between Bristol stool scale and fecal load of C. difficile was found for both spores and vegetative cells at all time points as counts were persistently high (P = non-significant). Weak positive correlations were found between stool frequency and fecal load of C. difficile spores and vegetative cells (r s = 0.22 and 0.24, P = 0.04 and 0.03, respectively). These findings indicate that quantitative colony counts were sufficiently high to detect C. difficile, irrespective of stool consistency, and suggest that semiformed stool should be sought for the pathogen in symptomatic patients with frequent stools.
Introduction
Clostridium difficile is a Gram-positive spore-forming anaerobic bacillus that is the major cause of antibiotic-associated diarrhea, accounting for 15–25% cases of nosocomial antibiotic-associated diarrhea. 1 Given the presentation of C. difficile infection (CDI), loose stools are usually sought for diagnosis and are thought to harbor higher bacterial loads of the pathogen than semiformed or formed stools.
Bristol stool scale was developed by Lewis and Heaton in order to establish a clinical tool to monitor changes in intestinal function (Fig. 1). 2 The scale was significantly correlated with this parameter in terms of stool form and defecatory frequency. The European Society of Clinical Microbiology and Infectious Diseases in its latest guidelines on CDI diagnosis and treatment include Bristol stool scale in the definition of diarrhea associated with this infection. 3 The scale is also included as a tool for the assessment of CDI severity in the guidance on the management and treatment of CDI published by Public Health England. 4

Bristol stool scale. Adapted from Ref. 2
A study by Caroff et al 5 examined the positivity of C. difficile diagnostic assays, enzyme immunoassays (EIAs) for glutamate dehydrogenase, and toxin A/B followed by a nucleic acid amplification test (NAAT), in correlation with the Bristol stool scale. They demonstrated that a correlation between Bristol stool scale and results of C. difficile assays was lacking with the organism being recovered more frequently from semiformed stools (Bristol scores of 5 or 6) than from liquid stools (Bristol score of 7) with NAAT. EIA had 50% lower chance of detecting C. difficile in semiformed stools versus NAAT. The authors questioned whether the low fecal load of the organism or its toxin concentration in semiformed stools may have been associated with false-negative results obtained with EIA. Therefore, we aimed to address this issue through assessing the correlation between Bristol stool scale and the quantitative fecal load of C. difficile in order to elucidate whether low bacterial loads, particularly toxin-producing vegetative cells, may be associated with false-negative results of EIA testing for C. difficile toxin as well as whether semiformed stools carry sufficient amounts of the organism to be sought for CDI diagnosis. A secondary objective of the study was to assess the correlation of stool frequency with fecal C. difficile spores and vegetative cells to find out whether the presence of correlation may support the finding of the primary objective.
Methods
This is a descriptive observational study where we used data from a randomized, open-label study that we conducted to compare the effect of fidaxomicin versus oral vancomycin on C. difficile bacterial load of both spores and vegetative cells. 6 The study protocol was approved by the Hartford Hospital Institutional Review Board, and all patients provided written, informed consent prior to entry into the trial. In this study, three stool samples were collected from enrolled patients who tested positive for C. difficile by NAAT. Samples were collected at baseline (before initiation of intervention), at a midpoint during the therapeutic course (days 3–5) and at the end of the therapeutic course (days 10–13). Bristol score was recorded for each collected sample. Samples were cultured, depending on availability, on either chromID C. difficile plates (bioMérieux SA)or cycloserine-cefoxitin-fructose agar (CCFA) plates (BD) since no difference in C. difficile recovery and quantitative colony counts was reported between the two types of media. 7 ChromID and CCFA plates were incubated under anaerobic conditions in BD GasPak anaerobic chambers (BD) at 37°C for 24 and 48 hours, respectively. Each sample was plated twice, once without treatment and once after treatment with ethanol in order to kill vegetative cells and keep spores. Thus, untreated plates contained colonies (colony forming unit, CFU) that represent both spores and vegetative cells, while ethanol-treated plates contained spores only. Vegetative cells' CFUs were counted by subtracting the number of spores' CFUs in the ethanol-treated plate from the total number of CFUs in the untreated plate. Log10 CFU/g of stool for both spores and vegetative cells was then calculated and plotted. The lower limit of quantification of the culturing assay is 40 CFU/g stool. Stool frequency was determined based on a number of bowel movements over the last 24 hours prior to each sample collection.
Given the unequal distribution of the data as tested by Shapiro–Wilk test for normality, Spearman correlation coefficient (rs) was calculated to measure the strength of the relationship between Bristol stool scale and the bacterial load of C. difficile in stool at each of the three time points. The same test was used to evaluate the correlation of stool frequency with C. difficile fecal counts. A P value of ≥0.05 was considered significant, and hence, a correlation between the two variables was deemed existent.
Results
A total of 83 stool samples were collected from 30 patients at the three sampling points over 24 months: 28 samples were collected at baseline, 30 samples at the midpoint of therapy, and 25 samples at the end of therapy. Reasons for the decline in the number of samples from the midpoint of therapy going forward were either loss to follow-up or treatment failure and subsequent exclusion from the study. An additional sample from a patient who failed therapy was collected at the day of attrition, which was within the midpoint window. In the case of another CDI patient, a baseline sample failed to be delivered to our laboratory; however, samples at subsequent sampling points were collected and analyzed. The mean age of patients was 67.3 ± 15.3 years; 20 (66.6%) of them acquired their infection from the community and only 7 (23.3%) of the infections were because of the North American pulsed-field gel electrophoresis type 1 (NAP1) strain of C. difficile (identified by ribotyping and commonly designated NAP1/BI/027), which is known to be more virulent and associated with more severe diseases than other strains of the organism. 1
Figure 2 shows scatter plots of log10 CFU/g of stool for spores and vegetative cells at the three sampling points. Bristol stool scale and log10 CFU/g of stool for C. difficile spores and vegetative cells showed rs values of −0.14 and −0.05 (P = 0.53 and 0.77, respectively), 0.14 and −0.32 (P = 0.52 and 0.08, respectively), and 0.25 and −0.26 (P = 0.25 and 0.22, respectively) at baseline, at the midpoint of therapy, and at the end of therapy, respectively. The non-significant P values of the correlations of baseline samples indicate that a correlation between Bristol stool scale and log10 CFU/g of stool for both spores and vegetative cells was lacking at the diagnostic phase of CDI. Likewise, no correlation was found between Bristol stool scale and quantitative fecal load of C. difficile during and after completion of CDI therapy. Stool frequency showed a weak, although statistically significant, correlation with fecal load of C. difficile with rs values of 0.22 (P = 0.04) and 0.24 (P = 0.03) for spores and vegetative cells, respectively, which indicates the presence of higher bacterial CFUs with more frequent stools (Fig. 3).
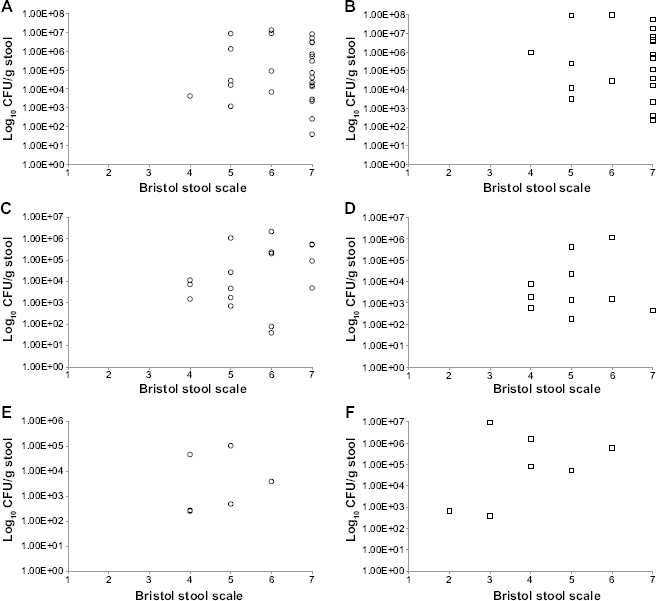
Scatter plot of Bristol stool scale versus spores and vegetative cells colony counts at different time intervals. Bristol stool scale versus log10 CFU/g stool of (
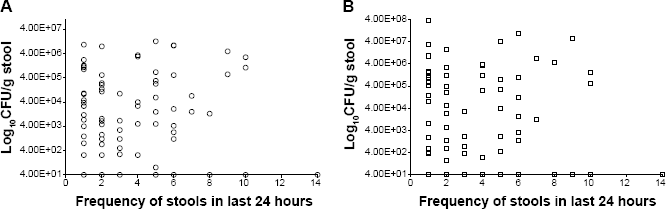
Scatter plot of frequency of stools versus spores and vegetative cells colony counts. Frequency of stools versus log10 CFU/g stool of (
Discussion and Conclusions
To our knowledge, this is the first study to examine the correlation between fecal loads of C. difficile with Bristol stool scale and stool frequency. In the context of clinical disease, it would appear that the quantitative CFU of C. difficile was sufficiently high to detect the pathogen, irrespective of stool consistency, when diagnosing symptomatic patients presenting with frequent bowel movements. In addition, semiformed and formed stools can also harbor the organism at detectable quantitative levels. Both of these findings explain why NAAT (a test that detects the organism, whether in vegetative or in spore form, rather than the toxin of the organism) was more successful in detecting C. difficile in semiformed stool (Bristol scores of 5 or 6) as the organism may have presented in quantitative amounts in the study by Caroff et al. 5 Moreover, C. difficile detectability by EIA may not be associated with the fecal load of the organism, but rather with the concentration of its toxins since EIA is a test for C. difficile toxins. Therefore, a study examining the correlation between Bristol stool scale and the concentration of C. difficile toxins in stool may further aid answering the lower detectability of C. difficile by EIA in semiformed stools.
Author Contributions
Conceived and designed the experiments: AKT, DPN. Analyzed the data: AKT, DPN. Wrote the first draft of the manuscript: AKT, DPN. Agree with manuscript results and conclusions: AKT, DPN. Jointly developed the structure and arguments for the paper: AKT, DPN. Made critical revisions: AKT, DPN. All authors reviewed and approved of the final manuscript.
Footnotes
Acknowledgments
We thank Seth Housman, PharmD, MPA for his involvement in the data collection. We also thank Lucinda Lamb and Mary Anne Banevicius for the development of the C. difficile culturing technique. We thank them, as well as the staff and fellows at the Center for Anti-Infective Research and Development, for their help in processing the samples.
