Abstract
Cancer patients undergoing chemotherapy are particularly susceptible to drug-drug interactions (DDIs). Practitioners should keep themselves updated with the most current DDI information, particularly involving new anticancer drugs (ACDs). Databases can be useful to obtain up-to-date DDI information in a timely and efficient manner. Our objective was to investigate the DDI information sources of pharmacy practitioners in Asia and their views on the usefulness of an oncology-specific database for ACD interactions. A qualitative, cross-sectional survey was done to collect information on the respondents' practice characteristics, sources of DDI information and parameters useful in an ACD interaction database. Response rate was 49%. Electronic databases (70%), drug interaction textbooks (69%) and drug compendia (64%) were most commonly used. Majority (93%) indicated that a database catering towards ACD interactions was useful. Essential parameters that should be included in the database were the mechanism and severity of the detected interaction, and the presence of a management plan (98% each). This study has improved our understanding on the usefulness of various DDI information sources for ACD interactions among pharmacy practitioners in Asia. An oncology-specific DDI database targeting ACD interactions is definitely attractive for clinical practice.
Keywords
Introduction
Cancer patients undergoing chemotherapy are particularly susceptible to drug-drug interactions (DDIs). 1 In general, DDIs are thought to be the cause of approximately 20% to 30% of all adverse drug reactions, 2 and the risk of DDIs has been found to increase markedly from 5.6% to 84% when the number of concomitant medications is increased from 2 to 6. 3 The clinical consequences of these interactions are significant because anticancer drugs (ACDs) possess narrow therapeutic ranges and are inherently toxic. 1 It has been estimated that at least one potential drug interaction occurs in 27% of cancer patients. Of these interactions, 13% involves ACDs and 86% are classified as being of major or moderate severity. 4 It is thus important for healthcare professionals to keep themselves informed with the most current drug interaction information, particularly involving new ACDs that are constantly being brought to the market.
Oncology clinicians may not have the time or necessary skills to constantly update, critically evaluate and apply the enormous amount of published literature available to their clinical practice. 5 Studies done on information sources used by oncology practitioners have concentrated on general cancer information instead of oncology DDIs.5,6 It has been reported that a higher proportion of practitioners in the United States (US) use electronic databases (51%–79%) as sources for DDI information rather than printed references (9%–24%); 7 and approximately 53% and 25% of prescribers and pharmacists rely on a colleague for such DDI information. 7 This is a worrying phenomenon since healthcare professionals may lack the ability to identify potential DDIs.8–12 The practitioners' ability to identify DDIs is inversely affected by the number of drugs on patients' medication profiles. 8 Thus, it is essential that clinicians find appropriate sources of ACD interaction information at the point of patient care. The practitioner's ability to keep abreast with the latest ACD-related information is likely to be hindered by inherent delays in traditional literature publication processes. 13 Online drug databases can be useful for obtaining up-to-date DDI information in a timely, efficient and accessible manner. 14 Hence our study objectives were to investigate the current sources of DDI information among practitioners, the usefulness of an oncology-specific database which targets ACD interactions, and the parameters that are essential if such a database was available for clinical practice.
Methods
A qualitative, cross-sectional research design was employed. A structured questionnaire consisting of 10 questions collected information regarding the respondents' practice characteristics, frequency of encountering ACD interactions in their daily practice, and their current sources of DDI information. Their opinions on the usefulness of an online ACD interaction database were also asked together with the features which they deemed important for such a database. These included the accuracy of DDI content, usefulness in clinical practice and user-friendliness. In addition, they had to rate the importance of several DDI parameters as “absolutely essential”, “essential”, “not essential, but good to have” or “not essential at all”. The “neutral” option was also provided. The DDI parameters were pharmacokinetics of the chemotherapy and interacting drugs, mechanism of interaction, severity, substantiation, and presence of a management plan.
The self-administered questionnaire was distributed to the participants of The 2nd Asia-Pacific Oncology Pharmacy Congress (APOPC) held in Bangkok, Thailand, from 11–13 September 2008. Participants were healthcare professionals from the Asia-Pacific region, US and Australia. Returned questionnaires were coded with an identification number to ensure confidentiality. Survey implementation was approved by the APOPC organizing committee, and replies from the individual participants were considered as consent to the questionnaire.
Returned questionnaires were included for analysis if the questions regarding respondents' sources of ACD interaction information, or useful features in an online database were answered, and at least half (5/10) of the questions were answered. Responses with minor missing data, such as practice demographics and frequency of encountering DDIs were also included.
Data were analyzed using SPSS 17.0 (Chicago, IL, USA). Descriptive statistics were used to describe the categorical variables. Kruskal-Wallis test was performed to detect for statistical significant differences in sources of interaction information and parameters related to the usefulness of an ACD database among 4 groups of respondents (inpatient practice only, outpatient practice only, both inpatient and outpatient practices, other practice settings). Mann-Whitney rank sum test was used for comparisons between any 2 groups. All tests were two-tailed and p-values below 0.05 were considered as statistically significant.
Results
Respondent characteristics
Of the 209 distributed questionnaires, 105 (50%) were returned and analyzed. Three (1%) were excluded from analysis because majority of the questions were not answered. Of the remaining questionnaires that were usable, only those that were completed by pharmacists working in Asia were included for analysis, since majority of the respondents were pharmacists by profession (96%) and practicing in Asia (93%). Our results would then portray a clearer picture on the opinions of pharmacists in this part of the world regarding DDIs and the usefulness of an oncology-specific database which targets ACD interactions. As a result, a total of 91 completed questionnaires were used for our analyses. Table 1 shows the practice characteristics of the respondents. Approximately half practiced in inpatient and outpatient settings, out of which 31 (34%) and 21 (23%) were solely in either inpatient or outpatient practices respectively. Fifty-two respondents (57%) had 5 to 20 years of practice experience, and a quarter frequently encountered DDIs with ACDs (more than 25% of their time) in their daily practice.
Practice characteristics of the survey respondents (n = 91).

Percentages may not add to 100% due to missing values and rounding of data.
Sources of ACD Interaction Information
Electronic databases (70%), drug interaction textbooks (69%) and drug compendia (64%) were most commonly used by the pharmacy practitioners to check for ACD interactions (Table 2). On the other hand, medication package inserts (41%) and standard textbooks (42%) were seldom used. Sixteen respondents (18%) did not utilize any computerized forms of information sources, such as electronic databases or specific softwares, to check for ACD interactions. Among those who did, electronic databases seemed to be more frequently used (Tables 2 and 3). The proportion of pharmacists who consulted with their colleagues was a little less than half (44%), edging the use of standard textbooks (42%) and product information (41%). Inpatient pharmacists were more likely to utilize standard textbooks as DDI sources for ACDs than outpatient pharmacists (52% versus 14% respectively, P = 0.007).
Healthcare professionals' sources of anticancer drug interaction information (n = 91).

Percentages may be over 100% due to multiple selections.
Sources of drug interaction information based on the practice settings of healthcare professionals.

Percentages may be over 100% due to multiple selections.
P < 0.05 between the 2 groups indicated by asterisks (*).
Usefulness of an Oncology-Specific DDI Database for ACD Interactions
Majority of the pharmacy practitioners (93%) indicated that it would be useful to have an online ACD database catering towards DDIs. Only 2% disagreed. The rest felt that it was not essential, but a bonus, to have such a database.
Overall ratings of DDI information accuracy (98%) and usefulness in clinical practice (97%) were higher than that for user-friendliness of the database (88%). One respondent in other practice settings indicated that clinical usefulness was “not essential, but good to have” (Table 4). On the other hand, more inpatient and outpatient pharmacists considered user-friendliness to be a less essential factor.
Factors for consideration in an online drug interaction database specific to oncology.
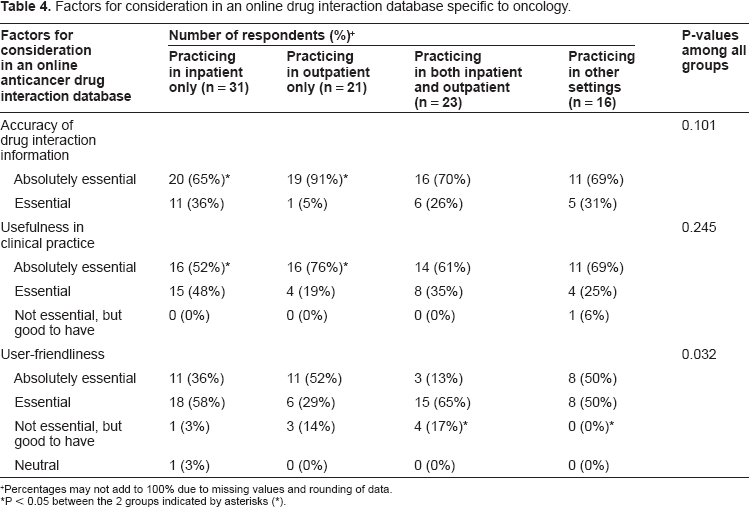
Percentages may not add to 100% due to missing values and rounding of data.
P < 0.05 between the 2 groups indicated by asterisks (*).
Almost all the pharmacy practitioners considered DDI content accuracy as an important factor for the database. A larger proportion of outpatient pharmacists (91%) indicated this as “absolutely essential”, in contrast to only 65% of inpatient pharmacists (P = 0.013). The trend was similar for clinical usefulness of the database, where a higher proportion of outpatient pharmacists (76%) rated as “absolutely essential” compared to 52% of inpatient pharmacists (P = 0.043). Outpatient pharmacists could have viewed such a database as their main source of DDI information at the point of dispensing, and hence considered these 2 factors as more important elements in the database. On the other hand, user-friendliness of the database was considered to be less important by pharmacists working in both inpatient and outpatient practices than those who were in other practice settings (P = 0.006). All the pharmacists in the latter group agreed that user-friendliness was important for such a database, but there were 4 pharmacists in the former group (17%) who indicated that this factor was only “good to have”. In fact, among all the respondents, 9 pharmacists working in inpatient and outpatient settings (10%) were either neutral or considered this factor to only be of added value to the database, compared to pharmacists practicing in other settings, who deemed this factor as an essential or absolutely essential component for the database.
Interaction Parameters for an Oncology-Specific DDI Database
The most important DDI parameters, indicated by ratings of “essential” and “absolutely essential”, were the mechanism and severity of the detected interaction, as well as the presence of a management plan (98% each). A significantly higher percentage of outpatient pharmacists rated the mechanism of interaction as “absolutely essential” (76%), compared to only 42% of inpatient pharmacists (P = 0.014) (Table 5). One inpatient pharmacist also felt that the mechanism was not essential but a value-added component in the database.
Drug interaction parameters that are important in an oncology-specific drug interaction database.

Percentages may not add to 100% due to missing values and rounding of data.
P < 0.05 between the 2 groups indicated by asterisks (*).
Interestingly, pharmacokinetics of the drugs was viewed to be less important (95%) than the mechanism and severity of interaction, or the management plan (98% each). Substantiating studies (85%) and reference sources (90%) fared the most poorly. In fact, some pharmacists felt that pharmacokinetics (2%) and substantiating studies (1%) were not needed at all. On the other hand, all the pharmacy practitioners agreed that the other 4 parameters would be “good to have” as DDI information. Some respondents also gave useful feedback with regards to other DDI parameters that could be considered for inclusion in the database. The reader is referred to Appendix 1 for details on these additional parameters.
Discussion
This study has shown that the mechanism of interaction, severity and presence of a management plan were regarded as the most important DDI parameters in an oncology-specific database. This was probably because these parameters directly relates to the interaction effects and how the interactions can be managed at the point of patient care. However, the respondents in this study considered the presence of a proposed management plan to be more crucial than the other parameters. This was reflected by the presence of “not essential, but good to have” ratings for the mechanism and severity of the DDI, in contrast to those for the proposed management plan, which were “essential” and “absolutely essential”. In addition, more outpatient (76%) than inpatient (42%) pharmacists considered the interaction mechanism as an “absolutely essential” component in the database (P = 0.014), probably due to the difficulty of consulting DDI resources at the point of patient care; unlike inpatient practitioners who could still refer to other DDI resources for supporting evidences of the interaction mechanisms.
Interestingly, a consistently higher percentage of outpatient practitioners rated the DDI parameters as “absolutely essential” compared to inpatient practitioners, except for the severity of interaction. More inpatient than outpatient practitioners considered severity as “absolutely essential”, even though the percentages were not statistically different (P = 0.495). A high probability exists for inpatient pharmacists to identify interactions when they screen the drug orders for patients. A study by Ducharme and Boothby had identified a substantially higher number of DDIs occurring in the outpatient setting compared to hospitalized inpatients. 15 Severity could have played a higher weightage in the clinical practices of inpatient pharmacists, probably because they see DDIs as being a preventable drug-related problem, and severity would play a more important role in their decisions to dispense or not dispense the affected drug combination. Moreover, recent studies have shown the existence of inconsistent severity ratings among different drug compendia.16–19 Hence, it is imperative that the severity data provided by such a database be accurate and reliable for clinical practice.
The pharmacy practitioners in our study seemed to be more concerned about the accuracy of DDI content and clinical usefulness of the database than its user-friendliness. Granted the fact that user-friendliness is subjective and depends on the practitioner's familiarity with the database, approximately 88% of the respondents still considered this as an important factor. User-friendliness was more highly desirable among pharmacy practitioners who were working in other settings (e.g. academia, retail, home care, and private practice), probably because they relied heavily on electronic databases (81%) and softwares (44%) as resources for DDI information, but had to work independently in their workplace. A user-friendly database could possibly enable them be more efficient in terms of time-management when checking for a DDI. Desirable features of a good online database include ease-of-use and good navigation features. 20 Database usability has also become increasingly important in clinical practice with the availability of software programs for handheld devices such as personal digital assistants (PDAs). One respondent in our study indicated that a software program was preferred over an online DDI database. We postulate that this could be a reason since databases which are in the form of software programs would also offer an alternative version for handheld devices. However, ease-of-use comes at a price which may compromise the scope and completeness of drug information, 21 thus it was expected that content accuracy and clinical usefulness was still viewed as being more important than user-friendliness.
From our results, a larger proportion of outpatient than inpatient practitioners considered content accuracy and clinical usefulness to be “absolutely essential”. We assumed that respondents who rated the factors as “absolutely essential” would consider them of higher priority than “essential”. Thus, we hypothesized that these 2 factors played more important roles for outpatient practitioners and could be related to the ease-of-access to the various DDI resources in the inpatient and outpatient settings. Pharmacists working in outpatient settings could be subjected to time constraints and greater difficulty in accessing drug information resources because of their contact with patients at the point of dispensing. Studies conducted by pharmacists have suggested that the outpatient setting may be suitable for improving the quality of patient care.22–24 The outpatient practitioners in our study could have seen this database as their main source of ACD interactions at the point of dispensing, and therefore considered these factors to be of higher priority, while those in inpatient practice probably saw this database as being complementary to other available resources.
An important finding in our study was that there were lower percentages of inpatient and outpatient practitioners who used drug information softwares (45%–57%) than DDI textbooks (61%–67%) or compendia (67%–77%) for ACD interaction information. A similar trend was seen with inpatient practitioners when comparing electronic databases (58%) with DDI textbooks (61%) and compendia (77%). This could be due to the existence of certain environmental barriers in hospital institutions: 5 (a) connection or firewall issues which impede access to the internet/intranet; (b) restricted access to the internet/intranet in clinical areas; (c) limited number of computers available in the wards or treatment areas for clinical purposes; (d) old or slow computers, and (e) economic reasons such as costs of online journal subscriptions. These barriers might not have been as prominent in other non-clinical settings.
Despite these environmental barriers, a large proportion of pharmacy practitioners in this study still used electronic references such as e-databases (e.g. PubMed) and softwares (e.g. Micromedex) as ACD interaction resources. This is not surprising since clinicians tend to perceive these sources as being readily available and time-efficient at the point of patient care. 5 The advent of the internet and portable electronic devices such as PDAs have allowed clinicians to access online databases and drug information programs for evidence-based oncology information. 5 The advantage of these resources over hardcopy books is their currency of information. 5 In general, larger proportions of pharmacy practitioners from all practice groups in our study consulted electronic resources for DDI information compared to standard textbooks. However, a lower proportion of practitioners used drug information software (48%) compared to drug compendia (64%) and other drug interaction textbooks (69%). This proportion was also lower than that reported in a study on US practitioners (79%). 7 Our result was expected since the study assessed sources of general DDI information used by prescribes in a variety of specialties. 7 Our study specifically targeted ACD interactions. From our knowledge, drug information softwares used in clinical practice, such as Micromedex, Lexi-Comp, and Facts and Comparisons, among others, are not specific to ACD interactions. The low proportion of practitioners who use these softwares to check for ACD interaction information is consistent with the high percentage of respondents (93%) who indicated a need for an online ACD interaction database. In this aspect, an oncology-specific database which caters towards ACD interactions would definitely be a useful tool for pharmacy clinicians in Asia.
Limitations and Future Work
The respondents in our study were pharmacists practicing in Asia, thus our results may not reflect the views of other healthcare professionals, and may also differ from practitioners in other parts of the world. However, the results of our study are still useful since majority of patients are seen by pharmacists as the last line of practitioners during their consultations with the healthcare team for the dispensing and counselling of their medicines. The opinions of this subgroup of healthcare professionals can provide an insight to the development of future drug information systems which will hopefully provide a “slice” to the Swiss cheese model of safety incidents, 25 so that patient safety can be improved.
Despite distributing the surveys before the commencement of a plenary lecture at the conference, where we hoped to capture most of the practitioners, only half were completed, possibly introducing non-response bias to our study. We postulated that those who responded were more interested in oncology DDIs, probably because of their lack of experience in this area. Tan et al. had previously identified that when less experienced cancer clinicians were exposed to unfamiliar situations, they tended to refer to their more experienced colleagues for guidance and quick information. 5 Thus, the relatively high percentages of respondents in our study who had a limited duration of clinical practice (71% practiced for 10 or less years, of which 40% had less than 5 years of experience) could have also biased our results to a higher than expected proportion (44%, range 38%–61%) who relied on consultations with other colleagues as information sources for ACD interactions, compared to other studies done on US prescribers (14%–53%).7,26 Furthermore, even though majority encountered DDIs less than one-quarter of the time, the idea of having an ACD interaction database could have attracted their attention, and prompted them to do the survey. Nevertheless, our results would still apply to the cohorts of pharmacy practitioners who enter oncology practice, either fresh from graduation or from another specialization.
Certain conclusions in this study, particularly those regarding the usefulness of the oncology-specific database, as well as the importance of the various DDI parameters, were made based on the assumption that respondents who rated “absolutely essential” would consider that component to be of higher importance than “essential”. Due to the nature of the Likert-scaled questions in our survey, and the different practice settings of our surveyed population, the interpretations of these 2 anchor points (“absolutely essential” versus “essential”) could vary among the respondents, leading to subtle differences in their responses. However, we attempted to control for this difference in the designing of the questionnaire by arranging the parameters for each of those sections as separate parts of a single question. For example, participants would be asked to rate the importance of the 3 different factors of the database (information accuracy, usefulness in clinical practice, and user-friendliness) as one full question with the same set of Likert scales. This design would inevitably allow them to compare the factors in terms of importance. An “essential” rating for both clinical usefulness and user-friendliness would imply that these factors were equally important, while an “absolutely essential” rating for information accuracy would imply that this factor was considered to be more crucial than the other 2 factors. Thus, we could assume that a rating of “absolutely essential” would consistently be a notch higher than “essential” in terms of importance in all our survey responses.
In addition, a “ceiling effect” might have occurred since this study was carried out in an oncology congress meeting, whereby majority of the responses from the surveyed population could have skewed towards the higher priority end of the spectrum, thus masking the actual differences in opinions of the pharmacy practitioners. Various statistical techniques have been published in literature to control for this “ceiling effect”, such as ordinary least squares (OLS) regression, Tobit models, censored least absolute deviation (CLAD) approach, two-part models (TPM), latent class models (LCM), and the likelihood ratio test; among which the latter 3 approaches seemed to be more robust in accounting for ceiling effects.27,28 These approaches could be considered in future studies when this survey is administered to a larger group of healthcare professionals from a wider international audience (e.g. physicians and nurses practicing outside Asia). Although our aim was to obtain the opinions regarding ACD interaction sources and the usefulness of an oncology-specific DDI database from the core group of healthcare professionals practicing in the oncology setting, another way to minimize the ceiling effect would be to target healthcare professionals from other specializations who encounter patients with cancer in their daily practice. Patients with cancer tend to suffer from depression due to the emotional impact of the diagnosis of this terminal illness, biological stressors and side effects of chemotherapy,29–31 while those with brain tumors or metastases may be predisposed to suffering from seizures,32,33 thus requiring other medications such as antidepressants or anticonvulsants. In these cases, suitable groups of healthcare professionals in which the survey could be targeted would include psychiatrists and neurologists. Their opinions would be valuable since they might hold different points of view from the oncology practitioners surveyed in this study.
Lastly, this survey was administered in English and not translated to other Asian languages such as Chinese, Malay or Thai. Since this conference was held in Thailand, many of the practitioners who attended the conference could be from the host country, and their English language abilities could differ. We assumed that attendees of the conference would have a certain level of understanding in the English language since all the sessions were carried out in English. However, we did take an effort to ensure that there were no difficult phrases in the survey, and technical terms were not included. We can only postulate that those who did not understand the survey would not have completed it, and this could have contributed to the low response rate of our study.
Conclusion
This study has improved our understanding on the usefulness of various information sources for ACD interactions among healthcare professionals, especially pharmacists, working in Asia. Popular forms of DDI resources include electronic databases and specific drug information softwares (48%–70%), drug interaction textbooks (69%) and drug compendia (64%). Furthermore, a large proportion of practitioners also refer to colleagues for consultations on ACD interactions (44%).
In addition, we have demonstrated the need for an oncology-specific database which caters towards ACD interactions. From our knowledge, this is the first and only known study which has identified the need for such a database in clinical practice, as well as determined the DDI parameters which pharmacy practitioners deem as clinically-relevant. The potential of a DDI database for clinical practice is definitely attractive to improve the awareness of ACD interactions among healthcare professionals, and can ultimately improve the pharmaceutical care of patients by reducing drug-related problems such as drug interactions.
Disclosures
This article is written within a project financed by the start-up grant awarded by the National University of Singapore. The authors report no conflicts of interest.
Footnotes
Acknowledgements
We would like to thank Ms. Vivianne Shih from the National Cancer Centre Singapore, and the student helpers at the 2nd APOPC for their help in the distribution and collection of the survey questionnaires.
Other parameters for inclusion in an oncology-specific drug interaction database.

| Drug interaction parameters | Hypothesized rationales of why respondents in our study suggested the parameters for inclusion |
|---|---|
| 1) Risk or incidence of the interaction |
It is known that the potential for interactions increases with the number of drugs in a prescription. 34 Cancer patients typically receive a combination of multiple chemotherapeutic agents in their treatment regimens so as to improve clinical response. 2 However, this may also pose a risk of patients manifesting drug interactions with the drugs in the regimens. |
| 2) Interactions with combination chemotherapy regimens |
Approximately 36%–56% of prescribers consider the risk of an interaction during selection of their drug products. 26 With the use of multiple anticancer drugs (ACDs) in combination regimens, prescribers should exercise more caution when selecting other drugs to be used concurrently. Thus, the database should include the risk or incidence of interaction, and also cater towards interactions with chemotherapy regimens. |
| 3) Interactions with traditional Chinese medicines (TCMs), complementary and alternative medicines (CAMs), parenteral and enteral nutritional products |
A recent survey on US community pharmacists showed that approximately half of them did not have adequate knowledge on oral chemotherapy drug interactions. 35 In another study, it was reported that oncology practitioners were generally dissatisfied with data on CAM interactions with cancer therapy. 6 In Asia, the proportion of oncology practitioners who felt inadequate about their knowledge in ACD-CAM interactions was higher (80%), with younger oncologists being more concerned about ACD-CAM interactions. 36 |
| 4) Non-interacting alternative drugs |
Since our survey population consisted of young practitioners, they may not have had adequate knowledge and experience in ACD interactions. Furthermore, a number of them were in community, hospice and outpatient practices. Thus, ACD interactions with other non-conventional medications could also be considered in the database. |
