Abstract
Epidemiology has indicated a possible increase in lung cancer among stainless steel welders. Chromium (Cr) is a primary component of stainless steel welding fume. There is an initiative to develop alternative welding consumables [nickel (Ni)- and copper (Cu)-based alloys] that do not contain Cr. No study has been performed to evaluate the toxicity of fumes generated from Ni- and Cu-based consumables. Dose-response and time-course effects on lung toxicity of a Ni- and Cu-based welding fume (Ni-Cu WF) were examined using an in vivo and in vitro bioassay, and compared with two other well-characterized welding fumes. Even though only trace amounts of Cr were present, a persistent increase in lung injury and inflammation was observed for the Ni-Cu WF compared to the other fumes. The difference in response appears to be due to a direct cytotoxic effect by the Ni-Cu WF sample on lung macrophages as opposed to an elevated production of reactive oxygen species (ROS).
Introduction
Millions of workers worldwide are exposed to welding fumes on a daily basis. Welding generates particulate matter composed of a complex of metals, some of which are toxic, such as manganese (Mn), chromium (Cr), and nickel (Ni). The presence of Cr and Ni in the fumes may cause lung injury and inflammation,1–6 lung tumor formation,7,8 immune dysfunction,8,9 and systemic toxicity. 10 Similarly, the presence of Mn in welding fumes raises concerns over its ability to cause potential adverse neurological responses11–13 as well as Parkinson's disease-like dopaminergic dysfunction. 14
Epidemiology and animal toxicology studies have shown stainless steel welding fume to be toxic to the lungs. 15 Cr is a primary component of stainless steel welding fume. Significant quantities of hexavalent (Cr6+) have been measured in fumes during stainless steel welding. Cr6+ has been classified as a human carcinogen, 16 and welding fumes that contain Cr have been shown to be mutagenic in cell-based studies.17–19 Recent epidemiology studies have indicated a possible increase in mortality from lung cancer among stainless steel welders.20–23 Because of this, recent federal legislation reduced the permissible workplace exposure limit (PEL) for Cr from 52 to 5 μg/m.3,24 Achieving this new PEL has not always been practical during repair or fabrication of stainless steel components in certain US. industries (ie, shipbuilding) where welding in confined spaces is common. 25 Moreover, local exhaust ventilation may be difficult to utilize or ineffective because many stainless steel welding operations are done in enclosed spaces or awkward positions. The best control approach may be to minimize the exposure at the source.
More research is required to help identify and develop methods by which current welding practices and processes may be modified to protect a significant number of welders while sustaining workplace productivity with minimal or no added costs. There is an initiative in the welding industry to develop new welding consumables that have the same weldability characteristics of stainless steel consumables but contain negligible amounts of Cr. New and alternative consumables [nickel (Ni)- and copper (Cu)-based alloys] have been developed. Initial laboratory studies indicate that the weld mechanical properties and weldability performance were similar in nature when comparing stainless steel and a specific Ni- and Cu-based consumable. 25 Importantly, the levels of airborne Cr were reduced by two orders of magnitude when welding with a Ni- and Cu-based welding consumable as opposed to stainless steel welding. Although Cr concentrations were reduced, significant levels of Ni, Cu, and other potentially toxic metals were present in the fume generated from the new consumable substitute. Currently, no toxicological study has been performed that has evaluated the health effects of fumes generated from the welding of Ni- and Cu-based consumables that are being proposed to serve as alternatives to Cr-containing stainless steel consumables. Exposure to elevated levels of Ni in welding fume is of concern because Ni is classified as a lung carcinogen (International Agency for Research on Cancer, 1990) and has caused lung irritation, inflammation, and adverse immune responses in humans and laboratory animals. 26
The overall objective of the study was to evaluate the potential adverse pulmonary responses of welding fume generated from a Ni- and Cu-based consumable. A comprehensive chemical and physical characterization of a Ni- and Cu-based consumable was performed. In addition, the dose-response and time-course effects of a Ni- and Cu-based welding fume on pulmonary toxicity were examined in an in vivo animal bio-assay model and compared with two other well-characterized welding fumes, one of which contained significant amounts of Cr [gas metal arc-stainless steel (GMA-SS)] and the other that did not [gas metal arc-mild steel (GMA-MS)]. Because differences in pulmonary responses were observed when comparing the different welding fumes, a mechanistic in vitro study was performed that examined the effects of the welding particles on lung macrophage viability and production of reactive oxygen species (ROS).
Materials and Methods
Welding fume samples and characterization
The two gas metal arc welding fume samples were generated and provided by Lincoln Electric Co. (Cleveland, OH, USA). Bulk samples of the welding fumes were generated in an open front fume chamber (volume = 1 m3) by a welder using an appropriate electrode and collected on 0.2 μm nuclepore filters (Nuclepore Co., Pleasanton, CA, USA). The fume samples were generated by (1) gas metal arc welding with a mild steel E70S-3 electrode (GMA-MS) and (2) gas metal arc welding with a stainless steel ER308L Si electrode (GMA-SS) using argon and CO2 shielding gases as previously described. 1 The welding fume generated from the Ni- and Cu-based consumable (Ni-Cu WF) was produced and provided by the Welding and Joining Metallurgy Group at The Ohio State University (Columbus, OH, USA) during shielded metal arc (SMA) welding using a consumable with a target composition of Ni-7.5Cu-1Ru that contained other alloying agents. The fume samples were collected onto electrostatic filter medium (PE 1306NA; Hollingsworth and Vose, East Walpole, MA, USA). The particle size of the individual welding fume samples was characterized by scanning electron microscopy and found to be of respirable size with count mean diameters of <2 μm as previously described. 1
Replicate welding fume samples were analyzed for Cr(VI) levels using NIOSH method 7605. 27 Briefly, 5 mL of extraction solution (3% Na2CO3/2% NaOH) was added to each 5 mg sample, and the tubes were sonicated in a bath for 30 minutes. This procedure extracts both soluble and insoluble Cr(VI) present in the fumes. Analysis used a Dionex HPIC-AS7 column with 250 mM (NH4)2SO4/100 mM NH4OH mobile phase and a post-column reagent (2.0 mM diphe-nylcarbazide/10% methanol/1N H2SO4) with absorbance detection at 540 nm. Four concentrations of standards were made from a certified Cr(VI) solution, covering a range of 0.4-4 μg/mL. The estimated limit of detection is 0.02 μg, and the method range is 0.05-20 μg of Cr(VI). Analysis of other elements present in the welding fume samples was performed as previously described using inductively coupled plasma-atomic emission spectroscopy (ICP-AES) using NIOSH method 7300 modified for microwave digestion. 28
In addition, portions of the different welding fume samples (GMA-SS, GMA-MS, and Ni-Cu WF) were suspended in distilled water, pH 7.4, and sonicated for one minute with a Sonifier 450 Cell Disruptor (Branson Ultrasonics, Danbury, CT, USA) to determine particle/metal solubility. The three particle suspensions (total samples) were incubated for 24 hours at 37 °C, and the samples were centrifuged at 12,000 ×
In vivo toxicology study
Animals
Male Sprague-Dawley rats from Hilltop Lab Animals (Scottdale, PA, USA), weighing 250-300 g and free of viral pathogens, parasites, mycoplasmas, Helicobacter, and CAR Bacillus, were used for all exposures. The rats were acclimated for one week after arrival and were provided HEPA-filtered air, irradiated Teklad 2918 diet, and tap water ad libitum. All animal procedures used during the study were reviewed and approved by the NIOSH Animal Care and Use Committee. The animal facilities are specific pathogen free, environmentally controlled, and accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International.
In vivo welding fume treatment
The welding fume sample was prepared in sterile saline and sonicated to disperse the particulates. Rats were lightly anesthetized by an intraperitoneal injection of 0.6 mL of a 1% solution of sodium methohexital (Brevital, Eli Lilly, Indianapolis, IN, USA) and intratracheally instilled with 0.5 and 2.0 mg/rat of GMA-MS, GMA-SS, or Ni-Cu WF in 300 μL of sterile phosphate buffered saline (PBS). Vehicle control animals were intratracheally instilled with 300 μL of sterile PBS.
To estimate how the intratracheal instillation particle dose used in the study correlated with a “real-world” worker exposure to welding fumes, the exposure was calculated. Importantly, the calculations made here do not account for particle clearance, but provides an estimate of the plausible welder exposure concentrations that our exposure paradigm mimics. The daily lung burden of a welder was estimated, assuming eight hours of continuous welding, a worker minute ventilation of 20,000 mL/minute, a particle deposition efficiency in the alveolar region of 15%, 29 and a fume concentration of 5 mg/m3 (previous threshold limit values for eight hour day for welding fume). The following calculations were used:
Fume concentration × minute volume × exposure duration × deposition efficiency
= Daily deposited dose
5 mg/m3 × (20,000 mL/minute × 10−6 m3/mL) × (8 hours × 60 minutes/hour) × 0.15
= 7.2 mg deposited per day
Next, assuming an average worker weighed 75 kg and the average weight of the rats used in the study was 0.40 kg:
7.2 mg deposited/75 kg worker = × mg/0.40 kg rat
From the doses used in the study, 0.5 and 2.0 mg:
0.5 mg/0.0384 mg = 13.0 days or ~2.6 weeks of exposure (based on five day work week)
2.0 mg/0.0384 mg = 52.1 days or ~10.4 weeks of exposure (based on five day work week)
Bronchoalveolar lavage
At 3 (
Cellular evaluation
Total cell numbers recovered by BAL were determined using a Coulter Multisizer II and AccuComp software (Coulter Electronics, Hialeah, FL, USA). Cells were differentiated using a Cytospin 3 centrifuge (Shandon Life Sciences International, Cheshire, England). Cell suspensions (5 × 104 cells) were spun for five minutes at 800 rpm and pelleted onto a slide. Cells (200/rat) were identified after labeling with Leukostat stain (Fisher Scientific, Pittsburgh, PA, USA) as lung macrophages, polymorphonuclear leukocytes or neu-trophils (PMNs), lymphocytes, and eosinophils.
Biochemical parameters of injury
Using the acellular first fraction of BALF, albumin content, an index to quantify increased permeability of the bronchoalveolar-capillary barrier, and lactate dehydrogenase (LDH) activity, an indicator of general cell damage and toxicity, were measured as previously described. 3 Albumin content was determined colorimetrically at 628 nm based on albumin binding to bromcresol green using an albumin BCG diagnostic kit (Sigma Chemical Co., St. Louis, MO, USA). LDH activity was determined by measuring the oxidation of lactate to pyruvate coupled with the formation of NADH at 340 nm. Measurements were performed with a COBAS MIRA auto-analyzer (Roche Diagnostic Systems, Montclair, NJ, USA).
In vitro lung macrophage study
In vitro welding fume preparation
For primary rat macrophage treatment experiments, welding fume particles were suspended in sterile filtered 1 × PBS at a 1 mg/mL stock, vortexed, and diluted into cell culture media at a low (50 μg/mL) or high dose (250 μg/mL), based on previous in vitro studies of GMA-MS and GMA-SS fumes.30,31 Welding fume stocks were prepared freshly for each experiment. Equal volumes of 1 × PBS were used as control conditions for each experiment.
BAL and alveolar macrophage isolation
Before BAL, untreated rats were deeply anesthetized with an overdose of sodium pentobarbital (>100 mg/kg body weight; Sleepaway, Fort Dodge Animal Health, Wyeth, Madison, NJ, USA) and then exsanguinated by severing the abdominal aorta. The lungs were then lavaged with three separate 6 mL volumes of calcium- and magnesium-free PBS. The recovered samples were centrifuged, and the supernatants were discarded. Cell pellets were resuspended in 1 mL PBS, and cell counts were performed using a Coulter Multisizer II and AccuComp software (Coulter Electronics, Hialeah, FL, USA) to determine the number of lung macrophages in each sample. The cells were then centrifuged again, and pellets were resuspended at the desired macrophage concentration, depending on the assay. For the intracellular ROS and viability assays, cells were resuspended in warm minimal essential medium (MEM) with Eagle's salts, 10% fetal bovine serum, and 50 mg/mL penicillin/streptomycin (Invitrogen Life Sciences, Grand Island, NY, USA). Cells were then plated at 2 × 105/well in 96-well dishes and incubated at 37 °C in a 5% CO2 incubator for one hour to allow attachment. Before treatment with welding fume suspensions, cells were washed three times with medium to remove unattached cells from the wells.
Electron spin resonance
Electron spin resonance (ESR) spin trapping was used to detect short-lived free radical intermediates. Hydroxyl radicals were measured using the addition-type reaction of a short-lived radical with a compound (spin trap) to form a relatively long-lived paramagnetic free radical product (spin adduct), which can then be studied using conventional ESR. For radical measurements, freshly lavaged rat alveolar macrophages were suspended in PBS at a concentration of 1 × 106/mL and mixed with welding fumes (1 mg/mL) in a suspension of PBS and the spin trap 5,5-dimethyl-1-pyrroline-
Intracellular ROS
Plated lung macrophages were treated with the cell-permeable fluorogenic probe DCFH-DA (2′,7′-dichlorodihydrofluorescein diacetate, Cell Bio-labs, Inc., San Diego, CA, USA) at a final concentration of 1 mM in serum-free MEM for 45 minutes at 37 °C. Cells were washed twice with PBS, and MEM was added back to the wells, along with 50 μg/mL or 250 μg/mL welding fume suspensions. Upon ROS production, DCFH-DA is oxidized to form DCF, which is highly fluorescent. The cells were incubated at 37 °C for four hours, and plates were read at 485 nm excitation/530 nm emission to measure any change in fluorescence, indicating ROS production. To ensure that the fluorescent signal was due to DCF product and not autofluorescence that may emit from the welding fume suspensions, separate wells of MEM and each fume were included in the plates, and these readings were subtracted from their respective wells that contained treated cells. All conditions were run in triplicate wells per rat and macrophages from five rats were used. All values were normalized to represent the fold-change over PBS-treated control wells.
Macrophage viability assay
Plated lung macrophages were treated with 50 or 250 μg/mL welding fume suspensions for 4 and 24 hours. Changes in cell viability at each time point were assessed using the MultiTox-Fluor Cytotoxicity Assay according to the manufacturer's directions (Promega, Madison, WI, USA). A cell-permeable protease substrate, GF-AFC (glycyl-phenylalanyl-amino-fluorocoumerin), is cleaved by live cells to produce fluorescent AFC. Thus, the fluorescent signal is proportional to the number of viable cells. To ensure that the fluorescent signal was due to substrate products and not autofluorescence or interference by the welding fume suspensions, separate wells of MEM and each fume were included in the plates, and these readings were subtracted from their respective wells that contained treated cells. All conditions were run in triplicate wells per rat, and macrophages from five rats were used.
Statistical analysis
Results are means ± standard error of measurement. Statistical analysis was performed using GraphPad Prism 6 Software (La Jolla, CA, USA). For the
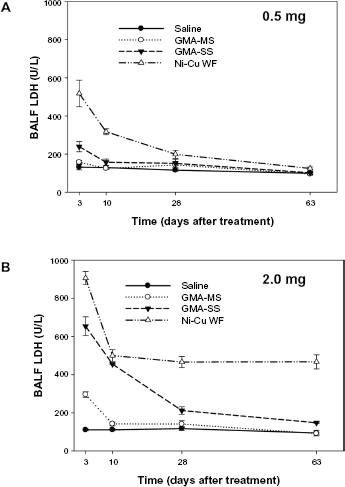
LDH activity in acellular BALF at 3, 10, 28, and 63 days after intratracheal instillation of (A) 0.5 mg or (B) 2.0 mg of GMA-MS, GMA-SS, or a Ni-Cu WF. Control animals were treated with sterile saline. Values are means ± standard error (

Total albumin in acellular BALF at 3, 10, 28, and 63 days after intratracheal instillation of (A) 0.5 mg or (B) 2.0 mg of GMA-MS, GMA-SS, or a Ni-Cu WF. Control animals were treated with sterile saline. Values are means ± standard error (
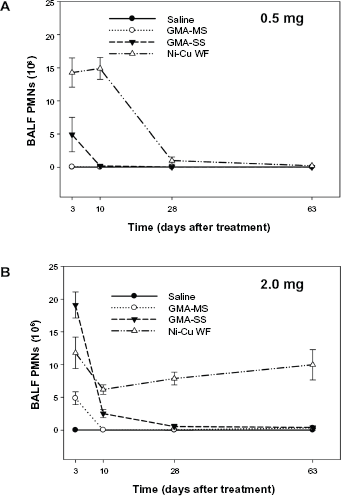
Total number of neutrophils (PMNs) recovered from BALF at 3, 10, 28, and 63 days after intratracheal instillation of (A) 0.5 mg or (B) 2.0 mg of GMA-MS, GMA-SS, or a Ni-Cu WF. Control animals were treated with sterile saline. Values are means ± standard error (

Total number of lung macrophages recovered from BALF at 3, 10, 28, and 63 days after intratracheal instillation of (A) 0.5 mg or (B) 2.0 mg of GMA-MS, GMA-SS, or a Ni-Cu WF. Control animals were treated with sterile saline. Values are means ± standard error (
Results and Discussion
The pulmonary toxicity potential of three chemically distinct welding fumes was examined and compared in an animal bioassay. The animals were treated by intratracheal instillation using two doses (0.5 and 2.0 mg/rat) of the different welding fume samples. These doses were chosen based on results from a previous dose-response welding fume study. 1 In the previous study, it was found that an intratracheal instillation welding fume dose of 0.2 mg/rat (regardless of fume type) induced no pulmonary response, whereas doses ≥5 mg/rat caused massive lung inflammation and prominent particle lung burden in all fumes studied. One goal of the current study was to choose two doses that fell between the 0.2 and 5.0 mg/rat dose range used in the earlier study.
Our previous study also demonstrated that stainless steel welding fume was more pneumotoxic and persisted in the lungs longer than a mild steel welding fume after treatment with a dose of 2.0 mg/rat. 1 The difference in pulmonary response was likely due to the difference in chemical composition of the two fumes. It was not surprising that the stainless steel fume generated the greater pulmonary response as it contained two highly cytotoxic metals, Cr and Ni, which were not present in the mild steel fume. The study revealed that the absence of a persistent pulmonary response to mild steel was similar to what was observed after the intratracheal instillation of iron oxide, a metal particle which has been characterized as a nuisance particle with little inflammatory and toxicity potential in the lungs. The overall goal of the current study was to compare the pulmonary responses of a newly developed Ni-Cu WF that contained a minimal amount of Cr with a Cr-containing stainless steel fume (GMA-SS) that is known to cause lung injury and inflammation in an animal model and a mostly non-toxic mild steel welding fume (GMA-MS) that contained no Cr.
The welding fume samples were chemically and physically characterized (Table 1). The fume samples were quite different in chemical composition but were similar in size as the particles were in the respirable size range with count mean diameters of <2 μm. Because the fumes were collected in bulk onto filters, the count mean diameter are somewhat larger than the mass mean aerodynamic diameter (MMAD) of welding fume collected during generation, which is generally between 0.2 and 0.4 μm.2,3,33–35 In addition, all three welding fumes were observed to be relatively insoluble with soluble/insoluble ratios of 0.006, 0.020, and 0.004 for the GMA-MS, GMA-SS, and Ni-Cu WF samples, respectively. Owing to the similarity in particle size and solubility of the fume samples collected for the current study, any observed difference in pulmonary responses would likely be attributed to the metal profile of the fumes.
Welding fume characterization.

The GMA-SS fume was composed of mostly Fe (57%) with significant amounts of Cr (20%), Mn (14%), and Ni (9%) present, whereas the GMA-MS sample was composed of only Fe (83%), Mn (15%), and small amounts of Cu (Table 1). The chemical composition of the Ni-Cu WF was more complex than the two GMA fumes, and likely because of the presence of fluxes incorporated into the electrode as it was generated during SMA welding. In GMA welding, the electrode is bare, containing no coating or core. The shielding is supplied by a combination of inert gases and protects the weld from reacting with constituents of the atmosphere. In SMA welding, the shielding is provided by decomposition of the electrode covering that contains fluxing agents and scavengers to protect the weld from oxidation by reaction with atmosphere components. Because of the use of fluxes, several alkali metals were present in the Ni-Cu WF, such as potassium (K), strontium (Sr), magnesium (Mg), calcium (Ca), and barium (Ba). Besides Ni and Cu, other prominent metals present in the Ni-Cu WF included aluminum (Al) and titanium (Ti). Importantly, the Ni-Cu WF had substantially less total Cr and Cr6+ compared to the GMA-SS fume (Table 1).
In comparing the pulmonary responses of the three welding fumes, different parameters of lung injury and inflammation were examined at 3, 10, 28, and 63 days after treatment with the 0.5 or 2.0 mg/rat doses. In the assessment of lung injury, LDH and albumin were measured in the acellular fraction of BALF recovered from the treated animals (Figs. 1 and 2). For both parameters, the responses were similar. At the lower dose (0.5 mg), the Ni-Cu WF group induced a statistically significant increase (
In the evaluation of lung inflammation, PMNs and lung macrophages were recovered by BAL and counted at multiple time points after exposure to the different welding fume samples (Figs. 3 and 4). After treatment with the lower fume dose (0.5 mg), significantly more (
Based on the findings from the current animal lung toxicology study, the Ni-Cu WF has the potential to induce adverse pulmonary responses in exposed workers after inhalation. Lung inflammation and injury were significantly elevated compared to the reference welding fume samples (GMA-MS and GMA-SS) at early time points after treatment with 0.5 mg of the Ni-Cu WF sample. Importantly, and unlike the response to the GMA-MS and GMA-SS fume samples, the significant increase in inflammation and injury was observed to persist over a 2 month period when the dose of the Ni-Cu WF sample was increased to 2.0 mg. In regards to the reference welding fume samples, the results from the current study confirm previous observations; in that the GMA-MS fume at the higher dose had only a slight transient effect on lung injury and inflammation and no effect at the lower dose. This finding correlated well with earlier studies that examined the pulmonary effects of GMA-MS fume after a single intratracheal instillation1,4 or after exposure to a relatively high concentration of 40 mg/m3 of fume by inhalation for 10 days. 35 These results and findings from the current study suggest that the pneumotoxic potential of GMA-MS fume is quite low. As was observed previously, intratracheal instillation of the GMA-SS fume induced significant lung injury and inflammation early after treatment with the response returning to control values for most parameters by four to five weeks. 1 Previous studies have shown stainless steel welding fume to be highly reactive and to enhance leukocyte production of ROS and inflammatory cytokines in comparison to the GMA-MS fume.31,36,37 However, unlike what was observed in the persistence of the adverse lung effect caused by the Ni-Cu WF sample, it has been demonstrated in animal studies that the potential for chronic pulmonary toxicity of the GMA-SS fume is low if exposure is ceased. 1
Because of the observed differences in pulmonary response among the three welding fumes, an
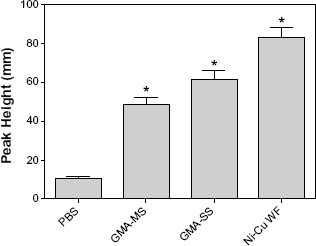
The generation of short-lived hydroxyl radical (•OH) upon reaction of rat lung macrophages treated with different types of welding fume. ESR spectrum recorded after five minute incubation at 37 °C and filtration. Sample contained rat lung macrophages (1 × 106/mL), welding fume (1 mg/ml), 100 mM DMPO in PBS (pH 7.4). ESR settings were center field, 3468 G; scan width, 100 G; time constant, 40 milliseconds; scans, 5; modulation amplitude, 1 G; receiver gain, 1 × 104; frequency, 9.748 GHz; and power, 127 mW. Values are means ± standard error of measurement (
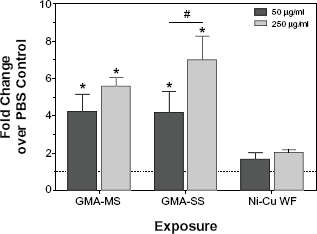
Intracellular ROS production in rat lung macrophages following treatment with welding fumes. Plated rat lung macrophages were pre-treated with DCFH-DA, exposed to welding fumes, and plates were read after four hours to measure intracellular ROS. values are means ± standard error of measurement (
In the examination of cytotoxicity (Fig. 7), all three welding fumes reduced lung macrophage viability within four hours at the high dose (250 μg/mL), and there was no significant effect at the low dose (50 μg/mL). By 24 hours, however, Ni-Cu WF exposure caused significant loss of viability (
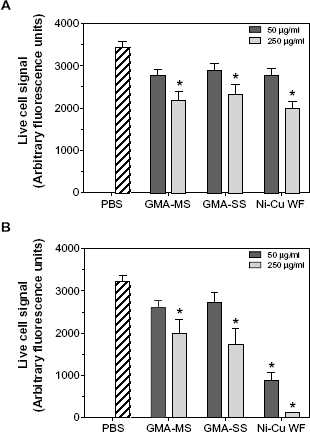
Rat lung macrophage viability following treatment with welding fumes. Plated rat lung macrophages were treated for (A) four hours or (B) 24 hours with welding fumes, then treated with MultiTox-Fluor reagent for one hour at 37 °C, and plates were read at 400ex/505em to determine fluorescence signal from live cells. Values are means ± standard error of measurement (
Conclusions
The toxic potential of welding fume is highly dependent on metal composition. Metal solubility does not appear to play a significant role in the pulmonary responses observed in this study as all three fume samples were determined to be relatively insoluble. The observed increase in lung injury and inflammation are likely due to the presence of cytotoxic metals (eg, Cr and Ni) in the GMA-SS fume, which were not present in GMA-MS fume. Even though only trace amounts of Cr were present, the lung toxicity potential of the Ni-Cu WF sample as determined by our animal bioassay was the greatest among the three welding fumes because of an elevation in response early after treatment as well as the persistence of the response over time. Based on our
Author Contributions
JMA, MAB, SSL, and JRR conceived and designed the experiments. JMA, MAB, TGM, MK, SSL, and JRR analyzed the data. JMA wrote the first draft of the manuscript. MAB, MK, TGM, SSL, and JRR contributed to the writing of the manuscript. JMA, MAB, TGM, MK, SSL, and JRR agreed with manuscript results and conclusions. JMA, MAB, SSL, and JRR jointly developed the structure and arguments for the paper. JMA, MAB, TGM, MK, SSL, and JRR made critical revisions and approved the final version. All authors reviewed and approved the final manuscript.
Disclaimer
The findings and conclusions of this paper have not been formally disseminated by NIOSH and should not be construed to represent any agency determination or policy.
Footnotes
Acknowledgments
The authors would like to gratefully acknowledge Dr. Boian Alexandrov of the Welding and Joining Metallurgy Group at The Ohio State University for providing the Ni-Cu WF. The authors also thank Natalie Fix and Katherine Dunnick for their technical assistance during the study.
