Abstract
Background:
Atrial fibrillation (AF) is the most common arrhythmia globally and is increasingly recognized as being influenced by environmental exposures. While air pollution is a well-established risk factor for cardiovascular disease, its specific relationship to AF remains undetermined. This systematic review aims to synthesize current evidence on the association between air pollution and AF, identifying key pollutants, exposure patterns, and population vulnerabilities.
Methods:
Following PRISMA guidelines, a comprehensive search of PubMed, Embase, CINAHL, Web of Science, and other databases was conducted to identify cohort, case-crossover, and time-series studies examining the effects of ambient air pollutants (PM2.5, PM10, NO2, SO2, CO, and O3) on AF outcomes in adults. Data on study design, pollutant levels, AF metrics, and population characteristics were extracted and the Joanna Briggs Institute Critical Appraisal Checklist was used to evaluate the selected study quality.
Results:
Thirty-one studies met inclusion criteria, encompassing over 63 million participants across North America, Europe, and Asia. PM2.5 was the most studied and most consistently associated pollutant, with 21 of 29 studies showing significant positive associations with AF risk. Several studies identified elevated AF incidence even at pollutant levels below WHO air quality guidelines. Subgroup analyses revealed that individuals with comorbidities (eg, diabetes, obesity, hypertension), women, and older adults may be more vulnerable to pollution-related AF. Study methodologies varied widely in terms of pollutant measurement, AF detection, and temporal resolution, affecting comparability.
Conclusions:
This review affirms a significant association between air pollution, particularly PM2.5 and NO2, and the risk of atrial fibrillation. The findings suggest that even pollutant concentrations deemed “safe” may increase AF risk, particularly in vulnerable populations. Future research should standardize exposure assessment and explore understudied pollutants and effect modifiers. These results underscore the importance of air quality regulations as a public health intervention to reduce AF burden and support cardiovascular health.
Plain Language Summary
Atrial fibrillation (AF), the most common sustained cardiac arrhythmia, is associated with increased risk of stroke, heart failure, and mortality. Recent evidence suggests that environmental exposures, particularly air pollution, may play a significant role in AF incidence and exacerbation. This narrative review synthesizes the current literature linking air pollution, especially fine particulate matter (PM2.5), nitrogen dioxide (NO2), and ozone (O3), to AF pathogenesis and clinical outcomes. Several population-based and time-series studies demonstrate a consistent association between short- and long-term exposure to PM2.5 and increased rates of hospital visits, emergency department utilization, and AF episodes. Proposed pathophysiologic mechanisms include systemic inflammation, oxidative stress, endothelial dysfunction, autonomic imbalance, and direct myocardial effects. Vulnerable populations, including older adults, individuals with pre-existing cardiovascular conditions, and those in socioeconomically disadvantaged or highly polluted areas, appear to face heightened risk. This review emphasizes the need for integrated cardiovascular and environmental health surveillance and highlights a critical gap in physician awareness and training regarding environmental determinants of cardiovascular disease. Policy solutions, including improved emissions regulation and patient-centered interventions like air filtration and public health advisories, are discussed. In light of growing evidence, AF should be recognized not only as a clinical and public health priority but also as an environmentally influenced condition, requiring transdisciplinary collaboration across cardiology, public health, environmental science, and policy.
Introduction
Air pollution is now recognized as one of the greatest threats to human health globally, with its impact extending across nearly every organ system. Among the various pollutants, fine particulate matter, especially particles less than 2.5 microns in diameter (PM2.5), has been identified as a leading contributor to the global burden of disease, surpassing other more widely recognized risk factors such as tobacco use, poor diet, and hypertension. 1 Impacts are largely driven by systemic absorption of these microscopic particles, triggering inflammation, oxidative stress, endothelial dysfunction, and autonomic imbalance. The result is a sharp increase in adverse cardiovascular outcomes, including ischemic heart disease, stroke, heart failure, and arrhythmias.2,3
In urban settings around the world, air pollution has emerged as a primary environmental determinant of cardiovascular morbidity and mortality. 2 Studies consistently show that exposure to PM2.5, nitrogen dioxide (NO2), and ozone (O3) is linked to increased rates of hospitalization, emergency department visits, and death from cardiovascular causes. Although the Clean Air Act of 1970, enforced by the Environmental Protection Agency (EPA), has significantly reduced the levels of key air pollutants in the United States, contemporary exposure remains far from safe.3,4 Current research suggests that even PM2.5 concentrations below the EPA’s existing thresholds can be harmful, particularly for vulnerable populations including older adults, people with pre-existing cardiovascular conditions, and those living in socioeconomically disadvantaged areas. 5
Fossil fuel combustion continues to be the dominant source of anthropogenic air pollution and is estimated to be responsible for approximately 10.2 million premature deaths per year, with a substantial proportion attributable to cardiovascular disease. 6 These deaths are preventable and highlight a critical intersection between environmental policy, energy systems, and public health. In this context, reducing fossil fuel reliance and improving air quality are not only environmental imperatives but also key strategies for cardiovascular disease prevention and global health equity.
Emerging mechanistic evidence provides additional context for why air pollution may precipitate or exacerbate atrial fibrillation. Fine particulate matter and gaseous pollutants can induce oxidative stress and systemic inflammation, which in turn promote atrial structural remodeling, including atrial fibrosis and cardiac remodeling, a key substrate for AF initiation and maintenance. Pollutants also disrupt autonomic nervous system balance, leading to heightened sympathetic activity, reduced heart rate variability, and increased vulnerability to triggered electrical activity. 7 The underlying mechanisms by which air pollution contributes to cardiovascular damage begin with pulmonary irritation, initiating a cascade involving activation of the sympathetic nervous system and a subsequent “spillover” effect, whereby pollutants penetrate the pulmonary barrier and trigger systemic responses including inflammation, endothelial dysfunction, and thrombosis. 3 Particulate matter (PM) exposure, especially PM2.5, has been associated with the development of hypertension and other forms of cardiovascular disease. In addition, several studies have demonstrated that air pollution negatively impacts heart rate variability (HRV), a key indicator of autonomic nervous system balance. Reduced HRV is a known risk factor for cardiovascular morbidity and mortality, further emphasizing the connection between pollution exposure and cardiovascular risk. 8 Collectively, these biologic pathways offer a coherent explanation for the epidemiologic associations observed between air pollution and AF, underscoring why even low-level exposures may be clinically relevant.
Given the public health implications of both air pollution and AF, this review was conducted to systematically evaluate the existing evidence on the association between air pollution and AF. Specifically, this review aimed to answer the following questions: (1) What are the effects of air pollution on AF? (2) Which air pollutants are associated with the development or exacerbation of AF?
Methods
The review was prospectively registered in PROSPERO (CRD42022368145). This systematic review has been conducted under the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) and registered in Prospero.
Data Source and Search Strategies
We searched the databases of PubMed, Embase, Cumulative Index to Nursing and Allied Health Literature (CINAHL), Web of Science, Google Scholar, Greenfile, and Science Direct for all relevant studies. The following keywords were used to guide the search: “arrhythmia,” “dysrhythmia,” “ventricular fibrillation,” “atrial fibrillation,” “atrial flutter,” “ventricular tachycardia,” “atrial tachycardia,” “bradycardia,” “bundle branch block,” “heart block,” “long QT syndrome,” “premature atrial contraction,” “premature ventricular contraction,” “atrial tachycardia,” “sinus tachycardia,” “sick sinus syndrome,” “sinus arrhythmia,” “Wolff-Parkinson-White syndrome,” “Adams-Stokes disease,” “air pollution,” “particulate matter,” “ozone,” “carbon monoxide,” “nitrogen dioxide,” “sulfur dioxide,” “PM2.5,” “PM10.” The full strategy is listed in The Open Science Framework (https://osf.io/nwmy3/files/osfstorage).
Inclusion and Exclusion Criteria
Studies were included if they met the following criteria: (1) studies examining the short- or long-term effects of ambient air pollutants (eg, PM2.5, PM10, nitrogen dioxide, sulfur dioxide, carbon monoxide, and ozone) on atrial fibrillation; (2) studies examining the short-term association must be time-series or case-crossover studies; (3) studies providing the relative risks (RR), odds ratios (OR), or hazard ratio (HR) and the corresponding 95% confidence intervals; (4) studies published in peer-reviewed English language journals.
Exclusion criteria were as follows: (1) studies that do not include arrhythmia as an outcome; (2) review articles or letters; (3) conference abstract; (4) studies that look at other environmental risk factors such as heat, extreme weather conditions, other forms of pollution, and toxicity.
Study Selection
Two independent reviewers (CY and RM) screened titles or abstracts of studies from the electronic search for potential studies. Any discrepancies between the 2 reviewers were resolved by consultation of a third reviewer (EC). Reviewers verified use of eligible studies by accessing full articles and evaluating articles based on inclusion criteria.
Data Extraction
Preliminary data was first extracted by 2 reviewers (CY and RM) that included title, author, year, journal, country of origin, study design, type of arrhythmia, and type of air pollutants. From there, data focused on atrial fibrillation was extracted by multiple reviewers (CY, RM, ZC, and JK). After data extraction by 1 reviewer, a second reviewer reassessed and extracted any additional data if needed.
A structured data collection form was used to derive the following information from each study: title; year the study was conducted; name of the first author; publication year; country where the study was conducted; demographic and characteristics data of subjects (gender, age); study design; data source; exposure measurement; sampling method; diagnostic criteria defined by individual studies for AF/atrial arrhythmias; accuracy, sensitivity and specificity of the device in diagnosing AF/atrial arrhythmias. We additionally assessed study strengths and weaknesses. The JBI Sumari Critical Appraisal Checklist was used to assess the quality of the selected studies (Figure 1). Patients and the public were not involved in any way of this systematic review.

Flowchart diagram showing results of the systematic literature search.
Results
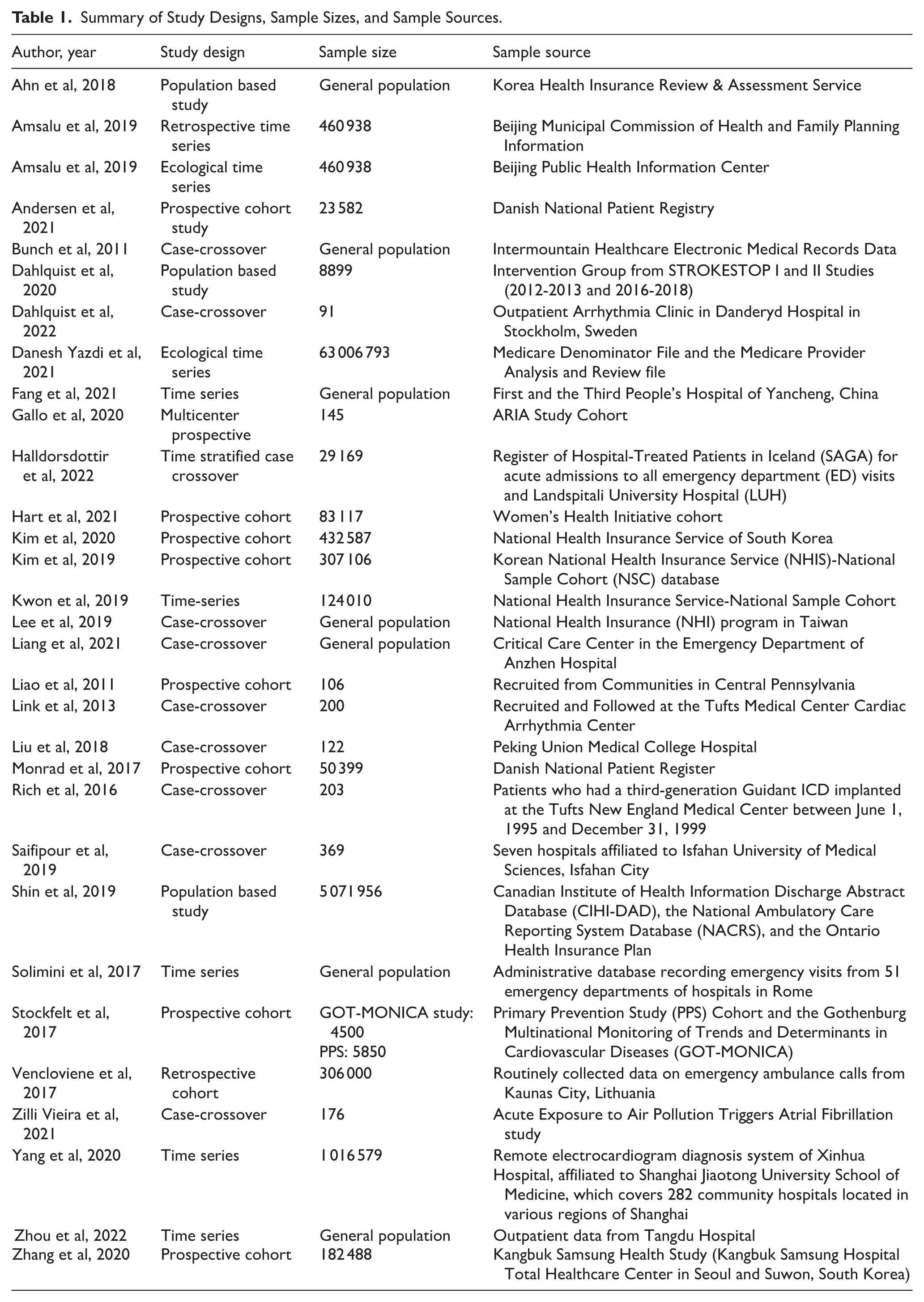
Thirty-one studies met inclusion criteria, encompassing over 63 million participants across North America, Europe, and Asia. PM2.5 was the most studied pollutant (Table 1). The studies spanned several regions (Table 2). In North America, there were 7 studies conducted in the United States and 1 in Canada.9-16 In Europe, there were 2 studies from Denmark, 3 from Sweden, 2 from Italy, 1 from Iceland, and 1 from Lithuania.17-25 In Asia, 7 studies were conducted in China, 5 in South Korea, 1 in Iran, and 1 in Taiwan.26-39 The included studies employed a variety of designs, including cohort studies, case-crossover studies, and time-series analyses. Of the 13 cohort studies, 10 were prospective,11,12,17,18,20,22,24,29,30,37 and 3 were retrospective in nature.15,25,26 There were 10 case-crossover studies,9,13,14,16,19,21,32-35 and 8 time-series studies.10,23,27,28,31,36,38,39
Summary of Study Designs, Sample Sizes, and Sample Sources.
Summary of Studies and Pollutants.

ED + Outpatient = 1; Admission + Outpatient = 3; Admission + ED + Outpatient = 1.
Mixed: combination of >2 outpatient visits, hospitalizations, and ED visits.
Sampling methods varied significantly across studies (Table 1). Seven studies utilized data from the general population, often sourced from health insurance claims or electronic medical records.15,17,22,31,32,40,41 These studies did not report total population numbers. Eight studies used samples from existing cohort studies,11,16,18-20,24,37 while twelve studies relied on patient data from insurance registries or hospital admissions.10,15,17,21,22,28-32,35,37 Two studies gathered retrospective data from cardiovascular hospital admissions,27,39 1 from a national patient registry, 17 5 from insurance registries,15,26,29,31,32 1 from ambulance call records, 25 1 from a remote EKG diagnosis system, 36 and 1 from emergency room data. 23 While many studies restricted recruitment to a single site type, 12 studies consisted of a mix of inpatient, outpatient, and emergency department sites. Five studies actively recruited participants: 1 study recruited participants from an outpatient clinic for ICD (implantable cardioverter-defibrillator) data collection, 2 recruited ICD patients directly, and 2 recruited inpatients from hospitals12-14,34 (Table 1).
Of these, 10 studies reported participant age using the mean and standard deviation, while another 10 studies provided age breakdowns by percentage.9,17,19,25,28,31,33-35,37 The mean age of participants across studies ranged from the 50s to 60s. The gender distribution was predominantly male, with 17 of the 31 studies reporting male-majority samples.9,13,14,16,19-21,24,26,27,29-32,37-39 Two studies exclusively included female participants.11,17 In 7 studies, the gender breakdown was based on the number of AF events rather than the total sample size, as the sample number was not provided.12,14,21,23,32,33,35,38
The characteristics of study participants, including age distribution and male-to-female ratios, are detailed in Supplemental Appendix 1. One study specifically examined patients aged 75 to 76 years from the STROKESTOP I study, in addition to the general population from STROKESTOP II, with both datasets combined for final analysis.18,19 Mean participant ages ranged from the 50s to 70s across studies. Some studies, such as Stockfelt 2017, utilized data from multiple cohorts, including the Multinational Monitoring of Trends and Determinants in Cardiovascular Diseases (MONICA) project and the Primary Prevention Study (PPS). 24 While most studies had a near-equal gender distribution, 2 studies focused exclusively on female subjects.11,17
Study Quality and Risk of Bias
The quality of each selected study was adequate with limited risk of bias using the Joanna Briggs Institute (JBI) appraisal tool (Table 3).
Significant Findings for Included Studies and JBI Appraisal Scoring.

Statistics are provided in the full version of the table (Supplemental Appendix 2).
Air Pollution Exposure Assessment
The studies varied in methods of air pollution measurement, ranging from using multiple monitoring stations27,39 to relying on a single central location within a city 19 (Table 4). Some studies measured exposure at the zip code level, 10 while others used participants’ residential addresses to estimate exposure.22,29,39 One study uniquely measured individual exposure in real time, linking elevated PM2.5 levels with P-wave complexity and prolonged PR intervals—both predictors of AF. 12 Additionally, the frequency of air pollutant measurements varied from annual, daily, and hourly to real-time data. Air pollutant concentrations were most reported as averages with standard deviations, but some studies provided additional metrics such as ranges, medians, and interquartile ranges (IQRs).
Descriptive Statistics of Studied Pollutants.

Original units from studies were retained. While 1 µg/m3 is equal to 0.001 ppb and both units measure density, they differ in scale and the conversion factors depend on the molecular weight of the substance being measured. Thus, while they represent the same mass concentration, they are used in different contexts depending on the scale of measurement.
Different means are reported from different areas in Utah (SLC = Salt Lake City).
Atrial Fibrillation Event Reporting
The metrics for reporting AF events varied. Some studies reported the number of events such as hospitalizations, emergency room visits, outpatient visits, or electrocardiogram (EKG) and ICD-detected events (Table 4; Supplemental Appendix 1). The total number of AF events is recorded in Supplemental Appendix 2. In a few studies, the number of AF episodes detected by ICDs was averaged per patient. Three studies specified first-time AF events such as initial hospitalizations, emergency room visits, or outpatient encounters.29-31 Twenty-two studies included data on comorbidities, and subgroup analyses were often conducted to explore associations between pollutants and AF in different demographic and health categories, such as sex, age, body mass index (BMI), and the presence of other comorbidities.9,11-13,15,16,18-20,22-24,26,29-32,34,35,37,42,43
Air Pollutant and Atrial Fibrillation Associations
Significant associations between air pollution and AF outcomes are summarized in Table 4, with detailed statistics available in Supplemental Appendix 2 Particulate matter (PM2.5) was the most commonly studied pollutant, featured in 29 of the 31 studies. Among these, 21 studies reported statistically significant associations, with odds ratios (OR) greater than 1 and 95% confidence intervals. PM2.5 was also linked to changes in cardiac electrophysiology, such as increased P-wave complexity and PR interval duration. 12 Exposure to PM2.5 was typically reported as annual averages, but in some studies, 24-hour averages18,19 or 12- to 36-month periods 14 were used.
Other pollutants were also frequently studied, including nitrogen dioxide (NO2), ozone (O3), and sulfur dioxide (SO2). NO2 was investigated in 23 studies,10,11,13-15,17-19,21-24,26-28,30-32,34,35,38,39,44 with 15 showing significant associations with AF. Ozone was studied in 16 articles,10,13-15,18,19,26-28,30-32,34-36,38,39 of which 9 reported significant findings, while SO2 was examined in 15 studies,13,14,21,26-28,30-32,34-36,38,39 with 7 showing significant associations. PM10 was studied in 16 articles,13,18-21,23-26,28,30-32,34-38 with significant results found in 10 studies. O3 was commonly measured as a daily maximum 8-hour average, while PM10 was often reported similarly to PM2.5, with annual averages. In studies by Dahlquist et al, all pollutants were measured as 24-hour averages.18,19 Some studies, such as Danesh 2021, reported exposure distribution across person-years for PM2.5, NO2, and O3. 10 Although most studies reported odds ratios (OR), 2 studies used hazard ratios (HR) to assess the association between air pollution and AF outcomes29,30 (Supplemental Appendix 1).
Other Factors Affecting Atrial Fibrillation
Several studies revealed that, while no direct overall significance between air pollutants and AF episodes was found, specific demographic factors (ie, age) impacted AF. For instance, 1 study identified myocardial infarction as a significant effect modifier for the association between air pollution and AF in patients across inpatient, outpatient, and emergency settings. 17 Additionally, while older age is typically associated with a higher risk of arrhythmia, Yang observed an increased AF risk in patients younger than 65 years, suggesting that factors beyond age-related immune weakness may contribute to AF risk. 36 Furthermore, Dahlquist et al, reported a significant association between PM10 and AF incidence in diabetic patients compared to non-diabetics, while O3 was significantly associated with AF in patients who were diabetic or overweight. 18
The temporal relationship between air pollution exposure and AF onset was explored with a study which found that emergency ambulance calls for AF were more frequent in the morning and early afternoon, with carbon monoxide (CO) as a key factor during these periods. PM10, on the other hand, was associated with AF episodes occurring late in the evening and early morning during the summer, suggesting that time of day and seasonality may influence the relationship between air pollution and AF. 25
Discussion
Our findings contribute to the growing body of evidence linking air pollution exposure to an increased risk of atrial fibrillation (AF), a common and serious cardiac arrhythmia with significant clinical and public health consequences. While much of the prior research has focused on traditional AF risk factors such as hypertension, obesity, and diabetes, emerging data—including this analysis—suggest that environmental exposures, particularly ambient air pollution, may play a critical and underrecognized role in AF pathogenesis. Fine particulate matter (PM2.5) was found to be the air pollutant most consistently and significantly associated with episodes of atrial fibrillation (AF). These findings supported the proarrhythmic effects of PM2.5, particularly by studies using electrocardiogram (EKG) monitoring or implantable cardioverter-defibrillators (ICDs) for AF detection.
Even in studies that did not achieve statistical significance, strong positive associations between air pollution and AF were frequently observed. For instance, Zhou (2022) reported a statistically significant association for nitrogen dioxide (NO2), while other pollutants also demonstrated positive but non-significant trends. 17 Similarly, Zhang (2020) identified positive correlations across all measured pollutants, and Rich (2016) found associations between sulfur dioxide (SO2), NO2, carbon monoxide (CO), and paroxysmal AF, with ozone (O3) being the only pollutant to reach statistical significance.14,37 A particularly noteworthy finding from Dahlquist (2022) was the observed positive association between PM2.5 and AF at exposure levels below the World Health Organization (WHO) air quality guidelines. 19 This raises important concerns about the adequacy of current “safe” thresholds, suggesting that even low-level exposure to PM2.5 may increase AF risk. 4 These results highlight the need to reevaluate air quality standards and strengthen public health protections, especially for vulnerable populations. Although the majority of studies focused on PM2.5, PM10, O3, NO2, and SO2, a few studies explored less common pollutants such as black carbon (BC) and oxidants (Ox). Zilli Vieira et al demonstrated significant results for BC, which highlights the need for further investigation into a wider range of pollutants beyond the commonly studied ones. 16 These findings suggest that other environmental toxins could also play a role in the development of AF and should be included in future research.
Demographic factors (eg, age,) and existing health conditions (eg, diabetes or overweight) were important factors correlated with risk of AF.17,18,36 These findings underscore the importance of accounting for individual patient characteristics in assessing AF risk due to air pollution exposure and suggest subpopulations with greater susceptibility to poor air quality. This review also found that emergency ambulance calls for AF were more frequent in the morning and early afternoon, with carbon monoxide (CO) as a key factor during these periods. 25 PM10, on the other hand, was associated with AF episodes occurring late in the evening and early morning during the summer, suggesting that time of day and seasonality may influence the relationship between air pollution and AF.
Implications and Future Directions
These associations have important implications for healthcare delivery and health policy. First, it highlights the need to integrate environmental risk assessment into cardiovascular disease prevention strategies. Healthcare providers, especially those managing patients with or at risk for AF, should be aware of the cardiovascular effects of air pollution and incorporate environmental exposure histories into clinical decision-making. Education about minimizing personal exposure—such as staying indoors on high pollution days or using air purifiers—may be a useful adjunct to traditional medical management, especially for vulnerable populations such as older adults and those with preexisting cardiovascular disease.
From a policy perspective, these findings underscore the urgency of enforcing and strengthening air quality regulations. The Clean Air Act has led to significant improvements in U.S. air quality over the past several decades, but current standards may not fully account for the cardiovascular risks posed by fine particulate matter (PM2.5) and other pollutants. 45 Policymakers should consider revisiting the National Ambient Air Quality Standards (NAAQS) and other guidelines to align with the latest scientific evidence demonstrating health harms at lower pollution levels. Moreover, regulatory frameworks should explicitly address the cumulative burden of air pollution in more vulnerable and disadvantaged communities, where exposures are often higher and healthcare resources more limited. 46
Beyond environmental regulation, healthcare systems have a role to play in addressing air pollution as a modifiable health determinant.47-50 Hospitals and health organizations can advocate for cleaner transportation, renewable energy adoption, and community air monitoring initiatives. Integrating environmental health indicators into electronic medical records and public health surveillance could help identify at-risk populations and guide targeted interventions. Additionally, further research is warranted to evaluate whether improved air quality can reduce the incidence or severity of AF and related complications, such as stroke and heart failure.
Future research should focus on mean annual concentrations of air pollutants and regional weather conditions to better understand the influence of climate on the association between air pollution and AF. Several studies in this review reported significant AF associations at pollutant concentrations below current WHO air quality guideline levels, raising important questions about the adequacy of existing standards. However, because exposure metrics, concentration ranges, and analytical approaches varied substantially across studies, we were unable to determine how consistently low-level exposures were associated with AF or to quantify the number of studies demonstrating effects below guideline thresholds. Future work using more standardized study designs will be essential to evaluate concentration–response relationships more precisely. Such analyses, including pooled assessments of AF risk at pollutant concentrations below WHO and country-specific regulatory limits, would provide a valuable contribution to the field and help inform revisions to air quality standards and clinical risk stratification.
There is also a need to explore other less commonly studied pollutants, such as black carbon and oxidants, which have shown potential significance in this review. Investigating the role of comorbidities, such as age, gender, and pre-existing health conditions, as effect modifiers for AF risk in relation to air pollution exposure is critical for developing personalized preventive strategies and targeted interventions. Addressing these areas in future studies will improve our understanding of the complex relationship between environmental pollution and AF, ultimately informing public health initiatives and policy decisions aimed at reducing pollution-related cardiovascular risks. To meaningfully reduce air pollutant exposure and the burden of associated health issues like atrial fibrillation, a rapid and coordinated transition away from fossil fuels toward clean, renewable energy sources is essential.
Limitations of the Studies
There were several limitations in the studies reviewed. First, many studies gathered AF data from a mixture of outpatient, inpatient, and emergency department visits, making it difficult to distinguish between more severe AF events that require hospitalization and less severe episodes seen in outpatient settings. This heterogeneity in included studies means that both symptomatic and asymptomatic AF were captured in the results. While future studies should qualify differences between the occurrences, the physiologic triggering of AF, regardless of symptomatology, by air pollutants remains an important association. Additionally, for studies using ICDs to detect AF, some relied on “predictors” rather than recording actual AF events, which may have affected the accuracy of the findings. The variability in air pollution measurement techniques, such as the use of central monitoring stations versus zip code-level data, also contributed to potential lack of fine discrimination of local air pollutant concentrations. Furthermore, the lack of stratification based on mean annual pollutant concentrations and climate conditions may have limited the ability to detect significant associations in regions with lower pollution baselines or distinct weather patterns. Importantly, substantial heterogeneity existed across studies in terms of exposure metrics (eg, PM2.5, PM10, NO2, ozone), modeling approaches, lag structures, and exposure time windows, which resulted in differing effect estimates and limited comparability. This methodological variability, combined with differing outcome definitions and study designs, precluded quantitative pooling of results; accordingly, a meta-analysis was neither appropriate nor feasible for this scoping review.
Conclusion
This systematic review reaffirms the association between air pollution and atrial fibrillation, with multiple factors influencing AF risk, including pollutant type, comorbidities, and demographic variables. As the prevalence of AF continues to rise, it is crucial to identify vulnerable populations and determine whether current air quality standards adequately protect against AF risk. The findings suggest that even air pollutant levels considered “safe” may still pose a risk to susceptible individuals, warranting further investigation and potentially stricter air quality regulations.
Footnotes
Acknowledgements
There are no additional acknowledgments.
Author Contributions
CY – conceptualization, software, formal analysis, writing – original draft, visualization; MRF - validation, writing – review and editing, supervision; ZC – investigation; RM – investigation; BS – software, methodology, data curation; EC – methodology, writing – review and editing, supervision.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available at https://www.dropbox.com/scl/fi/4dhh8svzjarlgaogmfdyt/Appendix-A-and-B.docx?rlkey=fvmoms6lx9r2o4yiv10a5b1b7&st=w08eapz6&dl=0
