Abstract
The development of gallium(III) compounds as anti-cancer agents for both treatment and diagnosis is a rapidly developing field of research. Problems remain in exploring the full potential of gallium(III) as a safe and successful therapeutic agent or as an imaging agent. One of the major issues is that gallium(III) compounds have little tropism for cancer cells. We have combined the targeting properties of folic acid (FA) with long chain liquid polymer poly(ethylene glycol) (PEG) 'spacers’. This FA-PEG unit has been coupled to the gallium coordination complex of 1,4,7,10-tetraazacyclo-dodecane-N, N′, N′, N′′-tetraacetic acid (DOTA) through amide linkages for delivery into target cells overexpressing the folate receptor (FR). In vitro cytotoxicity assays were conducted against a multi-drug resistant ovarian cell line (A2780/AD) that overexpresses the FR and contrasted against a FR free Chinese hamster ovary (CHO) cell line. Results are rationalized taking into account stability studies conducted in RPMI 1640 media and HEPES buffer at pH 7.4.
Introduction
The anti-cancer properties of gallium(III) have been extensively investigated since 1971 (Hart et al. 1971). Gallium has numerous ways to induce cell death, including DNA binding and modification (Hedley et al. 1988), enzyme inhibition (especially ribonucleotide reductase) (Chitambar et al. 1988), and ion transport disruption (such as calcium efflux from mitochondria), a known trigger of cellular apoptosis (Collery et al. 1996).
In general, the poor pharmacokinetic properties of gallium salts investigated have prevented their widespread use in chemotherapy. Efforts to develop gallium complexes to improve its profile, by addressing the problems of hydrolysis, poor absorption, poor solubility, rapid renal excretion and little tropism for cancer cells are currently underway (Keppler and Jakupec, 2004; Desoize, 2004). Complexes such as those produced by the groups of Keppler (Keppler et al. 2006), Sharma (Sharma et al. 2007), Low and Green (Low and Green et al. 1996) have been successful in increasing plasma concentrations of gallium, providing better antiproliferative effects or improved imaging of cancer cells (when using gallium radioactive isotopes (67Ga γ, 68Ga β+)) (Greenwood and Earnshaw, 2005). Problems still remain however especially in regards to renal retention times and cancer cell targeting.
Folic acid (FA) (see Fig. 1) is a vitamin potentially capable of delivering agents specifically to folic acid-receptor (FR) positive tumors (Lee and Sudimack, 2000). FRs are membrane glyco-proteins (Anderson et al. 1990) overexpressed by a number of tumor cell types such as ovarian, breast, cervical, colorectal, renal and nasopharyngeal cancers (Antony, 1996). Cells overexpressing the FR bind FA-drug conjugates tightly (Kd ~ 0.42 × 10–9 M) (Shen et al. 1995) and endocytose them inside (Anderson et al. 1988), provided that chemical modification of the FA upon conjugation does not disrupt recognition by the FR (Liu et al. 2005). Targeting the FR is attractive because in addition to being overexpressed in tumor lines, it is down-regulated (and inaccessible to blood circulation) in healthy adult cells (Anderson et al. 1988).
FA with its three major structural components including the α- and γ-carboxylic acid group of the glutamate moiety indicated.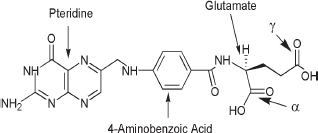
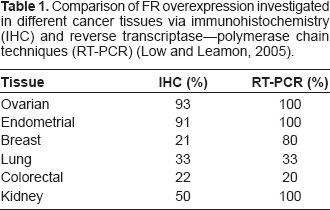
Comparison of FR overexpression investigated in different cancer tissues via immunohistochemistry (IHC) and reverse transcriptase–-polymerase chain techniques (RT-PCR) (Low and Leamon, 2005).
We set out to synthesize, purify and evaluate in vitro a new FA bioconjugate of gallium and compare its activity to the unconjugated gallium analog. We began by initially complexing gallium (III) to the 1,4,7,10-tetraazacyclo-dodecane-N, N′, N′, N′′-tetraacetic acid (DOTA) ligand (Doyle et al. 2006). We have previously described the synthesis and solid state structure of this system (see Fig. 2). Preceding literature reports have demonstrated good kinetic and thermodynamic stability provided by the DOTA ligand in its coordination chemistry and we wished to exploit this coupled with folate receptor (FR) targeting conjugates. In addition to coupling the gallium-DOTA complex to folic acid we wished to include poly(ethylene glycol) (PEG) polymer linkers between the FA and gallium complex since 'PEGylated’ FA conjugates have been shown to have greater affinity for the FR than free FA (Low and Lee, 1994). Pathways that can break down or efflux certain FA conjugates are inhibited by the polymer-FA conjugate and renal retention times of certain pharmaceuticals have been improved by conjugation to PEG units (Anderson et al. 2005). Hence, the use of FA-PEG conjugates yields synergistic traits that are of particular interest.
Crystal structure of Ga(HDOTA) [20].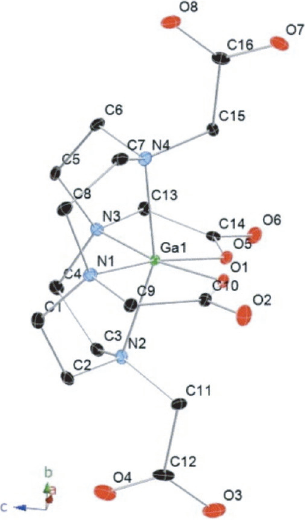
Conjugation of PEG to FA through the glutamate moiety produces two regioisomer products at the α- and γ-carboxylic acid functional groups that need to be separated. FA modified at the α-carboxylic acid loses its affinity for the FR, making it unsuitable as a targeting agent (Yan and Ratnam, 1995). This separation is difficult when using polydisperse PEG units. Such PEG units are typically all that are commercially available but are approved for use by the FDA (Qui and Bae, 2006). A facile route to separation was previously reported by us and this route was used here to allow access to pure α-FA-PEG-NH2 for subsequent conjugation to the gallium-(HDOTA) complex (Doyle et al. 2008).
In vitro cytotoxicity assays were conducted against adriamycin resistant ovarian cancer cell line (A2780/AD), which overexpresses the FR, and contrasted against a non-FR expressing Chinese hamster ovary (CHO) control line.
Experimental
Chemicals
The following reagents were purchased and used without further purification: Folic acid (FA) (98%, Sigma), N, N‘-Dicyclohexylcarbodiimide (DCC, ≥99%, Fluka), polyethylene glycol bis(amine) (PEG, MW: 2000) (Fluka), N-Hydroxysuccinimide (NHS, ≥97%, Fluka), and 1,4,7,10-tetraazacyclododecane-N, N’, N”, N’”-tetraacetic acid (DOTA, 98%, Strem Chemicals), N-Hydroxysulfosuccinimide sodium salt (sulfo-NHS, 98.5%, Fluka), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide (EDC, 98%, Alfa Aesar) and trifluoroacetic acid (99%, Aldrich). GaCl3 (99.9%) was purchased from Alfa Aesar and dissolved in 100 mM ammonium acetate (pH 4.8) to make a stock concentration of 1.325 M. Dimethylsulfoxide (DMSO) (min. 99.9%, Sigma) was dried by running the solvent through a column of 4 å molecular sieves (Mallincrodt) dried previously overnight at 120 °C. Solvents used for HPLC and growth media are filtered with 0.45 μm filter (Fisher). Pyridine (99.9%) was obtained from Fisher. Triethylamine (99.5%) was purchased from Sigma Aldrich. 3′-azido-3′-deoxythymidine (AZT; used as internal control in cytotoxicity assays) was purchased from Sigma Aldrich. All other reagents and buffers used were of reagent grade or higher. Ultra pure water (18.6 Mω) was used through out the investigation. All syntheses except for
Physical measurements and instrumentation
An Agilent 1100 reverse phase high pressure liquid chromatography (HPLC) with manual injection and automated fraction collector was fitted with a Zorbax C18 analytical column (42 × 10 mm) for analytical trace analysis with a flow rate of 0.7 ml/min. Purification was achieved using a C18 (9.4 × 250 mm) semi-preparative column using a flow rate of 2 ml/min. Detection was by ultra violet monitoring at 280 nm. The linear gradient used was: (1) 90% 5 mM Na2HPO4 (pH 7.0) and 10% acetonitrile over 10 minutes; (2) 40% 5 mM Na2HPO4 and 60% acetonitrile over 20 minutes. Ion exchange chromatography (IEC) was conducted on an Akta Prime Plus with Primeview 5.0 software. The ANX (1 ml) and the PD10 Sephadex G-25M desalting (10 ml) columns were purchased from GE Healthsciences. 1 H nuclear magnetic resonance ( 1 H NMR) was performed on Bruker Avance DPX 500 MHz. A Shimadzu LCMS-2010 A mass spectrometer and An Applied Biosystems Voyager-DE linear Matrix Assisted Laser Desorption Ionization–-Time of Flight mass spectrometer (MALDI-TOF) were used for mass spectrometry analysis. Infrared (IR) analyses were performed as KBr pellets on a Nicolet Magna-IR 850 series II spectrophotometer. A Perkin Elmer ELAN 6100 was used to conduct inductively coupled plasma analysis (ICP). Centrifugation was performed using a Sorvall Legend RT centrifuge typically as 10 minute runs at 4000 rpm at 4 °C. Optical densities were measured with a Thermo Multiskan EX 96-well plate reader equipped with Ascent Software version 2.6 with 450 nm filter.
Chemical synthesis
Synthesis of GaHDOTA (1)
Synthesis of γ-FA-PEG-NH2 (γ-2)
FA (0.0441 g, 0.100 mmol) was dissolved in 3 ml of dry DMSO. To this solution, 0.0127 g of NHS (0.110 mmol) was added. The mixture was stirred for 5 minutes after which 0.023 g (0.110 mmol) of DCC was added. The solution was then stirred overnight. The activated FA was filtered through a 0.45 μM filter to remove the dicyclohexylurea side product. The FA-NHS solution was then added dropwise to PEG2000 (0.200 g, 0.100 mmol) previously dissolved in 3 ml DMSO. 100 μL of pyridine was then added and the reaction stirred overnight. Approximately 25 ml of chilled isopropanol (–78 °C) was added forming a light yellow precipitate. The precipitate was obtained via centrifugation. The γ- and α-isomers of
Only the isolated γ-isomer (γ-2) was used for subsequent coupling.
Synthesis of γ-FA-PEG-H3DOTA (γ-3)
The sulfo-succinamide ester of DOTA was prepared by activating 0.121 g (0.300 mmol) of the ligand with 26.5 μL (0.150 mmol) EDC and 0.0260 g (0.120 mmol) sulfo-NHS in 2 ml water.
Synthesis of γ-FA-PEG-Ga(HDOTA) (γ-4)
Cell lines and culture conditions
Adriamycin resistant ovarian cancer cell line (A2780/AD) and Chinese hamster ovary (CHO) cell line were cultured as adherent monolayers in RPMI 1640 (Invitrogen) growth media containing L-glutamine and FA supplemented with 10,000 units penicillin and 10 mg/ml streptomycin (Sigma), 10% (v/v) fetal bovine serum (Sigma). CHO cells were obtained from the ATCC. The A2780/AD cell line used for testing was provided by the Fox Chase Cancer Centre, Philadelphia. Cells were incubated and grown in a VWR mammalian incubator at 5% CO2 and 95% humidity. The presence of the FR in the A2780/AD line (and indeed absence in CHO cells) was followed by RT-PCR and confocal microscopy (Doyle et al, unpublished results). All preparations for cell culture and assays were conducted in a sterile environment under a Labconco Purifier I Laminar flow hood. Cells were cultured in Millipore 250 mL culture bottles with vented lids.
Drug cytotoxicity
The proliferation of the exponential phase cultures of A2780/AD and CHO cells was assessed by colorimetric assay. WSK-8 (Dojindo) was performed according to manufacturer's instructions.
Adherent cell cultures were harvested by stripping of culture flasks by a non-enzymatic cell stripper (Mediatech) after a 30 minute incubation period. The cells were then collected. The cell densities were adjusted using FA-free RPMI 1640 media to 3.0 × 104 cells/ml to guarantee exponential growth for the period of drug exposure. To each well, aliquots of 100 μL were inoculated. After a 24 hour incubation time to facilitate adherence, the FA free RPMI media was removed and replaced with 200 μl of fresh media containing different concentrations of
Stability studies
2 mM solutions of
Results and discussion
Chemical synthesis
The synthesis of Synthesis of 1 involving chelation of Ga(III) to DOTA in ammonium acetate buffer (pH 4.8).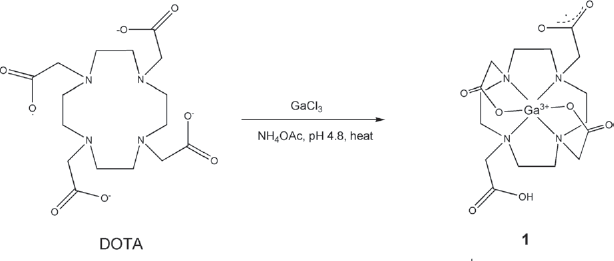
FA was activated by a reaction with DCC/NHS to couple it to PEG. Synthesis and separation, via IEC of the regioisomers of the α- and γ-isomers of 2.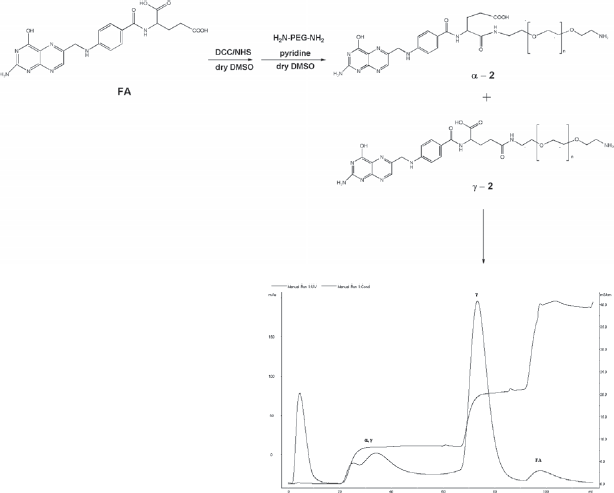
Synthesis of γ-3 using EDC/sulfo-NHS as coupling agents.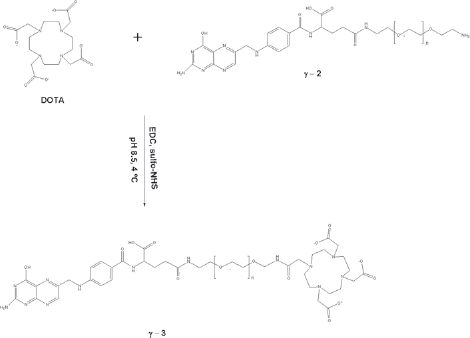
Coupling of Synthesis of γ-4 illustrates the coupling of γ-2 and 1 using DCC and NHS as coupling agents in dry DMSO.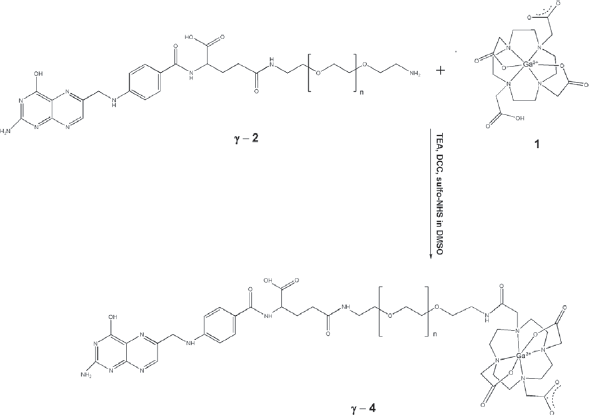
Mass spectra
A MALDI-TOF mass spectrum of the commercial PEG displayed a mass range of 1900–2100 m/z due to the polydispersity of the polymer. The observed mass of MALDI-TOF mass spectrometry analysis of a) γ-2 and b) γ-4 showing a central peak at ca. 2400 m/z and 2846 m/z respectively. ICP also confirmed the presence of gallium in γ-4.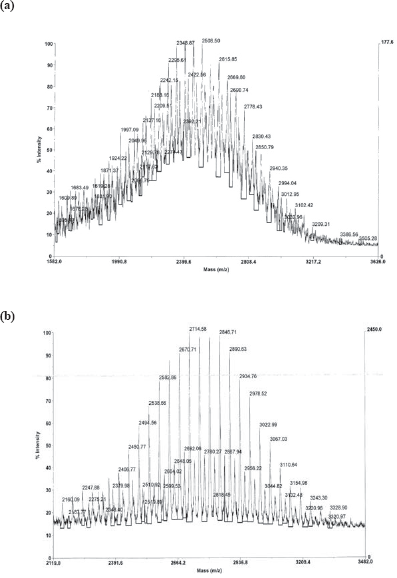
In vitro biological activity
IC50 concentrations for 1, γ-2, γ-3, γ-4 and DOTA against A2780/AD ovarian cells and Chinese hamster ovary (CHO) cells. (–) indicates no IC50 was recorded. AZT was used as a control and returned an IC50 concentration of ~ 6–8 mM consistent with literature values (Doyle et al. 2008).
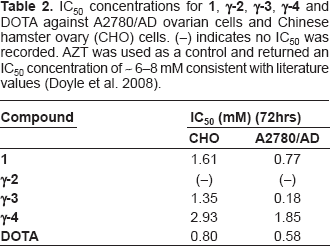
Interestingly gallium compounds were noted as less toxic than free DOTA containing controls. This was the case in both cell lines. The IC50 concentrations for both compounds containing gallium (namely
Toxicity of the FA-PEG containing DOTA compound (
Toxicity of
Gallium decomplexation from a ligand such as DOTA that renders such thermodynamic and kinetic stability is possible even with a reported stability constant of log K ~ 21.33 (Clark and Martell, 1991) (compared to say to open-chain multidentate ligands like ethylenediamine (log K ~ 17.2) (Harris and Martell, 1976)). A key factor in this release is an increase in DOTA's electron density due to inductive effects contributed by the ethylene bridges (Hancock and Martell, 1995).
A plausible explanation for the possibility of gallium release concerns the formation of the FA conjugate. The stability of
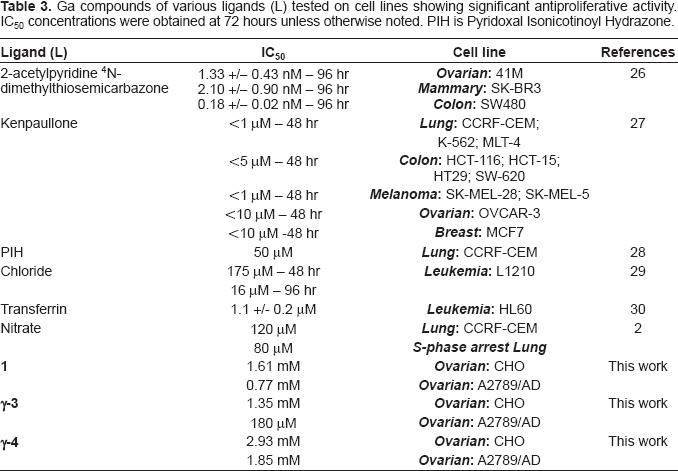
Ga compounds of various ligands (L) tested on cell lines showing significant antiproliferative activity. IC50 concentrations were obtained at 72 hours unless otherwise noted. PIH is Pyridoxal Isonicotinoyl Hydrazone.
To prove that gallium has indeed been “freed” from its macrocyclic cage, stability studies in HEPES buffer and RPMI 1640 media over 72 hours coupled with HPLC and ICP techniques were conducted. New peaks were observed between 48 and 72 hours indicative of gallium release (see supplemental material). These peaks were analyzed via ICP and gallium was noted. In addition, a slight precipitate could be removed by filtration (0.22 μM filters) and ICP confirmed the presence of gallium in the collected solid fraction. Attempts to identify the new species were unsuccessful by electrospray mass spectrometry and 1 H NMR and attempts to obtain crystals for X-ray structural analysis also proved unsuccessful. It is likely that both soluble and insoluble gallium salts (such as gallium hydroxides) are forming and/or gallium is complexing with compounds found in the RPMI media. The comparable observed cytotoxicities on both cell lines can then be ascribed to gallium leaching from DOTA over 72 hours.
Conclusion
We have successfully synthesized, characterized, and investigated the in vitro cytotoxicity studies of DOTA based gallium complexes and conducted controls to track the source of the toxicity. These results demonstrate that while a ligand of extraordinary kinetic and thermodynamic stability gallium can 'leach’ from DOTA over a 72-hour period. What is also clear is that DOTA itself has between [500–800 μM] toxicity, an interesting note in and of itself. Toxicity in both lines could be explained by the uptake, by diffusion, of free DOTA or gallium-DOTA, or the presence of uncomplexed DOTA and/or the release of gallium from the conjugate as applicable in the FA-PEG containing systems. Clearly the fact that free DOTA has greater toxicity than the gallium complexed forms described herein, make them unsuitable as anti-cancer agents themselves. There is however a significant difference on FR containing cells over non-FR containing cells in terms of selectivity, as well as sufficient stability, to suggest that coupling the γ-emitting 67Ga isotope or the β-emitting 68Ga isotope to the FA-PEG conjugate unit may provide a suitable route to targeting radioisotopes of gallium to cell lines for use as diagnostic agents. This work is currently being investigated in the group.
Supporting Material
HPLC stability traces and IC50 graphs showing exponential plots.
Footnotes
Acknowledgments
The authors wish to thank Syracuse University and the iLEARN program for funding. We also thank Karen L. Howard (State University of New York, ESF) and Chris Incarvito (Yale University) for assistance obtaining MALDI-TOF mass spectra and Colin Fuss (CESE, SU) for conducting ICP.
