Abstract
Background
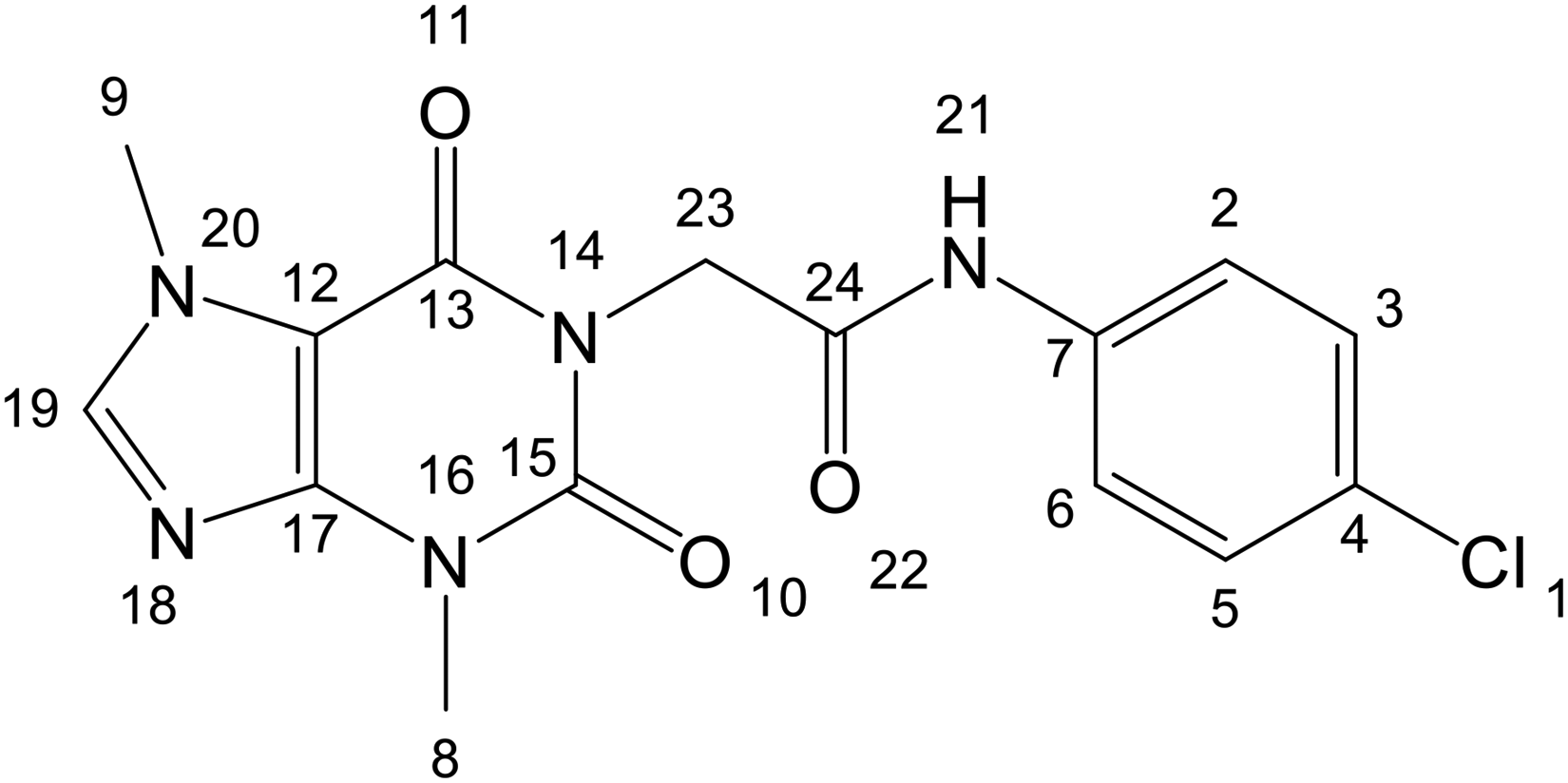
Theobromine derivatives have emerged as promising anticancer agents, but their therapeutic potential is limited by poor bioavailability. This study investigates a chitosome-based delivery system for a novel theobromine derivative, N-(4-Chlorophenyl)-2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)acetamide (
Methods
Results
The
Conclusion
Keywords
Introduction
The World Health Organization (WHO) reports that in 2022, there were an estimated 20 million new cancer cases and 9.7 million cancer-related deaths worldwide. An estimated 53.5 million people were alive within five years of a cancer diagnosis. 1 Modern cancer treatment increasingly uses functional materials designed to target specific subcellular sites, improving precision and efficacy by selectively binding and inhibiting key molecules in cancer cells.2–4 A variety of innovative strategies are being explored to enhance the effectiveness and specificity of cancer treatment. These include nanotechnology, 5 gene therapy, 6 photodynamic therapy, 7 and hyperthermal therapy. 8
The epidermal growth factor receptor (EGFR) signaling pathway plays a crucial role in regulating cancer cell proliferation, survival, differentiation, and migration.9–12 Overexpression of EGFR has been widely associated with the initiation and progression of multiple cancer types. 13 EGFR activates multiple signaling pathways that drive uncontrolled proliferation and apoptosis evasion, making it a key target in cancer therapy.14,15 However, conventional EGFR inhibitors often face limitations such as poor aqueous solubility, suboptimal bioavailability, 16 and the development of drug resistance.17,18 Nanodelivery systems offer a promising strategy to overcome these barriers by improving the solubility and stability of EGFR inhibitors, enhancing their bioavailability, and enabling targeted delivery to tumor tissues through passive mechanisms that exploits the enhanced permeability and retention effect, whereby the leaky vasculature and impaired lymphatic drainage of tumor tissues facilitate the accumulation and prolonged retention of nanocarriers.19,20 In contrast, active targeting involves functionalizing nanocarrier surfaces with specific ligands—such as folate, antibodies, peptides, or transferrin—to promote selective localization within tumor tissue, thereby achieving higher intratumoral drug concentrations and reducing systemic side effects. 21 Such approaches can enhance drug accumulation at the tumor site, minimize systemic side effects, and potentially improve the therapeutic index of EGFR-targeted therapies, while nanocarriers also help overcome EGFR- and cancer-related drug resistance.22,23
Several types of nanocarriers have been developed and investigated in improving pharmacological effects of cancer diagnostic and therapeutic agents including micelles, 24 liposomes,25–27 solid lipid nanoparticles,28,29 nanostructured lipid carriers, 30 nanoemulsions, 31 dendrimers,32,33 polymeric nanoparticles, 34 microspheres, 35 quantum dots 36 carbon nanotubes 37 and inorganic nanoparticles. 38
Nanoencapsulation can improve the solubility, absorption and bioavailability of poorly water soluble drugs in addition to protecting the encapsulated drug from harsh gastrointestinal conditions.39,40 Chitosomes are hybrid nanocarrier systems composed of conventional liposomes coated with chitosan, a natural cationic polysaccharide. 41 This modification enhances the stability, mucoadhesiveness, and cellular uptake of liposomes due to the positive surface charge imparted by chitosan. Compared to unmodified liposomes or polymeric nanoparticles, chitosomes offer improved biocompatibility, prolonged circulation time, and enhanced interaction with negatively charged cancer cell membranes. 42 These properties make them particularly suitable for delivering anticancer agents with low aqueous solubility or requiring controlled release. 43
The combined use of in silico and in vitro methods offers an efficient framework for drug discovery.44,45 Computational modeling such as docking 46 and molecular dynamic (MD) simulations47,48 predict molecular interactions, 49 and pharmacokinetic behaviors, 50 while experimental assays validate these predictions and offer mechanistic insights.51,52 This combined approach accelerates candidate optimization, improves precision,,53–56 and reduces reliance on costly in vivo studies.57–59 Our research group has effectively applied this dual approach across multiple domains, including antiviral drug development against SARS-CoV-2,28,57,59 targeting microbial virulence mechanisms,60,61 advancing anticancer strategies,62–64 and improving the delivery of bioactive compounds.65–67
In 2023, our team semi-synthesized a bioactive theobromine derivative, N-(4-Chlorophenyl)-2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)acetamide
15
(
Experimental
In Silico (Molecular Docking) Studies
Target Preparation
The molecular structure of chitosan (PubChem CID: 71853) retrieved from PubChem, protonated, and subjected to energetic minimization using GB/VI electrostatics with a 15 Å distance cutoff and a dielectric constant of 2. Structural optimization was carried out using Hamiltonian AM1 and MMFF94x.
Ligand Preparation
Docking Protocol
Preparation of Liposomes and T-1-PCPA Loaded Chitosomes
Lipoid S 100 was generously provided by Lipoid, Germany, while cholesterol and chitosan were procured from Sigma Aldrich, USA. Blank liposomes were created through the thin film hydration technique. In summary, 100 mg Lipoid S 100, 2 mg cholesterol, and 40 mg Tween 80 were dissolved in 1.5 mL absolute ethanol, stirred on a water bath at 50 °C until complete ethanol evaporation and thin film formation. Film rehydration was conducted using either 10 mL deionized water or 0.05% chitosan (CS) for liposome and chitosan-coated liposome (chitosome) preparation, respectively. The mixtures were then ultrasonicated (SonicaR 2200 EP S3, Soltec) at 60% amplitude for 5 min. The resulting nanodispersions were stored in the refrigerator for further investigation.
Physiochemical Characterizations
Particle Size, PDI, and Zeta Potential
Particle size (PS), polydispersity index (PDI), and zeta potential (ZP) of the prepared
Entrapment Efficiency (EE%) and Drug Assay
Entrapment efficiency was determined by separating the unentrapped
The EE% was calculated using the following equation:
The drug assay of
Stability Study
A short-term physical stability study was carried out for
Transmission Electron Microscope (TEM)
The formulation of
In Vitro Biological Evaluations
Cytotoxicity
The anti-cancer effects of the
Selectivity
The Selectivity Index (SI) values were calculated as described earlier
70
using the following formula: SI = IC50 for cancer cells / IC50 for non-cancerous cells. In this analysis, IC50 values were determined for both non-cancerous cells (Vero) and cancer cells (HCT-116 and A549) in response to the tested compounds (
Migration Assay
Migration assay was conducted to evaluate the effectiveness of the
Flow Cytometry (Cell Cycle and Apoptosis)
Flow cytometry analyses were conducted to delve into the impact of the
Statistical Analysis Section
GraphPad Prism 6 software (GraphPad, San Diego, CA, USA) was used to analyze all results. The data were represented as mean ± standard deviation (Mean ± SD) from at least three independent experiments (n = 3). One-way analysis of variance (One-way ANOVA) followed by Tukey post hoc multiple comparison tests were elected to analyze the significant difference between all groups. P < .05 was considered statistically significant.
Results and Discussion
Molecular Docking
Docking of T-1-PCPA Against Phosphatidylcholine-Chitosan Complex
The primary rationale of the molecular docking studies was to assess, at the molecular level, whether
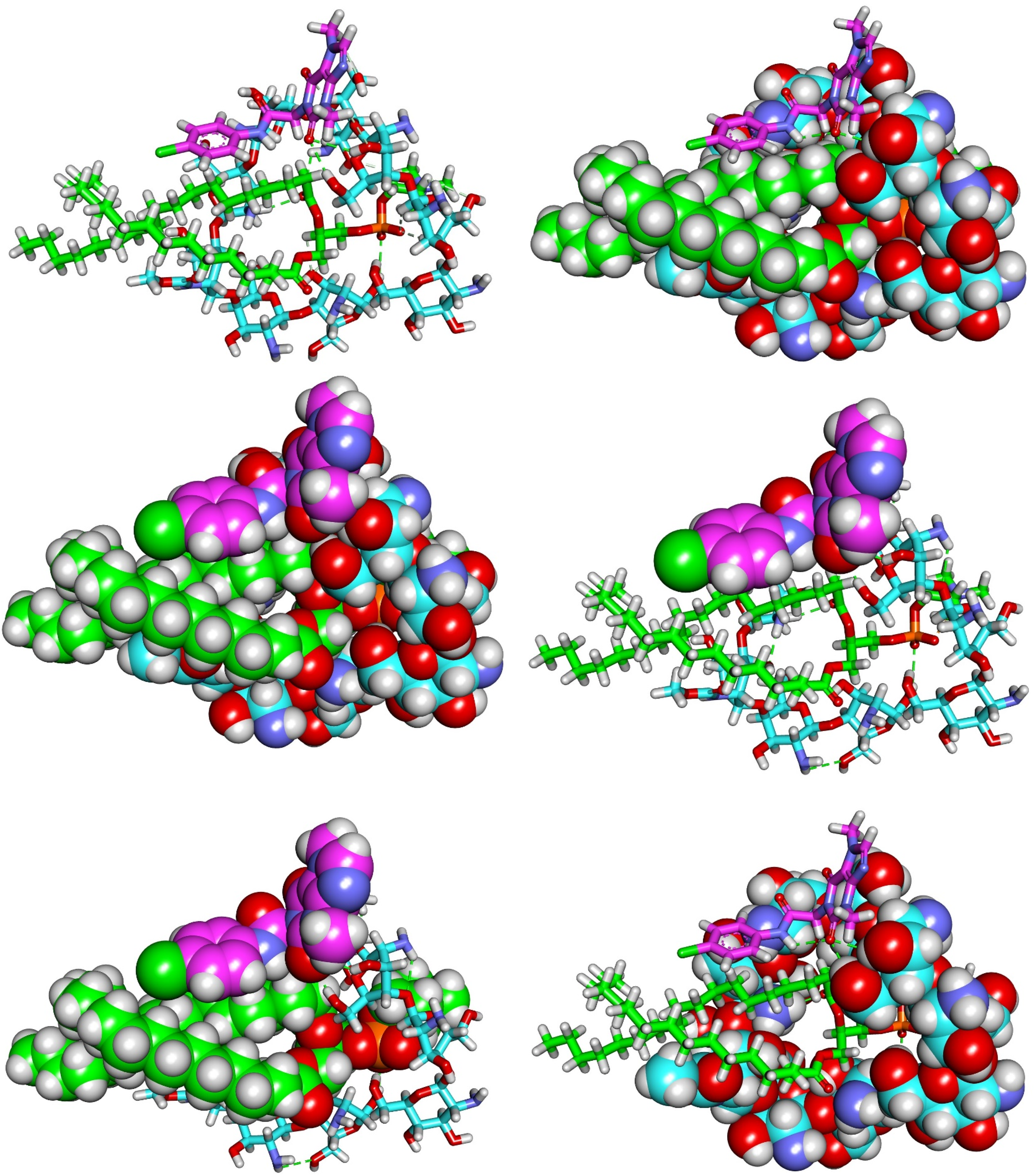
Binding Mode of
Distances, and Angles of the Hydrogen Bonds Between T-1-PCPA, Phosphatidylcholine (PC) and Chitosan (CS) Through the T-1-PCPA Loaded Chitosomes.

Docking of T-1-PCPA Against EGFR
Docking studies were carried out to investigate the binding mode of

Superimposition of the Docked Ligand (Erlotinib) (Pink) and the Original Ligand (Green) with RMSD Value of 1.85 Å.
Erlotinib and

Preparation of the T-1-PCPA -PC-CS Complex
Polymer coating improves the performance of liposomes, with chitosan being especially useful due to its hydrophilic, biocompatible, and biodegradable nature. Chitosomes combine the strengths of both systems, enabling controlled drug release, greater stability, mucoadhesion, and enhanced tumor uptake.
75
Furthermore, chitosan possesses anti-cancer activity as it can interfere with tumor metabolism and as result cell growth is inhibited.
76
Chitosan-coated nanocarriers can be attached to anionic glycoproteins in oral mucin through electrostatic interactions this will in turn increase the intratumoral retention rate of the nanocarriers. This mucoadhesive feature is especially desired for targeting tumors that are characterized by mucin-dominant mucosae.
77
Chitosomes have shown to be effective in improving the pharmacological efficacy of different chemotherapeutic agents in different types of cancer cell lines.
78
During the formation of the

A Schematic Diagram Showing the Preparation
Figure 5 shows the key parts of the prepared formulation. It highlights how
Physiochemical Characterization of the T-1-PCPA Loaded Chitosomes
Particle Size, Polydispersity Index (PDI) and Zeta Potential Evaluation
Previous reports recommended that nanocarriers of particle size lower than 200 nm have shown effective tumor retention by enhanced permeation and retention effect.79–81 As Table 2 illustrates, particle size of the prepared chitosomes did not exceed the appropriate range for effective tumor targeting. Incorporation of
Colloidal Characteristics of Blank and

The PDI is a key measure of how uniform the nanodispersion is. Lower PDI values mean particles are more evenly sized. In this study, the formulations showed PDI values between 0.108 and 0.145, indicating excellent uniformity. Such consistency in particle size supports the stability and reliability of the chitosomes, reflecting a well-controlled and reproducible process.82,83
The ZP serves as a crucial parameter indicative of the stability of formulations, particularly in colloidal systems. Formulations exhibiting ZP values around ±30 mV are widely considered stable due to the repulsive forces among particles that prevent coalescence and aggregation.84,85 In our study, the prepared chitosomes exhibited a moderately positive ZP (16.1 ± 1.45 mV), which, although slightly lower than the ±30 mV benchmark, still falls within the range associated with stability. This positive surface charge, conferred by the chitosan coating, contributes to electrostatic repulsion among particles, thereby minimizing aggregation and maintaining dispersion stability. Such electrostatically mediated stability is a key feature of chitosan-coated colloidal systems. 41
Beyond stability, the physicochemical properties of nanoparticles, particularly PS and ZP, are critical in governing biosafety and cellular uptake. The chitosomes demonstrated a mean particle size of 75-110 nm, which lies in the optimal range for endocytosis-mediated uptake. Nanoparticles below 200 nm can efficiently interact with cell membranes, penetrate tissues, and avoid rapid clearance by the mononuclear phagocyte system (MPS), thereby enhancing tumor accumulation through the enhanced permeability and retention (EPR) effect. The moderately positive ZP also favors biosafety, as it facilitates interaction with negatively charged cancer cell membranes and promotes adhesion and internalization, without triggering significant cytotoxicity or aggregation in biological fluids.
While the present work provides important in vitro evidence in two cancer cell lines, further in vivo biosafety and long-term stability assessments are planned. These future studies will build upon the current findings and are expected to comprehensively validate the therapeutic promise of the drug-loaded chitosomes.
Encapsulation Efficiency %
Chitosomes have been recognized for their ability to enhance Encapsulation Efficiency (EE%), primarily attributed to the stabilizing influence of chitosan on the membrane bilayer.
86
In the case of
Previous studies reported the effectiveness of chitosomes in encapsulating chemotherapeutic agents and cancer treatment. Moya-Garcia et al reported that docetaxel was successfully loaded in chitosomes that were internalized into the cytoplasm of human laryngeal cancer cell with a higher cytotoxicity of compared with human stromal cells and control treatments. 77 Shukla et al 88 reported that metformin loaded chitosomes demonstrated enhanced internalization, cytotoxicity and reduction in tumor volume in malignant pleural mesothelioma. 88 Also, Alomrani et al 42 reported that the cytotoxicity study using a colon cancer cell line (HT-29) showed that 5-fluorouracil loaded chitosomes were more effective in killing cancer cells in a sustained manner compared with uncoated liposomes and the 5-fluorouracil solution. 42
Transmission Electron Microscopy (TEM)
The encapsulation of
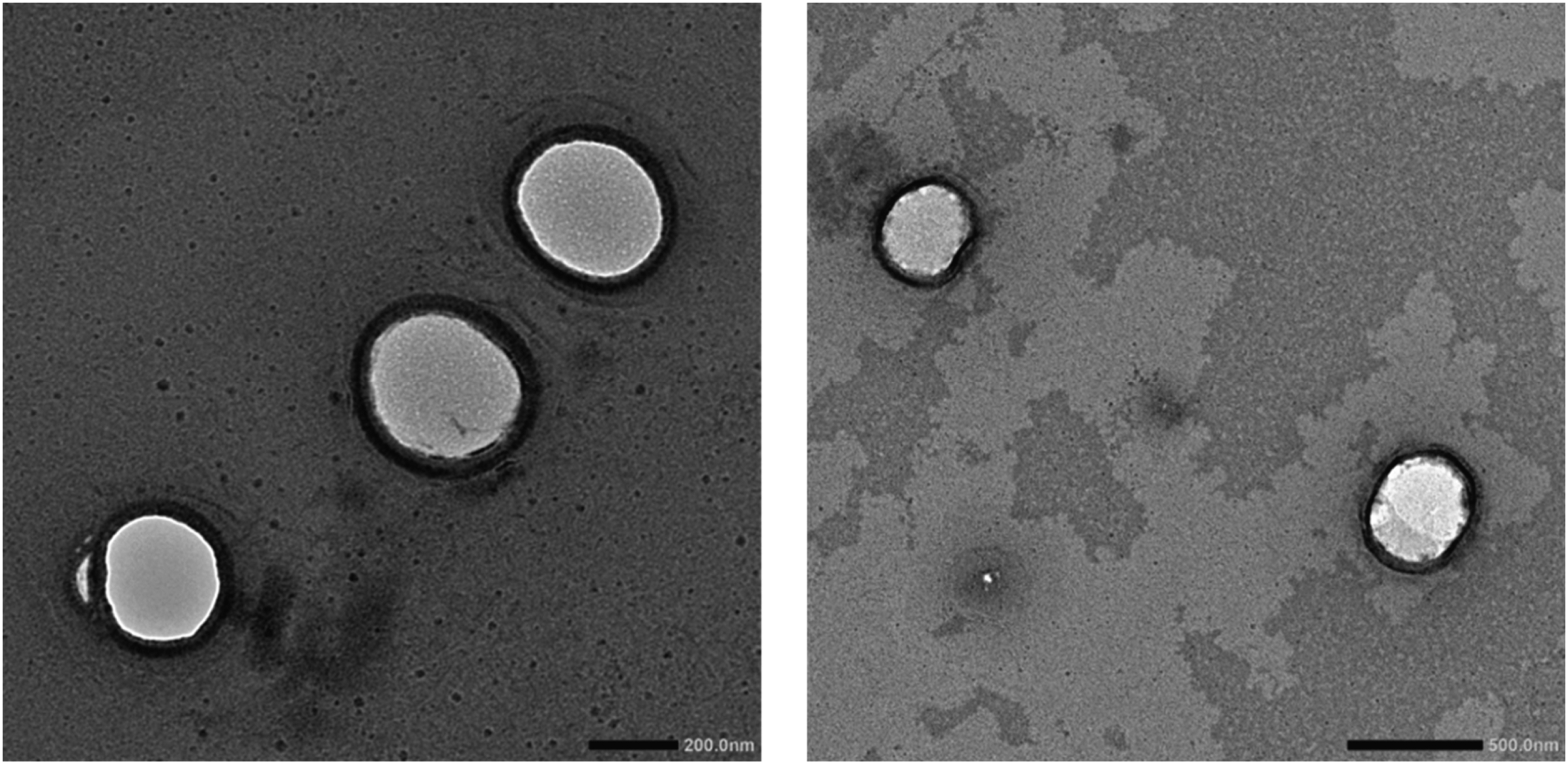
TEM Micrograph of
In Vitro Anti-Cancer Evaluations
Cytotoxicity
The presented study (Table 3) investigated the cytotoxic effects of

Data are presented as the mean ± standard error of the mean (SEM) from three independent replicates.
Safety and Selectivity
The SI is a key measure for evaluating how effectively and specifically a drug formulation targets cancer cells compared to normal cells. It is calculated as the ratio of the IC50 for normal cells to that for cancer cells. A higher SI indicates a greater preference for targeting cancer cells, suggesting that the drug is more effective and specific in its action.
In this study, SI values were calculated for the

Data are presented as the mean ± standard error of the mean (SEM) from three independent replicates.
In conclusion, the SI values confirm the superior selectivity of the
Cancer Cell Migration Assay
The cell migration assay was conducted to evaluate the effect of the
As shown in Table 5, both groups started with comparable wound areas: the control group had a slightly larger initial wound area (982.6 µm²) than the treated (977.6 µm²). However, the initial wound width varied, with the control group measuring 383.3 µm, while the treated group displayed a wider wound width of 748.6 µm.
In Vitro Effects of

After 48 h, as shown in Figure 7, the control group showed a modest decrease in both wound area and width, indicating active cell migration and partial wound closure. In contrast, the

In vitro Migration Abilities of (A) Untreated HCT-116 Cells and (B) Treated HCT-116 Cells with a Sub IC50
Cell Cycle Assay
The cell cycle analysis, conducted using flow cytometry, evaluates the distribution of treated and untreated cancer cells across various phases (G0/G1, S, and G2/M), providing insight into whether the formulation induces cell cycle arrest. The analysis of cell cycle distribution, as reflected in the DNA content percentages of G0-G1, S, and G2/M phases, provides valuable insights into the impact of the

Flow Cytometry Analysis of HCT-116 Cell Cycle Phases After the
Effect of

Significant P value significant P value < .05 & by using One-way ANOVA followed by Tukey's post hoc multiple comparison tests.
In comparison, untreated control HCT-116 cells showed 46.92% of cells in the G0–G1 phase, 33.52% in the S phase, and a higher proportion—19.56%—in the G2/M phase, reflecting more active mitotic progression. These shifts in phase distribution between treated and control groups indicate that the
Apoptosis Assay
The apoptosis assay, conducted using flow cytometry, detects programed cell death and distinguishes between early and late apoptotic populations, as well as necrotic cells. Together, these complementary assays offer a more comprehensive understanding of the anticancer effects of T-1-PCPA-loaded chitosomess. The presented data reveals distinct differences in apoptotic and necrotic events between the

Apoptosis and Necrosis Induction in HCT-116 Cells Following Treatment with
Effect of

Significant P value significant P value < .05 & by using One-way ANOVA followed by Tukey's post hoc multiple comparison tests.
In
The pronounced elevation in both early and late apoptosis following treatment suggests a potent pro-apoptotic effect of the
The observed increase in early (21.55%) and total apoptosis (39.55%) suggests that
Despite the encouraging in vitro findings, several limitations must be considered. A critical next step is in vivo validation to assess the pharmacokinetics, biodistribution, and therapeutic efficacy of the
Conclusion
This study evaluated the anticancer potential of a theobromine derivative, N-(4-Chlorophenyl)-2-(3,7-dimethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-purin-1-yl)acetamide (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X251384515 - Supplemental material for Exploring the Anticancer Potential of Chitosome-Encapsulated Theobromine Analogue: Integrated Computational, Physiochemical and In vitro Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X251384515 for Exploring the Anticancer Potential of Chitosome-Encapsulated Theobromine Analogue: Integrated Computational, Physiochemical and In vitro Study by Ahmed M. Metwaly, Heba Abd-El-Azim, Mariam Zewail, Aisha A. Alsfouk, Eslam B. Elkaeed, Hazem Elkady and Ibrahim H. Eissa in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank Research Center at AlMaarefa University for supporting this work.
Ethical Approval
Not applicable.
Statement of Informed Consent
Not applicable.
Funding
This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2025R116), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Statement of Human and Animal Rights
Not applicable.
Sample Availability
Upon request,
Supplemental Material
Contain detailed methodology, including tables, photos, and graphs, of the biological investigations, such as anticancer assays, selectivity studies, flow cytometry analysis, and migration assays.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

