Abstract
Exosome-mediated signaling is important in mediating the inflammatory response. To exert their biological or pathophysiological functions in the recipient cells, exosomes deliver a diverse array of biomacromolecules including long and short coding and non-coding RNAs, proteins, and lipids. Exosomes secreted by antigen-presenting cells can confer therapeutic benefits by attenuating or stimulating the immune response. Exosomes play a crucial role in carrying and presenting functional major histocompatibility peptide complexes to modulate antigen-specific T cell responses. Exosomes from Dendritic Cells (DCs) can activate T and B cells and have been explored for their immunostimulatory properties in cancer therapy. The immunosuppressive properties of exosomes derived from macrophages and DCs can reduce inflammation in animal models for several inflammatory disorders. This review focuses on the protective role of exosomes in attenuating inflammation or augmenting immune response, emphasizing studies on exosomes derived from DCs and macrophages.
Introduction
Inflammatory response is a well-regulated process of integrated and complex network of cellular communication. Multiple modes of information exchange are employed through the secretion and subsequent receptor-mediated detection of biomolecules, including cytokines, chemokines, and even metabolites. Uni- and bidirectional communication exists between immune and non-immune cells during the process of initiating, maintaining, and resolving inflammation. 1 Most of the interactions between immune cells are mediated by cyto-kines released in response to different stimuli. Recent evidence has shed light on a novel mode of intercellular communication mediated by exosomes, a class of Extracellular Vesicles (EVs) in regulating inflammation.
Cells secrete different types of EVs that can be classified based on their size and subcellular origin. EVs formed by budding from the plasma membrane of cells are referred to as microvesicles or ectosomes. Exosomes differ from ecto-somes in that they are derived from inward budding of the inner endosomal membrane to form multivesicular bodies, followed by the fusion of multivesicular bodies with the plasma membrane. 2 These vesicles range in size from 30 to 150 nm. Composed of a lipid bilayer, they contain both transmembrane and cytosolic proteins and enclose mRNAs and miRNAs.3–8 In a recent editorial, the International Society for Extracellular Vesicles put forth a minimal set of biochemical, biophysical, and functional standards that should be used to attribute any specific biological cargo or functions to EVs. 9 After isolating exosomes, the purity of the exosomes can be tested using electron microscopy and western blotting. It has been reported that exosome processing including centrifugation, dehydration, and fixation for transmission electron microscopy may alter the size and morphology of vesicles. The cup-shaped morphology for exosomes shown in some studies has been attributed to differences in sample processing.10,11 Proteins highly enriched in exosomes are used as markers to identify exosomes. Commonly used exosomal markers are Hsp70, tet-raspannin family glycoproteins CD63, CD81, and CD914. Cells use these vesicles to communicate with both adjacent and distant cells. The molecules present on the surface of EVs enable them to target recipient cells. These EVs can induce signaling by receptor-ligand interaction or can be internalized by endocytosis and/or phagocytosis, or even fuse with the membrane of the target cell, thereby delivering vesicular contents into the cytosol of the recipient/target/acceptor cell. 8
The biological effects mediated by exosomal uptake in recipient cells depend on the donor cells secreting the exo-somes.12,13 The exosomal contents vary depending on the source and the physiological conditions of cells releasing them. Changes due to infection, inflammation, or transformation as in the case of tumor cells can influence and thereby alter the composition of exosomes. However, not everything that is present in the parent cell is incorporated into the exo-somes, suggesting that this well-regulated process is dynamically altered by signaling cues. This variability in biomolecules within the exosomes is the basis for biological consequences upon their uptake, as well as their biomarker utility. Though the function of exosomes in normal cellular homeostasis is still being elucidated, their role in pathogenesis is well established; it is known that exosomes can be pathogenic or protective. 14 Here, we will focus primarily on the protective role of exo-somes in attenuating inflammation or augmenting immune response, emphasizing studies on exosomes derived from Dendritic Cells (DCs) and macrophages.
Immunomodulatory Function of Exosomes
Innate and adaptive responses collectively contribute to immune response. The evolutionarily conserved innate immune system is found in all multicellular organisms, while the adaptive immune response is found only in vertebrates. 15 The activation of the innate immune system is mediated by a limited number of receptors that can recognize pathogen-associated molecular patterns or damage-associated molecular patterns.16,17 Toll-Like Receptors (TLRs) are well characterized examples in mammals. Often referred to as pattern recognition receptors, these receptors are germline encoded. In adaptive immune systems, the antigen receptors are not germline encoded but generated through somatic hypermutations. 18 This results in a wide range of antigen receptors in the adaptive immune system, compared to the limited number of receptors for pathogen-associated molecular patterns or damage-associated molecular patterns in the innate immune system. 19 T and B cells are the two main cell types in the adaptive immune system. B cells can recognize antigens, while the T cells utilize the process of antigen presentation for antigen recognition. DCs and macrophages are Antigen-Presenting Cells (APCs). These APCs internalize foreign antigens and load the processed antigens on to major histocompatibility complexes I and II (MHCI and MHCII) molecules for presentation to naïve CD8+ and CD4+ T cells, respectively, imparting the memory of the antigen to the T cells. 20
Exosomes play a crucial role in carrying and presenting functional MHC-peptide complexes to modulate antigen-specific CD8+ and CD4+ responses.6,21 This presentation can be direct or occur as a cross-presentation. In direct presentation, MHC-peptide complexes on the exosomes are directly engaged by antigen-specific T cells, leading to T-cell activation. In cross-presentation, APCs acquire antigens carried by exosomes and after additional processing of these antigens, present their peptides to T cells. Cross-presentation can also occur when antigenic peptide-MHC complexes are together transferred onto DCs and then presented to T cells (termed cross-dressing). 6 These features thus form the basis of protective or therapeutic benefits conferred by exosomes.
Immunomodulatory Role of Exosomal RNA
RNA-sequencing analysis using exosomes from human plasma samples showed the presence of a wide variety of RNA species within these circulating vesicles. 22 These include mRNAs and noncoding regulatory RNAs including miRNAs and long noncoding RNAs.3,23–27 The functionality of RNA in the recipient cell after uptake4,28 has been demonstrated. RNA sequencing of naïve and activated macrophages has shown that inflammation alter the transcriptome. The timing of transcription factor activation, as well as the localization of nascent transcripts (chromatin-associated, nucleoplasmic, and cyto-plasmic) play a role in spatial and temporal regulation of transcriptome changes resulting from inflammation.29,30 Whether inflammation-induced alterations are reflected in exosomal transcriptome was determined in a separate study by sequencing exosomal RNA derived from naïve and lipopolysaccharide (LPS)-stimulated macrophages. Pathway analysis showed significant changes in both the adaptive and innate immune processes; specifically, pathways related to NF-κB activation and TLR cascades differed between exosomal mRNAs from naïve cells compared with those from LPS-stimulated cells. 31 Exosomal miRNAs miR-21 and miR-29a secreted by tumor cells can bind to TLR8 and TLR7 in immune cells, leading to NF-κB activation and secretion of inflammatory cytokines. 32
RNA sequencing and profiling studies have shown that miRNAs are abundant in exosomes.2,4 It is also known that the repertoire of exosomal miRNAs differ from that of the donor cell. 33 Recent studies have explored the mechanisms determining the incorporation of a subset of cellular miRNAs into exosomes suggesting that the sorting of specific miRNA species to exosomes may be actively regulated. One study showed that endogenous mRNAs modulate miRNA sorting to exosomes and transfer to acceptor cells. 34 This study implied that exosomal miRNA secretion is a mechanism by which cells rapidly dispose miRNAs in excess of their targets to adjust miRNA:mRNA homeostasis. Based on current research, four potential modes/mechanisms for miRNA sorting into exosomes are known. The sorting mechanism for exosomal miRNAs includes the following steps: (1) The neural sphingomyelinase 2 (nSMase2)-dependent pathway: overexpression of nSMase2 increased the number of exosomes secreted and exosomal miRNAs. Conversely, inhibition of nSMase2 expression reduced the number of exosomal miRNAs. 35 (2) The miRNA motif and sumoylated heterogeneous nuclear ribonucleoproteins (hnRNPs)-dependent pathway: sumoylated hnRNPA2B1 recognized the GGAG motif (EXOmotif) in miRNA sequences and caused specific miRNAs to be packed into exosomes. 36 This short EXOmotif sequence is overrepresented in miRNAs that are commonly enriched in exosomes allowing exosomal miRNAs to be specifically recognized (and bound) by a transport protein, hnRNPA2B1. This motif controls the loading of these miRNAs into exosomes. 36 This specific 4 nucleotide sequence (GGAG) was overrepresented in miRNAs enriched in T-cell-derived exosomes. (3) The 3’ miRNA sequence-dependent pathway: adenylation and uridylation of miRNA at its 3’ end can contribute to direct miRNA sorting into exosomes suggesting that the 3’ end may harbor critical sorting signal. 37 (4) The miRNA-induced silencing complex (miRISC)-related pathway: mature miRNAs can interact with assembly proteins GW182, and AGO2 to form miRISC complex. miRISC components co-localized with multi vesicular bodies and there is a correlation between AGO2 and exosomal miRNA sorting. Knockout of AGO2 can decrease the types or abundance of the preferentially exported miRNAs. 38 In summary, specific sequences present in certain miRNAs may guide their incorporation into exosomes; certain enzymes or proteins may control the sorting of exosomal miRNAs 39 and mRNA targets of miRNA being sorted may also play a role. 34
It is now well established that endogenous miRNAs are transferred between immune cells and functional in recipient cells, thereby contributing to the inflammatory response. Exosome-mediated transfer of miRNAs between T cells and APCs is enhanced by the formation of a functional immune synapse. Though first described in cells of the adaptive immune system including T and B cells, the description now encompass interactions involving innate immune cells such as Natural Killer (NK) cells. 40 The immune synapse promotes exchange of miRNA-loaded exosomes between a T cell and its cognate APC. 3
Several of the LPS-responsive miRNAs detected in exosomes have validated mRNA targets that encode proteins involved in TLR signaling, chemokine signaling, and the transforming growth factor-beta (TGF-β) pathway. 41 Comparison of miRNA profile in exosomes purified from THP-1 cells, a human-derived monocytic cell line, and RAW 264.7 mouse macrophage, with and without LPS stimulation showed that the exosomal biomolecular signature will differ between cell types and between species. Thus, the species and the physiological state of the cells secreting the exosomes will influence the molecules in the vesicles. 31 The same study showed that there was a striking shift in the most abundant noncoding RNA populations after LPS stimulation of RAW 264.7 cells. The presence of pre-miRNAs and snoRNAs suggests that exosomes deliver molecules that could induce temporal epigenetic regulation in recipient cells regulating the course of inflammatory gene expression. 30 The influx of inflammatory-relevant pre-miRNAs in LPS-stimulated cells could indicate the need for a rapid response to inflammation rather than regulation at the nuclear level. Mature miRNAs may fine-tune the regulation of inflammation by altering the mRNA levels of inflammatory proteins immediately, while pre-miRNAs offer a second wave of regulation at a later time point. Exosomes have the advantage of delivering proteins and miRNAs that are primed to act directly and immediately along with mRNAs that can be readily translated. 31
A recent study showed the presence of mRNA fragments enriched in the 3’ untranslated regions (UTR) in exosomes. 42 The 3'UTRs of mRNAs have multiple regulatory roles and harbor sequences for numerous RNA-binding proteins that modulate the stability and translational efficiency of mRNAs. They also have multiple miRNA target-binding sites that guide the RNA-induced silencing complex (RISC), resulting in miRNA binding by seed sequence complementarity, inducing degradation or translational repression. It was suggested that the mRNA fragments transported by exosomes may act as competing RNAs to regulate stability, localization, and translational activity of mRNAs in target cells. 42
Immunomodulatory Role of Exosomal Proteins
Cells secrete a variety of vesicles that are heterogeneous in size and biogenesis. Protein markers are often used to differentiate EVs. A recent study systematically performed comparative analysis of the protein composition of all EVs recovered in the different steps of the differential ultracentrifugation protocol that is classically employed to isolate exosomes. 43 Human primary monocyte-derived DCs were used as source cells because of their ability to promote immune responses. Quantitative proteomic analysis demonstrated that some protein markers are unique to exosomes, whereas some others are generic. They identified three different tetraspanins -CD63, CD81, or CD9 - as specific protein markers for exosomes. 43
Exosomes are enriched in proteins and they do not contain a random set of proteins, as would be the case for cell debris; the proteome of exosomes includes cytosolic, nuclear, endosomal, and membrane proteins. 44 Databases compiling protein composition of exosomes from various studies include Exocarta, 45 Vescilepedia, 46 and EVpedia. 47 A number of endosome-associated proteins (Rab GTPase, SNAREs, Annexins, and flotillin) and proteins involved in exosome biogenesis (ESCRT complex, ALIX, TSG101), tetraspanins mentioned above, heat shock proteins (HSP70, HSP90), and MHC are present in exosomes. 48 Exosomes are carriers of important soluble mediators such as cytokines, and for cyto-kines that lack an N-terminal signal peptide, exosomal release represents a form of leaderless secretion. 49 A list of cytokines secreted by EVs has been reported. 49 LPS stimulation of RAW 264.7 mouse macrophage cells can induce the secretion of cytokines into culture media after 24 hours. Exosomes derived from RAW 264.7 mouse macrophages after LPS stimulation showed increased cytokines, predominantly chemokines. Of the 16 cytokines secreted by RAW 264.7 cells after LPS stimulation, 10 were detected in RAW 264.7 cell-derived exosomes. 31 Systematic studies have not been conducted to determine the complete spectrum of EV-associated cytokines. Additionally, the extent to which vesicular localization of cytokines affects conventional cytokine measurements has not been addressed. 49
Therapeutic Benefits of Exosomes
Exosomes can confer therapeutic benefits by attenuating or stimulating the immune response. 5 DC-derived exosomes (Dex) can amplify immune responses in vivo, by transferring peptide-MHC complexes from DCs that have been exposed to an antigen to another DC that have not been in contact with the same antigen (Fig. 1). 5 The ability of the exosomes to stimulate immune responses is dependent on the maturation state of the DCs secreting them. Mature DCs carry more MHCII, intercellular adhesion molecule-1 (ICAM-1), and costimulatory molecules and are more potent T-cell stimulators.50,51 Maturation state is also known to influence the miRNA profile of DCs-derived exosomes. 52

Immunomodulatory effects of exosomes derived from APCs.
The use of Dex has enabled immunotherapy and overcome some of the challenges associated with the use of DCs in clinical settings. In addition to being amenable to regulated manufacturing process and long-term storage, administering exosomes eliminate the risks associated with in vivo replication and lodging of cells in microvasculature. 15 Dex derived from tumor peptide-stimulated DCs can prime tumor-specific cytotoxic T lymphocyte responses in vivo, and a single intradermal injection resulted in delayed tumor growth or a complete eradication of established murine tumors. 53 Strategies harnessing DCs or their functions driving tumor-associated antigen (TAA)-specific T-cell responses have been successful in cancer immunotherapy. 7 Exosomes maintain the ability of DCs to present TAA and to activate TAA-specific responses.54,55 The occurrence of antigen-presenting MHC I and II molecules along with the ICAM for adhesion and integrins for docking facilitates in vivo efficacy of exosomes. Two studies proved the feasibility of large-scale Dex production and confirmed the safety profile for Dex administration in patients.56,57 Clinical trials were conducted on patients with advanced stage melanomas 56 or non-small cell lung carcinomas (NSCLC) 57 expressing melanoma-associated antigen. These studies using the first generation of Dex in end-stage cancer demonstrated that Dex exerted NK-cell effector functions in patients, but only minimal or no melanoma-associated antigen-specific T-cell responses were observed.56,57
The low immunogenic capacities of Dex led to the development of second-generation Dex, aimed at boosting NK and T-cell immune responses. Dex secreted by interferon-γ (IFN-γ)-treated DCs express higher levels of CD40, CD80, CD86, and ICAM-1 molecules compared with Dex from immature DCs, which in turn enhanced the immunogenicity. 58
A phase-II clinical trial was conducted on patients with inoperable NSCLC without tumor progression. IFN-γ-Dex loaded with MHC class I- and class Il-restricted cancer antigens were used as maintenance immunotherapy after induction chemotherapy. In this study that enrolled 22 patients, the primary endpoint, which was to observe at least 50% of patients with progression-free survival at four months after chemotherapy cessation was not met. However, this study confirmed that these Dex can boost the NK cell arm of antitumor immunity in patients with advanced NSCLC. 59
Exosomes derived from APC have been shown to confer immunosuppressive effects in different disease models including Rheumatoid Arthritis (RA). 60 RA is a systemic autoimmune disorder characterized by synovial inflammation and hyperplasia. DCs transduced with adenovirus expressing the IL-10 gene or treated with recombinant murine IL-10 suppressed the onset of murine collagen-induced arthritis.61,62 Several studies demonstrated positive outcomes using exo-somes derived from DCs engineered to over express certain genes including IL-10, IL-4, FasL, and indoleamine 2,3 diox-ygenase (IDO). A single dose of these exosomes systemically delivered after the onset of carrageenan-induced arthritis in mice effectively ameliorated disease progression, an effect that was absent with the direct injection of recombinant murine IL-10. 63 DCs genetically engineered to express the IL-4 were effective in reducing inflammation in murine arthritis. Exosomes derived from these DCs were shown to be effective in reducing the severity and the incidence of established arthritis when delivered systemically and the effects were significantly higher than those observed after repeated injection of recombinant IL-4. 61 Exosomes derived from FasL-expressing DCs, similar to the effect of their parent cells, were able to suppress collagen-reactive T cells and inhibit the progression of murine carrageenan-induced arthritis after systemic injection. 62 FasL-expressing DCs also showed an anti-inflammatory effect in a murine delayed hypersensitivity model upon local administration. 64 Exosomes from DCs expressing IDO reduced inflammation, inhibited T cell activation, and suppressed T-cell responses to auto- and alloantigens by tryp-tophan starvation and/or production of toxic metabolites. 65 Exosomes derived from DCs treated with IL-10 suppressed trinitrobenzene sulfonic acid (TNBS)-induced colitis. 66 Thus, these studies point to the immunosuppressive effects of exosomes derived from modified DCs, suggesting their potential therapeutic potential (Fig. 1).
Exosomes from LPS-stimulated macrophages significantly reduced paw swelling in mice caused by a single intraplantar injection of complete Freund's adjuvant (CFA). A single injection of exosomes attenuated thermal hyperal-gesia in a mouse model of inflammatory pain, suggesting an immunoprotective role for macrophage-derived exosomes (Fig. 1). In CFA-treated animals, injection of exosomes purified from LPS-stimulated macrophages induced a transient increase in thermal hypersensitivity that was not observed in saline-treated animals. By 24 hours, CFA-treated animals that received injections of exosomes from LPS-stimulated RAW 264.7 cells had increased paw withdrawal latency and hence reduced thermal hyperalgesia compared with CFA-treated animals that received PBS. At 48 hours, CFA-treated animals displayed reduction in thermal hyperalgesia in response to exosome administration from both LPS-stimulated and naïve macrophages indicating that the reduction in thermal hyper-sensitivity observed after 48 hours was independent of the inflammatory status of the macrophages from which these exosomes were derived. The absence of pain and swelling in saline-treated paws after exosome injection demonstrates that exosomal delivery does not produce a proinflammatory response. Attenuation of thermal hyperalgesia by macrophage-derived exosomes in CFA-treated animals could reflect the temporal regulation that exosomes can mediate by synergistically influencing multiple inflammatory pathways through the delivery of immediate-acting biomolecules such as cytokines and those that are translation dependent to induce changes in recipient cells by influencing gene transcription. 31
There are also reports of anti-inflammatory drugs inducing alterations in exosome composition. Sulfasalazine and methotrexate are used in the management of RA due to their anti-inflammatory properties. 67 Treatments of human synovial sarcoma cell line SW982 with sulfasalazine and methotrexate altered the protein profiles of exosomes. Combination of the two drugs suppressed part of the protein profile changes induced by IL-1β. Most of the identified proteins were immunity- or anti-oxidation-related proteins 68 and the authors suggest that exosomes may have a role in mediating the actions of anti-inflammatory drugs.
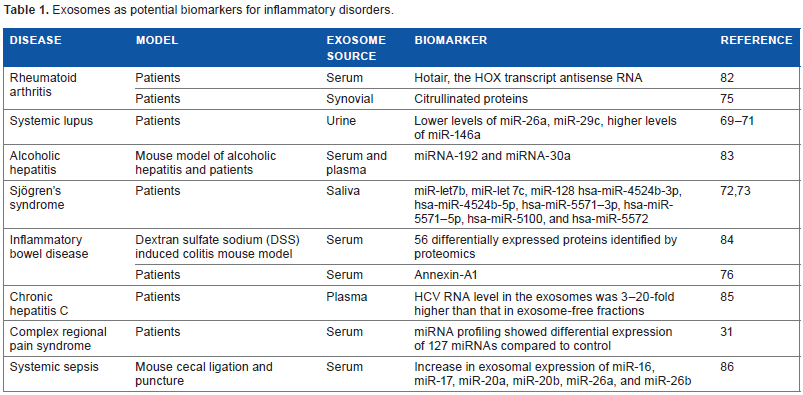
Exosomal contents can also serve as biomarkers for inflammatory disorders. The presence of exosomes in bodily fluids, a biomolecular composition rich in RNA, proteins, and lipids, along with the plasticity of exosomal content in response to various physiological stimuli and pathological states render exosomes an ideal biomarker for disease state. Table 1 lists the inflammatory disorders for which exosomes were investigated as potential biomarkers. miRNAs transported by exosomes may serve as biomarkers for several inflammatory and autoimmune disorders including systemic lupus69–71 and Sjögren's syndrome.72,73 The number of miRNAs that are differentially expressed in exosomes from patients with complex regional pain syndrome were much higher than that observed in whole blood (18 in whole blood 74 vs 127 in exosomes 31 ), suggesting that exosomes may be a better source for biomarker studies. Exosomes also transport important protein biomarkers such as citrullinated proteins in RA 75 and annexin-A1 in inflammatory bowel diseases. 76
Exosomes as potential biomarkers for inflammatory disorders.
Conclusions
Immense interest in understanding the role of exosomes, and all EVs in general, in both normal physiology and disease states is continuing to grow. Immunomodulatory functions of exosomes from DCs have been explored in clinical trials for cancer and will provide guidance, as therapeutic approaches are being pursued for other inflammatory disorders. Exosomes hold immense promise for therapeutic drug delivery. They confer a number of advantages over other vector-or liposome-based delivery methods. They act as natural, non-toxic membranous nanocarriers of bio-macromolecules for effective delivery of the desired compound. Exosomes isolated from the recipient's own body fluids or cell culture (autologous exosomes) can be used for loading biological drugs. 77 Exosomal composition is also being explored as biomarkers and as a noninvasive approach for early diagnosis of various disorders. 78 The role of exosomes in normal physiology is less explored. There is concerted effort from the EVs community to standardize the purification methods and this is crucial in validating published reports.79–81 Exosome biology will undoubtedly be important in the implementation of strategies to make precision medicine a reality for a broad spectrum of diseases.
Author Contributions
Wrote the first draft of the manuscript: BS, SA. Developed the structure and arguments for the paper: SA. Made critical revisions: SA. Both authors reviewed and approved of the final manuscript.
