Abstract
Ulipristal acetate (UPA) is a progesterone receptor modulator that is available for emergency contraception (EC) and can be taken up to 120 hours after unprotected intercourse. A meta-analysis of clinical trials comparing UPA with levonorgestrel (LNG) for EC, demonstrated that UPA has higher efficacy than LNG. This higher efficacy is supported by biomedical studies that have demonstrated that UPA is a more potent inhibitor of ovulation, being able to delay ovulation in the immediate preovulatory period, when LNG is no longer effective. A recent study that explored risk factors for failure of EC, demonstrated that obese women were at increased risk of EC failure, with either UPA or LNG. However, failure was significantly less amongst women receiving UPA than those receiving LNG. There is growing evidence therefore, that UPA should be the preferred oral method of EC.
Ulipristal Acetate
Ulipristal acetate (UPA) is a progesterone receptor modulator that has been developed and approved as an oral method of emergency contraception (EC) for use up to five days after sex; a time span that corresponds to the lifespan of sperm in the reproductive tract. A single dose of 30 mg UPA for EC (ellaOne®, HRA-Pharma, Paris, France) was approved by European Medicines Agency (EMEA) in May 2009 and by the Food and Drug Administration (FDA) in USA in June 2010 (Ella®). This clinical review covers findings from English language articles published between 2000 and 2011 on UPA for EC.
Mode of Action
Stratton et al demonstrated that giving a single dose of UPA (10, 50, 100 mg) in the follicular phase of the cycle could delay or prevent ovulation, and that this effect was dose dependant. 1 The time is the cycle when the probability of conception is greatest is the immediate pre-ovulatory phase, when the risk of conception is approximately 30%. 2 In a study by Brache et al, UPA was shown to be capable of delaying ovulation in this pre-ovulatory period, (even after the LH surge) when ovulation is imminent in the following 48 hours. 3 In this double- blind, placebo–controlled, cross-over study, subjects received 30 mg UPA (same dose in ellaOne®) or placebo, once ultrasonographic assessment revealed that the dominant follicle had reached a diameter of 18 mm (immediate preovulatory). All subjects who received placebo ovulated by five days, but in subjects who received UPA there was no ovulation by five days in 59% of women. This five day period is notable because it corresponds to the lifespan of sperm in the female reproductive tract. In contrast, the same team had previously shown, in a study of similar design, that the EC dose of levonorgestrel (1.5 mg), when given at the same point in the cycle was not capable of delaying ovulation any more than a placebo tablet. 4 These biomedical studies demonstrate clearly that UPA is a more potent inhibitor of ovulation, than levonorgestrel (LNG) and can delay ovulation even when taken in the immediate pre-ovulatory phase of the cycle, when LNG is no longer effective. This also suggests that UPA should be a more effective method of EC.
A study conducted to examine the effects of UPA (10, 50, 100 mg) when given after ovulation, demonstrated that when taken in the early luteal phase, UPA does not affect the length of the cycle, nor does it affect circulating levels of progesterone. 5 In this study, histological assessment of endometrium revealed that UPA did not retard the development of a secretory endometrium. Although there was a trend for thinner endometrium in UPA treated cycles (based upon ultrasound measurement), the endometrial thickness was still within the normal range for this phase of the cycle. Endometrial samples were also subjected to analysis of molecular markers of a secretory endometrium. There was a trend for these progesterone dependent molecular markers to be reduced by UPA, in a dose dependent manner. Taken together, this data would suggest that the dose of UPA in ellaOne®, may be too low to exert significant ‘anti-fertility’ effects on the endometrium. The mode of action by which UPA works as EC is likely to be exclusively through effects on ovulation alone.
Efficacy
To date, there have been two comparative studies of UPA and LNG for EC. Both studies were designed as ‘non inferiority studies’, powered to show that UPA was not inferior to LNG. The first study was conducted in the USA and recruited over 1600 women who presented within 72 hours of unprotected sex. 6 Women were randomized to received either 1.5 mg of LNG (taken as two separate doses of 0.75 mg, 12 hours apart) or 50 mg (non micronized) UPA (taken as 50 mg UPA followed by placebo 12 hours later). Of the 774 women followed up after LNG there were 13 pregnancies giving a pregnancy rate of 1.7% (95% CI 0–8 to 2.6). Of the 775 women followed up after UPA, there were 7 pregnancies, giving a pregnancy rate of 0.9% (95% CI 0.2–1.6). The lower pregnancy rate with UPA was not statistically significant, but nevertheless this study showed that UPA was not inferior to LNG. 6
The second study, was conducted in both Europe and USA, and recruited women presenting within 120 hours of unprotected sex. 7 Women were randomized to receive either a single 1.5 mg dose of LNG or a 30 mg (micronized) dose of UPA. Previous studies had demonstrated that 1.5 mg LNG was as effective as two separate doses of 0.75 mg, and a pharmoacokinetic study had shown that 30 mg micronized UPA was bio equivalent to the 50 mg non micronized dose.8,9
Out of 954 women followed up after LNG and 939 women followed up after UPA, there were 25 pregnancies in the LNG group and 15 in the UPA group, giving a pregnancy rate of 2.6% (95% CI 1.7–3.9) and 1.6% (95% CI 0.9–2.7) for LNG and UPA respectively. Once again the lower pregnancy rate with UPA was not statistically significant, but nevertheless this study showed that UPA was not inferior to LNG. 7
A non-comparative study using UPA (30 mg micronized) was conducted in the USA that examined use of UPA for EC in over 1200 women presenting between 48 hrs and 120 hrs after sex. A pregnancy rate of 2.1% was observed, which was significantly less than the 5.5% pregnancy rate that would have been expected in the absence of EC. 10 This study also confirmed that the efficacy of UPA did not decrease with time; pregnancy rates were 2.3%, 2.0% and 1.3% respectively for EC intake 48–72 hours, 73–96 hours and 97–120 hours after sex. 10
Both of the RCT's with LNG and UPA have been combined in a meta-analysis. 7 The meta-analysis was conducted in order to have a greater number of subjects and hence a greater power to show a statistical difference between the treatments, if indeed one exists. The meta-analysis showed that women presenting within 72 hours or within 120 hours had almost half the risk of pregnancy compared to women receiving LNG (Table 1). However, those women presenting within 24 hrs of unprotected sex, had almost one third the risk of pregnancy compared to counterparts receiving LNG. 7
Comparative efficacy of UPA and LNG for EC, in RCT's and meta -analysis according to time from unprotected sex to EC.
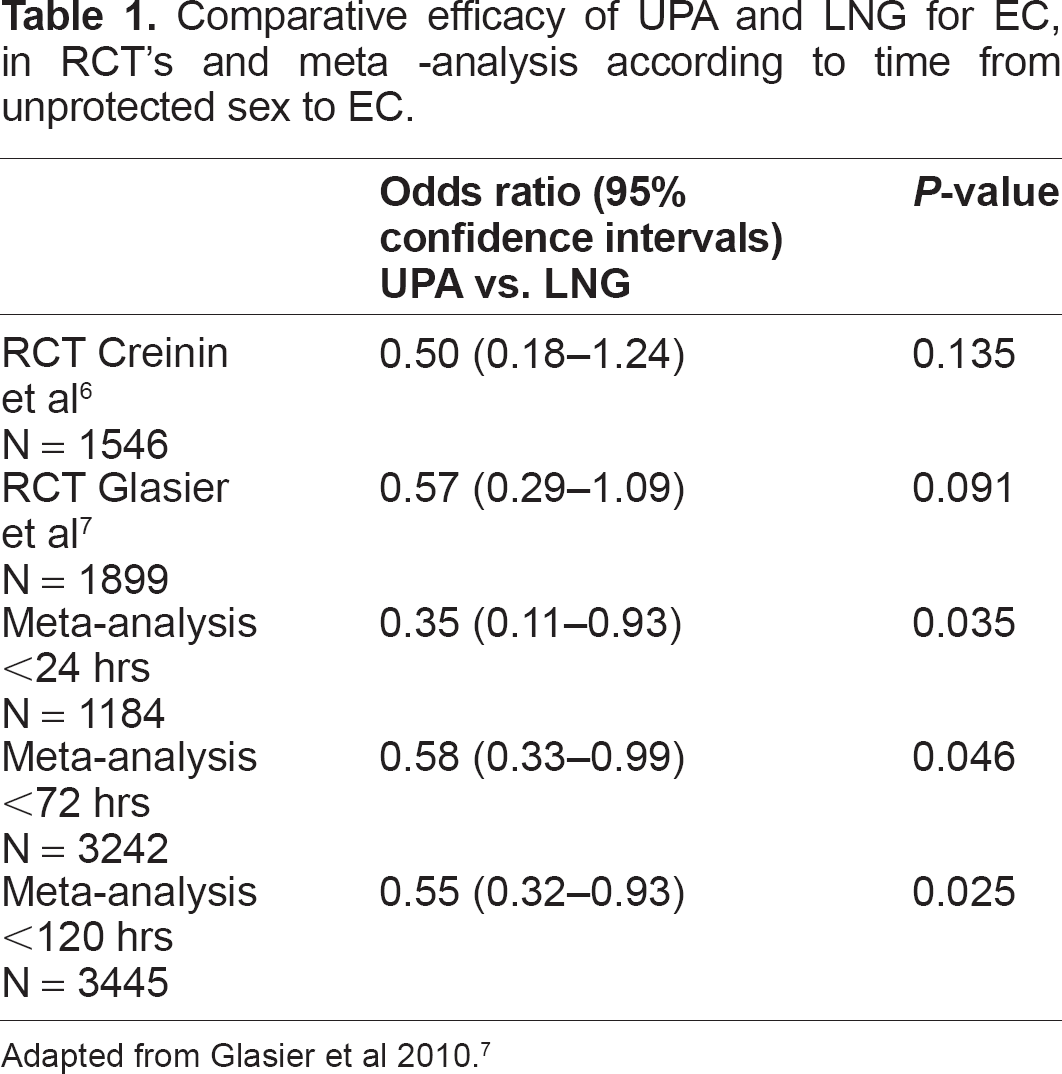
Adapted from Glasier et al 2010. 7
Safety Data
In both RCT's to date, the pattern of side effects reported by women has been the same for both LNG and UPA. The commonest reported side effects being headaches (19%), dysmenorrhoes, fatigue nausea, dizziness and abdominal pain (Fig. 1). 7 However, these side effects were generally considered ‘mild’ and it should be noted that in the double blind study of Brache comparing the effects of UPA and placebo on follicle development, that similar side effects were reported by women in both placebo and UPA groups. 3
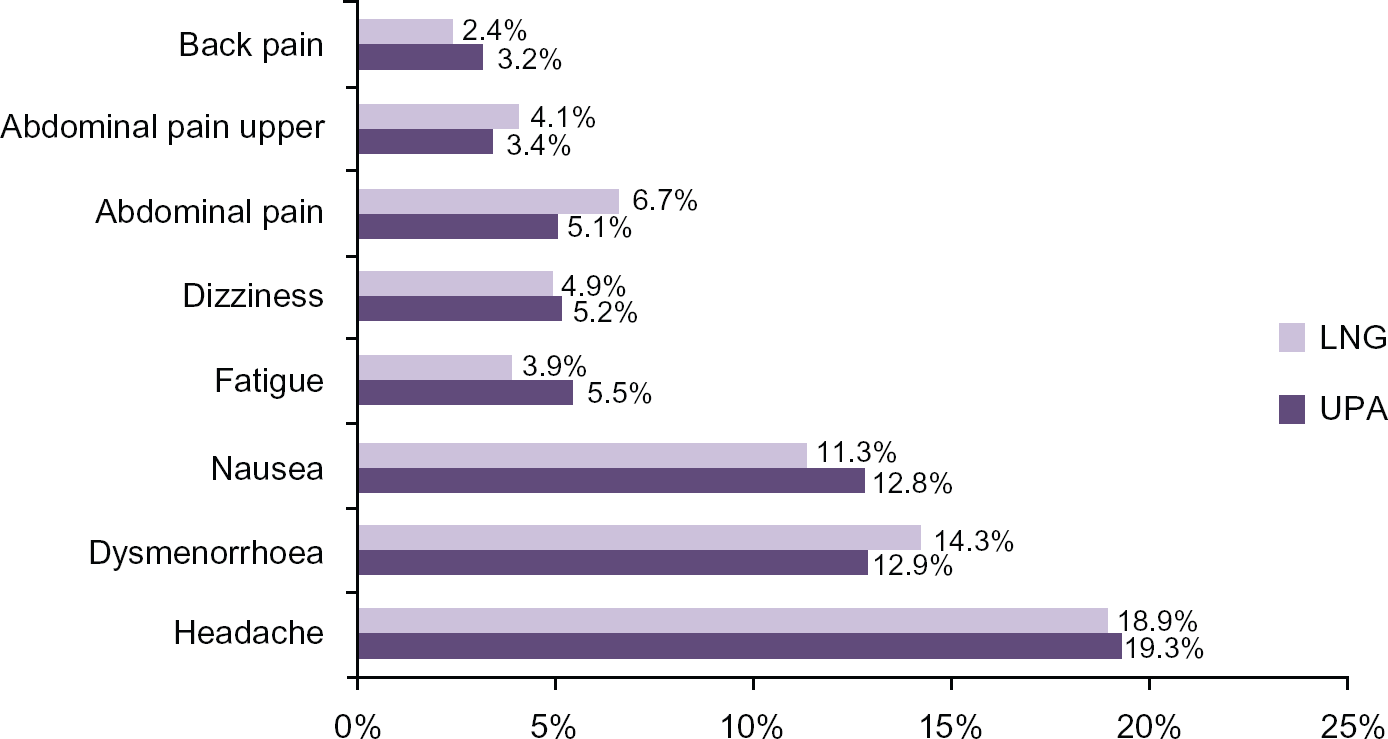
Percentage of women with adverse events—adapted from Glasier et al. 7
In the RCT' of UPA and LNG, there has been a trend for women treated with UPA, to have their period after treatment to be delayed by on average 2 days, compared to women treated with LNG to have their next menses arrive on average 1 day earlier than expected. 7 Most women (76%) receiving UPA or LNG (71%) did have their subsequent period within 7 days of the expected date. 7 Furthermore, treatment with UPA did not affect the volume of menses. 7 Thus, the advice for women who use UPA for EC is the same as for those using LNG, that if their period has not arrived by one week of the expected date that they should have a pregnancy test.
In the clinical trials to date, over 4000 women have been treated with UPA. Since ellaOne® became available as EC, more than one quarter of a million women had received treatment with it by May 2011 (HRA- personal communication). At present, there is no data on the effect of repeated doses of UPA within the same cycle and so repeat use of UPA for EC is not currently recommended.
To date, data on the outcome of pregnancies that have been exposed to UPA is limited. In the studies, the proportion of women who became pregnant was low (<2%), and the majority of these (three quarters) opted for a termination of pregnancy.6,7 The proportion of women in the studies who were pregnant after taking UPA and who had a miscarriage was no higher than what one would expect in a normal population. 7 The total number of women who became pregnant after UPA in the all the clinical trials and who chose to continue the pregnancy have been few in number and many of these women have been lost to follow-up. The outcome of those for whom follow-up is available has been a normal birth. 9 In order to obtain robust information on the outcome of future pregnancies exposed to UPA, an agreement between the European Medicines Agency (EMEA) and the market authorization holder, HRA Pharma, has been that a registry has been created to collect information on any pregnancy exposed to UPA, such as an unrecognised pregnancy before EC intake or following treatment failure.
It is not known whether UPA is excreted in human milk, since such studies have not yet been conducted. However, since UPA is a lipophilic compound, it could in theory be excreted in human milk. Current recommendations are therefore that breast-feeding should not take place for at least 36 hours following UPA intake. 11 For LNG, the corresponding recommendation is to avoid breast-feeding for at least eight hours but not more than 24 hours after LNG intake. 12
Risk Factors for Failure of EC
An analysis of the data from the two RCT's of LNG and UPA was undertaken to determine factors that were associated with a risk of failure of EC. 13 Three factors; intercourse taking place during the estimated fertile window, occurrence of further episodes of sex after EC and a raised body mass index (BMI) were significantly associated with failure of both UPA and LNG (Table 2). Women who had sex during the fertile window (5 days before estimated date of ovulation to one day after) had more than a threefold increase in the risk of failure of EC compared to women who had sex out with the estimated fertile window. This, together with the finding that women who had further episodes of sex were at higher risk of pregnancy, was not unexpected, since it has previously been shown in other studies.8,14 However, the finding that raised BMI was associated with a higher risk of EC failure was a new finding. When effect of BMI was analysed according to treatment groups, it was found that the impact of BMI was more pronounced amongst women who received LNG. 13 Women who were obese who received LNG had a statistically significantly higher risk of an unintended pregnancy than women receiving UPA (P = 0.002) (Table 3). Obese women (BMI ≥ 30 kg/m2) receiving LNG had almost four times the risk of an unintended pregnancy compared to normal or underweight weight women (OR 4.41 95% CI 2.05–9.44). For UPA, the corresponding odds ratio for failure of treatment in obese women compared to normal or underweight women was OR 2.62 (95% CI 0.89–7.00). Further analyses revealed that the BMI of women at which the expected pregnancy rate was no different to what one would expect in the absence of EC was 26 kg/m2 LNG compared with and 35 kg/m2 for UPA. If expressed in terms of weight alone, the corresponding figures were 70 kg for LNG and 88 kg for UPA. 13 This data is clinically important because it strongly suggests that obese women who require EC and who do not wish an IUD for EC should have UPA rather than LNG.
Factors associated with risk of failure of either UPA or LNG for EC (based upon data of Glasier et al 2011. 13

Fertile window (day of ovulation minus 5 to plus 1) compared to outside fertile window.
Pregnancy risk according to BMI group and treatment with either LNG or UPA.
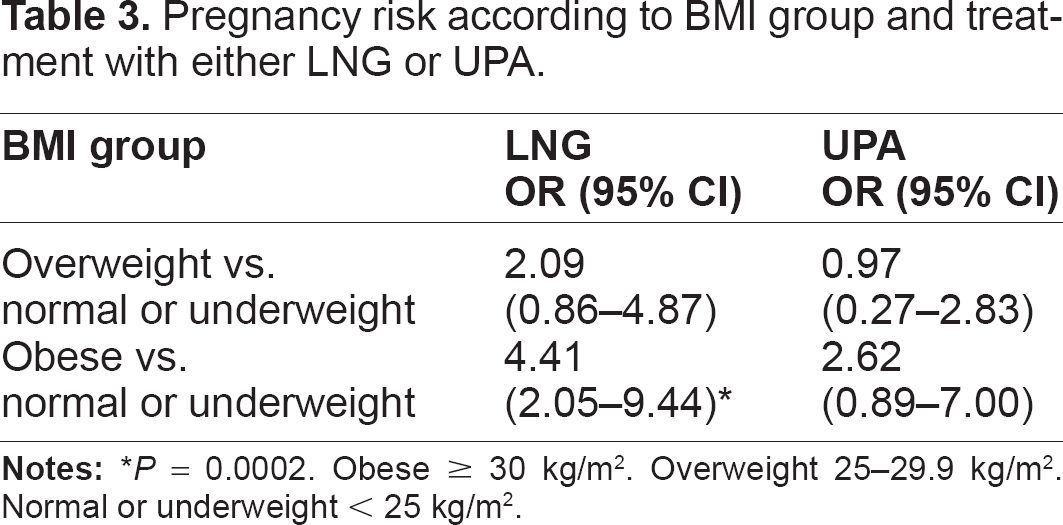
P = 0.0002. Obese ≥ 30 kg/m2. Overweight 25–29.9 kg/m2. Normal or underweight < 25 kg/m2.
Contra Indications
Contraindications to UPA are few. Use of UPA for EC is not advised in women with severe renal or hepatic impairment (due to route of metabolism of drug) and in women with uncontrolled severe asthma (UPA has some antagonist activity at the glucocorticoid receptor). Metabolism of UPA is mainly through the cytochrome enzymes (CYP3A4) is. 11 Although specific drug interaction studies have not been conducted, it is possible that inducers of these enzymes, eg, rifampin, St. John's Wort and certain anti-epileptics (carbamezapine, phenytoin, phenobarbital) may induce the metabolism of UPA and cause lowered plasma levels. Concomitant use is therefore not recommended. Enzyme induction wears off slowly and effects on the plasma concentrations of UPA may occur even if a woman has stopped taking an enzyme inducer within the last 2–3 weeks. Furthermore, inhibitors of the cytochrome enzymes such as antiretrovirals, itraconazole, clarithromycin, and grapefruit juice, may inhibit the metabolism of UPA and could lead to increased plasma levels. 11 Concomitant administration of UPA with drugs that increase gastric pH (eg, proton pump inhibitors, antacids and H2-receptor antagonists) is also not recommended since these may reduce plasma concentrations of UPA with a possible decrease in efficacy of UPA. 11 The medicinal product ellaOne® contains lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should also be advised not to take this medicine. 11
Hormonal Contraception after UPA
Given the finding that further unprotected sex after EC is associated with a significantly increased risk of pregnancy,13,14 it is important that women establish an effective method of ongoing contraception immediately after EC. This is often termed ‘quick-start'. The term ‘bridging contraception' is also used in the context of starting a temporary effective method until the chosen method is available. For example after EC, a woman may use an oral contraceptive pill as a ‘bridging’ method until she can have her chosen long term method such as an implant inserted.
Given the fact that UPA is a progesterone receptor modulator, there is theoretical concern that it might alter the effectiveness of progestogen containing contraception. The summary of product characteristics for ellaOne® recommends that women await their menses after treatment before commencing hormonal contraception. 11 The Clinical effectiveness Unit of the Faculty of Sexual and Reproductive Healthcare (FSRH), UK, have recently issued recommendations about quick starting contraception after EC. 15 They recommend that after UPA, that if a woman wishes to a quick-start the combined oral contraceptive pill (COCP), that she should also abstain/use barrier methods of contraception for 14 days before she can rely exclusively on the COCP for contraception. 15 This advice is based not upon evidence, but upon theory and assumptions about the time taken to clear UPA. The half- life of UPA is the region of 32 hours. 11 It is estimated that it takes between 5 to 7 half lives to eliminate UPA, which is approximately 7 days. Since it is generally accepted that it takes 7 days of COCP pills to have ovarian quiescence, the FSRH advise use of 14 days of additional contraceptive precautions ie, 7 days to clear UPA plus the 7 days of pills to achieve ovarian quiescence. 15 The FSRH advise that if a woman wishes to quick-start the progestogen only pill (POP) after UPA, that she should use additional contraceptive precautions for a total of 9 days 15 ie, 7 days to clear UPA plus 2 days to achieve a contraceptive effect of POP on cervical mucus.
Cost Effectiveness
A cost effectiveness analysis was conducted in the UK to determine how much extra it cost the National Health Service (NHS) to avoid an additional unintended pregnancy by using UPA rather than LNG. 16 This study calculated the drug costs and the direct costs of an unintended pregnancy (miscarriage, induced abortion or birth), using NHS data for 2008. This study factored in the risk of pregnancy after EC and the relative proportion that end in induced abortion, miscarriage or a birth, based upon clinical trial data from the meta analysis of the two comparative trials to date. 7 The calculated cost of preventing one additional unintended pregnancy with UPA (rather than LNG) was calculated to be £311. This was significantly less than the cost of an unintended pregnancy (£948) regardless of the outcome (birth, miscarriage) or the cost of an induced abortion (£672). The authors concluded therefore that UPA was a cost effective alternative to LNG. 16
Summary
It is clear from the studies to date that UPA is not only a method of EC that can be taken up to 120 hours but is a more effective EC than LNG, and is just as well tolerated as LNG. According to data from the meta-analysis, if UPA is taken within 120 hours of unprotected sex, it is associated with almost half the risk of pregnancy compared to if LNG is used for EC. If taken within 24 hours of sex, it is associated with almost one third the risk of pregnancy compared to LNG. UPA is also more effective than LNG amongst women who are of raised BMI. Clearly there are questions that do remain about its use, such as how many days after taking UPA should a woman who wishes to start hormonal contraception have to wait before she can rely exclusively on the hormonal contraceptive to protect her from pregnancy. Current guidelines on this area are based on theory and may be too restrictive. In spite of this, the current evidence clearly shows ever that UPA should be the preferred oral method of EC.
Footnotes
Author Contributions
Wrote the first draft of the manuscript: STC. Writing of the manuscript: STC. Developed the structure and arguments for the paper: STC. Made critical revisions and approved final version: STC. Reviewed and approved of the final manuscript.
Competing Interests
STC has received funding from HRA Pharma.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
