Abstract
Ulipristal acetate (UPA), a selective progesterone receptor modulator, when taken as a single 30-mg dose, is safe and effective for emergency contraception up to 5 days (120 h) following unprotected intercourse. This indication has been approved in Europe since May 2009 and was approved by the US FDA in August 2010. The older progesterone-only emergency contraceptive, levonorgestrel (LNG), is approved only up to 72 h after unprotected intercourse. UPA is effective in delaying or inhibiting ovulation, even if taken 24 to 48 h prior to expected ovulation, a time when LNG is no longer effective. A recent meta-analysis of two randomized clinical trials showed UPA to have a pregnancy risk 42% lower than LNG up to 72 h, and 65% lower in the first 24 h following unprotected intercourse. In a randomized trial enrolling women up to 5 days after unprotected intercourse, significantly more pregnancies were prevented with UPA than with LNG when taken beyond 72 h.
Half of all pregnancies in the USA are unintended. Of the 3.1 million unintended pregnancies in 2001, nearly 50% ended in elective abortion [1]. Emergency contraception (EC), sometimes referred to as the ‘morning-after pill’, helps prevent unwanted pregnancy following unprotected intercourse. Knowledge about EC is especially important for the 4.6 million women not using a regular contraceptive method and the 6.8 million women who rely on condoms for protection against pregnancy in case of slippage or breakage [2].
Up to now there are two forms of emergency contraceptive pills (ECPs) available in the USA: progestin-only and combined estrogen/progestin pills. They are approved for use only up to 72 h following unprotected intercourse. Progestin-only pills have largely supplanted combined estrogen/progestin regimens because of improved efficacy and reduced side effects.
Although approved for use up to 72 h following unprotected intercourse, efficacy of levonorgestrel (LNG) rapidly declines over time following intercourse [3,4].
A woman presenting beyond 72 h of unprotected intercourse can be proposed an insertion of a copper intrauterine device (IUD) (ParaGard®, Teva Pharmaceuticals, Petach Tikva, Israel), which is an off-label use for EC in the USA. The IUD is appropriate for women seeking to start long-term contraception. However, the IUD requires insertion by a trained clinician, is relatively expensive and invasive, and many women seeking EC are not seeking a long-acting contraceptive method.
There was, until recently, an unmet need for a highly effective ECP that is safe and highly effective up to 5 days following unprotected intercourse. Ulipristal acetate (UPA), a selective progesterone receptor modulator, is a new single-dose ECP which is effective up to 5 days after unprotected intercourse. It was approved in Europe in May, 2009 (ellaOne®, HRA Pharma, France) and in the USA by the FDA in August, 2010 (ella®, HRA Pharma, France). It is marketed in the USA by Watson Pharmaceuticals (CA, USA) and available to US women since December 1, 2010.
Overview of the market
Progestin-only pills
Levonorgestrel is currently approved in the USA, Europe and Asia for EC up to 72 h following unprotected intercourse (Plan B One-Step™, Teva Pharmaceuticals, Next Choice® and Watson Pharmaceuticals in the USA) and in Europe and Asia for EC up to 72 h following unprotected intercourse. Plan B One-Step and Next Choice are nonprescription in the USA if the patient is aged 17 years and older and available by prescription for females 16 and younger. They are kept behind the counter rather than on the shelf. The progestin-only ECPs have largely replaced the combined ECPs because they are more effective and have fewer side effects.
Combined estrogen & progestin pills
When dedicated ECPs are not available, combined oral contraceptives containing ethinyl estradiol and various progestins can be used for EC. The regimen for this is one dose followed by a second dose 12 h later, where each dose consists of two, four, five or six pills, depending on the brand
Prescriptive equivalents of common oral contraceptives for use as emergency contraception.

LNG: Levonorgestrel. Data taken from [102].
Copper T IUD
The copper T IUD is available for long-term contraception in the USA, marketed as Paragard. Insertion of Paragard® (Teva Pharmaceutical Industries Ltd, Israel) for EC up to 5 days following unprotected intercourse is an evidence-based, but off-label use. It is especially appropriate for a woman desiring both EC and long-term and highly effective reversible contraception. However, the IUD requires insertion by a trained clinician and is relatively expensive and invasive.
Ulipristal acetate
Ulipristal acetate is a selective progesterone receptor modulator that was approved in Europe (ellaOne) in May 2009 and in the USA (ella) in August 2010 as an ECP that can be taken from the first day on and up to 120 h following unprotected intercourse. It is only available by prescription in Europe as it is in the USA.
Introduction to the compound
Ulipristal acetate is an orally-active selective progesterone receptor-modulator. UPA was initially developed in the USA by the NIH and HRA Pharma licensed the rights to develop the compound in 2000.
Chemistry
Ulipristal acetate is a white-to-yellow crystalline powder. It is freely soluble in methylene chloride, soluble in methanol, acetone and ethanol, and insoluble in water. The structure of the active substance UPA is shown in
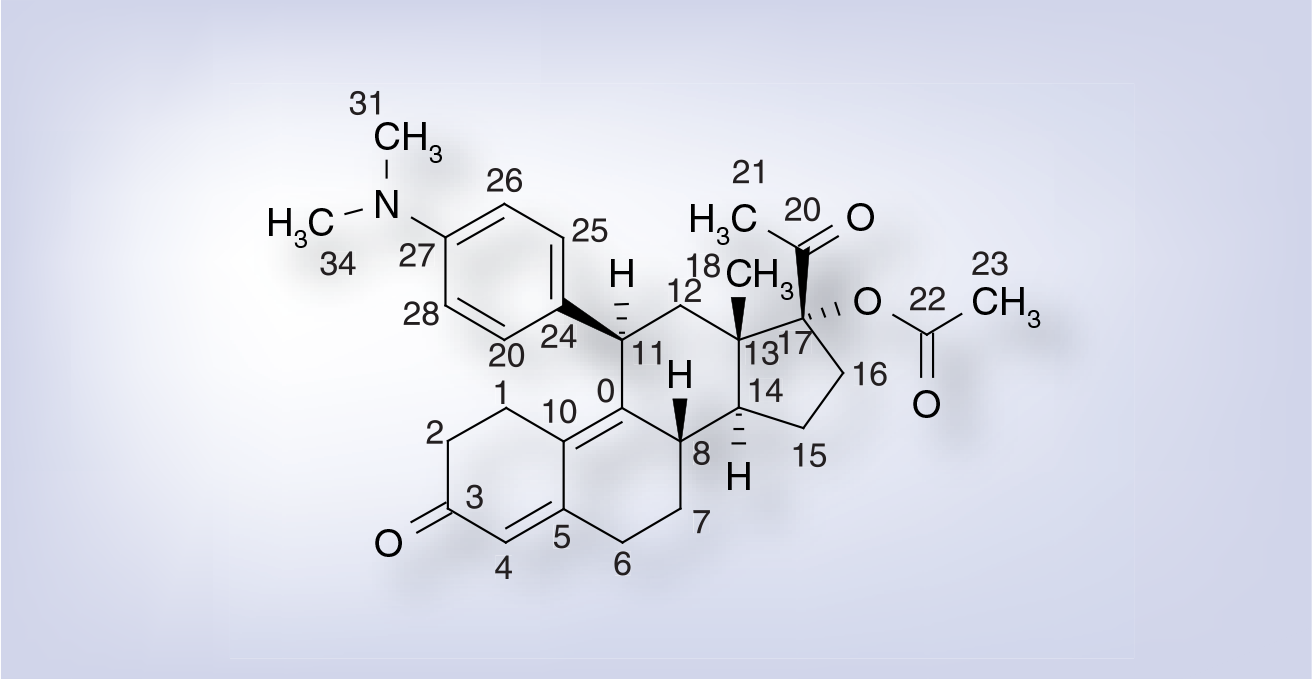
Ulipristal acetate.
Ulipristal acetate has five asymmetric carbons but only the following absolute configuration of the chiral centers are used: 8S, 11S, 13S, 14R and 17R.
Ulipristal acetate is a compound derived from 19-norprogesterone. UPA is a synthetic selective progesterone receptor modulator characterized by antagonistic and partial agonistic effects at the progesterone-receptor. UPA prevents progesterone from occupying its receptor, thus the gene transcription (normally turned on by progesterone) is blocked. Preclinical studies indicate that UPA displays potent affinity at the human progesterone and glucocorticoid receptors (1–6 times the affinity of progesterone and 1.5 times the affinity of dexamethasone) and shows in vivo antiglucocorticoid activity at doses approximately 50-fold greater than those needed for the antiprogestin effects. UPA binds to the androgen receptor with much weaker affinity (0.2 times the affinity of testosterone) and displays negligible affinity for the estrogen and mineralocorticoid receptors.
Pharmacodynamics
The mechanism of action of single-dose UPA is the delay or inhibition of ovulation. UPA is significantly more effective than LNG at delaying or inhibiting ovulation especially during the preovulatory period. When the leading follicle reaches 18–20 mm (and ovulation should occur within 48 h) and the probability of conception exceeds 30%, UPA prevents follicular rupture for at least 5 days following administration in 59% of cycles, compared with 12% in women administered LNG [5,6].
Pharmacokinetics & metabolism
Ulipristal acetate was evaluated with different formulations through Phase I, II and III clinical trials. The 30-mg micronized and the 50-mg nonmicronized formulations have similar absorption profiles according to indirect pharmacokinetic comparisons and demonstrated similar efficacy and safety profiles in clinical trials.
The main metabolites of UPA, CDB-3877 (mono-N-demethylated form) and CDB-3963 (di-N-demethylated form) are both pharmacologically active. CDB-3877 has affinity for the progesterone receptor nearly equivalent to its parent molecule. In vivo, CDB-3877 inhibited endometrial proliferation after oral administration with a lower magnitude of efficacy than UPA while CDB-3963 was only weakly active [7]. The pharmacokinetic properties of UPA are summarized in
Summary of pharmacokinetics for ulipristal acetate.
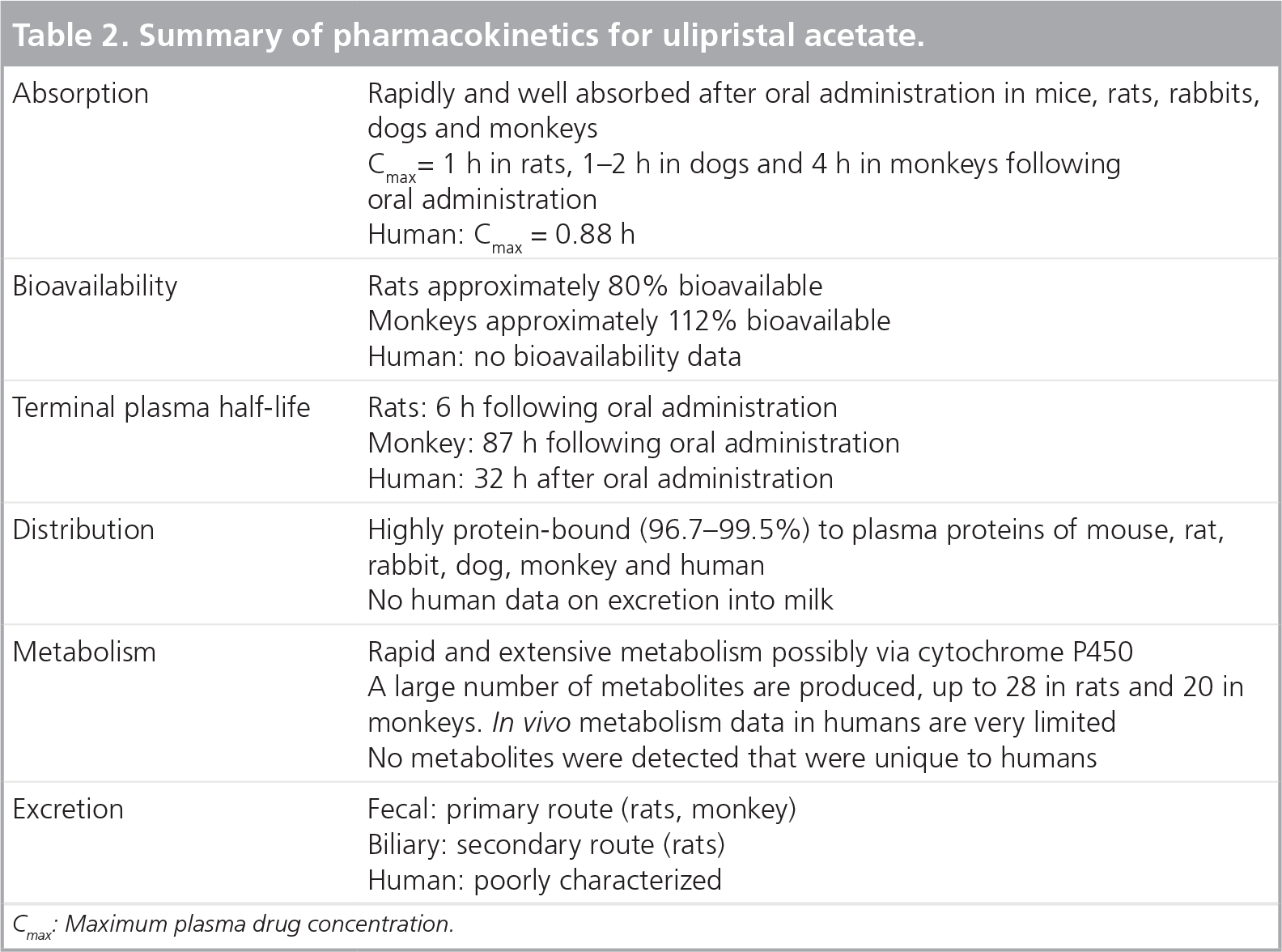
Cmax: Maximum plasma drug concentration.
Clinical efficacy
Phase I studies
The effects of single doses of UPA when administered at different points in the menstrual cycle have been studied in healthy women volunteers: an initial safety study in normally cycling women (mid-luteal phase administration) and two additional studies designed to determine the effect of giving the drug at different times in the menstrual cycle (follicular and luteal phases) on ovulation and endometrial maturation were conducted. No safety problems were observed with the administration of single doses up to 200 mg.
Phase II studies
The safety, efficacy and tolerability of UPA for EC have been characterized in two large-scale double-blind Phase II/III studies. The first study compared a single 50-mg dose of UPA unmicronized formulated in a gelatin capsule with the LNG reference method (two 0.75-mg doses taken 12 h apart), and the second compared gelatin capsules containing 10 mg micronized UPA with gelatin capsules containing 50 mg UPA unmicronized. These Phase II/III studies are listed in the following section.
A total of 1672 participants were enrolled in the first study carried out by Crenin et al., and equally distributed between UPA (n = 840) and LNG (n = 832) [8]. In the efficacy-evaluable population, defined as women treated with a known pregnancy status and excluding pretreatment pregnancies, there were 20 pregnancies overall (seven pregnancies among 773 subjects treated with UPA and 13 pregnancies among 773 subjects treated with LNG). The emergency contraceptive effectiveness of the two methods was examined over each 24-h period using Trussell's method (prevented fraction) [9]. The contraceptive efflectiveness of LNG was seen to decrease with time, as has been observed in the literature [4].
Overall, 76% of the subjects treated experienced an adverse event (77% UPA, 76% LNG). The most frequently reported adverse event in each treatment group was fatigue (38% UPA, 36% LNG), followed by headache (32% UPA, 33% LNG), and nausea (30% UPA, 24% LNG). Although nausea was more frequent in the UPA users, the incidence of vomiting was similar with UPA and LNG. Subjects in both treatment groups had similar bleeding and spotting experiences. Relative to the average cycle length reported by subjects on study entry, the treatment cycle was approximately 3 days longer for UPA subjects and approximately 2 days shorter following treatment with LNG.
Following the study by Creinin et al., the safety and efficacy of a lower dose of UPA (10 mg) was examined by protocol extension with identical eligibility criteria and study design [101].
The pregnancy rate for the participants administered UPA 50 mg was 1.3% (95% CI: 0.4–3.2) compared with 2.7% (95% CI: 1.3–5.0) for UPA 10 mg micronized.
Summary of Phase II/III studies of ulipristal acetate for emergency contraception.
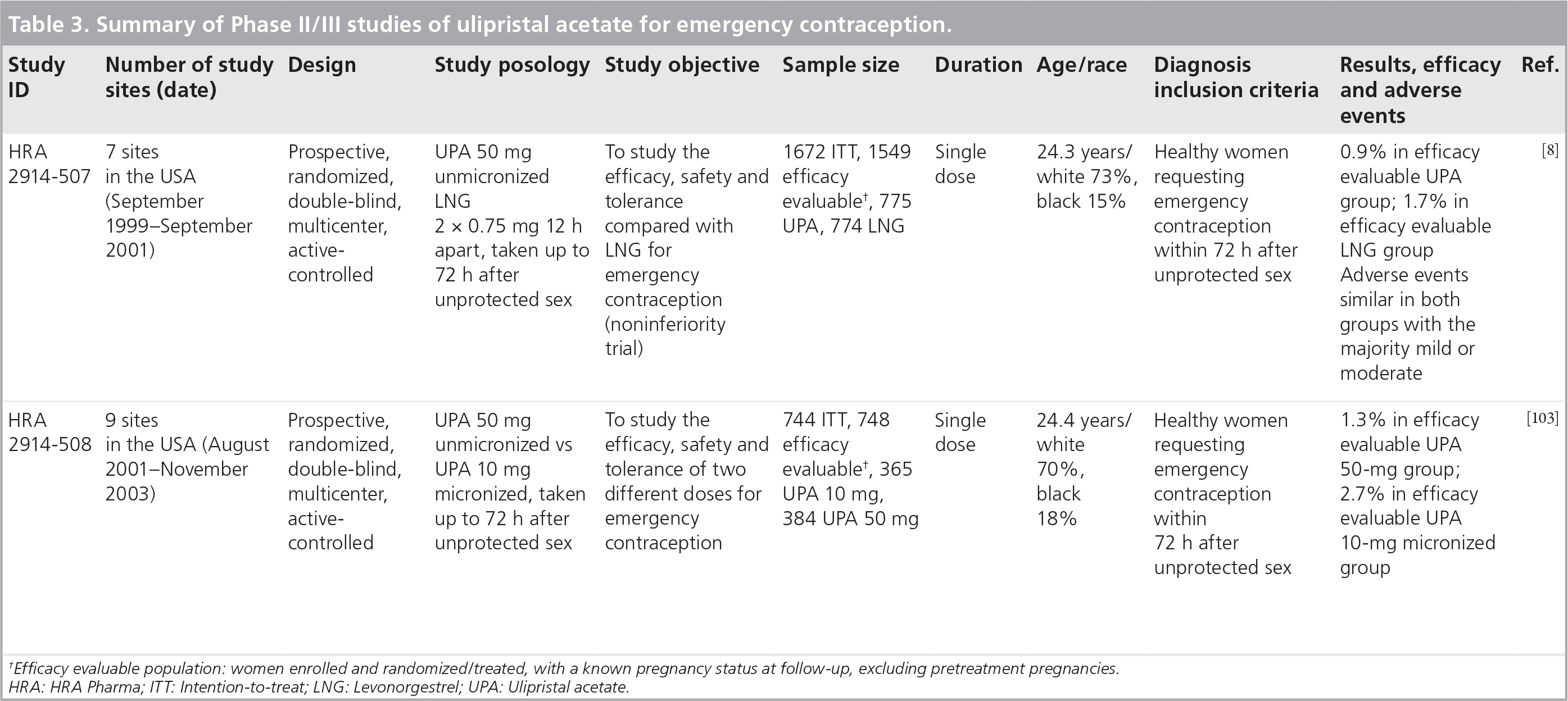
Efficacy evaluable population: women enrolled and randomized/treated, with a known pregnancy status at follow-up, excluding pretreatment pregnancies. HRA: HRA Pharma; ITT: Intention-to-treat; LNG: Levonorgestrel; UPA: Ulipristal acetate.
Phase III studies
The study entitled ‘UPA taken 48–120 h after intercourse for EC’ was an FDA pivotal multicenter, US prospective clinical trial [10]. The aim was to evaluate the efficacy and safety of UPA for EC taken as a single 30-mg micronized dose 48–120 h following unprotected intercourse in normally menstruating women.
A total of 1241 women were evaluated for efficacy with an overall pregnancy rate of 2.1% (95% CI: 1.4–3.1%), which was statistically less than the clinical limit of interest set at 4% for an emergency contraceptive. This threshold corresponds to a reduction by half of the expected 8% pregnancy rate in the absence of contraception as observed in previous international studies [3,4]. In addition, efficacy did not decrease over time with pregnancy rates of 2.3% (95% CI: 1.4–3.8%) at 48–72 h after unprotected intercourse; 2.1% (95% CI: 1.0–4.1%) at 72–96 h; and 1.3% (95% CI: 0.1–4.8%) at 96–120 h. Adverse events were mainly mild or moderate with the most frequent being headache, nausea and abdominal pain. Menstrual cycle length increased a mean of 2.8 days, but the duration of menstrual bleeding did not change.
The study by Glasier in 2010 entitled ‘UPA versus LNG for EC: a randomized noninferiority trial and meta-analysis’ was an international, prospective, randomized clinical trial performed in the UK and the USA [11]. The efficacy and safety of UPA as a single 30-mg micronized dose was compared with LNG 1.5 mg taken within 5 days of unprotected intercourse.
Among the 2221 women enrolled, a total of 1696 women received EC within 72 h of intercourse were evaluated for efficacy; 844 receiving UPA and 852 LNG. There were 15 pregnancies in the UPA group (1.8%; 95% CI: 1.0–3.0) and 22 in the LNG group (2.6%; 95% CI: 1.7–3.9). Of the 203 women who received EC between 72 and 120 h, there were three pregnancies, which were all in the LNG group. Adverse events were similar between groups, with headache the most common in approximately 19% of women
Summary of Phase III studies of ulipristal acetate for emergency contraception.
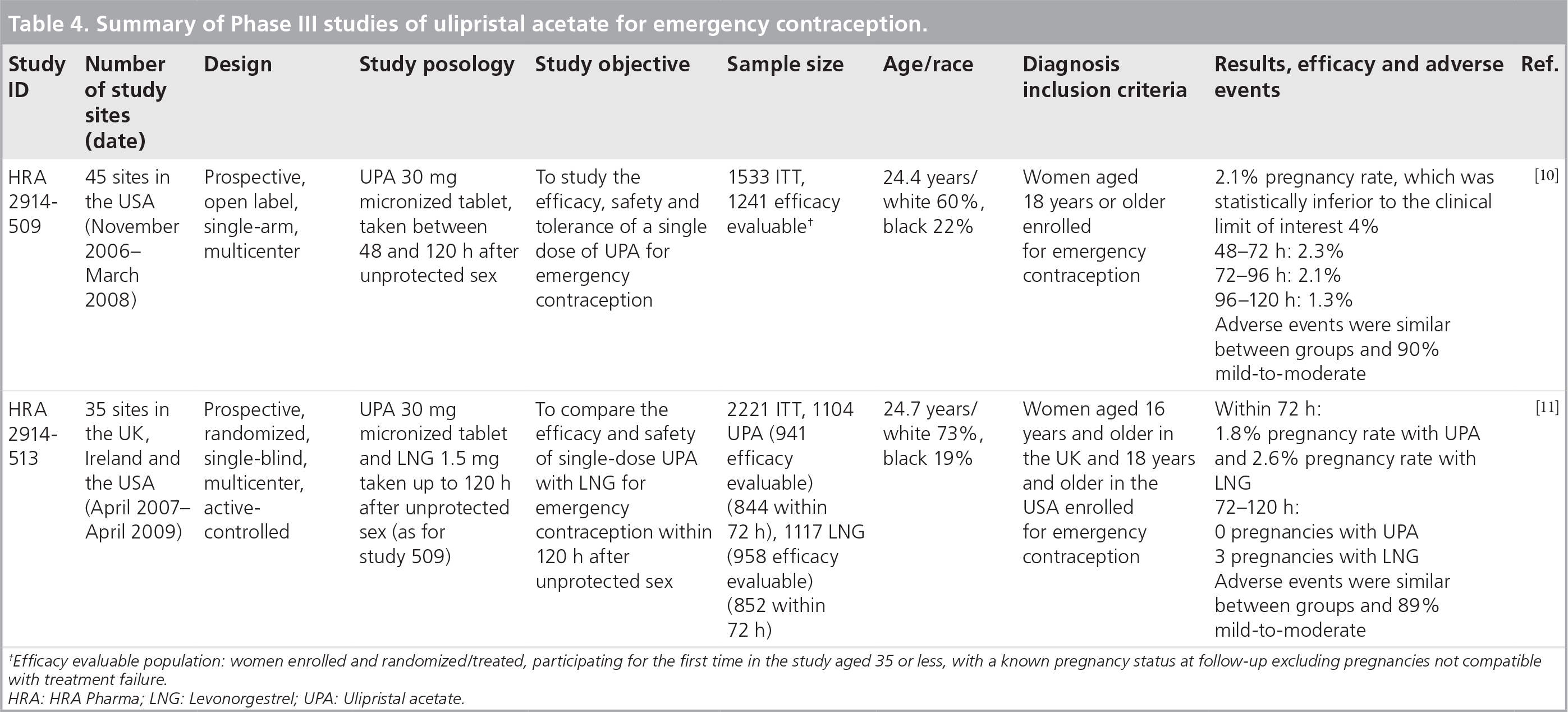
Efficacy evaluable population: women enrolled and randomized/treated, participating for the first time in the study aged 35 or less, with a known pregnancy status at follow-up excluding pregnancies not compatible with treatment failure.
HRA: HRA Pharma; LNG: Levonorgestrel; UPA: Ulipristal acetate.

Adverse events (>3%) compared with LNG.
Postmarketing surveillance
Since the launch of UPA in Europe and as of 18th November 2010, the Pharmacovigilance Department of HRA Pharma has received 50 spontaneous reports, of which 24 were pregnancies resulting from treatment failure.
Safety & tolerability
Ulipristal acetate was taken as a single dose for EC in a total of 4736 women. UPA was very well tolerated, as well as LNG, the currently US-approved ECP, and is associated with no greater risk of adverse events or menstrual disturbance.
Adverse events of ulipristal acetate verus levonorgestrel in the comparative Phase III trial.

Dizziness that resolved within 24 h.
Molar pregnancy.
AE: Adverse event; LNG: Levonorgestrel; SAE: Serious adverse event; UPA: Ulipristal acetate.
Data taken from [11].
Regulatory affairs
Ulipristal acetate was launched throughout Europe in September 2009 and was recently approved by the FDA in August 2010. It is marketed by Watson Pharmaceuticals in the USA and is available by prescription only to US women since December 1, 2010.
Conclusion
Emergency contraception is a woman's last chance to prevent unwanted pregnancy. Ulipristal acetate inhibits or delays ovulation during the preovulatory period when the follicle has reached 18-20mm size, while LNG is no more effective than placebo [12,13]. The efficacy of UPA even when it is taken just before ovulation is especially important because at this time of the cycle the probability of conception is at its peak and the frequency of sexual intercourse is also highest [13].
Unlike LNG, which is approved for use only up to 72 h (3 days) following unprotected sex, UPA maintains its high efficacy as an emergency contraceptive up to 120 h (5 days) after unprotected sex. UPA prevented twice as many unintended pregnancies compared with LNG in a meta-analysis of two large comparative clinical trials. Moreover, the risk of pregnancy was reduced by almost two-thirds in women taking UPA within 24 h of unprotected intercourse compared with LNG. The incidence of adverse events with UPA was similar to LNG with most being mild. A single dose of UPA used for EC does not have a significant effect on endometrial histology and histochemistry that could theoretically impair implantation of a fertilized oocyte [14].
Levonorgestrel is currently available in the USA for EC as a nonprescription medication for women 17 years of age and older and available by prescription for females 16 and younger, theoretically facilitating access and use. UPA is available by prescription only in Europe and was approved by the FDA on 13 August 2010. It was marketed in the USA by Watson Pharmaceuticals and is available by prescription only to US women since December 1, 2010. Unfortunately, over-the-counter status of LNG for EC has not reduced the unacceptable 50% rate of unintended pregnancy in the USA. A more effective emergency contraceptive drug combined with increased education of women concerning availability and use of emergency contraceptives is needed. UPA will provide women and healthcare providers with an alternative choice for EC that can be used up to 5 days after unprotected intercourse.
Pregnancy outcomes in emergency contraception trials
Overall pregnancies: 92
Outcome data available: 82
Spontaneous miscarriage: 15 (18.3)
Elective termination: 60 (73.1)
Live birth: 7 (8.5)
Ectopic pregnancy: 0
Future perspective
The development and marketing of more efficacious ECPs such as UPA has important medical and social benefits. The time window for use is extended from 3 to 5 days following unprotected intercourse. Moreover, UPA is more efficacious than LNG from the first day on following unprotected intercourse. Increased education and knowledge by both healthcare providers and women about EC is needed to reduce the currently unacceptably high 50% rate of unintended pregnancy. Commercial introduction of UPA is expected to be accompanied by direct-to-physician and direct-to-consumer advertising which should be helpful in this regard. It is certainly possible that following several years of prescription-only use, UPA will later be granted nonprescription status by regulatory authorities as occurred with LNG in the USA and Europe.
Executive summary

Ulipristal acetate for emergency contraception is a single-dose 30 mg oral tablet to be taken as soon as possible and within 5 days following unprotected sex.
It is a synthetic selective progesterone receptor modulator.
It can delay or inhibit ovulation, even within 24–48 h of expected ovulation after the luteinizing hormone surge has begun.
Its high efficacy as an emergency contraceptive is maintained up to 120 h (5 days) after unprotected sex.
Ulipristal acetate is a 30 mg micronized immediate-release tablet.
It is rapidly and well absorbed after oral administration.
Its bioavailability is 112% in monkeys. There are no human data.
It is highly protein-bound with a half-life of 32 h after oral administration.
It undergoes rapid and extensive hepatic metabolism by cytochrome P450 enzymes, especially CYP3A4.
Ulipristal acetate has consistent efficacy for the entire 5-day period in contrast to levonorgestrel (LNG).
Ulipristal acetate almost halves the risk of becoming pregnant compared to LNG when taken 120 h of unprotected intercourse.
Ulipristal acetate reduces pregnancy risk by almost two-thirds compared to LNG when taken within 24 h of intercourse.
Ulipristal acetate has a very good safety profile, as good as levonorgestrel.
The safety of ulipristal acetate has been evaluated in a large number of women (>4500 women exposed).
The most frequent adverse events are headache (18%), abdominal pain (12%), nausea (12%), dysmenorrhea (9%), fatigue (6%) and dizziness (5%) and were in great majority mild-to-moderate.
There is a potential reduction in efficacy if administered with other potent CYP3A4-inducing medications.
Use of barrier contraception is advised until the following onset of menses.
Single-dose 30-mg tablet taken up to 5 days following unprotected intercourse.
No food or drink restrictions.
Pregnancy should be excluded before prescribing ulipristal acetate. If pregnancy cannot be excluded on the basis of history and/or physical examination, pregnancy testing should be performed.
Repeated use of ulipristal acetate within the same menstrual cycle is not recommended.
Ulipristal acetate should not be used by breastfeeding women.
Acknowledgements
The author would like to thank Henri Mathe and Dr Delphine Levy for their editorial assistance.
Footnotes
The author is a consultant for HRA Pharma. The author has no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
