Abstract
The causes of chronic vulvar pain are many and when not accompanied by obvious physical signs they are difficult to unravel. Many common vulvar dermatoses can manifest without obvious erythema or mucocutaneous lesions, as can some less common disorders of both infectious and allergic origin. In addition localized and generalized vulvar dysesthesia, recently defined chronic pain syndromes of neurogenic origin, can also occur in the vulvar area.
Chronic vulvovaginal symptoms in early stages are often presumptuously labeled as vulvovaginal candidiasis (VVC) by patients and clinicians alike, which can delay accurate diagnosis. When presented with chronic vulvar pain unaccompanied by verifiable signs, the clinician must effect a sensitive integration of a detailed medical history, including temporal associations of all potential exposures, with more definitive diagnostic tools. Effective use of those available tools can improve initial diagnosis, thus allowing prompt initiation of effective therapy. Optimal therapy will recognize the significant psychological distress that accompanies any chronic pain syndrome.
Introduction
Although described in the literature as early as 1891, 1 syndromes of chronic vulvar pain did not begin to appear in the medical literature as a topic of research interest until the 1980s. 2 Still inadequately described, these syndromes can be challenging for both physician and patient alike.
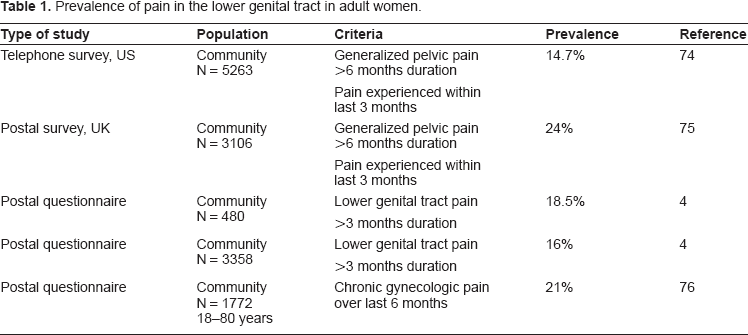
Recent investigations have proven that chronic pain of the vulva and vagina is not uncommon in women of reproductive age. Initial studies, specific to patients of gynecological clinics, found prevalence rates of up to 15%. 3 More recent research into community prevalence has found similar rates, ranging between 14.7% and 24%. (Table 1) Prevalence rates vary by the criteria by which chronic pelvic pain was defined, including type of pain, duration of consistent pain, and window in which symptoms were experienced.
Prevalence of pain in the lower genital tract in adult women.
Pain at presentation is variously described as burning or stinging; sharp, stabbing, or knife-like; or raw. Pain can also be reported as prickly, pruritic, or even as a sense of something crawling under the skin (formication). The pain may be continual, intermittent, or specific to certain behaviors or it may be intensely localized, diffuse, or referred. Sufferers sometimes describe irritation or exaggerated sensitivity.
Many women find this condition difficult to discuss with their clinicians. A community-based survey in Boston in 2003 found that nearly 40% of women suffering chronic vulvar pain did not seek treatment. 4 Sixty percent of women who seek treatment eventually see at least three different clinicians. 4 Of 173 women who sought treatment and reported a diagnosis, 34.7% were diagnosed with an infection, 30% with pelvic disorders, 9.8% with skin disorders, 9.2% with a chronic vulvar pain syndrome, 4.6% with hormonal disturbances, 1.2% with a mental disorder, 1.2% with dietary complaints, and 9.8% with various other disorders. 4 Most sufferers go undiagnosed.
Diagnostic difficulties arise from the fact that the debilitating pain, though persistent, is often unaccompanied by obvious physical signs or laboratory findings. Many patients are told that their problems are psychological, thereby simultaneously invalidating and increasing their suffering. 5 In addition, as symptoms prove recalcitrant to successive therapies, patients are exposed to multiple topical or systemic medications, which can further cloud diagnosis. 6
Correct diagnosis of chronic vulvar pain requires careful evaluation of the clinical presentation, particularly the presence or absence of mucocutaneous changes, the types of lesions observed, and tissue affected, as well as the description of sensory symptoms involved. The disparate embryologic origin of the vulva, combined with its occlusive nature and immunological makeup form a unique anatomical environment which can present the clinician with challenges in diagnosing and treating chronic vulvar pain. The contribution of diverse individual intimate behaviors and hygiene practices, sometimes dictated by unusual cultural norms, must also be taken into consideration. 7
Vulvar Anatomy
The vulva consists of a triangular mound of tissue bordered by the mons pubis, the perineum, and, at the thighs, the labiocrural folds. (Fig. 1) The vulva differs substantively from skin at other sites in numerous aspects with potential to influence pain induction and perception.
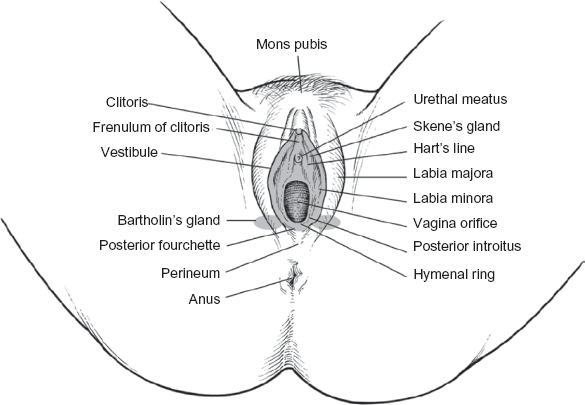
Anatomy of the vulva.
Vulvar epithelium is unique in that it is derived from two embryonic layers, the ectoderm and the endoderm, and thus displays dramatic regional differences in physiology. Thickness and degree of keratinization decrease steadily from the labia majora moving inward, becoming nonkeratinized from approximately the inner third of the labia minora. 6
Inner mucosal layers are indistinctly differentiated and loosely packed; polyhedral cells alter in size and organelle density as they migrate upward from the generative basal layer, but do not form clearly demarcated strata as observed in the skin. Cervicovaginal secretions moisten the vulvar vestibule. 6 Innervation of the vulvar area does not strictly correlate with embryonic development. 8
Vulvar epithelium is immunocompetent, with Langerhans cells being the most common immune cell. Allergic responses in vulvar skin are a prime contributor to chronic vulvar discomfort 9 ; however, antigen application to non-keratinized skin may induce tolerance. While immune responsiveness of the vulvar vestibule has not been studied, the possibility exists that response to contact sensitizers may differ between keratinized and nonkeratinized regions of the vulvar vestibule. 6
The vulva differs from skin at other sites also in the degree of blood flow, skin hydration, and occlusion. Blood flow in the epithelium of the labia majora is over twice that of the forearm 10 ; blood flow in vulvar skin is responsive to histamine treatment at doses at which forearm skin is not. 11 Vulvar skin is more hydrated, but water diffuses across the stratum corneum of the labia majora faster than across the stratum corneum of the forearm, an observation only partially the result of occlusion. 12 The increased hydration of occluded vulvar skin raises its friction coefficient, which may make vulvar skin more susceptible to mechanical damage.10,13,14 In contrast to the vast majority of the epidermis, the vulva is also characterized by chronic occlusion. 15
Sensitivity of the Vulva
The epithelium is a sensory organ; that of the vulvovaginal area, in fact, has increased nerve density as compared to epithelium at other anatomical sites. Cutaneous irritation is a syndrome with multiple potential etiological factors such as age, genetics, hormonal factors, skin dryness, race, skin pigmentation, pre-existing or underlying diseases, and cultural and environmental factors. 15 Certain individuals report more intense and frequently adverse dermatologic sensations (a phenomenon known in popular usage as sensitive skin), particularly upon exposure to cosmetics, toiletries, and topical medicaments.16,17 These “sensitive skin” patients report stinging, itching, burning, and dryness 18 over a wide spectrum of intensities 19 that is sometimes accompanied by erythema, desquamation, papules, wheals, and scaling. 18 Self-reports of skin sensitivity, however, are often not verifiable by clinical signs of irritation, 20 and patients who deny subjective discomfort can sometimes demonstrate strong objective response to a given irritant. 21 No correlation has been found between sensitive skin and alterations in skin hydration, blood flow, or transepidermal water loss (TEWL). 22
A general lack of correlation between subjective discomfort and objective skin findings has complicated the available body of research; however, a tentative connection has been observed between sensitive skin and atopic tendencies. Loffler et al found a significant association between the perception of sensitive skin and verifiable nickel allergy. 22 In addition, another study which surveyed 1039 individuals with regard to self-perception of sensitive skin further correlated self-perceptions and found significant association between self-perceptions of sensitive skin and a (self-reported) personal and family history of skin allergy. 16
Genital skin sensitivity, due to the difficulty of extensive testing in this sensitive area, has not been significantly explored. Preliminary studies have observed no correlation between self-reports of skin sensitivity and visible erythema. 23
Skin sensitivity has also been shown to be significantly variable within the same individual at different anatomic sites. 24 In a survey of 1039 consumers participating in separate focus groups, 68.4% claimed sensitive skin. Of those, 77.3% reported sensitive facial skin, 60.7% reported sensitive body skin, and 56.3% reported sensitivity in the genital area. 25 Testing of sensitivity specific to the vulvar area has been very limited, as elevated hydration of the vulvar area makes measurements difficult, 26 and the nature of the testing makes routine testing logistically unfeasible. 21 Developed methods, in addition, are in general less suitable to the vulvar area, making data less meaningful than in exposed, fully keratinized skin. 27
The relative permeability of vulvar skin is primarily the result of elevated hydration and reduced water barrier function but is also influenced by other variables.28–30 Ultimately, permeability is consistent with postulated mechanisms of tissue penetration. 21 Nonkeratinized vulvar skin exhibits clearly increased permeability related to the absence of keratin and loosely packed, less structured lipid barrier. 21 In addition, the inner epithelia are thinner, representing a shorter distance for substances to penetrate. 21 Buccal tissue is often employed in a surrogate model for vulvar testing, as it has very similar structure and biochemistry. Buccal skin has been demonstrated to be 10 times more permeable than keratinized skin. 15 Mucosal tissue is also more susceptible to a breach of integrity; buccal permeability, for example, is 40 times more permeable to nicotine, an irritant organic base. 31 Ultimately, permeability depends on regional differences in vulvar epithelial structure as well as lipid composition, tissue hydration, molecular structure of penetrants, and hydration of the vulvar tissue. Based on these observations and known structural differences, the vulvar mucosa is predicted to be significantly more vulnerable than exposed skin to topical penetrants. 6
At all stages of life, the vulva is more sensitive to a wider range of irritants and allergens than other skin sites. 28 Persistent hydration of the vulvar area may heighten susceptibility to hydrophilic irritants such as propylene glycol, a common constituent of topical steroid creams and anti-yeast preparations, which can produce a burning irritation reaction upon use. 21
Diagnosis of Chronic Pain Syndromes
Chronic genital pain in the absence of physical signs is frequently misdiagnosed by both patient and physician. In the absence of discharge or pain, diagnosis becomes significantly more challenging. Vulvovaginal conditions which can produce chronic pain without other obvious signs are discussed below.
Traditionally, diagnosis of dermatologic diagnosis is based largely upon visual recognition of cutaneous changes, but in the vulva exaggerated symptoms may be unaccompanied by the characteristic mucocutaneous lesions. 32 A vulvar biopsy submitted to a dermatopathologist may help to identify the dermatologic disorder. A dermatopathologist may be able to diagnose a significant skin disorder in the majority of otherwise clinically obscure vulvar pain cases. 33
Non-Neurogenic Sources of Chronic Vulvar Pain
Numerous common vulvar dermatoses can present as chronic vulvar pain without objective signs. 34 Contact dermatitis, however, causing significant itching and burning pain, was the principal diagnosis in 54% of 141 patients referred to a dermatologist for chronic vulvovaginitis. 9 Clinicians who provide care for women may be generally unaware of the high rate of vulvar contact dermatitis, and may underestimate the role of exogenous factors in vulvar inflammation. 6
Symptoms and signs of contact dermatitis are nonspecific. Patients complain of itching, burning, stinging, and irritation with either a sudden or gradual onset. A careful medical history may provide clues about exogenous factors that contribute to signs and symptoms. Clinicians should probe the use of new personal products, menstrual products, prescription or over-the-counter medications (particularly antifungals), and hygiene routines. 35
Contact dermatitis can be either irritant or allergen driven with numerous potential exposures to both types in the vulvar area (Table 2).
Differentiation of allergic and irritant contact dermatitis.

Hygiene practices are the most common cause of irritant vulvar dermatitis. Fastidious washing with harsh soaps or antiseptics or the use of rough washcloths can produce erythema and edema, which may be localized or diffuse. A detailed history which explores cleansing routines, use of any new personal care products, and past vulvar dermatoses should be obtained. Manifestations are usually localized to the keratinized epithelia. Successful treatment depends on identification of irritant and termination of exposure. 6
Allergic reactions should be considered if contact dermatitis does not respond to standard treatment (Table 2). Many components of cosmetics and medicaments as well as some type of preservatives and dyes, and contraceptives may cause allergic reactions (Table 2). Other possible culprits include benzocaine, lanolin, perfumes, and cosmetic ingredients. Anecdotal reports have implicated thiuram in rubber condoms; 4-phenylene diamine in black underwear, sanitary pads, and feminine hygiene sprays; however these reports are rare and not generally reproducible and may be contact irritation. 6
Allergic reactions differ from irritant reactions by the fact that allergic reactions typically occur at least 48 hours after exposure, and are characterized more by episodic itching than by pain (Table 2). Although persistent itching suggests other dermatoses such as lichen simplex chronicus or chronic candidiasis, ongoing use of topical medications may produce chronic itching due to the development of secondary allergic contact dermatitis. One study found that 38% of patients with chronic vulvar complaints evidenced allergic response to common vaginal medicaments. 36
Allergic contact dermatitis of the vulva can be difficult to distinguish from the irritant form. Vesiculation, a hallmark sign of delayed contact hypersensitivity, is sometimes masked on vulvar skin. 35 Patch testing is not routinely included due to the limitless nature of potential allergens; it can be useful, however, in diagnosis if a particular allergen is already suspected. 35 Medications should be discontinued to determine whether symptoms resolve and, in the case of topical antifungal use, whether vulvovaginal candidiasis (VVC) asserts itself. 35
Human seminal plasma allergy, a syndrome recognized fairly recently, causes chronic burning and swelling of the vulvar area as the result of coital exposure to semen. This condition is often misdiagnosed as VVC but proves resistant to treatment. It is believed to be an immunoglobin E (IgE)-mediated allergic response of the mast cells. Barring concomitant allergy to latex, resolution of symptoms with condom use is considered definitive diagnosis of seminal allergy.38,39
Menstrual exacerbation of vulvovaginal pain and pruritus may have several etiologies. One form of chronic premenstrual vulvovaginal pruritus is thought to be attributable to autoimmune progesterone dermatitis, an allergic reaction to endogenous progesterone. 40 Menstrual exacerbation of similar symptoms has also been credited to hypersensitivity to Candida 41 Allergies to menstrual pad components have been demonstrated in some individuals, particularly to colophony, and may cause pain and burning. 42
One rare allergic reaction which may occur in the vulvar area is aquagenic pruritus, characterized by severe prickly skin discomfort (immediate or delayed and without observable lesions) evoked by contact with water. 43 Although sometimes drug induced or secondary to polycythemia rubra vera, some idiopathic cases have been described. Recent research implicates the production of vasoactive intestinal peptide, a neurotransmitter, by vasoactive intestinal polypeptide (VIP)-immunoreactive epidermal cells upon exposure to water. 44
Treatment for both types of contact dermatitis includes discontinuance of potentially culpable substances and adoption of gentle cleansing with water alone until symptoms subside. Gentle rinsing with a squirt bottle is the mostly recommended method to clean after urination. Topical steroids may reduce inflammation in moderate cases. When contact dermatitis is secondary to treatment for another condition, the dermatitis must be allowed to resolve before an alternative treatment for the primary condition can be identified. Many patients are kept constantly irritated by overzealous application of sensitizing substances. 35 Superimposition of contact hypersensitivity on an underlying vulvar dermatosis should be suspected when a patient does not respond to prolonged therapy or symptoms recur after initial remission. Secondary allergic reactions must be resolved before the primary clinical disorder may be effectively managed. 6
Vulvar Pain of Neurogenic Origin
In chronic vulvar pain in an otherwise normal vulva, particularly after treatment for disorders of infectious or exposure-oriented dermatoses has proven ineffective, neuropathic pain syndromes should be considered. A collection of syndromes variously described under the heading “vulvodynia”, meaning vulvar pain, have recently been officially categorized by the International Society for the Study of Vulvovaginal Disease (ISSVD), classified by whether pain is primarily provoked or spontaneous and primarily localized or diffuse, terming the two groups localized vulvar dysesthesia and generalized vulvar dysesthesia. At this point, it is not completely clear whether these two groups represent independent diagnoses. It may be that initial pain may be provoked and localized but then progress to pain which is constant and diffuse. 45
Before contemplating a diagnosis of vulvar vestibulitis or vulvodynia, it is important to exclude any diagnosable skin disorder. Vulvar dermatologic disorders may result in discomfort that is out of proportion relative to the visible manifestations. A skilled dermatopathologist may be able to diagnose specific dermatologic disorders in over 60% of cases of otherwise obscure vulvar pain. 33 Such skin diagnosis is challenging and requires experience that may be beyond the training of the general surgical pathologist in the typical clinical laboratory.
Localized Vulvar Dysesthesia (Vestibulodynia)
Localized vestibulodynia is characterized by pain confined to the vulvar vestibule that occurs upon vestibular touch or attempted introital entry (e.g. intercourse, tampon insertion) with a lack of physical findings beyond varying degrees of vestibular erythema. Introital dyspareunia, the intensity of which may inhibit or prevent intercourse, is often the presenting symptom. Pain may occur in other situations that exert pressure upon the vestibule such as tampon insertion or withdrawal, bicycle or horseback riding, the wearing of tight clothing, or prolonged periods of sitting. Pain typically becomes chronic, although spontaneous remissions have been reported. 46
Vestibular tenderness is assessed by applying a cotton-tipped swab to the vulvar vestibule in a clock-like pattern. 46 Thresholds to pain provoked by pressure are markedly lower in vestibulodynia patients 47 and reliably distinguished patients with and without vestibulodynia who had a history of dyspareunia. 48 In some women, pain manifests at the first experience of intercourse; others experience onset after a history of pain-free sexual activity. In a study of 162 patients with strictly defined vestibulodynia, 20.4% experienced symptoms at the first act of coitus. 49
It is unclear whether these two groups arise from different etiologies or represent different presentations of the same pathogenic process. 49 A study of 111 patients found no significant differences between the two groups other than time of onset, with no differences observed between early- and late-onset patients with regard to recurrent Candida infection, human papillomavirus infection, or interleukin-1 receptor antagonist (IL-1RNA) genotype profile. 50 However, late-onset vestibulodynia was characterized more often by pain specific to the anterior region of the vestibule, implying a distinct etiology, 50 and a group of patients diagnosed with vulvar vestibulitis syndrome were successfully differentiated by carriage of different variants of the interleukin-1β gene. 51 In addition, some evidence exists that vulvodynia may be the result of a congenital neuronal hyperplasia. 52
It also may be that environmental or lifestyle factors can contribute to disease onset. A postulated association between oral contraceptive use and the development of vulvodynia is still unproven, with some studies supporting the hypothesis, and others failing to offer support.
The prevalence of vestibulodynia in the general population is unknown. Among a cohort of women presenting to a referral center for vulvovaginal disorders, prevalence was 20%, measured over a 4-year period. 53 It was observed to be 15% among patients seen over a 6-month period in a gynecology clinic, but 1.3% among 24 walk-in patients in a genitourinary clinic. 54 Patients ranged in age from 20 to 40 and were predominantly Caucasian; interestingly, a genetic profile recently associated with a higher risk of vestibulodynia (homozygosity at allele 2 of the IL-1RNA gene) 55 is very rare in the African-American population. 56 Available demographic statistics may be skewed by cultural differences in the likelihood of seeking intervention as well as by diagnostic delays.
The only available population-based survey, which was comprised of 4915 women aged 18 to 64 from ethnically diverse Boston communities, found that 40% of women with vulvar pain sought no treatment, while 60% of those who did so consulted three or more health care providers over several years before obtaining a diagnosis. 4 About 16% reported histories of chronic burning or knife-like vulvar pain or pain on vulvar contact experienced over a period of at least 3 months; 12.4% complained specifically of pain on vulvar contact. The survey did not fully distinguish between those whose pain is elicited by vestibular contact and those who have generalized vulvar dysesthesia.
Although etiology of chronic vulvovaginal pain is still controversial, localized vulvar dysesthesia is currently believed to be a chronic pain syndrome involving abnormal pain perception, postulated to result from sensitization of vestibular nerve fibers and establishment of a sympathetically maintained pain loop. 57 In theory, some triggering event (i.e. Candida infection, human papilloma virus (HPV) infection, toxins, semen, or laser surgery 51 precipitates an inflammatory reaction which is not adequately controlled. The prolonged inflammatory reaction induces prolonged firing of the sympathetic, Type C nerve fibers 57 ; this in turn causes the wide dynamic-range neurons in the brain to respond abnormally, such that mild stimuli are perceived as pain. 57
Doppler perfusion imaging has also revealed heightened erythema and increased superficial blood flow in the posterior vestibule of vestibulodynia patients, suggesting either the presence of classic inflammation or neurogenically induced vasodilation. 58
A possible genetic contribution to this inflammation has been recently identified. Carriage of homozygosity at the 2 allele of the IL-1 receptor agonist gene, a down regulator of inflammatory response, was found to be more frequent in vestibulodynia patients. 55 In addition, patient with the homozygous 2 allele at the Il-1 receptor agonist loci were shown to have increased intensity and duration of inflammatory reactions. 55 A pro-inflammatory variant of the melanocortin-1 receptor has also been found to be substantially more prevalent in vestibulodynia patients, 55 and the risk of vestibulodynia rises additively in women who carry both genes. 59 Impairment of interferon-alpha production 60 and natural killer cell function, 61 as well as reduced estrogen receptor expression in localized regions of the vestibular mucosa 62 have also been reported in vestibulodynia patients. Because estrogen both stimulates the antibody response and inhibits T-cell mediated inflammation, localized insensitivity to circulating estrogen may increase vulvar susceptibility to inflammation caused by infectious agents.
Although these lines of evidence support a pathogenic role for inflammation, they do not establish a causative relationship to nocireceptor sensitization and hyperproliferation. A complicating factor in identifying possible inflammatory triggers is the delay between first onset of symptoms and first diagnosis; inflammation associated with an initiating event either may subside by the time patients are evaluated or may persist only in the most severe cases. 57
Chronic pain can be a disease process itself, irrelevant to verification by physical signs. 63 Although triggering pathology like trauma or infection may be present, the pathological process results from disturbances in the central nervous system (CNS). 63 Support for this theory is found in the fact that local vulvar dysesthesia often occurs in association with other pain syndromes, including pelvic floor dysfunction, 64 irritable bowel syndrome, 63 interstitial cystitis, 65 fibromyalgia, 66 temporomandibular disorder, 63 migraines, 63 burning mouth syndrome, 67 and orofacial pain. 68 In addition, women with localized vulvar pain have heightened responses to noxious stimuli in the vulvar area as well as sites distant to the genitalia, including mechanical thermal and chemical stimuli. 63 The affected tissue is hyperalgesic to thermal tactile and pressure stimuli, sometimes involving a hyperpathic “after pain” that lasts for minutes after stimulus removal. Derangement of normal pain processing pathways as a main mechanism for these syndromes is supported by the fact that the genetic profile associated with vestibulodynia, homozygosity at the 2 allele of the receptor agonist gene, is also associated with syndromes of chronic inflammation at numerous other body sites. 49
Still debated is the contribution of psychosomatic disturbance to chronic vulvar pain. Women with vestibulodynia experience greater psychological distress and sexual dissatisfaction than healthy controls. Pain relief has been achieved by surgical excision of affected portions of the vestibule. Most studies fail to distinguish between predisposing impaired, precipitating, perpetuating, or simply the result of having an unmitigated pain syndrome. Nevertheless, some authors still espouse the historical view that vulvar pain is largely of psychogenic origin; mounting evidence supports physical dysregulation of the CNS. Optimal treatment, however, will necessitate recognition of the psychological sequelae of chronic vulvar pain and its effect on sexual health as well as provide appropriate therapy for all relevant issues.
No accepted curative therapy exists, and current approaches to management lack a clear etiologic basis. A dearth of rigorous randomized prospective trials exists for most therapies; evidence for efficacy derives largely from single-case studies or case series where each patient was her own control. Studies also differ in endpoints assessed, extent of recovery defined as success, and duration of follow-up. Preliminary interventions include symptom relief through topical medicaments like lidocaine 69 and cromolyn cream 70 and oral medications that decrease neuronal hypersensitivity (amitriptyline, desipramine, gabapentin, paroxetine, venlafaxine), 2 indicated for pain control rather than mood adjustment. Biofeedback has been employed with some success. 71 Surgical intervention is employed only for the most refractive cases, but has been shown to produce symptom relief in 60% to 90% of cases. 71 With treatment, about 50% will experience sustained improvement 63 Efficacy of treatment suffers due to a lack of appropriate differential diagnosis. 2 Optimal therapy is still unclear, as no particular therapy could be given more than a B evidence rating, meaning that evidence available is inconsistent and of limited quality. 2 Substantial research is in progress with regards to effective therapies for chronic vulvar pain, however, with frequent additions to the literature, but a thorough analysis of treatment options are outside the scope of this review.
Generalized Vulvar Dysesthesia (Dysesthetic Vulvodynia)
Dysesthetic vulvodynia is unprovoked vulvar pain with no apparent cause in an otherwise normal vulva. Patients are usually peri- or postmenopausal women who complain of unrelenting, poorly localized vulvar pain with a burning, sore character, or formication that is not linked to any particular activity or stimulus. Allodynia and hyperalgesia may elevate discomfort when wearing tight clothes or applying topical products. The pain is usually bilateral around the introitus and on the labia and may radiate to the perineum and mons pubis. Painful urination and frequent micturition may accompany the syndrome. Younger patients are more likely to report localized pain with a shooting or stabbing character; this is often secondary to neuropathy caused by nerve compression or trauma. Clitorodynia maybe a localized form of the syndrome. 35
The term vulvodynia, which means vulvar pain, is a symptom and not a disease entity. As stated, many conditions that cause vulvar pain are diagnosable and treatable. True dysesthetic vulvodynia is idiopathic; there is no diagnostic test. The vulva is normal except for some degree of atrophy consistent with the patient's age or a diffuse erythema caused by rubbing or other forms of self-treatment. In rare cases, the condition may be superimposed on other dermatological conditions. In such instances, resolution of mucocutaneous lesions will not entirely eliminate vulvar pain. 35
The etiology of dysesthetic vulvodynia is unknown but is likely to be a neurological disorder. Cutaneous hypersensitivity to endogenous vulvovaginal C. albicans may contribute to symptoms in some patients. 72 Diffuse vulvar pain is treated with low-dose tricyclic anti-depressants administered for their effects on pain rather than mood. The patients should be counseled that the medication is being prescribed as a pain treatment. Low doses are used to minimize side effects such as sedation.
A comparison of critical diagnostic criteria for unexplained vulvar pain is displayed in Table 3.
Comparative diagnosis of chronic vulvar pain syndromes.
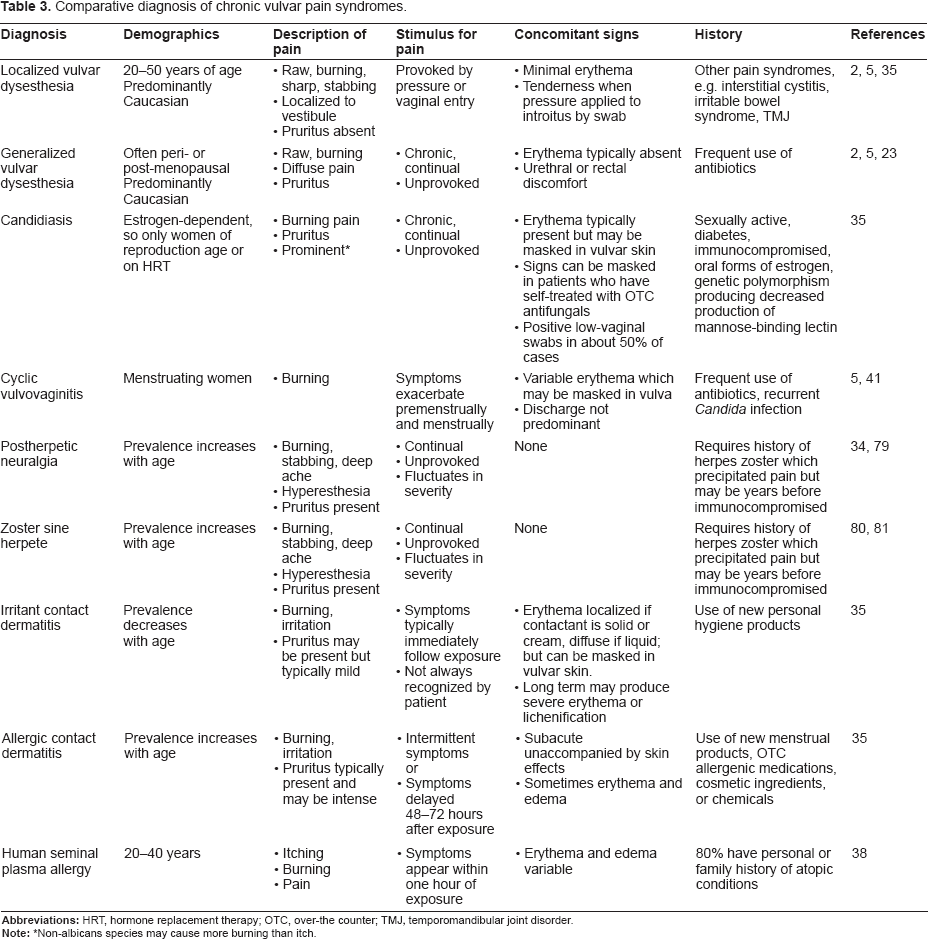
Non-albicans species may cause more burning than itch.
Conclusion
Vulvar discomfort without obvious signs (e.g. definitive discharge, mucocutaneous lesions, erythema) can be challenging. Many dermatologic disorders can present with similarly intense pain, and the clinician must effect a sensitive integration of a detailed medical history, including temporal associations of all potential exposures, with more definitive diagnostic tools.
Treatment of chronic vulvovaginal pain without verifiable signs has had a poor track record, partially due to the frequent assumption by both patients and their physicians that vaginal burning and/or itching is usually related to Candida infection. Symptoms are often treated on the basis of this presumption without any clinical evaluation. Accurate diagnosis of vestibulodynia is therefore a primary goal, particularly because Candida-related nosocomial blood stream infections are increasingly dramatically (207% from 1979 to 2000). 73 Effective use of the tools available for diagnosis will improve efficacy rates for current therapy options.
Numerous conditions can present as unexplained vulvovaginal pain with minimal objective clinical findings. Most cases of unexplained vulvar pain have a dermatologic origin (contact dermatitis, bacterial or yeast infection, lichen sclerosis, and mild lichen planus) and diligent efforts to exclude diagnosable vulvar pathology should be pursued before determining the pain to be of neurogenic origin only.
The hallmark of localized vulvar dysesthesia is the description of pain as raw, burning, sharp, knife-like pain as well as pain confined to the vulvar vestibule and provoked by pressure. This is distinguished from generalized dysesthesia, which involves chronic, diffuse vulvar pain that occurs without stimulation. Neurogenic origin is currently a diagnosis of exclusion, and other organic sources of the pain must be ruled out (e.g. candidiasis, contact dermatitis, shingles, and other vulvar dermatoses) by a careful history of temporal exposures and available clinical findings, paying particular attention to the presence or absence of itch and other signs of irritation.
Patch testing is not recommended unless allergic contact dermatitis is suspected since no relevant reactions either to standard allergens or to a series pertinent to vulvar disorders were found in vestibulodynia patients. However, a subset of women with vestibulodynia exhibited immediate-type hypersensitivity to seminal fluid (as assessed by plasma antibody titers to pooled semen samples) does exist. The majority of these patients reported that symptoms began with an episode of sexual intercourse, with symptoms temporally associated to sexual activity.
Vulvar pain of neurogenic origin is a debilitating syndrome, whose etiology is still unknown and possibly multifactorial. The prevailing theory postulates that the syndrome is a neuropathic disorder of abnormal pain perception triggered by some form of chronic inflammation. Possible triggers include infectious agents, excessive use of irritating topical products or medications, prior laser or cryogenic treatments for HPV infections, and Type I hypersensitivity to seminal fluid. Mounting evidence suggests that vestibulodynia-afflicted women are genetically predisposed to chronic inflammatory responses or may have impaired immune defenses against infectious agents. Evidence also exists for physical and psychological contributing factors. Rigorous randomized prospective trials on alternative therapeutic approaches are lacking.
Conservative interventions for which some evidence for efficacy exists are anesthetic symptom relief, pain modulation with low-dose tricyclic anti-depressants, and electromyographic biofeedback. Anti-fungal or interferon therapy may be of benefit in selected subsets of patients. Surgical excision of afflicted portions of the vestibule produces relief but is reserved for chronic recalcitrant cases after other treatments have failed. Patients with vestibulodynia benefit from supportive therapy and a multi-modal treatment approach may be optimum to address both physical symptoms and psychological sequelae. Appropriate therapy will recognize the complex psychological aspects of chronic pain. 63
Failure to consider vulvar dysesthesia as a possible source for chronic vulvar pain is the most common reason for misdiagnosis. 5 Research demonstrates that both forms of neurogenic vulvar pain are being increasingly diagnosed, as family physicians become aware of this condition and are able to recognize and diagnose it at the initial visit. Currently, however, vulvar pain syndromes apparently of neurogenic origin are diverse and ill-defined, and may represent a diversity of disease pathways that present similar clinical pictures. Effective treatments, however, will require understanding of distinct pathways and specifically targeted therapies. More research is required to elucidate etiologic mechanisms and devise evidence-based efficacious treatments for this complex disease.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgment
The authors are grateful to Ms. Z. Schwen, Strategic Regulatory Consulting (Cincinnati, OH) for assistance in this manuscript.
