Abstract
Ureteral stricture, regardless of etiology, remains difficult to treat. Mainstays of therapy include polymeric double J stents and percutaneous nephrostomy tubes, each with respective complications. Multiple retrospective studies have now been published using the Resonance metallic double J stent, which is the focus of this review. A literature search was completed utilizing Pub Med. Key words included metallic stent, Resonance stents, and ureteral stricture. All identified papers were included. The stent is generally well tolerated, with infections, hematuria, and voiding symptoms requiring removal in 0% –14% of patients. Stents remained in place for mean of 4 to 9.4 months with the exception of a single study evaluating ureteroenteric strictures, where average duration was 21 days. In most studies a subset of patients kept the stent in situ for > 12 months, indicating that for some, the Resonance stent is a viable option, though predicting which patients will do well remains difficult.
Introduction
Ureteral obstruction, whether of malignant or chronic benign etiology, has historically been a difficult problem to treat. Definitive management generally involves an open surgery, which is not appropriate for many patients. Conservative management has centered on double-J polymeric stents and percutaneous nephrostomy tubes. Long-term indwelling ureteral stents (IUS) were initially developed by Zimskind in the 1960s and quickly became a commonly used, if not fundamental, endourologic tool for management of either external or internal ureteral obstruction. Many materials have been used including polyethylene, silicone, and polyurethane. By far, polyurethane is the most widely used in current practice, owing to a tendency of polyethylene to fracture and for silicone to be too pliable. 1
After placement, stents begin to become encased in a biofilm, often within hours.2,3 Biofilm forms in three stages: endogenous protein deposition, bacterial attachment, and development of the “biofilm structure.” Endogenous urinary proteins (specifically Tamm-Horsfall protein and glycoproteins) deposit on the foreign body prior to any bacterial attachment. Bacteria first attach reversibly utilizing hydrophobic and electrostatic forces. Once held in proximity, bacterial polysaccharides create a more durable bridge. A biofilm suprastructure is erected, consisting of a bacterial polysaccharide matrix permeated with channels transporting oxygen and nutrients to the underlying bacteria. 4 Biofilm formation on foreign bodies within the urinary tract is often ubiquitous, ranging from 68% to 90%, though rates of bacteriuria are near 30%. 4 Bacteria immersed in biofilm become difficult to treat due to a combination of effects. First, the layers of the film are difficult to penetrate with antibiotics. Secondly, gene expression is altered in bacteria within a biofilm, decreasing antibacterial targets or bacterial metabolism, subsequently impairing antibacterial efficacy. Interestingly, a study by Paick et al found that while no plastic stents were colonized with bacteria prior to 2 weeks, 44% had been colonized after 2 weeks, with E.coli and Enterococcus being the two most commonly isolated organisms. 5
The same biofilm that facilitates attachment of bacteria increases risk of encrustation. Particularly with urease producing bacteria, an alkaline environment facilitates formation of struvite and hydroxyapatite crystals which become incorporated into the biofilm itself. Progressive encrustation can make stent exchange more difficult and potentially occlude the lumen. 4 Moreover, stents are susceptible to tissue ingrowth or stent migration, and cause a variety of patient complaints including hematuria, bladder spasms, dysuria, and flank pain. To avoid obstruction secondary to encrustation and to decrease bacterial load, stents typically are changed under general anesthesia or heavy sedation every 3–6 months. Contrasting with stent obstruction due to encrustation in benign disease, polymeric stent failure in malignant obstruction is often due to increased external compression, with cited rates of failure ranging from 35.7% to 58%.6–8
Percutaneous nephrostomy (PCN) tubes generally provide reliable drainage and are frequently used after IUS failure or as an initial alternative to ureteral stents. PCN tubes must also be changed periodically, usually by interventional radiology, and require external drainage appliances. Comparing PCN tubes to IUS in one study, major complication rates did not differ significantly, though stents were more prone to obstruction while PCN tubes are more prone to dislodgement. 9 Quality of life surveys comparing stent to PCN tubes have shown no statistically significant difference in most patients; 10 however, many patients still prefer internal drainage despite stent changes and risk of stent-specific complications.
Metallic Stents
Metallic stents were developed to alleviate the need for frequent stents changes and in theory would be less susceptible to external malignant compression. In the 1990s the Wallstent (Schneider, Zürich, Switzerland) was introduced as a self-expanding segmental cobalt alloy mesh stent. 11 The Wallstent is typically placed over a wire after balloon dilation of the stricture and is permanent, fundamentally contrasting with the more temporary plastic stents. Placement of the stent causes ureteral edema and hyperplasia for 4–6 weeks, after which the stent is actually incorporated into the ureteral wall. Hyperplastic tissue can temporarily obstruct the lumen, often prompting prophylactic double-J stent placement through the mesh stent for the first few weeks. An early study by Lugmayr and colleagues reviewed 23 patients with malignant ureteral obstruction. 12 Ninety-seven percent had successful placement, with decrease in patency to 83% at 30 weeks and 5 patients requiring double-J stent placement due to obstruction. In six subsequent studies, primary patency rates ranged from 14% to 58% at follow-up from 3 months to 2 years.13–17 In general, Wallstents did not produce durable stricture patency in patients with malignant obstruction, due to susceptibility to encrustation and tissue ingrowth, and they were often difficult to remove, sometimes requiring open ureteral reimplantation.
The Memokath 051 is a nickel-titanium alloy thermo-expandable segmental stent with metal composition and spiral configuration designed to decrease encrustation and susceptibility to tissue ingrowth. The thermal shape-memory design provides a malleable proximal end at temperatures <10 °C that resumes initial conformation when warmed to 50–55 °C. During placement, a sheath is advanced past the proximal end of the stricture over a 0.9 mm guidewire after dilation to 12F. The dilator and wire are removed. The sheath is pulled back to allow fluoroscopic visualization. Once the stent is in appropriate position, 50 ml preheated water is injected into the injection port, deploying the expandable proximal end. During removal, cold water is used to contract the proximal aspect of the stent, allowing endoscopic removal. Data overall is scant, with variable results, though recent publications have been promising. Kulkarni and associates published positive early results at 2 and 4 years’ experience. 18 Their 11-year results were published in 2008 with mean follow-up of 16 months. Seventy-four stents were placed in 55 patients: 28 patients with malignant obstruction and 27 with benign. Immediate complications occurred in 3 patients, including urine extravasation (1), poor expansion (1), and locking assembly failure (1). Stent migration occurred in 13 patients (23.6%), encrustation in 2 (3.7%), and fungal infection in 3 (5.5%). Fourteen (25.5%) patients needed reinsertion. There was no documentation of irritative voiding symptoms, encrustation, tissue ingrowth, or hematuria.
Similar results were published last year by Papatsoris and colleagues. 19 Memokath stents were placed in 73 patients (86 strictures) over 5 years, 55 of which were benign and 31 malignant. Stents were successfully placed in all patients and subsequently remained in situ for a mean of 11.6 months. At mean follow-up of 17.1 months, 79% remained in situ. Six stents (7%) failed, 2 for progressive malignant obstruction and 4 secondary to encrustation. Stent migration occurred in 13 (15%) stents. Conflicting results were found by Klarskov et al, who reported 22 of 37 stents (59.5%) had obstructed (12) or migrated (10) after a median 5 months, requiring stent removal. 20 Four stents were obstructed due to encrustation.
Resonance® Stent
Of particular interest recently, and the focus of this review, is the Resonance® stent (Cook Medical, Bloomington, IN)—a full length double-J stent constructed of spirally coiled nickel-cobalt-chromium-molybdenum approved in 2007 for use in malignant ureteral obstruction. The stent has no side ports or end holes, theoretically decreasing risk of tissue ingrowth. Urine passes around the stent itself, as with any double-J stent. Alternatively, urine may traverse the coils into the lumen under higher pressure above the stricture, and out again once past the stricture. The Resonance® stent can be placed antegrade or retrograde. For retrograde insertion, recommendations from the manufacturer for cystoscopic placement include first placing a guidewire into the renal pelvis under fluoroscopy. A coaxial system is then used with an outer 9F introducer sheath and 5F inner catheter, passed over the wire up to the renal pelvis. Both guidewire and inner catheter are removed and the stent is advanced under fluoroscopy through the 9F sheath. Once the coil is visualized in the renal pelvis, the pusher holds the stent in place while the sheath is pulled back until the coil is deployed in the bladder. For antegrade placement, a nephrostomy tube is placed 2 weeks prior to Resonance® stent insertion. An antegrade pyelogram is taken to identify anatomy and the stent is advanced antegrade using the 9F and 5F sheath/catheter system.
Initial data were promising; however, there have been no recent studies reviewing more recent publications. Specifically unknown are incidences of irritative voiding symptoms, hematuria, UTI, rates of stent occlusion and removal, need for secondary procedures for stent removal, and differential performance in benign and malignant strictures.
Borin and colleagues published the first case report in 2006, describing a 64-year-old female with ureteral compression secondary to metastatic breast cancer requiring percutaneous nephrostomy tube after failing double-J polymeric stent. 21 A Resonance® stent was placed and was indwelling for four months at time of publication. Since that time, multiple other studies have been published examining results in both benign and malignant ureteral obstruction. However, most studies to date are limited by retrospective design and small patient numbers, producing at times dramatic inter-study variability. An overview of the results and possible reasons for the variability in data are discussed below.
Overall performance
Overall, in the studies reviewed here, mean stent duration ranged from 21 days to 9.4 months. Removal of successfully placed stents ranged from 17.6% to 90%, with mean time in situ ranging 21 days to 9.4 months. However, within many studies there were a number of patients who kept the Resonance® stent indwelling for at least 12 months and in some cases up to 32 months. It remains unclear why some patients faired so much better than others. The high degree of inter-study and inter-patient variation was often attributed to etiology of the stricture (primarily malignant versus benign) though other factors were evaluated including prior radiation exposure, encrustation, migration, and recurrent urinary tract infections.
Recurrent obstruction
Results between patients with malignant and benign obstruction varied widely (Table 1). The majority of studies observed either patients with malignant obstruction exclusively, or a combination of malignant and benign obstruction. Several of the studies appear to be outliers, though with an absence of randomized controlled trials, it is difficult to draw definitive conclusions. When broken down into malignant versus benign, excluding Garg and associates’ study of uretero-enteric strictures, median stent duration in malignant disease ranged from 4 to 11 months versus 4.75 to 11.8 months in benign disease. Complicating generalizability of the data are the apparent contradictory findings of two specific papers comparing malignant to benign disease. In 2008 Nagele and colleagues published three-year follow-up data on 14 patients (18 stents) with ureteral obstruction, either malignant or benign, who were stented with 6F × 28 cm Resonance® stents. 22 Mean (median) stent duration at was 8.6 (9) months. Eight (44%) stents were removed, 4 with insufficient drainage and 4 due to infection, voiding symptoms, or hematuria. Several patients retained the stents for >12 months, including 2 for 16 months, without ill effect. Patients with benign disease kept the stent in place for a mean of 11.8 months compared to 7.3 months in patients with malignant obstruction. This contrasts with data published by Liatsikos and colleagues in 2009. 23 They placed 54 stents in 50 patients with benign (18) or malignant (25) obstruction, as well as in 7 patients with obstructed metal mesh stents. Successful stent placement was 100%, with balloon dilation required in 19 cases. Mean overall follow-up was 8.5 months (range 4–14). The authors reported 100% patency rate in malignant strictures at a mean follow-up of 11 months compared to 44% in benign strictures (failure occurred at mean follow-up of 2.6 months).
Results in Resonance® stent placement in malignant versus benign cases.

3 stents placed but had poor drainage on subsequent antegrade; renal unit managed with PCN, but no comment on whether stents were removed;
includes 7 Resonance® stents placed in obstructed metal mesh stents at a ureteroenteric stricture.
Given this variability, the results warrant interpretation within the clinical context in which the stent was placed. Malignant disease generally portends a poor prognosis, with a life expectancy typically measured in months. 6 Due to a chronic onset, malignant obstruction often does not produce the same flank pain as an acute obstruction. Therefore, rather than symptom relief, the goal is maximizing renal function during chemotherapy and/or minimizing recurrent procedures when the disease process almost inevitably progresses. Interestingly, the percentage of patients dying during follow up was quite different between studies, ranging from 0%–46% during follow-up of up to 12 months. This indicates probable heterogeneity in aggressiveness, stage, or clinical status of disease, a factor not specifically addressed in any study. Furthermore, type of cancer was intermittently reported (ie, colon, prostate, cervical), which again may add heterogeneity to disease parameters, accounting for some of the inter-study variability. A recent study by Izumi et al found ureteral obstruction secondary to gynecologic malignancy to have significantly better prognosis in terms of stent failure free rates than obstruction due to other malignancies. 24 Other factors that might impact stent patency were also not assessed. For example, gross bladder invasion by any primary cancer was an independent predictor of polymeric stent failure in a study by Ganatra et al. 6 None of the studies we reviewed documented presence or absence of bladder invasion at time of stent placement or removal, and none evaluated bladder invasion in association with stent failure. An additional point discussed by Modi and colleagues is that in their study, they document median follow up of 5 months (range 0–18 months). 25 However, in the stents which obstructed, median time to obstruction was only 1.5 months. They specifically note that 39 of 44 malignantly obstructed renal units had experienced prior plastic stent failure. Liatsikos et al report 100% stent patency at 11 months follow-up for malignant obstruction, but do not report prior failure of plastic stents, which could also account for some inter-study variability. 23 While replacing a plastic stent with a Resonance stent in a patient with MUO may seem like a reasonable next step, future studies may show prior plastic stent failure to be an unfortunately strong predictor of future failure with a metallic stent.
For benign strictures, a long-term, more cost effective solution is sought, as the estimated cost of annual management with traditional double-J stents is $23,999. 26 In the only study limited to benign disease, López-Huertas et al evaluated cost effectiveness of Resonance® stent in 13 patients, all with obstruction previously managed with double-J polymeric stents. Resonance® stents provided adequate initial drainage in 12 of 13 patients. 26 Eight of 13 (61.5%) patients were able to keep Resonance® stents in place for 12 months without significant encrustation. One stent was removed for obstruction at 5 months. In stents that were removed, average time to stent removal was 5.2 months. Calculations indicated a mean cost reduction of 43% per patient per year, or approximately $10,394 (43%) per patient per year as compared to more frequent polymeric stent changes. The most recently published data from Benson and colleagues reports no failures in 15 patients (18 ureters) with benign strictures with median follow up of 13 months, which stands out as the highest success rate with benign stricture disease to date. 27 They note 100% success in placing the stents without balloon dilation, possibly indicating a difference in the character of benign strictures treated in this study; Liatsikos documented balloon dilation rates of up to 35% and had markedly poorer results in their benign group.
Encrustation of Resonance® stents was of variable clinical significance between studies (Table 2). Removal for encrustation occurred infrequently overall, generally ranging 0%–3%, though Neagle and associates noted removal of 2 of 18 stents (11%) for encrustation. 22 Secondary procedures for stent removal were uncommon, occurring only in one study for two patients. 25 Interestingly, Liatsikos and associates noted macroscopic encrustation in 12 of 54 stents (22%), though by scanning electromicrograph (EM) and energy dispersive analysis by x-ray (EDAX), some degree of encrustation was noted on all stents. 23
Reported removal of Resonance® stents.
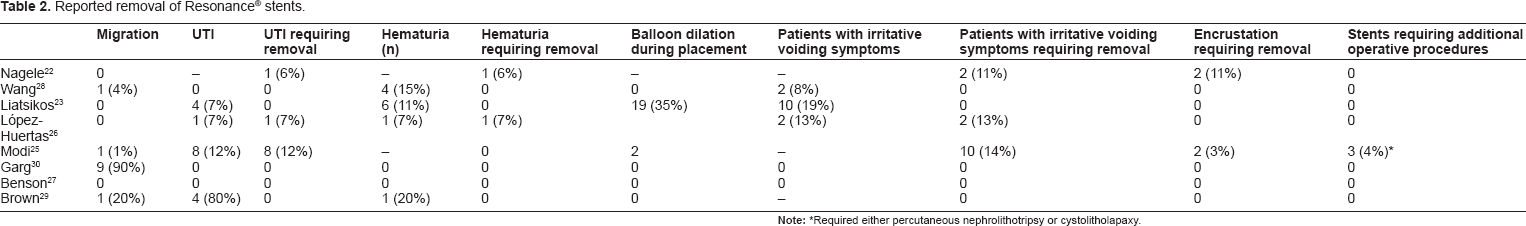
Required either percutaneous nephrolithotripsy or cystolitholapaxy.
Two studies noted obstruction due to tissue ingrowth. Liatsikos and associates found hyperplastic tissue ingrowth to be responsible for 7 of 8 stent failures in stents placed for benign disease and 7 of 7 failures in stents placed within mesh. 23 Modi et al found 1 patient to have tissue ingrowth, which required percutaneous removal. 25 Benson and associates are the only other group to describe urothelial ingrowth, which they reported to cause bilateral obstruction in one patient with progressive malignant obstruction. 27 However, the stents were left in place and so had not been physically examined at time of publication.
Two studies have examined results of prior pelvic radiation on Resonance® stent patency. Wang et al reported on 26 stents and observed a 50% rate of stent failure in patients who had undergone prior radiation therapy. 28 This is in contrast to Nagele et al who did not note any difference between radiated versus non-radiated renal units. 22 No other studies included pelvic radiation as a predictor of failure in multivariate analysis.
One study by Brown and colleagues noted obstruction in 4 of 5 stents within 4 months, all after having documented urinary tract infection. 29 Despite very small patient numbers, this prompts concern that the stents may be predisposed to obstruction after infection. However no other studies have described similar findings. Incidence of urinary tract infections prompting stent removal ranged from 0% to 12%, but no other study specifically described obstruction after infection.
Stent migration
In general, stent migration is an uncommon complication with Resonance® stent placement. The exception to that statement seems to be placement for ureteroenteric strictures. In the retrospective study by Garg and associates in 2009, 10 ureteroenteric strictures initially treated with polymeric single-J stents which were exchanged for Resonance® double-J stents. 30 Nine of 10 stents were removed due to migration, with mean time to migration of 21 days. Eight of the 9 patients had the distal curl of the stent protruding from the stoma and the other presented at 2 months with pyelonephritis. One of 10 patients was able to keep the stent in place for the full 12 months. Postulated reasons for the high rate of stent migration included increased weight of the metallic stents and motility of the bowel segment. Modi et al, 25 Brown et al, 29 and Wang et al 28 each documented one patient per case series with stent migration. Interestingly, Liatsikos et al placed Resonance® stents in 6 patients with ureteroenteric strictures and noted 50% failure rate among that group, though none of those failures were attributed to stent migration. 23 In fact, they did not observe migration in any of 54 stented renal units. López-Huertas et al 26 and Benson et al 27 also documented no stent migration in 13 or 42 stents, respectively.
Hematuria/bladder irritation
Hematuria occurred in 0%–6% of patients and necessitated removal of the stent in 0%–7% (Table 2). Irritative voiding symptoms occurred in 0%–17%, prompting removal in 0%–14% of patients. Nagele and colleagues placed 18 stents in 14 patients, of which 2 were removed for dysuria/pain and one for persistent hematuria. 22 Wang and associates noted mild hematuria in 4 of 26 Resonance® stents (15.4%), which resolved spontaneously without requiring stent removal. 28 Two of 26 patients had slight urgency/irritation type symptoms, also not requiring stent removal. In a review of 13 patients, López-Huertas et al removed 2 stents for persistent irritative voiding symptoms and one for persistent hematuria. 26 Liatsikos et al documented 6 of 50 patients (12%) to have gross hematuria, though all resolved spontaneously. 23 Ten of 50 patients (20%) had mild irritative symptoms. At the time of the study, only 6F × 28 cm stents were available. There was a trend toward significance for patients > 170 cm to keep the stent for a longer period of time, interpreted to indicate that appropriate sizing helps patients tolerate the stent. Modi et al noted 10 of 69 (14%) patients had Resonance® stents removed <12 months due to “stent symptoms.” 25 Benson and colleagues report no stent removals for refractory symptoms and no other stent complications though they do not comment specifically on symptoms which did not prompt stent removal. 27
Summary
Chronic ureteral obstruction continues to be a difficult clinical problem, whether the etiology is malignant or benign. Management options have traditionally been limited to open resection or reconstruction in patients who are surgical candidates versus double-J stents or percutaneous nephrostomy tubes in patients who are not. The Resonance® stent has an advertised in situ lifespan of 12 months and higher radial strength, in theory increasing resistance to external compression, decreasing cost/morbidity of stent changes, and precluding need for PCN tubes. Initial results were uniformly positive, with early data indicating stents could maintain good functionality with minimal encrustation despite lengthy periods of time in situ.
However, the Resonance® stent does fail for a variety of reasons in a significant portion of patients. Comparison to other metal stents and even between Resonance stent studies is difficult given the small number of applicable studies and the typically small number of patients in each publication. Furthermore, inter-study variability in reporting complications, definitions of failures, and follow up makes generalizability more difficult. While a potentially useful tool, further investigation and research are needed to further define guidelines for use and patient populations who most benefit from Resonance stent placement.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
