Abstract
Telcagepant (MK0974) is one of several calcitonin-gene-related peptide antagonists in development as a potential treatment for acute migraine attacks and is the first orally available drug in this class. Preclinical and clinical data are reviewed, which support the efficacy and tolerability of telcagepant for the treatment of migraine, particularly for patients unable to tolerate, or who have cardiovascular contraindications to, triptans.
Keywords
Introduction
Migraine is common neurological disorder recognized to affect at least 11% of the adult population. 1 The condition is characterized by recurrent episodic attacks of headache, lasting 4–72 hours, associated with nausea and/or vomiting, photophobia, and disability.2,3 The pathophysiology of migraine is complex and not fully elucidated. It is thought that during a migraine attack primary sensory neurons in the trigeminal ganglion are activated and release vasoactive neuroinflammatory peptides, including substance P, neurokinin A, and nitric oxide from nerve endings within the meninges. Consequent vasodilation of meningeal blood vessels results in plasma extravasation and mast cell degranulation. This in turn leads to stimulation of the trigeminocervical complex transmitting pain impulses to the trigeminal nucleus caudalis in the brain stem, which are relayed to the thalamus. Information from the thalamus passes to the cerebral cortex, which decodes the messages into pain.
Recent research has implicated calcitonin-gene-related peptide (CGRP) as a key molecule in the pathophysiology of migraine and other primary headaches.4–6 This 37-amino-acid neuropeptide was first identified in 1982. 7 It is a potent arteriolar vasodilator and mediates relaxation of these arteries via activation of the CGRP(1) receptors in human intracranial circulation. 8 CGRP receptors are ubiquitous throughout the human body, suggesting that the peptide is involved in physiological functions of all major systems. CGRP is widely distributed within the CNS. Identification of a dense supply of CGRP-containing fibres around the cerebral vessels, reported to originate in the trigeminal ganglion, increased interest in the role of the molecule in migraine. 9 Trigeminal nerve activation, putatively as a response to localized cerebrovascular constriction, results in antidromic release of CGRP and consquent vasodilatation. 10
Several studies have noted increased levels of CGRP during migraine attacks.4,11,12 Intravenous administration of human alpha-CGRP can induce headache in individuals susceptible to migraine.13,14 Following treatment with sumatriptan, eletriptan and rizatriptan elevated levels of CGRP revert to normal, coincident with reduction in migraine symptoms.12,15,16 These findings led to the development of CGRP antagonists to treat migraine by blocking the action of CGRP on the CGRP-receptor complex. Telcagepant (MK0974) (Fig. 1) is one of several CGRP antagonists in development as a potential treatment for acute migraine attacks and is the first orally available drug in this class.
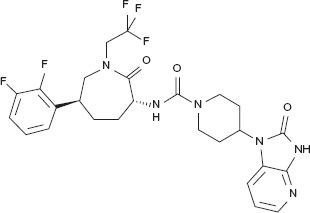
Chemical properties of telcagepant.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Mechanism of action
Telcagepant antagonises CGRP-induced relaxation, without affecting vascular tone.17–19 This peripheral inhibition of the vasodilatory effects of CGRP supported the potential for telcagepant to relieve the headache and associated symptoms of migraine in the absence of adverse coronary effects. However, the site of action of telcagepant and other CGRP antagonists in migraine is unclear (Fig. 2). A central mechanism has been proposed on the basis that a higher dose appears necessary to achieve a clinical effect than would be expected from the potency of telcagepant if it was acting peripherally. 20 The central hypothesis has been disputed on the basis that in-vitro receptor binding studies predicting telcagepant potency do not translate to in-vivo studies, which show that higher than expected doses are necessary to achieve blockade of known peripheral CGRP-mediated effects. 21 One explanation, which would explain the favorable tolerability profile of CGRP receptor antagonists, is that CGRP acts as modulator role in the CNS. 10
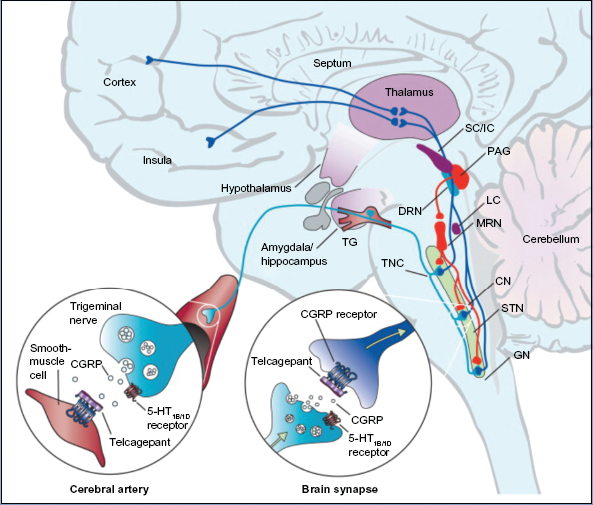
Brain areas expressing CGRP receptor that are possible sites of action of telcagepant in migraine treatment.
Metabolism
The primary route of elimination is hepatic metabolism, via cytochrome P450 3A. 22 Around 95% of the parent drug is metabolized, the major metabolite being a pyridine N-oxide.
Pharmacokinetics
Assessments of telcagepant in monkeys and rats suggest species dependent nonlinear pharmacokinetics following oral doses of telcagepant. 22 Telcagepant inhibited [125I]-hCGRP binding to membranes expressing the native human CGRP receptor in SK-N-MC cells with a Ki of 0.78 nM ± 0.05 nM and a Ki of 0.77 ± 0.07 nM on membranes from cells expressing the recombinant CGRP receptor. 23
Three separate clinical trials evaluated the pharmacokinetics and tolerability of telcagepant in healthy volunteers. Single oral doses of 300-mg or 400-mg telecagepant were studied in fasting healthy young men (n = 12), young men and women (n = 12), elderly men and women (n = 24), while multiple oral doses were studied in young men (n = 24). 24 A further two separate open-label studies assessed the oral (n = 19) and intravenous (n = 10) dose proportionality of telcagepant. 25
Following single oral dose administration in men and women, telcagepant was rapidly absorbed with median Tmax of 1 to 2 hours; plasma concentrations decreased from Cmax in a biphasic manner, with an apparent terminal half-life of 4 to 7 hours. 24 No clinically meaningful differences of single oral dose pharmacokinetics of telcagepant were noted in female or elderly subject pharmacokinetics compared to those observed in young male subjects. With multiple oral dose administration in men, steady state was achieved rapidly, within 3 to 4 days of dosing, with an accumulation ratio of approximately 2 for AUC0–24 h and Cmax. 24
Following intravenous dosing, the terminal half-life was approximately 8 to 9 hours. 25 Intravenous administration resulted in approximately dose proportional increases in exposure. 25 As with preclinical studies, the pharmacokinetics of oral telcagepant were non-linear, with greater than dose-proportional increases observed for some exposure measures following both single and multiple-dose administration. 24 This is thought to be a result of saturable metabolism/efflux of telcagepant in the gut.
Migraine is associated with delayed gastric emptying, reducing the absorption of oral medication can be reduced. 26 To study the pharmacokinetics of telcagepant during and between migraine attacks, a randomized, double-blind, placebo-controlled, two-period fixed-sequence study was undertaken in 23 migraineurs. 27 For the first part of the study, patients received study drug within approximately 2 hours of onset of a moderate to severe migraine attack, provided that this was at least 4 h after their last meal. In the second part, the study drug was administered to the same patients when they were migraine-free, at least 4 h after their last meal and at a similar time of day to their attack dose. Fifteen patients received 300 mg telcagepant in both periods and 8 received placebo. Geometric mean ratios during/between migraine attacks (90% CIs) for pharmacokinetic measures were: Cmax = 0.89 μM (0.71,1.11); AUC0–4h = 0.94 μM h (0.77,1.14); AUC = 1.07 μM h (0.91,1.25). The median T during migraine was 1.5 h versus 1.0 h between attacks. The mean half-life was 8.5 h during a migraine attack compared to between attacks (vs. 10.6 h).
Clinical Studies
A phase 2 randomized double-blind, parallel, dose-finding study used a two-stage adaptive design to identify potentially beneficial doses of telcagepant for acute treatment of migraine attacks. 28 This was a multicentre study comparing oral telcagepant (capsule formulations of 25, 50, 100, 200, 300, 400, and 600 mg) with oral rizatriptan (10 mg). The percentage of patients reporting the primary endpoint of pain relief at 2 hours was 68.1% for 300 mg telcagepant, 48.2% for 400 mg, and 67.5% for 600 mg (P = 0.015 versus placebo) compared to 69% for rizatriptan (P = 0.010 versus placebo) and 46.3% for placebo. Telcagepant was generally well tolerated and with no apparent increase in adverse events with increasing dose. The doses chosen for further clinical studies were 150 mg and 300 mg capsules. These doses are bioequivalent to 140 mg and 280 mg tablets, respectively. 29
Efficacy
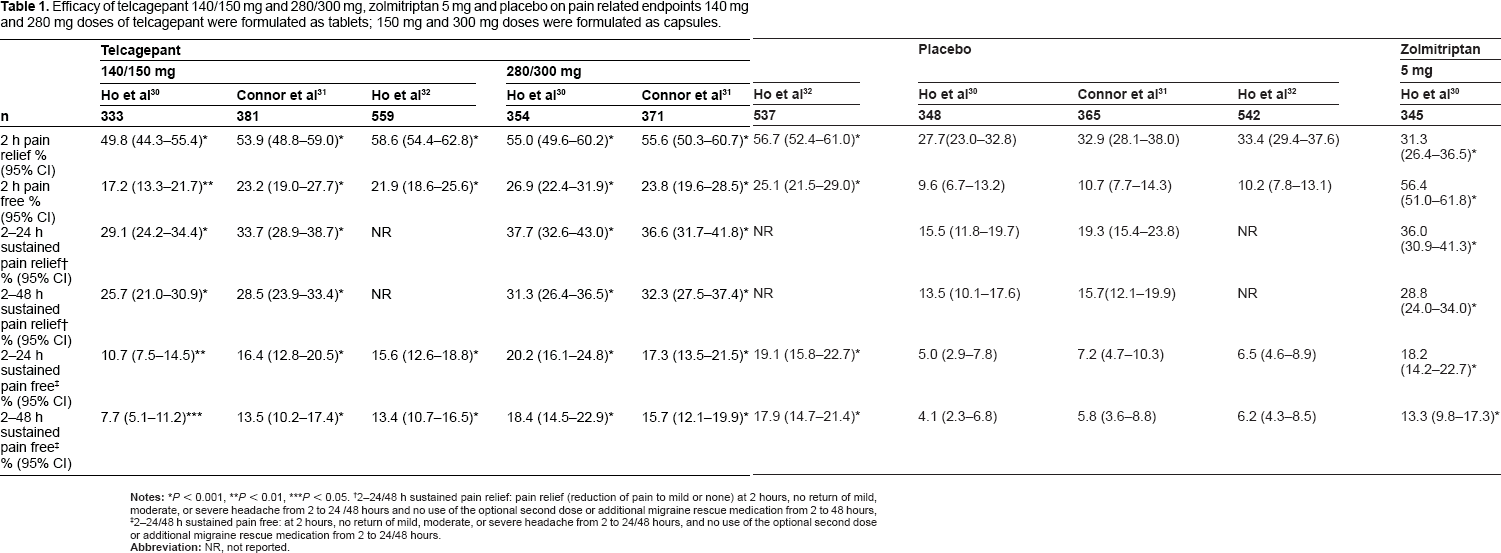
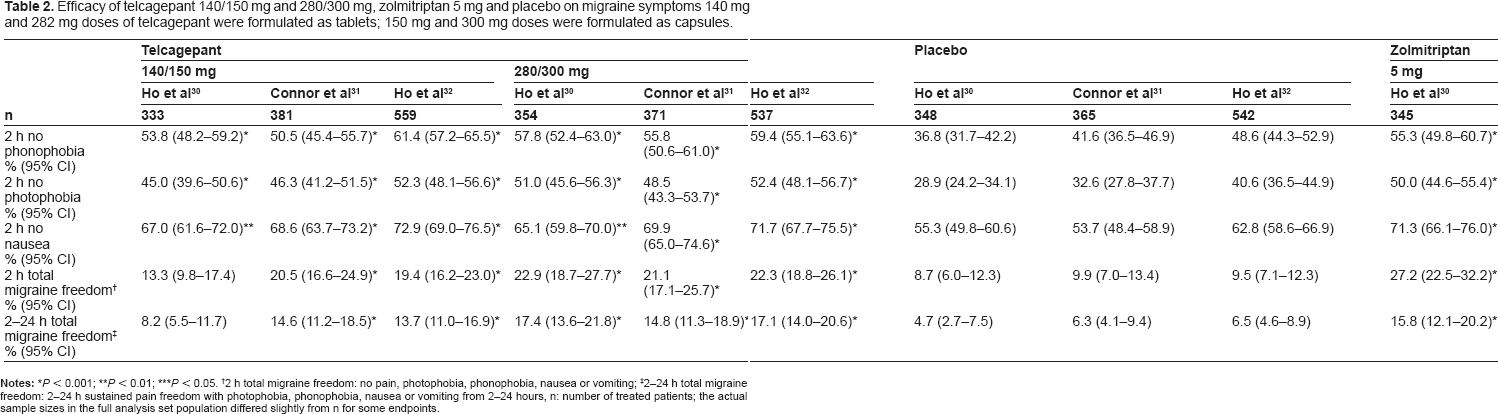
Three phase 3 efficacy studies of telcagepant for the acute treatment of migraine have been published. The first was a multicenter placebo-controlled parallel-treatment study in which 1380 patients were randomised to treat moderate or severe attacks with either oral capsules of telcagepant 150 mg or 300 mg, zolmitriptan 5 mg, or placebo. 30 No differences were reported at baseline in the characteristics of patients and treated migraine attacks in the different groups. The efficacy of telcagepant 300 mg and zolmitriptan 5 mg was similar for the percentage of patients achieving the five co-primary endpoints of pain freedom, pain relief, or absence of photophobia, phonophobia, or nausea at 2 h after treatment (Tables 1 and 2). Telcagepant 300 mg and zolmitriptan 5 mg were more effective than telcagepant 150 mg and placebo on all endpoints, including 24 h and 48 h sustained pain freedom. Telcageplant 150 mg was more effective than placebo for the primary endpoints. Telcagepant 300 mg and zolmitriptan 5 mg were more effective than telcagepant 150 mg and placebo on 2 h functional disability and 24 h migraine quality-of-life (Table 3).
Efficacy of telcagepant 140/150 mg and 280/300 mg, zolmitriptan 5 mg and placebo on pain related endpoints 140 mg and 280 mg doses of telcagepant were formulated as tablets; 150 mg and 300 mg doses were formulated as capsules.
P < 0.01,
P < 0.05.
2–24/48 h sustained pain relief: pain relief (reduction of pain to mild or none) at 2 hours, no return of mild, moderate, or severe headache from 2 to 24 /48 hours and no use of the optional second dose or additional migraine rescue medication from 2 to 48 hours,
2–24/48 h sustained pain free: at 2 hours, no return of mild, moderate, or severe headache from 2 to 24/48 hours, and no use of the optional second dose or additional migraine rescue medication from 2 to 24/48 hours.
Efficacy of telcagepant 140/150 mg and 280/300 mg, zolmitriptan 5 mg and placebo on migraine symptoms 140 mg and 282 mg doses of telcagepant were formulated as tablets; 150 mg and 300 mg doses were formulated as capsules.
P < 0.01;
P < 0.05.
2 h total migraine freedom: no pain, photophobia, phonophobia, nausea or vomiting;
2–24 h total migraine freedom: 2–24 h sustained pain freedom with photophobia, phonophobia, nausea or vomiting from 2–24 hours, n: number of treated patients; the actual sample sizes in the full analysis set population differed slightly from n for some endpoints.
2-hour functional disability and 24-hour migraine quality-of life.
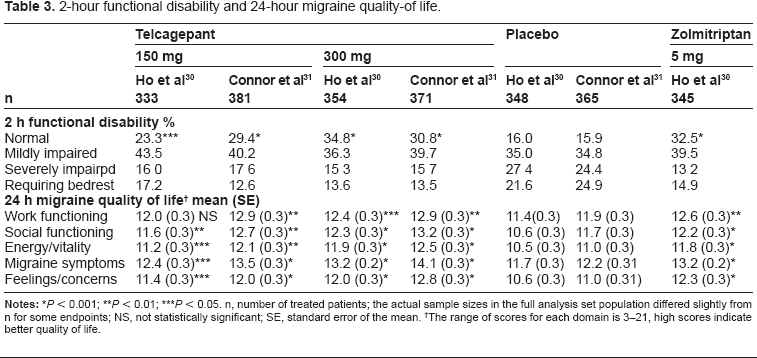
P < 0.01;
P < 0.05. n, number of treated patients; the actual sample sizes in the full analysis set population differed slightly from n for some endpoints; NS, not statistically significant; SE, standard error of the mean.
The range of scores for each domain is 3–21, high scores indicate better quality of life.
Another phase 3 randomized, double-blind, placebo-controlled, multicentre trial assessed oral capsules of telcagepant 50, 150, and 300 mg in 1294 patients with migraine. 31 Telcagepant 150 mg and 300 mg were significantly more effective than placebo for all five co-primary endpoints of pain freedom, pain relief, or absence of photophobia, phonophobia, or nausea at 2 h after treatment and for the secondary endpoints of 24 h and 48 h sustained pain relief and pain freedom, total migraine freedom at 2 h and 24 h functional disability at 2 h and migraine quality-of-life at 24 h (Tables 1–3). Both doses were significantly more effective than placebo.
A third phase 3 study evaluated oral tablets of telcagepant 140 and 280 mg, bioequivalent to oral capsules of telcagepant 150 and 300 mg respectively. 32 In this double-blind, placebo-controlled trial 1677 patients were randomized to treat up to four moderate-to-severe migraine attacks within 6 months. Control patients received placebo for three attacks and telcagepant 140 mg for one attack. Based on data from the first attack treated, telcagepant 140 mg and 280 mg were significantly more effective than placebo for all five co-primary endpoints of pain freedom, pain relief, or absence of photophobia, phonophobia, or nausea at 2 h after treatment and for the secondary endpoints of 24 h and 48 h sustained pain freedom and total migraine freedom at 2 h and 24 h (Tables 1 and 2). The percentage of patients with pain freedom at two hours and pain relief at two hours for each attack by treatment sequence was consistent across all four attacks (Fig. 3). Consistent efficacy was defined as ≥3 successes, and lack of consistent efficacy was defined as ≥2 failures, in treatment response. Telcagepant 140 mg and 280 mg were more consistently effective compared to the control group.
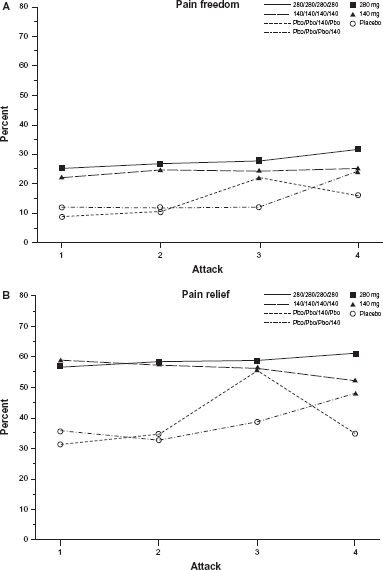
Observed percentage of migraine patients reporting (A) pain freedom at two hours and (B) pain relief at two hours by treatment sequence over four attacks. 32
Evaluation of efficacy was a secondary objective in a long-term safety study of telcagepant 280/300 mg and rizatriptan 10 mg. 33 Rizatriptan was more effective than telcagepant for the treatment of mild, moderate, or severe migraine attacks for 2 h pain free (OR 0.58 [95% CI 0.45–0.75]) and 2 h pain relief (OR 0.70 [95% CI 0.55–0.89]). For 24 h sustained pain freedom, the response was not significantly different (OR 0.80 [95% CI 0.63–1.02]). A posthoc analysis of response by triptan user subgroups, suggested that the differences were more clearly in favor of rizatriptan in triptan users while among nontriptan users, the two treatment groups were more similar. There was no evidence to suggest tolerance, as both telcagepant and rizatriptan demonstrated a consistent treatment effect over time.
A phase 2 randomized, double-blind, placebo-controlled study evaluated the efficacy and tolerability of telcagepant when co-administered with ibuprofen or acetaminophen for the acute treatment of migraine. 34 Patients treat ed a moderate or severe migraine headache with either telcagepant 280 mg and ibuprofen 400 mg (n = 145), telcagepant 280 mg and acetaminophen 1000 mg (n = 133), telcagepant 280 mg (n = 138), or placebo (n = 147). Compared to placebo, 2-hour pain freedom was greater in each active treatment group (P < 0.001): telcagepant/ibuprofen 35.2% (90% CI 28.6–42.2), telcagepant/acetaminophen 38.3% (90% CI 31.3–45.8), telcagepant 31.2% (90% CI 24.7–38.3), placebo 10.9% (90% CI 6.9–16.1). The results for telcagepant/ibuprofen and telcagepant/acetaminophen were numerically greater than, but not statistically significantly different from, telcagepant alone. Since the study had approximately 88% power to detect an additive effect of at least 15 percentage points (telcagepant combinations versus telcagepant alone) and 48% power to detect an additive effect of at least 10 percentage points, the authors recommended further studies powered to detect smaller additive benefits.
A phase 3 randomized, double-blind, placebo/ active-controlled, 2-period crossover (6 weeks per period) assessed oral telcagepant 300 mg versus placebo and oral paracetamol 1000 mg in adults with stable coronary artery disease and migraine. 35 Patients were randomized to either: telcagepant 280 mg (tablet)/300 mg (capsule) for up to 12 attacks, crossing over to acetaminophen 1000 mg for up to 12 attacks; or placebo for the first attack then acetaminophen 1000 mg for up to a further 11 attacks, crossing over to telcagepant 280/300 mg for up to 12 attacks. The primary efficacy endpoint was 2-hour pain freedom (telcagepant versus placebo for Attack 1, Period 1). Only 165 patients of the planned 400 were enrolled (<50% of the planned sample size). The proportions of patients with 2-hour pain freedom were 13/52 (25.0%) for telcagepant and 10/53 (18.9%) for placebo; the difference was not statistically significant.
Results are awaited from a six-month phase 2/3, randomized, double-blind, placebo-controlled clinical trial to evaluate the safety, tolerability, and efficacy of telcagepant for prevention of menstrually-related migraine in 4500 female patients with episodic migraine (NCT01125774). In this study, women were randomized to 140 mg telcagepant or placebo once daily at bedtime for 7 days, beginning at onset of menses, for 6 menstrual cycles.
Tolerability
Results from clinical trials confirm that telcagepant was generallywell tolerated, with an adverse event rate similar to placebo.
The most common adverse events reported in clinical trials with telcagepant are dry mouth, somnolence, dizziness, nausea and fatigue (Table 4).30–34 The majority of adverse experiences occurred during the first 48 hours after dosing and the tolerability profile up to 14 days after dosing is very similar to adverse experiences occurring within 48 hours.31,32
Adverse events reported within 48 h post-dose
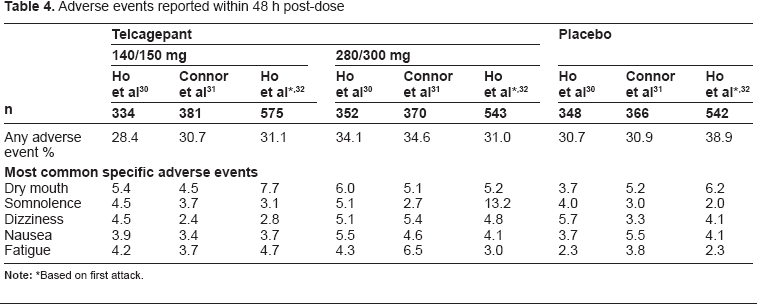
In the long-term 18-month tolerability study, the percentages of patients with adverse events possibly related to abuse liability were also similar between the treatment groups. 33 The three adverse events identified as having potentially greatest clinical significance for abuse liability (confusion, dissociation, euphoria) all occurred in the telcagepant group but were rare and generally isolated events that did not recur over subsequent attacks. Overall, these findings suggest that telcagepant does not have potential for abuse liability.
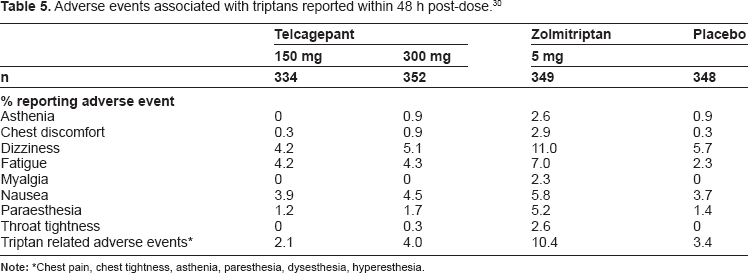
Compared with triptans, telcagepant appeared to have fewer of the adverse events commonly associated with triptans (Table 5).30,33
Adverse events associated with triptans reported within 48 h post-dose. 30
In a study of telcagepant 280 mg co-administered with ibuprofen 400 mg or acetaminophen 1000 mg for the acute treatment of migraine, all the active treatments were generally well tolerated although the combination treatments were associated with a higher percentage of adverse events: telcagepant/ ibuprofen 20.7%, telcagepant/acetaminophen 21.8%, telcagepant 15.3%, placebo 13.5%. 34 As in previous studies with telcagepant, adverse events which showed an increase over placebo in all active treatment groups were fatigue, dizziness, somnolence, and tremor.
Safety
With respect to cardiovascular safety, no consistent clinically relevant effects on electrocardiography parameters, blood pressure, or heart rate were observed in single and multiple-dose clinical pharmacology studies. 24
A preclinical study investigated the effects of telcagepant on human isolated coronary arteries. 18 Arteries with different internal diameters were studied to assess the potential for differential effects across the coronary vascular bed. Telcagepant alone did not induce contraction or relaxation of these coronary blood vessels. Pretreatment with telcagepant (10 nM to 1 microM) antagonized alphaCGRP-induced relaxation competitively in distal and proximal coronary arteries and coronary arterioles. alphaCGRP significantly increased cAMP levels in distal, but not proximal, coronary arteries, and this was abolished by pretreatment with telcagepant. These findings support the cardiovascular safety of telcagepant and suggest that it is unlikely to induce coronary side effects under normal cardiovascular conditions.
Several clinical studies support the cardiovascular safety profile of telcagepant. Two double-blind trials assessed the effect of high dose telcagepant on 24-hr systolic, diastolic and mean arterial blood pressure and heart rate in healthy volunteers. 36 Subjects were randomised to telcagepant 600 or 560 mg daily (dose equivalents) or placebo. In study 1, treatments were given in the morning for 8 consecutive days and in study 2 treatments were given in the evening for 5 consecutive days. In each study, 24-hr ambulatory blood pressure and pharmacokinetics were monitored on the first and final study days. For all parameters in both studies, telcagepant demonstrated no differences from placebo between the results from the first and final days.
A phase 1 double-blind, placebo-controlled, 2-period, single-dose crossover study assessed the safety, tolerability and effects of telcagepant on exercise tolerance in 61 patients with reproducible exercise induced angina. 37 Patients were randomized to a single oral dose of telcagepant 900 or 600 mg, or placebo treatment. There were no significant treatment differences in total exercise duration, maximum exercise heart rate, maximum ST segment depression (STD) or time to 1 mm STD among pooled data or upon stratification for dose.
In a phase 3 randomized, double-blind, placebo/ active-controlled, 2-period crossover of oral telcagepant 300 mg versus placebo and oral paracetamol 1000 mg in adults with stable coronary artery disease and migraine, the primary safety analysis was conducted on adverse events occurring within 48 hours, over all attacks and excluding those events attributable to placebo. 35 No adverse events observed within 14 days of dosing occurred in more then four patients. Six patients reported serious clinical adverse events within 14 days (four on telcagepant, two on acetaminophen). Three serious vascular adverse events were sent for adjudication (two on telcagepant, one on acetaminophen), all of which were adjudicated as non-thromboembolic events and occurred more than 48 hours after dosing. There were no clinically relevant changes in laboratory measures and no instances of elevated transaminase levels ≥ 3 times the upper limit of normal.
Further studies have compared the effects of telcagepant with triptans. A preclinical study conducted in human isolated coronary arteries compared telcagepant with zolmitriptan. 19 Zolmitriptan caused a concentration-dependent contraction while telcagepant evoked no change in contractile tone.
A phase 1 randomised, double-blind, placebo-controlled and active-controlled study assessed the safety, tolerability, and effects on blood pressure of concomitant oral telcagepant and oral sumatriptan in 24 patients with migraine. 38 Mean arterial blood pressure was the same following administration during the interictal period of a single dose of sumatriptan 100 mg alone or sumatriptan 100 mg with telcagepant 600 mg. During the 2.5 h following treatment sumatriptan alone or with telcagepant raised semirecumbent mean arterial blood pressure, whereas telcagepant alone did not.
In a placebo-controlled phase 2 dose-ranging study of 25–600 mg telcagepant versus rizatriptan only one serious adverse experience (appendicitis) was reported during the study and occurred in a patient who received placebo treatment. 28
In the three phase 3 studies there were no drug-related serious adverse experiences.30–32 Laboratory abnormalities were uncommon and no clinically relevant differences were seen between treatment groups. Other assessments, including the percentage of patients who exceeded predefined levels of change on laboratory parameters, vital sign measurements, electrocardiography measurements, and physical examinations, indicated no clinically meaningful differences between treatment groups. Specifically, there were no clinically relevant increases in the transaminases alanine aminotransferase (ALT) and aspartate aminotransferase (AST) three or more times the upper limit of normal.
However, elevated transaminases have been noted in other clinical trials in the programme. In a phase 1 study a transaminase elevation of 3.1 times the upper limit of normal was observed in a single subject who received 2 doses of telcagepant 400 mg 2 hours apart for 5 days. 24 The subject's transaminase declined below the times the upper limit of normal with subsequent dosing.
A phase 2 migraine double-blind placebo controlled study (NCT00797667) evaluated the efficacy of telcagepant for prophylactic treatment of migraine. The study was not completed following the identification of two patients with significant elevations in serum transaminases. 39
As both these studies involved consecutive daily dosing with telcagepant, the exposure achieved was higher than would be expected with intermittent acute treatment for migraine, suggesting that the potential for hepatic toxicity may be time and dose-dependent.
A phase 3 long-term safety study of oral telcagepant 280 mg and oral rizatriptan 10 mg, telcagepant was used by 640 patients who treated 19,820 migraine attacks over 18 months. 33 Three patients in the telcagepant group experienced transaminase elevation ≥3 times the upper limit of normal: one patient with coincident muscle injury had increased AST; one patient had increased ALT 2 months after dosing; and one patient had increased ALT and continued to treat with telcagepant without further elevations. These events were asymptomatic, transient, and temporally unrelated to dosing with study medication. A larger proportion of patients in the telcagepant group reported elevation in total bilirubin compared with patients in the rizatriptan group (11/640, 1.7% vs. 1/311, 0.3%). However, all but one of the telcagepant patients had a high-normal or elevated total bilirubin at baseline, including three patients with preexisting hepatic abnormalities (two patients with Gilbert's syndrome and one patient with hepatic steatosis).
In the study of telcagepant/ibuprofen, telcagepant/ acetaminophen, one patient in the telcagepant 280 mg group had ALT and AST elevations three or more times the upper limit of normal 11 days postdose. 34 The patient had been taking 2 g/day of non-study acetaminophen for pharyngitis for 1 week approximately 2 weeks prior to the elevations and also had a history of unexplained transient elevated liver function tests. The authors comment that while they cannot definitively exclude the possibilitythat telcagepant led to the transaminase elevations, it is more likely that they were related to the acetaminophen administration in a patient with possible underlying liver disease.
To date, none of the patients in the trials has fulfilled Hy's law criteria. This is an indicator of the potential for a drug to cause serious liver injury, defined as transaminases three or more times the upper limit of normal together with total bilirubin two or more times the upper limit of normal, in the absence of any non-drug cause. However, ongoing trials are carefully assessing hepatic risk.
Place in Therapy
In current practice, triptans (almotriptan, eletriptan, frovatriptan, naratriptan, rizatriptan, sumatriptan and zolmitriptan) are the drugs of choice for the acute treatment of migraine in patients who fail to gain adequate response from analgesics.40–42 Despite their excellent efficacy profile, triptans are not suitable for all patients, particularly those with cardiovascular risk factors. The comparative efficacy and favorable cardiovascular safety profile of telcagepant suggest that it should be the drug of choice for the treatment of migraine in patients with, or at risk of, cardiovascular disease.
Intolerance of adverse events is an additional limiting factor of triptan use, although the adverse events profiles vary between the seven triptans available. 43 A comparative review of clinical trial data indicates that telcagepant will be an effective alternative (Table 6). 44
Efficacy and tolerability of triptans and telcagepant. 44
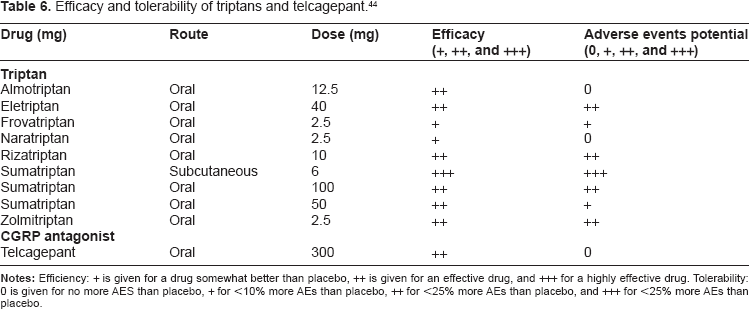
Headache recurrence or relapse, defined as a worsening of headache after pain free or mild pain has been achieved with a drug within 24 h, affects 12%–44% of patients taking an oral triptan. 43 There are limited data to suggest that thi s may be less likely to occur with telcagepant. In placebo-controlled trials treatment with telcagepant resulted in a significantly higher percentage of patients achieving sustained response than either rizatriptan or zolmitriptan.28,30 In the long-term safety study of oral telcagepant 280 mg and oral rizatriptan 10 mg, the initial 2-hour efficacy advantage of rizatriptan over telcagepant was diminished at later time points indicating that telcagepant may provide greater sustained effect in some patients. 33 Additional support for this observation is the greater numerical efficacy for telcagepant compared to rizatriptan with respect to the proportions of patients treating an average of 4 or more attacks per month, treating on 4 or more consecutive days, and monthly headache rates.
Telcagepant may also have a place for the treatment of migraine in patients who fail to respond to triptans. A post-hoc analysis of data from a randomized placebo-controlled trial of telcagepant and zolmitriptan suggests that patients responding to telcagepant may be different from those who respond to triptans. 45 Patients who indicated that they had a poor historical response to triptans or who did not take triptans had a substantially better response to telcagepant 300 mg than to zolmitriptan. Conversely, patients who indicated a good historical response to triptans responded better to zolmitriptan than to telcagepant 300 mg and 150 mg. Further research is necessary to confirm or refute these observations.
Conclusions
Calcitonin-gene-related peptide (CGRP) is a key molecule in the pathophysiology of migraine and other primary headaches. Telcagepant antagonises CGRP-induced relaxation, without affecting vascular tone. Although the mechanism by which telcagepant relieves the headache and associated symptoms of migraine is unclear clinical trials confirm comparative efficacy to rizatriptan and zolmitriptan, with better tolerability and in the absence of adverse coronary effects. These attributes position telcagepant as an effective treatment option for patient with migraine who are unable to tolerate triptans or who cardiovascular risk factors or diseases. Elevated transaminases have been reported in a small number of patients receiving telcagepant but none has fulfilled Hy's law criteria, an indicator of the potential for a drug to cause serious liver injury. A Food and Drugs Administration (FDA) review of the results of the clinical trial program is awaited.
Disclosures
This paper is unique and not under consideration by any other publication and has not been published elsewhere. The author has acted as a paid consultant to, and/or her department has received research funding from Addex, Allergan, AstraZeneca, BTG, Endo Pharmaceuticals, GlaxoSmithKline, Menarini, Merck, Pozen, and Unipath. She received no financial support or assistance from Merck in the preparation of this manuscript. The author confirms that the views presented are her own. She has permission to reproduce any copyrighted material.
