Abstract
Background
Bipolar disorder (manic depression) is a serious, long-term mental illness that affects about 1% of adults at some time during their life. It usually develops in late adolescence or early adulthood and affects men and women from all backgrounds. People with bipolar disorder experience wild mood swings that interfere with daily life and damage relationships. They can also have psychotic symptoms: they may see or hear things that are not there. During depressive episodes, affected individuals may feel helpless, worthless, and suicidal.
Treatments for bipolar disorder include drugs to stabilize mood swings (for example, lithium and anticonvulsant medications), antide-pressants to treat depressive episodes and antipsychotic drugs to treat manic episodes. The development of second-generation atypical antipsychotics (SGAs) has increased the hopes of psychiatrists. SGAs, however, cannot be considered a unique pharmacological class since each SGA has many complex pharmacologic actions, only some of which are shared with other SGAs. Even though, many antipsychotics have similar efficacy on average, prescribers may be able to achieve better than average results by considering differences in selecting a specific drug for a specific patient. Clinicians know that each patient is unique and in order to achieve best outcomes for the individual patient, the better therapy is the therapy tailored for the single patient.
Objectives
With this article we provide information on a relatively new antipsychotic aripiprazole released in 2002 by Bristol-Myers Squibb for the treatment of schizophrenia; for acute manic and mixed episodes associated with bipolar disorder in 2004; as an adjunct for major depressive disorder on November 2007; and to treat irritability in children with autism on 2009. Compared with other first line atypical antipsychotics aripiprazole has a unique profile due to partial agonism at dopamine D2 and D3 and serotonin 5-HT1A receptors, and antagonism at 5-HT2A receptors. This paper describes the development of aripiprazole, its unique properties and its metabolically-friendly profile including its receptor binding affinities, pharmacokinetics, central nervous system activity results of clinical efficacy and relevant clinical trials, in particular safety, efficacy and patient acceptability are also examined. The available literature on aripiprazole of the last six years is reviewed.
Introduction
Bipolar disorder is a highly prevalent neuropsychiatric syndrome affecting ~2% to 4% of the general population. Is a lifelong episodic illness requiring long-term treatment. Epidemiologic, comorbidity, cost-of-illness and mortality studies have reported dramatic illness associated morbidity and premature mortality in this population. Many patients have persistent difficulties in social adjustment. In addition, the majority of patients who experience episodes of mania are likely to have relapses and recurrences. During the past decade, several new pharmacologic agents and psychosocial strategies have been reported to be efficacious across various phases of bipolar disorder. 1
The most commonly used pharmacological treatments for acute manic episodes are lithium, valproate and antipsychotics. 2 Although carbamazepine is also indicated for the treatment of mania, its use is generally limited to lithium-refractory or lithium-intolerant patients. 3 The antimanic efficacy of atypical antipsychotics has been shown in a number of controlled clinical trials. In acute mania, atypical antipsychotics show similar efficacy to lithium and may have a faster onset of action and better efficacy in treating psychomotor agitation.4,5 Although multiple drugs are efficacious for the treatment of acute mania, most have noteworthy limitations. In addition to acute adverse events (AEs), some atypical antipsychotics, most notably clozapine and olanzapine, consistently produce weight gain and metabolic abnormalities that may impair medication adherence and increase risk for both diabetes and cardiovascular disease.6,7
Aripiprazole has a novel pharmacological profile: partial agonism at dopamine D2 and D3 and serotonin 5-HT1A receptors, and antagonism at 5-HT2A receptors, 8 has been shown to improve symptoms of acute mania in placebo-controlled studies9,10 and was superior to placebo in maintaining efficacy for up to 100 weeks in patients with bipolar mania. 11
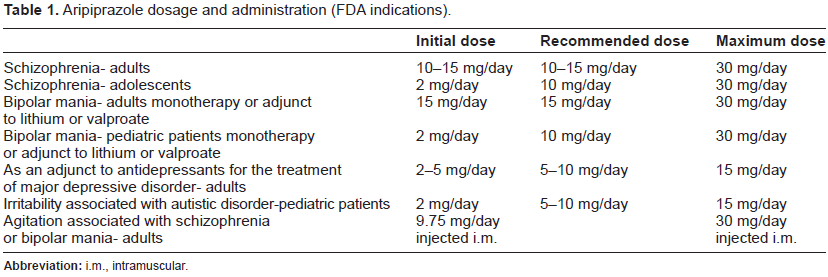
Aripiprazole it's a newer “atypical” or “second-generation” antipsychotic with many noteworthy receptor binding properties that contribute to its unique clinical profile and was approved by the US Food and Drug Administration (FDA) in 2002. Aripiprazole was approved by the United States Food and Drug Administration in November 2002 for the treatment of schizophrenia and subsequently for acute mania or mixed states and then as adjunctive maintenance treatment of bipolar disorder when added to lithium or valproate-treated patients and as adjunctive therapy in major depressive disorder. Finally an intramuscular preparation was approved for acute agitation in schizophrenia and manic phase as well (Table 1). 12 Its introduction was heralded by some as a “third-generation” antipsychotic, as it was the first dopamine partial agonist anti-schizophrenia drug to be marketed. Since then, there has been greater appreciation of the more complex nature of receptor binding affinities, and aripiprazole can also be classified as a medication with significant 5HT2A-antagonism, and with still other additional secondary binding characteristics which may be clinically important in individual patients. 13
Aripiprazole dosage and administration (FDA indications).
Methods
We conducted a PubMed search of all English-language articles published between January 2006 and January 2012 and the internet via Google. The key search term was aripiprazole combined with: bipolar disorder, safety, efficacy, pharmacology, pharmacokinetics, pharmacodynamics, mania, maintenance, patient acceptability, adverse effects. The search was augmented with a manual review of relevant article reference lists. The inclusion criteria were search randomized controlled trial (RCTs) about acute and long-term use of aripiprazole in bipolar disorder in monotherapy. The exclusion criteria were about use of aripiprazole in polytherapy in bipolar disorder and reviews. We found 327 papers, but only three studies are included in this review.
This review presents the available evidence regarding the mechanism of action, pharmacokinetics, clinical efficacy and adverse effects of aripiprazole.
Mechanism of Action
Aripiprazole is the first next generation atypical antipsychotic that is active against positive and negative symptoms of schizophrenia,14,15 has a low propensity for extrapyramidal side effects,14,16 causes minimal weight gain or sedation and produces no elevation in serum prolactin levels15–17 or prolongation of QTc interval on ECG. 15 The mechanism of action of aripiprazole differs from that of currently marketed typical and atypical antipsychotics. Previous preclinical studies have provided evidence that aripiprazole is a dopamine-serotonin system stabilizer with potent partial agonist activity at dopamine D2 and 5-HT1A receptors and antagonist activity at 5-HT2A receptors. 8
Dopamine (D)2 partial agonists (DPAs) activate D2 receptors in a manner that is less than the full agonist dopamine yet more than full antagonists (eg, most known antipsychotics) their novel pharmacologic actions may have important clinical utility in mood disorders and as augmenting agents to reduce the side effects of D2 antagonists. 17
All currently available antipsychotic agents, both conventional and atypical, are dopamine D2 receptor antagonists. Blockade of dopamine receptors in the mesolimbic pathway is thought to mediate antipsychotic efficacy, in particular the ability to decrease positive symptoms. 18 However, D2 receptor blockade in the mesocortical, nigrostriatal, and tuberoinfundibular pathways is correlated with a dysfunctional reward system and increased liability for extrapyramidal symptoms (EPS) and hyperprolactinemia–-unwanted side effects of antipsychotic therapy.
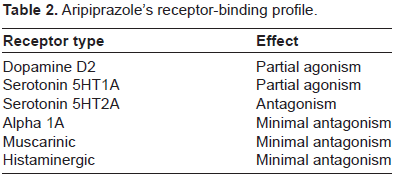
The development of D2 partial agonists is a logical strategy for the treatment of schizophrenia and bipolar disorder because the patho-physiologic mechanism of schizophrenia is thought to be based on too much dopamine activity in some regions of the brain and too little dopamine activity in other regions. A D2 partial agonist can act as a functional antagonist in areas of high levels of dopamine, such as the mesolimbic pathway, but not in areas of normal dopamine levels, such as the nigrostriatal and tuberoinfundibular pathways. 19 The resul is just right AP actions, accompanied by a decrease in negative and cognitive symptoms without the production of motor side effects or prolactin elevation (Table 2).20,21
Aripiprazole's receptor-binding profile.
Pharmacokinetics
The mean time to peak plasma concentration (Cmax) for aripiprazole is 3 h following multiple-dose administration of 10 or 15 mg. 22 The bioavailability of the parent drug is ~85%, and the mean elimination half-life of a single dose of aripiprazole is ~80 h. Based on the half-life, steady state is expected to be achieved by day 14.22,23 Aripiprazole and its active metabolite dehydroaripiprazole has been found to have high plasma protein binding affinity. 22 Elimination of the drug is primarily hepatic, mainly by the CYP3A4 and CYP2D6 enzyme systems. Hence, dosage adjustment of aripiprazole is recommended when it is coadministered with CYP3A4 and CYP2D6 inhibitors and with inducers of CYP3A4. It seems that there is no clear ethnic difference between Asian and Western subjects in terms of mean plasma pharmacokinetics of oral formulations.23,24 Whereas there is limited data on the correlation between dose and plasma concentration, the variability of the pharmacologic active sum of aripiprazole and dehydroaripiprazole has been shown in one study to be ~25%-30% less than aripiprazole alone, suggesting that the pharmacokinetic variability of aripiprazole may be partly determined by metabolism to dehydroaripiprazole. 25 The Cmax of intra-muscular aripiprazole indicated more rapid attainment of plasma concentrations than oral aripiprazole (78% and 5%, respectively). 26 It was found that the Area Under the Curve (AUC) in the first 2 h was 90% higher after intramuscular administration than after oral administration. 26 Pharmacokinetic variability of aripiprazole seems not to be involved with dose and gender differences.25,27
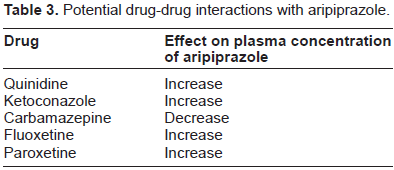
Pharmacokinetics of aripiprazole allows a once daily administration. It has been shown to exhibit a low potential of interaction with other medications but co-administration with inhibitors or inductors of cited hepatic enzymes (eg, paroxetine, fluoxetine and carbamazepine) should be monitored carefully28,29 (Table 3).
Potential drug-drug interactions with aripiprazole.
Pharmacodynamics
It has been proposed that aripiprazole's efficacy in schizophrenia is mediated through a combination of partial agonism (agonism/antagonism) at dopamine D2 and serotonin 5-HT1A receptors and antagonism at serotonin 5-HT2A receptors.8,30 Aripiprazole has demonstrated properties of an agonist in an animal model of dopaminergic hypoactivity and properties of an antagonist in animal models of dopaminergic hyperactivity. Aripiprazole also shows high affinity for dopamine D2 and D3, serotonin 5-HT1A and 5-HT2A receptors and moderate affinity for dopamine D4, serotonin 5-HT2c and 5-HT7, alpha1-adrenergic, histamine H1 receptors and the serotonin reuptake site. Aripiprazole also displays 5-HT1A partial agonism and 5-HT2A antagonist activity in non-clinical studies. 27 The emerging evidence indicates that 5-HT1A activity of antipsychotics may be correlated with the clinical observation of efficacy in the reduction of negative symptoms in patients with schizophrenia. 31 In a recent human positron emission tomography (PET) study, D2 occupancy levels were significantly correlated with plasma drug concentrations, and even the lowest dose (10 mg) led to 85% D2 occupancy. 32 However, it remains unproven in an in vivo setting whether aripiprazole is a D2 partial agonist, or a functionally selective D2 ligand. A recent report proposed that aripiprazole may function mechanistically as a functionally selective ligand, inducing regionally specific differential signaling, rather than as a simple partial agonist. 33 The authors suggested that functional selectivity at D2 receptors, perhaps combined with actions at nondopamine receptors, may contribute to the atypicality of aripiprazole. 33 In contrast, other atypical antipsychotics are shown to behave as ‘pure’ antagonists. As with most agents used to treat psychiatric and neurological disorders, the neurochemical basis for clinical observations cannot be explained at present at this level of detail. It is perhaps a balance of functional selectivity and classical pharmacology that defines the basis of drug action.
It seems likely that the favorable safety and tolerability profile of aripiprazole, including its low incidence of parkinsonian symptoms, lack of prolactin elevation, decreased adrenergic and anticholinergic side effects and decreased weight gain, is also mediated by receptor binding profile.8,30,31
Switching
It is common in clinical practice to switch from an initial antipsychotic to another due to inefficacy or poor tolerability. It is estimated that switching because of suboptimal antipsychotic efficacy or tolerability occurs in 30%-50% of patients a year in outpatient clinics. 34
Switching to aripiprazole from other antipsychotics was well tolerated using all three strategies described in the 8-week study of Casey et al 35 with a 66%-81% completion rate in the three groups. These groups included either immediate initiation of aripiprazole with immediate discontinuation of current antipsychotic therapy (group 1); immediate initiation of aripiprazole while tapering off current antipsychotic therapy (group 2); cross-tritration; uptitration of aripiprazole while tapering off current antipsychotic medication (group 3). The most common reason for discontinuation, worsening schizophrenia, was comparable across all three treatment groups (10% in groups 1 and 2, 8 in group 3). In the study, the most frequently reported AE was insomnia, which was reported by 28%, 32% and 33% of patients in treatment groups 1, 2 and 3, respectively. Notable differences were observed only for nausea and vomiting (less frequent in group 3 than in groups 1 and 2). Most frequently reported AEs were mild to moderate in intensity. Another naturalistically designed 26-week switch study was done 36 in which the patients randomly received either aripiprazole or an atypical antipsychotic standard of care (SOC) treatment (ie, olanzapine, quetiapine or risperidone). This study showed that aripiprazole was significantly superior in terms of patient preference, changes in body weight, prolactin and lipid profiles compared to SOC treatment. The rates of EPS-related AEs for aripiprazole (13.5%) were twice that observed in the SOC treatment group (5.6%). 37 A switching study with an intramuscular acute formulation of aripiprazole 38 examined patient outcomes after treatment with intramuscular aripiprazole or intramuscular haloperidol and subsequent switch to the respective blinded oral formulations for 4 days (aripiprazole 10-15 mg/day, n = 153; haloperidol 7.5-10 mg/day, n = 151). Incidence of EPS-related AEs was lower for aripiprazole (1.3%) than forhaloperidol (8.0%). Nausea and vomiting occurred more frequently in patients receiving aripiprazole (3.9% and 2.6%, respectively) than in those receiving haloperidol. 37
Given these findings, there are no prominent tolerability issues associated with the switch to aripiprazole from other antipsychotics regardless of preexisting psychopatholgy, previous antipsychotics or type of formulation. However, we could not overlook that partial agonism at the D2 receptor may have unintended consequences for some patients already being treated with another antipsychotic, an increased dopamine effect owing to the addition of aripiprazole in patients who have been subject to chronic dopamine blockade, as well as the displacement of lower binding affinity antipsychotics by aripiprazole at the dopamine receptor. In fact, numerous case series reported the worsening of psychosis while tapering off the previous atypical antipsychotics. 38
It may not be appropriate to switch to aripiprazole abruptly. A low dose (5 mg/day) may be preferred if the previous medication is switched because of metabolic issues such as weight gain, dyslipidaemia or hyperprolactinaemia, but the patient is doing well from a psychiatric standpoint. Aripiprazole 10-15 mg/day may be suitable if the change is due to inefficacy or partial efficacy of current treatment. Whatever dose is selected, aripiprazole should be added to the current treatment regimen, and other medications should be maintained for 2-3 weeks while aripiprazole is adjusted to a target dose of 10-30 mg/day and reaches steadystate plasma concentration. 42
Intramuscular formulation
Zimbroff et al. evaluated the efficacy of intramuscular (i.m.) aripiprazole for the treatment of agitation in patients with bipolar I disorder in a trial involving 301 patients. 39 Patients experiencing acute agitation were randomized to receive i.m. aripiprazole 9.75 or 15 mg (1 or 1.5 vials), i.m. lorazepam 2 mg or i.m. placebo. The primary efficacy end point was the change in the PANSS-Excited Component (PEC) score from baseline to 2 h. The PEC consists of five items from the PANSS total scale (hostility, lack of cooperation, excitement, poor impulse control and tension), and each item is scored on a scale of 1 (absent) to 7 (extreme).
Secondary end points included response rates and mean changes in other scores for the evaluation of agitated behavior.
Consistent with the findings in patients with schizophrenia and its related disorders, 40 i.m. aripiprazole was effective in treating agitation in patients with bipolar I disorder. Mean improvements in PEC scores at 2 h were significantly greater with i.m. aripiprazole (9.75 mg, -8.7; 15 mg, -8.7) and i.m. lorazepam (-9.6) than with placebo (-5.8, P value 0.001). Significant differences between active treatment and the placebo groups in mean change in PEC scores were evident by 45 min and 1 h after injection (P < 0.05). Over-sedation (ACES score 8-9) was less frequent with i.m. aripiprazole 9.75 mg (6.7%) and i.m. placebo (6.8%) than with aripiprazole 15 mg (17.3%) and lorazepam (19.1%). Aripiprazole was also effective in reducing agitation according to secondary efficacy measures. 40
Long Term Efficacy in Chronic Bipolar Disorder and Maintenance Studies
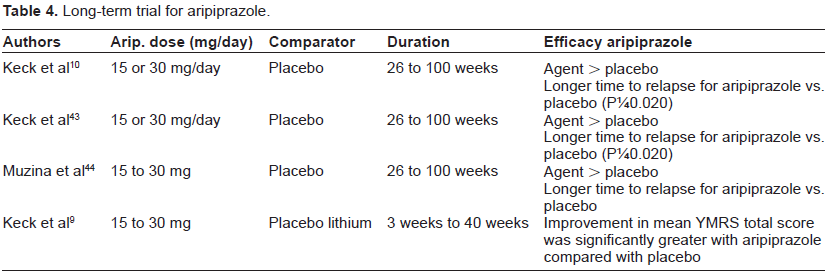
The most recent advances in bipolar treatment concern the European Medicines Agency (EMEA) and the US Food and Drug Administration (FDA) approval of olanzapine, risperidone, quetiapine, ziprasidone and aripiprazole for the treatment of acute mania, the approval of quetiapine and the olanzapine-fluoxetine combination against acute bipolar depression and the approval of olanzapine, quetiapine and aripiprazole for the maintenance phase. This is an important development, because the treatment of BD is as difficult and complex as the illness itself 41 (Table 4).
Long-term trial for aripiprazole.
Once control of manic symptomatology is achieved, the primary goal of pharmacotherapy is to maintain efficacy and to prevent recurrence of symptoms. There are three common ways in which patients may enter long-term or maintenance treatment with aripiprazole.
First, a patient may be initiated on oral aripiprazole plus adjunctive medication, if required. Second, a patient may start on IM aripiprazole. Third, stable and euthymic patients may receive a medication switch to aripiprazole following treatment-emergent side-effects with another atypical agent. 35
Three studies of the efficacy and tolerability of aripiprazole monotherapy for the maintenance treatment of bipolar I disorder were identified: a 26-week, randomized, double-blind study and its 74-week extension phase (for a total of 100 weeks of double-blind treatment),10,43 and a randomized, double-blind comparison of aripiprazole with placebo and lithium (internal comparator) for up to 12 weeks. 9
Overview of current clinical data for aripiprazole in mania.
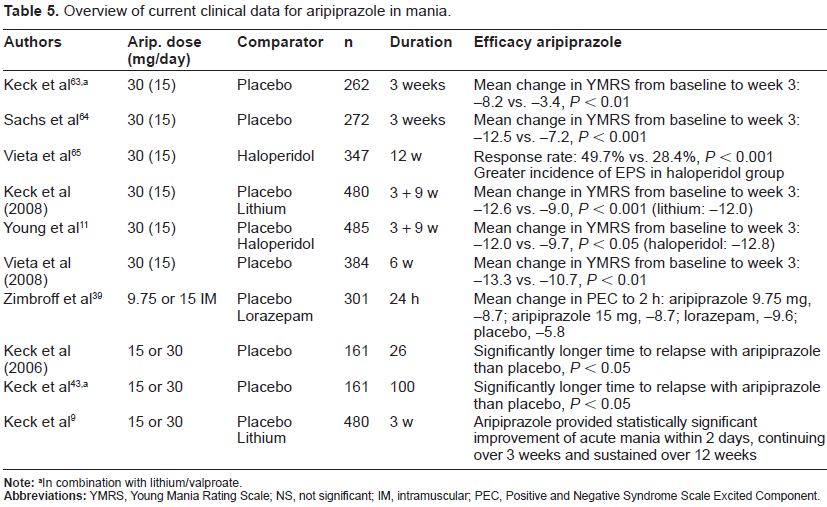
In combination with lithium/valproate.
The clinical study that evaluated long-term efficacy and tolerability of aripiprazole in patients with bipolar mania was composed of an initial period of 26-week treatment with aripiprazole or placebo, followed by a 74-week extension study.43,44 In the first RCT, patients were stabilized with 15-30 mg of aripiprazole for 6-18 weeks and then randomized to a 1:1 ratio to aripiprazole or placebo for an additional 26 weeks. At the end of the study 49% of the placebo group and 72% of the aripiprazole group had not experienced a relapse to a mood episode. The difference was significant only concerning manic relapses. The same was true for the final YMRS score while there was no difference concerning the MADRS score. There was also a superiority of aripiprazole in comparison to placebo concerning the CGI and the PANSS scores. 10 In the 100-week study showed a significantly longer time to relapse with aripiprazole when compared with placebo. Although earlier studies used 15 or 30 mg/day as an efficacious dose, recent data and clinical experience suggest that 15 mg/day may be appropriate for the treatment of mania as well as for maintenance of efficacy long-term. Furthermore, in the 100-week study, where patients could receive 15 or 30 mg/day, 45% of patients were on 15 mg/day at endpoint. Dose adjustment and use of adjunctive medications during long-term therapy can be managed using similar principles as for treatment of manic episode. 43 In terms of long-term safety and tolerability, aripiprazole has a low potential for weight gain, excessive sedation/somnolence and prolactin elevation. 43 Over the 100-week study period, there were no statistically significant differences between placebo- and aripiprazole- treated patients with regard to the change in combined fasting and nonfasting glucose, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, triglycerides or total cholesterol levels, whereas prolactin levels were stable in aripiprazole-treated patients and increased in placebo-treated patients, possibly due to a deterioration of their status. The reductions observed in rating scale scores at endpoint (YMRS total score, Positive and Negative Syndrome Scale total score and cognitive subscale score, Clinical Global Impression for Bipolar Disorder Severity overall score and mania subscale score) were greater in the aripiprazole group than in the placebo group (p value 0.02 for all comparisons).42,43 A post-hoc subgroup analysis of 28 patients (14 under placebo and 14 under aripiprazole) with rapid-cycling bipolar I disorder from the previous maintenance study suggested that aripiprazole was more effective than placebo in the prophylaxis of rapid-cycling patients against manic/mixed episodes. 44 The mean end-point aripiprazole daily dosage was 25.3 mg for the 26-week phase and 23.6 for the 100-week phase. The time to relapse was significantly longer with aripiprazole vs. placebo treatment, marginally at 26 weeks (P = 0.033; HR = 0.21; 95% CI: 0.04-1.03) and clearly at 100 weeks (P = 0.017; HR = 0.18; 95% CI: 0.04-0.88). The YMRS total scores increased in both groups and this increase was numerically smaller with aripiprazole vs. placebo from at week 26 and week. The same hold true for the MADRS total scores which increased in both treatment groups with no statistically significant difference with aripiprazole vs. placebo at week 26 (effect size 0.251) or week 100 (effect size 0.403).44,45
A randomized, double-blind, active- and placebo-controlled study compared the long-term efficacy and tolerability of aripiprazole and lithium monotherapy in the acute and maintenance treatment of bipolar I disorder. 9 Patients with acute bipolar mania or mixed states who required hospitalization were randomized to receive aripiprazole 15 to 30 mg/d, lithium 900 to 1500 mg/d, or placebo for 3 weeks. At the completion of 3 weeks of double-blind therapy, those who had been randomized to receive placebo were blindly switched to aripiprazole, whereas aripiprazole- and lithium-treated patients continued the assigned regimens. All patients continued double-blind treatment to week 12, at which time they could enter a 40-week, double-blind extension phase. Improvement in mean YMRS total score was significantly greater with aripiprazole compared with placebo from baseline to day 2 (-4.3 vs. -2.8, respectively; P = 0.003) and up to week 3 (-12.6 vs. -9.0; P < 0.001). Lithium was associated with significant improvement in YMRS total score compared with placebo at week 3 (-12.0 vs. -9.0, respectively; P = 0.005). 9
The available evidence supports the efficacy and tolerability of aripiprazole in the maintenance treatment of bipolar disorder. Aripiprazole has not been found to adversely affect body composition or metabolic parameters in most (>70%) treated individuals.
In conclusion, both short and long-term controlled clinical trials or in switch studies involving aripiprazole demonstrated improvement in positive in the maintenance of bipolar disorder. Aripirazole's efficacy in clinical trials shows consistency with his unique pharmacological features.
Safety
The common side effects during aripiprazole treatment include akathisia, tremor, headache, dizziness, somnolence, sedation fatigue, nausea, vomiting, dyspepsia, constipation, light-headedness, insomnia, restlessness, sleepiness, anxiety, hypersalivation and blurred vision.
The uncommon side effects, and those whose frequency is not precisely known, include uncontrollable twitching or jerking movements, seizures, weight gain, orthostatic hypotension or tachycardia, allergic reaction (such as swelling in the mouth or throat, itching, rash), speech disorder, agitation, fainting, transaminasaemia, pancreatitis, muscle pain, stiffness, or cramps and very rarely neuroleptic malignant syndrome and tardive dyskinaesia. Neuroleptic malignant syndrome (NMS) is a rare and potentially life-threatening neurologic emergency commonly associated with the use of antipsychotic medications. NMS refers to the combination of hyperthermia, rigidity, and autonomic dysregulation that can occur as a serious complication of the use of antipsychotic drugs. As a class, atypical antipsychotic agents have lower associated rates of extrapyramidal symptoms and are less likely to induce NMS as compared to conventional antipsy-chotics, but there are some case reports about NMS by aripiprazole.46–48
Elderly patients with psychosis associated with Alzheimer's disease and treated with aripiprazole are at increased risk of death compared to placebo due to cardiovascular (for example, heart failure, sudden death) or infectious (for example, pneumonia) problems or stroke. 49
Marder et al. 50 assessed the occurrence of EPSs from spontaneous reports and by using the Simpson-Angus Scale (SAS), Abnormal Involuntary Movement Scale (AIMS), and Barnes Akathisia Scale (BAS). In the study of De Leon et al. 30 spontaneously reported EPS-related events consisted of dystonic, parkinsonian, akathisic, dyskinetic, and residual events. No significant differences were observed between patients receiving any dose of aripiprazole and placebo regarding the incidence of EPS-related events (21.1% vs. 19.4%, respectively) or extrapyramidal syndrome (6.0% vs. 5.8%, respectively). EPS-related events were significantly higher in the haloperidol group compared with placebo (43.5%, P < 0.001). Mean changes at end point in SAS score from baseline were significantly higher in the haloperidol group (n = 183) than in the placebo group (n = 402) (P 0.001).30,50
Aripiprazole appears to be associated with minimal overall weight gain. In the meta-analysis by Marder et al. 50 patients in the aripiprazole 20-mg/d, 30-mg/d, and combined aripiprazole groups showed a significant but minimal mean increase in body weight from baseline (<1 kg) compared with placebo (P 2 0.01). A 26-week, double-blind, randomized, multi-center study in 317 patients compared the long-term effects of aripiprazole (15-30 mg/d) versus olanzapine (10-20 mg/d) on body weight. Clinically significant weight gain, defined as 7% increase in body weight was observed in more patients receiving olanzapine throughout the study. At end point (week 26), patients receiving olanzapine exhibited a mean weight increase of 4.23 kg versus a mean weight decrease of 1.37 kg in the aripiprazole group (P < 0.001).51,55
Registered placebo-controlled, clinical trial data 52 show that discontinuation rates of aripiprazole treatment in an adult population owing to adverse events (AEs) among approved indications vary from 6% to 12% (schizophrenia 7%; bipolar mania 11% for mono-therapy and 12 for adjunct therapy; MDD 6%; agitation associated with schizophrenia or bipolar mania (i.m. injection) 0.8%), compared to 2%-10% in cohorts receiving placebo (schizophrenia 9%; bipolar mania 10% for monotherapy and 6 for adjunct therapy; MDD 2%; agitation associated with schizophrenia or bipolar mania 0.5%). Similar rates of discontinuation owing to AEs have been observed in pediatric populations in registration studies for aripiprazole (schizophrenia 5% versus placebo 2%; bipolar mania 7% versus placebo 2%).
Adverse events occurring at an incidence of ≥2% and at least twice that for placebo across approved indications for adult population included akathisia, anxiety, sedation, restlessness, tremor, EPS, blurred vision, pain in the extremities, myalgia, muscle spasm, vomiting, dry mouth, hypersalivation, weight gain, dizziness and insomnia. The most common AE associated with aripiprazole (>5% in the aripiprazole-treated groups and a combined incidence at least twice that for placebo) was akathisia. Adverse events occurring at ≥5% in aripiprazole-treated subjects or placebo-treated groups across several indications for pediatric population include somnolence, EPS, fatigue, nausea, akathisia, tremor, blurred vision, hypersalivation, dizziness, nasopharyngitis and agitation. The most common AEs associated with aripiprazole (>5% in aripiprazole-treated groups and a combined incidence at least twice that for placebo) were EPS and somnolence. 53
Adverse event data from published studies in the elderly population are comparable to data from the adult studies. In randomized controlled trials (two flexible dose and one fixed dose study) 54 of dementia-related psychosis, aripiprazole was generally well tolerated. Nevertheless, there was an increased incidence of cerebrovascular AEs (eg, stroke, transient ischemic attack), including fatalities, in aripiprazole-treated subjects (n = 8/597 versus placebo n = 2/343). Similar risks have been associated with the entire class of atypical antipsychotics that also have black box warnings related to use in elderly patient populations.
The AEs reported in the 26-week, double-blind trial 10 comparing aripiprazole and placebo in adult patients with schizophrenia were generally consistent with those reported in the short-term, placebo-controlled trials. Adverse events occurring in ≥5% of the aripiprazole-treated subjects with an incidence at least twice that of placebo were tremor, insomnia, akathisia, vomiting and nausea (tremor was the most distinguishable between the two groups; 8% for aripiprazole versus 2 for placebo).
In common with second generation antipsychotics, in some studies aripiprazole shows that have less probability to cause acute extrapyramidal symptoms (EPS), tardive dyskinesia or raise prolactin levels above those seen in patients taking a placebo. In recent years the main side effects of second generation antipsychotics has been weight gain.
Evidence from clinical trials clearly shows that aripiprazole is weight neutral, 56 presumably because of aripiprazole's weak affinity for histamine H1 and adrenergic α1 receptors.
In other studies aripiprazole does not cause sedation because it has a relatively low affinity for H1 receptors, but other studies found these side effects. In the early acute phase of schizophrenia this may be considered disadvantageous because the sedating properties of a medicine can be useful in managing behavioural disturbance. However, in the medium to long term this effect may be advantageous, as many patients find the sedative side effects of antipsychotic medication a barrier to recovery. 57
A minority of patients taking aripiprazole have experienced an akathesia-like syndrome, nausea, and/or vomiting in the first one to two weeks of treatment. Good practice is to advise patients of the risks of these adverse effects occurring, and the likelihood that they will disappear within the first few weeks of treatment (as is common when initiating antidepressants).57 Some case reports describe acute dystonia followind a single dose of aripiprazole.58,59
In conclusion, aripiprazole seems to have less probability to experience AEs, but as SGA cause in any cause some adverse effects and in some cases very severe.
Patient Acceptability
There is a growing recognition of the importance of patient satisfaction in evaluating pharmacological and psychological treatments. 56 To date relatively few trials evaluating drug treatments have examined patients’ satisfaction with medication. In an eight-week naturalistic study of 833 patients with schizophrenia in 14 European countries, 68 per cent of people treated with aripiprazole and 66 per cent of caregivers rated aripiprazole as ‘slightly better’ or ‘much better’ than prior antipsychotic therapy. 60 It would inform clinical practice if future studies considered the patient subject experience as an important outcome variable, using a validated tool such as the Satisfaction with Antipsychotic Medication Scale. 61
In the 26-week study by Pigott et al. 54 comparing aripiprazole with placebo, aripiprazole was well tolerated, with no evidence of marked sedation, hyper-prolactinemia, or prolonged QTc. Extrapyramidal symptoms were comparable in the aripiprazole and placebo groups. Modest mean weight loss at endpoint was evident in both groups.
In the 52-week, open-label, randomized extension 62 more patients receiving olanzapine than patients receiving aripiprazole experienced significant weight gain (at least 7% increase in weight) at all measured time points, as well as greater mean change and mean percentage change from baseline in weight.
In the clinical trials comparing aripiprazole with placebo among patients with bipolar disorder, aripiprazole appeared to be well tolerated. In the study reported by Keck et al. 63 there were no differences in discontinuation rates between aripiprazole and placebo attributable to adverse events. However, gastrointestinal (GI) adverse events including nausea, dyspepsia, vomiting, and constipation, were more common with aripiprazole than with placebo. Somnolence was reported in 20% of patients receiving aripiprazole and in 5% of patients on placebo. Anxiety was reported in 18% of aripiprazole patients and in 10% of patients randomized to placebo. Akathisia was reported in 11% of the patients receiving aripiprazole, and in 3% of patients assigned to placebo. Changes from baseline on the scales measuring extrapyramidal effects, including akathisia, were small, but statistically greater among those receiving aripiprazole than placebo. No problems were identified for body-weight, prolactin elevation, QTc prolongation, vital signs, or laboratory analyses. Similar tolerability outcomes were seen in the study report from Sachs et al; 64 however, the incidence of treatment emergent akathisia was higher at 17.6% for aripiprazole and 4.5% for placebo. A significant difference was observed between the two groups at endpoint in mean change from baseline in akathisia rating scores. At endpoint, there was no significant difference between aripiprazole and placebo in the number of patients with clinically significant akathisia rating scale scores. The spontaneous reports of akathisia among the patients receiving aripiprazole were generally mild to moderate in severity and primarily occurred early in treatment, and rarely led to discontinuations or reduction in dose. The report by Sachs et al 64 also described the time course of somnolence, nausea, dyspepsia, and constipation as being limited mainly to the first week of treatment.
In the 26-week maintenance phase of the double-blind study comparing aripiprazole with placebo, 10 there were no adverse events in the aripiprazole group with at least a 10% incidence and twice the incidence of the placebo group. Treatment emergent adverse events reported by patients receiving aripiprazole at an incidence of at least 5% and twice the incidence of placebo were tremor, akathisia, vaginitis, and pain in the extremities. The analysis of movement disorder scales demonstrated no significant differences between aripiprazole and placebo. There was a statistically significant difference in favor of aripiprazole in mean change from randomization to endpoint in prolactin level. Although the incidence of elevated creatine phosphokinase (CPK) was slightly higher in patients receiving aripiprazole (6.76% vs. 4.11%) and there were 5 patients receiving aripiprazole with potentially clinically significant elevated CPK levels, none of these patients had associated symptoms consistent with neuroleptic malignant syndrome. There were no problems identified regarding QTc, vital signs, weight, or physical examinations.
In the comparison of aripiprazole and haloperidol, 65 the most frequent treatment-emergent adverse events were extrapyramidal syndrome, akathisia, and depression for haloperidol (35.5%, 23.1%, and 14.2% respectively), and insomnia, akathisia, and depression for aripiprazole (13.7%, 11.4%, and 11.4% respectively). It should be emphasized that anticholinergic agents were not permitted for symptomatic or prophylactic treatment of extrapyramidal symptoms during the study, contributing to the rates of extrapyramidal syndrome reported in the study and contributing to the high discontinuation rate for patients receiving haloperidol. Other differences between aripiprazole and haloperidol include serum prolactin elevations (mean decrease for aripiprazole but a mean increase for haloperidol). No differences were seen for mean change in bodyweight. There were no differences observed in QTc, vital signs, or laboratory measures.
In summary, aripiprazole appears to be well tolerated in patients with bipolar disorder. Although akathisia and GI complaints emerged at the start of treatment in some patients, GI symptoms in particular may be time-limited in many instances. Haloperidol was associated with greater rates of extrapyramidal symptoms than aripiprazole. There is a lack of comparative safety data with other second-generation antipsychotics among patients with bipolar disorder. 13
In a very recent head-to-head comparisons of metabolic side effects of second-generation antipsychotics (amisulpride, aripiprazole, clozapine, olanzapine, quetiapine, risperidone, sertindole, ziprasidone, zotepine) in randomized controlled trials weight change was chosen as the primary outcome, glucose and cholesterol changes as secondary outcomes.66,67 These studies found that aripiprazole was associated with lower weight gain, less elevation of glucose, and with the least effects on the lipid status (together with ziprasidone).
Patients prefer antipsychotics with a low side effect burden, and sometimes value side effects over efficacy for psychosis. Most clinicians prioritize efficacy for psychosis, and try to find the most efficacious agent that a patient can tolerate. This sometimes creates a lack of therapeutic alignment between patient and prescriber. 34
Caution in Prescribing
However there are some case studies that report some adverse effects of dopamine partial agonist. A letter to editor68,69 reports worsened agitation (paranoia and anger) with aripiprazole when it was added to a stable doses of antipsychotic medication. It is possible that in the context of postsynaptic dopamine receptor up-regulation (in response to chronic dopamine blockade), aripiprazole's agonist effects may enhance dopamine neurotransmission in limbic areas, resulting in intensification of psychosis. Aripiprazole initiation may produce overactivation of D2 receptors, which might worsen a patient's condition. Pae et al 70 aimed to find predictors of worsening psychosis with aripiprazole in patients whose previous antipsychotic therapy was immediately discontinued. They found lower baseline disease severity was associated with significant worsening during the first month of aripiprazole treatment. Other case reports describe the links between aripiprazole and the onset or worsening of compulsive behaviours, such pathological gambling and compulsive eating,71–73 hypersexuality 74 and compulsive shopping.75,76 The emergence of these unexpected changes in behaviour are attributable to the initiation or continuation of aripiprazole, relating to reductions in impulse control. It's possible that these behaviours are reward based and are reinforced by dopaminergic stimulation via the mesolimbic tract and involve the dopamine 3 (D3) receptor. The present cases suggests that clinicians should ask about compulsive behaviour with this antipsychotic, because patients may be reluctant to disclose this voluntarily. A review in 2006 75 estimates the extent of under-reporting of adverse drug reactions (ADRs) to spontaneous reporting systems and to investigate whether there are differences between different types of ADRs. The median under-reporting rate across the 37 studies was 94% (interquartile range 82%-98%). The statements suggesting that aripiprazole does not cause certain symptoms needs to be tempered.
Conclusion
A number of double-blind, randomized, controlled trials have confirmed the clinical efficacy of aripiprazole in bipolar disorder and schizophrenia. Aripiprazole is the prototype of the ‘third generation’ atypical antipsychotics, or dopamine-serotonin-stabilizers and is characterized by a relatively low risk of inducing metabolic adverse effects, causing sedation and other side effects such as hyperprolactinemia. As a partial agonist at dopamine D2 receptors, aripiprazole acts as a functional antagonist in the mesolimbic dopamine pathway, where excessive dopamine activity is thought to cause positive symptoms, but acts as a functional agonist activity in the mesocortical pathway, where reduced dopamine activity is thought to be associated with negative symptoms and cognitive impairment. 78
Aripiprazole is established as efficacious in the maintenance treatment of BD on the basis of mono-therapy and adjunctive data.
Compared with typical antipsychotics, aripiprazole presents a lower risk of akathisia, and compared with atypical antipsychotics, lower risk of increased prolactin levels and QTc interval prolongation. A consistent finding with aripiprazole is minimal change in body composition and metabolic parameters. Moreover, prolactin elevation is not encountered, nor is there any evidence of mood destabilization. Aripiprazole is associated with EPS (notably akathisia) which is usually mild and not treatment limiting and in rare case may cause neuroleptic malignat syndrome.
Aripiprazole has proven to be an effective medication for the acute treatment of manic and mixed episodes, as well as for the prophylactic–-maintenance phase of bipolar disorder in patients recovering from a manic/mixed episode. Choosing the appropriate dosing and tapering strategy, addressing the side effects, controlling withdrawal symptoms from previous medications and using adjunctive medications when necessary are key to successful treatment with aripiprazole. 79
Aripiprazole is one of the available treatments for the manic phase and prophylaxis of bipolar disorder, with relatively low potential to cause adverse events in terms of physical health. Conversely, so far we have only negative evidences–-in randomized controlled trials–-of the efficacy of aripiprazole for bipolar depression. When the clinician decides to use aripiprazole and agitation is present, initiation of the treatment of a manic episode should start with i.m. injection.
Aripiprazole oral, at a starting dose of 15 mg/day, may also be considered but in this case the use of an adjunctive medication, such as lorazepam and/or a classic mood stabilizer, is often necessary. Adjunctive medications may also be of value for anxiety or insomnia. When switching to aripiprazole in a stable patient with side effects from another medication, it is often recommended–-whenever possible–-to decrease the first medication very slowly and to initiate aripiprazole at a low dose (eg, 5 mg/day) if the switch is due to tolerability issues or medium dose (eg, 10-15 mg/day) if the switch is due to partial or lack of efficacy. Again, adjunctive medications (eg, an antihistamine agent, to prevent histaminergic rebound) may play an important role in this scenario. Bipolar Disorder is usually treated with polypharmacy; combination studies with extended maintenance phases with lithium and possibly anti-depressants would be interesting for future research. It's important to pay attention to this issue in the prescribing of aripiprazole in combination to other drugs. Aripiprazole has also received approval for the treatment of bipolar disorder as adjunctive therapy or monotherapy (manic or mixed episodes) as well as an augmentation therapy of major depressive disorder (MDD) by the US FDA. The overall safety and tolerability of aripiprazole is favorable compared to other atypical antipsychotics across the approved indications. Aripiprazole showed a minimal propensity for clinically significant weight gain and metabolic disruption. However, extrapyramidal side effects, such as akathisia, are reported and may limit its clinical use in some cases, particularly in patients with bipolar disorder and MDD. Although studies to date have demonstrated the potential for aripiprazole in the acute treatment phase and maintenance treatment of BPAD, additional data are needed to better clarify its role in the treatment of this disabling disorder, particularly in the maintenance phase. Due to the lesser incidence of metabolic side effects, aripiprazole is an attractive alternative to olanzapine, yet there are no head-to-head trials comparing either these two agents or aripiprazole to other atypical agents used in BPAD such as quetiapine or risperidone. Long-term maintenance studies comparing aripiprazole to lamotrigine and carbamazepineare also lacking. Additionally, no studies have been done comparing aripiprazole in combination with a mood stabilizer with two primary mood stabilizers such as lithium and valproate or carbamazepine. No research has examined its role in BPAD II.
In conclusion, aripiprazole has a less severe side effect profile compared to the medications it is compared against, but the fact remains that it still has side effects, many of which may be underreported, some of which are lethal (eg, neuroleptic malignant syndrome), that need to be monitored. The benefits of the medication must be couched by its side effects. Further studies are needed in order to replicate and validate these results and determine the effectiveness of aripiprazole in clinical practice.
Author Contributions
Conceived and designed the experiments: MPR. Analysed the data: MPR. Wrote the first draft of the manuscript: MPR. Contributed to the writing of the manuscript: MPR. Agree with manuscript results and conclusions: MPR. Jointly developed the structure and arguments for the paper: MPR. Made critical revisions and approved final version: MPR.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
