Abstract
Diabetes is associated with numerous complications. One of the most debilitating microvascular sequelae is painful diabetic peripheral neuropathy (PDPN). PDPN results from a multi-faceted pathogenesis involving direct axonal degeneration; free radical mediated cellular apoptosis, and microvascular perfusion abnormalities. While tight glycemic control has been shown to modulate the history of this diabetic complication, practicing clinicians have access to numerous published practice recommendations for treatment. Of the frequently utilized medication classes, anticonvulsants, antidepressants, anesthetics, and the neuromodulators are perhaps the most widely understood. The gabapentinoids are considered by many as first line therapy. Others recommend tricyclic antidepressants first-line. The provider treating PDPN must consider the medication side effects and monitoring parameters, the co-morbid disease states, and the ultimate effects on diabetic control. Finally, the clinician must address patient expectations of treatment. Goals may include increased functionality, decreased pain and improved sleep. Here multiple treatment modalities and evidence-based guidelines are reviewed.
Keywords
Introduction
The diagnosis of painful diabetic peripheral neuropathy (PDPN) comprises a subgroup of diabetic patients. It is estimated that there are 246 million people with diabetes mellitus worldwide. Of those individuals, approximately 10%-20% will develop PDPN over the course of their disease. This risk increases with chronicity of disease and poor glycemic control. Descriptive wording of this condition frequently incorporates adjectives such as lancing and burning pain. The impact on an individual's functionality, quality of life and sleep can therefore be devastating. Treatment goals target increasing functionality, quality of life, and sleep, while decreasing pain. This article reviews treatment options, recent guidelines from around the globe, and future direction for research.
Pathophysiology of Painful Diabetic Peripheral Neuropathy and implications for therapy
The pathophysiology of PDPN reflects an imbalance of the biochemical effects of glucose control. Large afferent nerves transmit information on proprioception, temperature and vibration. Small afferent fibers transmit information on nociception, touch and warmth sensation. In general, neuropathic pain results from damage at the level of the primary afferent nerves. Second and third order neurons become sensitized, recruited, and hypervigilant with central sensitization. As a result of the primary afferent damage peripheral sensitization arises due to axonal degeneration, sprouting, and Wallerian degeneration. Several identified risk factors exist. See Table 1.
Risk Factors for PDPN development. 113
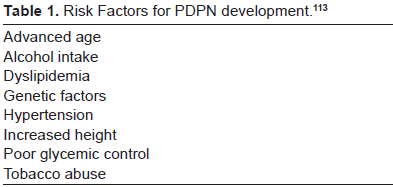
The neurotoxic effects of these factors are theorized to be a result of accumulation of glycosolated products affecting neuronal integrity and interfering with neuronal repair. Furthermore, excess glucose is converted to sucrose and fructose through aldose pathways with accumulation resulting in alteration of sodium and potassium adenosine triphospate channels impairing nerve conduction and breaking down neuronal axons.
Chronic hyperglycemia results in oxidative stress with increased production of free radicals. This causes direct vascular damage and nerve ischemia that leads to progressive nerve damage. There are multiple types of diabetic associated neuropathies–-autonomic with orthostasis, gastroparesis, and peripheral sensation alone without pain. Although there may be some overlap, this article focuses on PDPN.
Diagnosis
The diagnosis of PDPN often lags behind the onset of symptoms. This reflects an abnormality in pain signal generation, propagation and perception. Neuropathic pain may be experienced in many ways. Frequent descriptors include burning, tingling, scratching, or stabbing; an atypical sensation of hot or cold, dull and aching; or a sudden sharp, lightning bolt, pain. And finally, allodynia, or a painful response to a normally non-noxious stimulus, such as bedsheets or socks, is another well-known presentation of PDPN. Symptoms classically progress in a symmetric pattern from the distal extremity proximally and tend to result in the classic “stocking and glove” distribution pattern.
Key sensory information can be obtained via simple testing with a 10 g monofilament. Failure to feel the monofilament pressed over the sole of the foot can be diagnostic of neuropathy. Decreased vibratory sense can be tested with a 128 Hz tuning fork placed against the metatarsal joint. Extinguished sensation when compared to the upper extremity confirms sensory damage. Physical skin changes of neuropathy incorporate thickened cracked skin, dystrophic nails, calluses and abnormal wear signs to the skin, such as skin breakdown or open sore development. These are not unique to neuropathic disease however and careful examination of the individual's vascular status is warranted. Additionally, several validated screening tools have been developed to assist the clinician in distinguishing between nociceptive versus neuropathic pain in the face of diabetes.
In 2011, the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain released new assessment guidelines for neuropathic pain, updating the 2004 version. Their recommendations included incorporating a screening tool into the history process. Following this, a standardized clinical exam should be performed to capture the 10%-20% of individuals that may not be found by history alone. These clinical exams may include touch, temperature, vibration and pain assessments. More quantitative sensory testing can be performed but outcomes for neuropathic pain are variable and this is an area ripe area for future research. Specifically, recommended are visual analog scales or neuropathic rating scales to assess pain intensity. For treatment, the recommendation is to use patient's global impression of change or the clinician's global impression of change with the measures of 30% reduction and 50% reduction being the standard for numbers needed to treat calculation. Measurement of secondary outcomes such as mood, sleep, functionality, and quality of life is also recommended by the IMMPACT (Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials) group. 1 Consequences of PDPN include foot ulceration and amputation as a result of abnormal sensory response.
Current Pharmacotherapy
Treatment
The pharmacological management of PDPN is complex as is illustrated by the unique pathophysiology of this neuropathic pain syndrome described earlier in this manuscript. While there are common links of pathogenesis to other neuropathic syndromes, there additionally exists a role of potential hypoperfusion, opening the possibility of unorthodox treatments that may not be efficacious for other neuropathic pain problems.2,3
Currently, only duloxetine and pregabalin are Food and Drug Administration (FDA) approved for the treatment of PDPN. Many other pharmaco-therapeutic modalities have proven useful in well-designed randomized controlled trials. 4 Largely these agents are included within the classifications of antidepressants, anticonvulsants, antipsychotics, anesthetics/antiarrhythmics, vasodilators, anti-dementia agents, and opioids. Presented here are available options, clinical pearls, and level of evidence for these possible treatments organized by pharmacologic category.
Antidepressants
Perhaps no other class of medications has received as much attention, research funding, and use for PDPN as the antidepressants.5–7 While antidepressants may be classified in a variety of ways, we will use the classifications of selective serotonin reuptake inhibitors (SSRI), serotonin-norepinephrine reuptake inhibitors (SNRI), tricyclic antidepressants (TCA), and atypical antidepressants (those which do not conveniently fall within one of these groups).
Antidepressants largely exert their activity via modulation of serotonin, norepinephrine, and dopamine.8,9 It has been hypothesized that, based on the understanding of the descending inhibitory pain pathways, a component of at least two of the three of these neurotransmitter systems must be involved to exhibit a clinically meaningful response in pain reduction for PDPN.10,11 While this has yet to be fully elucidated, available clinical trial data does support this theory.
The TCA antidepressants (Table 2) have long been used for the treatment of PDPN and are especially useful in those patients with concurrent insomnia. 5 These agents have varying impact in the reuptake inhibition of both serotonin and norepinephrine at the termination of first order primary afferent neurons. Amitriptyline is by far the most widely used of this class, largely due to provider familiarity and cost. However, TCA antidepressants may further be classified into their chemical classification of tertiary or secondary amines. Interestingly, the previous “go-to” TCA antidepressants, amitriptyline and imipramine, actually undergo demethylation to their active metabolites, nortriptyline and desipramine, respectively. 12 This has real world clinical implications as secondary amines (nortriptyline and desipramine) exhibit less postural hypotension and other central nervous system and anti-cholinergic mediated side effects, yet maintain their clinical efficacy.13–15 Classification of TCA antidepressants as either tertiary or secondary amines is presented in Table 2.
Tricyclic antidepressants.
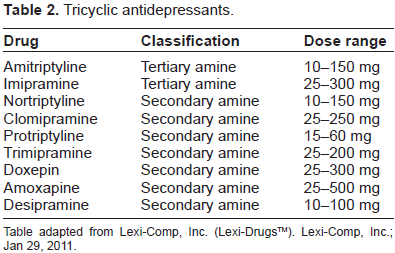
Table adapted from Lexi-Comp, Inc. (Lexi-Drugs™). Lexi-Comp, Inc.; Jan 29, 2011.
As depression and anxiety are oftentimes experienced by patients with chronic illness, especially pain and diabetes, it is important to note that TCA antidepressants do possess antidepressant and anxiolytic properties which may make them useful tools in the treatment of concurrent pain and psychiatric illness.16,17 A common misconception, however, is the dosing necessary to show efficacy for the symptoms of neuropathic pain, depression, or anxiety. Typically doses start as low as available for the TCA medications (for example 10 mg at bedtime) and are titrated to effect weekly by doses of 10-25 mg. The dose-response curve will begin to plateau between 100 mg and 150 mg daily for most of these agents. Yet, data suggests that antidepressant and anxiolytic effects of TCA antidepressants are not usually experienced until daily doses reach the 75 mg to 100 mg range for most agents.18,19 Some disagreement exists regarding the necessity of baseline and periodic electrocardiogram monitoring for patients beginning TCA antide-pressant therapy for PDPN. Judicious review of other potential QTc-prolonging drugs or a family history of Long QT syndrome is warranted. 20
The SSRI antidepressants have largely been disappointing in PDPN clinical trials with the exception of paroxetine and citalopram.21–23 Unfortunately, the level of evidence to support these agents raises concern about their mainstream use over more well-studied options. With the difficulty to differentiate symptom improvement of pain versus symptom improvement of depression or anxiety, it is questionable if these agents truly have an evidence-based role in the treatment of PDPN. Perhaps as we learn more about these individual agents’ pharmacology and the pathogenesis of PDPN, their role will become clearer.
In contrast with the SSRI antidepressants, the SNRI antidepressants have proven unequivocally effective for the treatment of PDPN.24–27 Unlike the TCA antidepressants, this class of agents provide analgesia for neuropathic pain symptoms, as well as antidepressant and anxiolytic effects, at similar dosage ranges. This enables the provider to treat these commonly co-morbid conditions simultaneously, without the burden of either the dose-related side effects of the TCA antidepressants or the addition of an additional antidepressant. While the SNRI antidepressants do possess serotonin reuptake inhibition as previously discussed, the norepinephrine reuptake activity is either equal to, or surpasses that of the serotonin modulation. 28 Unlike the other SNRI antidepressants, venlafaxine appears to exhibit more selectivity for serotonin at doses less than or equal to 75 mg daily and loses this selectivity as the dosage increases. 29 Duloxetine, the first FDA approved treatment for PDPN, is still under patent protection and the cost may be a consideration as many third party payers are requiring failure of either venlafaxine or a TCA antidepressant prior to approving reimbursement for this agent. Of particular interest, the SNRI antidepressants may cause elevations in blood pressure directly proportional to the noradrenergic activity they possess. This phenomenon appears to be more clinically relevant with the titration of venlafaxine to higher dosages which require consideration and monitoring. 30 Duloxetine may also result in modest blood pressure elevations, clinically insignificant increases in blood glucose, as well as clinically significant elevations in liver transaminases. 31 While the prescribing information for this drug recommends monitoring for this adverse effect of duloxetine, no clear guidelines on frequency of monitoring exist. The SNRI antidepressants, along with their dosage ranges are provided in Table 3.
Serotonin norepinephrine reuptake inhbitors.

Table adapted from Lexi-Comp, Inc. (Lexi-Drugs™). Lexi-Comp, Inc.; Jan 29, 2011.
The atypical antidepressants include trazodone, bupropion, mirtazapine, and vilazodone. Each of these agents possesses its own unique pharmacodynamic profile with varying levels of evidence for efficacy in the treatment of PDPN.32–35 Trazodone, which is frequently utilized in low doses for insomnia and higher doses for refractory depression, has a primary mechanism of action of serotonin modulation, which should be considered when using with other serotonin active agents. Of the atypicals, bupropion has perhaps the most robust available data in the treatment of PDPN. While effective as an antidepressant at similar doses used in neuropathic pain, some dose-related side effects warrant mention. Bupropion is a stimulating, or “activating”, antidepressant which results in significant insomnia when dosed later than 4:00 or 5:00 P.M. Additionally, due to the largely dopaminergic and noradrenergic activity of the drug, an increased risk of seizures should preclude its use in those with epilepsy or taking other drugs known to lower seizure threshold. 36 Compared to other antide-pressants, bupropion has some attractive attributes in the patient with diabetes, namely a low incidence of sexual side effects and a neutral to slightly negative weight profile. 37
In contrast with bupropion, mirtazapine possesses noradrenergic, serotonergic, and histaminergic activity. Interestingly, mirtazapine may additionally possess a small binding affinity to the mu-opioid receptor. These mechanisms of action result in a pharmacodynamic profile of weight gain and sedation. While evidence exists to support mirtazapine's use as an adjuvant analgesic in the treatment of PDPN, the side effect profile largely limits its use in the patient with diabetes. 38
Nefazodone has largely been abandoned in clinical practice today due to liver toxicity and significant drug interactions. 39 This, plus sporadic availability and only anecdotal reports of effectiveness in PDPN, should preclude its use altogether. 40
Vilazodone is a newer antidepressant with a similar chemical structure to trazodone, although pharmacologically it acts as both a serotonin reuptake inhibitor as well as a serotonin partial agonist. 41 Currently no data supports its use in PDPN nor are any clinical trials underway.
It should be noted, even when initiating antidepres-sant medications for PDPN, that appropriate assessment and follow-up is crucial. All of the medications discussed above may increase suicidal ideation. Therefore, consider a practice of two to three week follow-up with patients initiated on these agents. 42
Anticonvulsants
The anticonvulsants have more recently become considered first-line for the treatment of PDPN, largely due to a reduced side effect profile compared to antidepressants and the generic availability of gabapentin. 4 Anticonvulsants, as a class, typically possess more drug interactions, with the exception of the gabapentinoids (pregabalin and gabapentin), compared to their antidepressant counterparts. Anticonvulsants are largely classified as either first generation or second generation, with classification dependent more on when the medication was introduced versus true differences in pharmacodynamics or pharmacokinetics.43,44
Of the first generation anticonvulsants, none are FDA approved for the treatment of PDPN, however a tremendous amount of well-controlled studies exist to support their use individually. These medications include carbamazepine and valproic acid salts.45,46 Although anecdotal evidence exists to support the use of ethosuximide and phenytoin, given their side effect profiles these agents are rarely used. Carbamazepine, the first anticonvulsant to receive FDA approval for a neuropathic pain syndrome (trigeminal neuralgia or Tic Delareux), is a potent sodium channel antagonist. Most of the clinical studies performed with carbamazepine have been in neuropathic pain syndromes other than PDPN, however, those that do exist are significantly dated. Carbamazepine requires therapeutic serum drug monitoring as well as judicious evaluation for Stevens-Johnson syndrome and hyponatremia. 46 Other considerations include monitoring for elevated liver transaminases and agranulocytosis. Carbamazepine exhibits a pharmacokinetic phenomenon known as “auto-induction” in which the medication actually induces its own metabolism.47,48 While an incredibly effective neuromodulator for neuropathic pain states, carbamazepine is usually reserved as a last-line agent given the concerns mentioned above.
Valproic acid and divalproex both possess data from randomized controlled trials (RCT) to support use in the treatment of PDPN. 45 Unfortunately, these agents frequently cause significant weight gain and glucose intolerance, limiting their use in this patient population. Additionally, concerns with hepatoxicity and teratogenicity also limit its use. 45
The second generation anticonvulsants are much more widely used in practice today and are supported by a large clinical database of experience. Those agents with positive data to support use in PDPN, listed in order of market availability, include gabapentin, felbamate, topiramate, lamotrigine, zonisamide, oxcarbazepine, levetiracetam, pregabalin, lacosamide. Due to the amount of evidence supporting the use of these agents, level of support for each agent is presented in Table 4.
Anticonvulsants.

Table adapted from Lexi-Comp, Inc. (Lexi-Drugs™). Lexi-Comp, Inc.; Jan 29, 2011.
It is important to note that while most, if not all, of these agents possess some positive data to support their use in the treatment of PDPN, most recently the abundance of data refuting efficacy has surpassed that of supporting efficacy for the drugs lamotrigine and levetiracetam.49,50 Additionally, while still available through limited-distribution channels, felbamate has largely become burdensome to obtain and is limited to refractory epilepsy treatment.
The gabapentinoids (gabapentin and pregabalin) have become the widely recognized treatments for PDPN. 4 Although gabapentin's efficacy has been called into question due to concerns about manufacturer malfeasance, a recent Cochrane Collaborative review has reaffirmed its continued use for this pain syndrome.51,52 Both of the gabapentinoids are relatively easy to dose and titrate, possess very few drug interactions, and do not result in end organ toxicity. For both of these agents, the most commonly recognized adverse effects include peripheral edema, somnolence during titration, and weight gain.51,53 It is currently unknown if the weight gain seen with these agents is due solely to fluid retention and edema versus true changes in adipose composition.54,55 Currently data exist to support use of pregabalin in patients who have failed gabapentin, however, no data exist to support the use of both of these agents simultaneously.56,57 These agents are oftentimes mis-perceived to act via GABA-mimetic activity, however, they in fact inhibit a specific sub-type of the calcium channel (Caα2δ).51,53
The carbonic-anhydrase active anticonvulsants (topiramate and zonisamide) possess multiple mechanisms of action including sodium and calcium channel blockade, glutamate receptor antagonism, as well as weak inhibition of the carbonic anhydrase system.58,59 Both agents have proven effective for PDPN in RCT studies, however, topiramate was unable to reach its primary endpoint during registration trials and was subsequently dismissed as a reasonable treatment for PDPN.60–63 These agents both have shown variable effects on weight which warrants interest in the diabetic population. In fact, both have shown hemoglobin A1c reductions similar to several currently marketed medications for glucose lowering.64–66 The use of these two agents is limited by neurocognitive effects (word finding difficulties), paresthesias, and risk of nephrolithiasis. Additionally, a rare, but serious risk of acute secondary angle-closure glaucoma should result in patient education to discontinue should symptoms arise.67,68 Zonisamide should not be used in patients with a documented sulfa medication allergy while topiramate is not prone to this sensitivity. 44
Oxcarbazepine, a derivative of carbamazepine, exhibits similar sodium channel blockade with a reduced incidence of severe hepatotoxicity and blood dyscrasias. The risk of a Steven Johnson Syndrome rash still exists, as well as hyponatremia. No serum drug monitoring is required and the medication is usually well-tolerated when titrated quickly.69,70 The most recent American Academy of Neurology (AAN) practice guidelines recommend against its use due to lack of data compared to other treatments. 4
Lacosamide, the newest anticonvulsant with promising data in PDPN, is unique in that its proposed mechanism of action for PDPN has not been previously described for other anticonvulsants.71–74 In contrast to carbamazepine, oxcarbazepine, and others with sodium channel antagonism, lacosamide changes the configuration and expression of voltage gated sodium channels via binding to collapsing response mediator protein-2 (CRMP-2). 73 This phosphoprotein has significance in the pathogenesis of neuropathic pain as it is thought to mediate differentiation and outgrowth of primary afferent axons. Although lacosamide's previously described positive RCT data supports its use, a recent systematic review questions lacosamide's widespread utility given a relative lack of clinically meaningful separation from placebo. 75 Additionally, aspartame is present as an ingredient in the tablet formulation which may theoretically pose a problem in the use of this agent for patients with diabetes. Lacosamide, like all other anticonvulsants, should be monitored for increased suicidal ideation as this adverse effect has been reported. Additionally, lacosamide may cause dose-dependent PR interval prolongation. Subsequently, baseline and steady-state ECG monitoring is recommended. 71
While still somewhat controversial, patients with PDPN who receive chronic anticonvulsant therapy should be considered for bone densitometry screening given the effects of anticonvulsants on osteoclastic activity (especially those anticonvulsants with CYP 450 inducing profiles).76–78 Recommendations are forthcoming on this practice and should guide providers on patient selection and frequency. Several anticonvulsants may also decrease the clinical efficacy of oral contraceptives and patients should be counselled on seeking alternative contraceptive methods, especially given the teratogenicity of these agents. 44
Antipsychotics
The introduction of the “atypical” antipsychotics, so called because of their diverse pharmacology separate from that of pure dopaminergic blockade, resulted in the investigation of these agents for the treatment of pain of a neurogenic origin. Numerous atypical antipsychotics have been studied for chronic noncancer pain syndromes, although few have focused on PDPN.79–81 Animal models support the newer, less dopamine selective antipsychotics in symptoms consistent with painful diabetic neuropathy (hyperalgesia, allodynia) although randomized controlled trials (RCTs) of significant power continue to be lacking. 79 Given the generally negative metabolic profiles of these agents (weight gain, insulin resistance), consideration for their use in this patient population should be thoughtfully considered.
Anesthetics/anti-arrhythmic
The anesthetic anti-arrhythmics are perhaps the most under-utilized of the potential strategies for alleviation of neuropathic pain associated with diabetes, although careful monitoring by experienced practitioners is paramount. These agents are all potent voltage-gated sodium channel antagonists, similar to several of the anticonvulsants and tricyclic antidepres-sant medications which have proven effectiveness for PDPN. 82 Lidocaine, mexilitine, and tocainide have all shown benefit for PDPN in smaller, RCTs. Some pain clinicians recommend the administration of a lidocaine infusion to assess the potential response to oral mexilitine, a lidocaine congener.83–85 Others report success with periodic lidocaine infusions without transition to an oral alternative. Mexilitine is specifically mentioned as a reasonable second-line treatment option for refractory neuropathic pain in recent practice guidelines, although specific commentary on its use in PDPN is lacking. While tocainide does possess positive data in PDPN, its significant car-diotoxicity largely limits is use. Perhaps the easiest method to incorporate the utility of the anesthetic anti-arrhythmics is the use of the 5% lidocaine patch. The effectiveness of this drug and administration technique is supported by well-designed studies and possesses a favorable side effect burden.86–88 The topical lidocaine patches should be applied to the painful area no longer than 12 hours daily, with a maximum number of patches applied concurrently not to exceed three. Additionally, these patches may be cut in order to more accurately apply the drug where needed. Unfortunately, many clinicians are unfamiliar with the safe use of the orally administered anesthetics and clear dosage and monitoring recommendations are lacking. Most recently, the American Academy of Neurology (AAN) joint guidelines for the treatment of PDPN recommend against the routine use of mexilitine based on a paucity of controlled data. 4
Vasodilators
As the numerous pathophysiologic theories for PDPN emerged over the past two decades, several treatments, such as direct vasodilators, were investigated for analgesia in this pain syndrome. Given the concerns with changes in the microvasculature and resultant small fiber hypoxia due to diabetes, treatment utilizing vasodilatation seems intuitive. Several direct vasodilators have been studied without positive results with the exception of isosorbide dinitrate nasal spray and glyceryl trinitrate transdermal patches.2,3 Isosorbide dinitrate spray is recommended as a second-line therapy option for PDPN based on the AAN evidenced based guidelines. 4
Opioids
The Opioids analgesics have unequivocally shown effectiveness in the treatment of PDPN. While these agents, specifically tramadol, oxycodone, and morphine, are recommended as second-line therapy in recent practice guidelines, their long-term utility has yet to be elucidated.4,89 Tramadol, a unique mu-opioid agonist with serotonergic reuptake inhibition, is supported by a large clinical database in PDPN studies.90,91 Although tramadol remains a non-controlled substance in many regions, this medication does possess opioid activity and appropriate monitoring and consideration should be given to its abuse liability. Tramadol should be prescribed cautiously in those with a history of seizure disorder and concurrent use with other serotonin-active agents discouraged. Tramadol as a parent drug possesses little analgesic activity and must be metabolized via CYP 450 2D6 to its O-desmethyl metabolite. Numerous frequently utilized medications inhibit this pathway, namely the SSRI antidepressants.
While not specifically listed in the evidence-based practice guidelines published to date, methadone may have a unique place in the treatment of PDPN. Methadone, a potent mu-opioid agonist, additionally possesses numerous other pharmacologic properties such as serotonin and norepinephrine reuptake inhibition, and n-methyl-d-aspartate inhibition, proposed mechanisms of central sensitization, allodynia, and neural plasticity associated with long-standing diabetes. 92 This medication, while effective for nociceptive, neuropathic, and mixed pain syndromes, should be prescribed by experienced clinicians familiar with its dosing and pharmacokinetics. Dose assessment and titration should occur weekly with particular attention paid to electrocardiogram and potassium monitoring. Specific recommendations exist to guide the clinician in developing a monitoring strategy.
Tapentadol, a new centrally-acting opioid, has positive RCT data to support its use in PDPN. 93 In contrast to tramadol, tapentadol is significantly more potent as an analgesic and is more selective for norepinephrine reuptake inhibition. Caution should be exercised using this agent concurrently with SNRI antidepressants based on package labeling although no significant hemodynamic effects have been reported to date. Additionally, while unexpected, patients should be educated on the signs and symptoms of serotonin syndrome.
Treatment of chronic noncancer pain with opioids continues to be debated. Long-term complications include hypogonadism, hyperprolactinemia, immunosuppression, hyperalgesia, and tolerance. Respiratory depression is rare except during initiation and titration, however, newer evidence suggests an increased risk of nocturnal hypoventilation, even during stable dosing, in those patients who are obese, snore, or have variants of sleep apnea. 94 Misuse and abuse of these agents is growing dramatically and necessitates a risk mitigation and monitoring plan by providers.
Future area of research
A promising area in need of additional study is botulinum toxin. A report in 2004, listed a case series of 4 patients, one of which had PDPN, treated successfully with Botox A injections. 95 The authors postulated that the toxin was not only working on the acetylcholine channels as previously thought but also involving other mechanisms. Since that time many studies have started to look at Botox in pain syndromes. However, in a large evidence based review published in 2011, there was only one small (N = 18) study looking at PDPN. They authors gave the treatment a “C” rating for this positive evidence due to the small number of patients involved and study design. 96 In order to see just how helpful this treatment could be, a large randomized controlled trial is needed.
Natural Products
Nonpharmacologic treatment
As the old adage says “prevention is the best medicine.” The landmark Diabetes Control and Complications Trial (DCCT) showed that HgbA1C is directly related to the development of microvascular complications including peripheral neuropathy. Tight glycemic control effects extended well beyond the study. Follow up 13-14 years later showed the prevalence of neuropathy for those in the tight glucose control group was 9%-25% as opposed to 17%-35% (P < 0.001) for the conventional treatment group. 97
Finding quality studies of nonpharmacologic treatments is often difficult as evidenced by the paucity of definitive reviews in the literature, however the use of natural products and herbals for PDPN has become more commonplace. While acetyl-L-carnitine has been studied in other pain syndromes such as fibro-myalgia, data in PDPN is still lacking. Alphalipoic acid, B complex vitamins and topical capsaicin have all shown promise in the treatment of diabetic neuropathy.
Vitamin B was studied in a Cochrane Review in 2008 which included 13 trials of 741 participants. 98 Findings were mixed with one study showing a slight increase in vibratory perception in those taking benfotiamine (a B1 deriviative) for 8 weeks. Another trial found that B complex in higher doses over 4 weeks of treatment decreased pain over lower doses. B vitamins are generally well tolerated with few side effects.
Capsaicin, obtained from the red chili pepper, is an irritant widely used in the treatment of PDPN. Proposed mechanisms include both direct counter-irritant effects, as well as its depletion of neurokinin-active peptides, namely substance P.99,100 Topical application of capsaicin results in symptoms of burning for most patients which lasts one to two weeks. This may serve as a barrier to adherence and continued need for education and reassurance provided to the patient. Once the burning subsides following application, topical capsaicin is a cost-effective means to address mild to moderate PDPN.
Perhaps the most promising of these strategies is alpha lipoic acid at doses of 600 mg daily. The Neurological Assessment of Thioctic Acid in Diabetic Neuropathy (NATHAN 1) trial showed no change in primary end point (composite score) from baseline with 4 years of treatment, however there was clinically meaningful improvement in neuropathy impairment scores and prevention of progression of neuropathy. The treatment was well tolerated. 101
Monochromic infrared energy therapy has also been evaluated. The theory revolves around increasing blood flow and potentially nitric oxide translating to better circulation and decreased symptoms overall. This form of light therapy has had mixed clinical findings to date.102–105 There may be an increased protective awareness for some and a decrease in pain on average of up to 2 points on the visual analog scale.
Chinese herbal medicine has predominantly been looked at in Chinese studies. Cochrane Collaboration published a review in 2011 that looked at 39 trials of 2890 patients. Overall the treatments were heterogeneous and there was felt to be inadequate reporting of adverse effects making safety of these treatments difficult to ascertain. 106
A small study of acupuncture in the treatment of diabetic neuropathy was able to show significant improvement over sham treatment. 107 In addition, a Westernized version of acupuncture known as percutaneous nerve stimulation (PENS) has shown significant decreases in pain on the Visual Analog Scale compared with sham treatment and was recommended to be considered a treatment by the recent American Academy of Neurology guideline. 4 An added benefit noted in several acupuncture studies is an improvement in patient-reported sleep. Side effects of acupuncture and PENS are generally mild and include pain at insertion site, minor bruising or bleeding, and vasovagal response.
Review of Guidelines
Guideline review
Recently, a number of new guidelines have been released on the topic of chronic neuropathic pain, some specifically regarding diabetes.
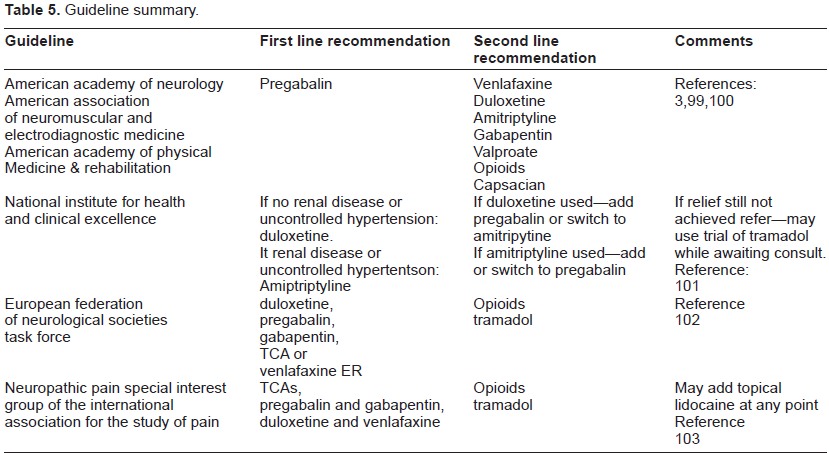
The American Academy of Neurology, the American Association of Neuromuscular and Electrodiagnostic Medicine, and the American Academy of Physical Medicine and Rehabilitation released a combined guideline in 2011. This guideline was specific for PDPN. It recommended pregabalin as the only Level A treatment due to Class I studies with improved pain and quality of life data. Medications receiving Level B recommendations included venlafaxine, duloxetine, amitriptyline, gabapentin, valproate, opioids and capsaicin. Other therapies received scrutiny but were not recommended due to ineffectiveness, lack of evidence, or side effects.4,108,109
An update of the National Institute for Health and Clinical Excellence (NICE) guidelines for neuropathic pain in the non-specialist setting was released in 2010. In individuals with PDPN, the recommended first line treatment in their setting based on evidence and cost is duloxetine, if not otherwise contraindicated due to renal function or uncontrolled hypertension. Amitriptyline is the other first choice if that is the case. On follow-up if good relief is accomplished but side effects are limiting, the recommendation turns to a trial of imipramine or nortriptyline. In second line treatment, one may combine or change to alternate medication. If the first line medication was duloxetine, the guideline recommends adding or changing to pregabalin. One could also switch to amitriptyline. If amitriptyline was the first line medication used, add or switch to pregabalin. However, if adequate pain control is not obtained after the second line treatment is titrated to full effectiveness, NICE recommends referral to subspecialty service and, while awaiting referral, a trial of tramadol. Recommendations against opioids in the non-specialist setting are clearly and specifically stated. 110
Additionally, in 2010, the European Federation of Neurological Societies Task Force revisited the 2005 recommendations regarding neuropathic pain. On the subject of PDPN, the guidelines similarly recommend duloxetine, pregabalin, gabapentin, TCA or venlafaxine ER first line, reserving opioids and tramadol for second line usage. 111
Finally, for this grouping of most current guideline updates is the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain 2009 consensus guidelines based on the available evidence. First line medication recommendations include TCAs, pregabalin and gabapentin, duloxetine and venlafaxine. Second line alternatives include treatment with opioids and tramadol. Consider lidocaine patches or topical gel if localized pain (first or second line). 112
See Table 5 for summary.
Guideline summary.
Conclusions
The current recommendations give the clinician the freedom to assess the patient and individual circumstances, choose the class of medication most suited to the situation and adjust as needed. PDPN proves to be a difficult process to treat in which many patients only receive a 30%-50% reduction in pain. Promotion of functionality, quality of life, sleep improvement, and pain reduction as opposed to alleviation are appropriate goals. Setting appropriate expectations with patients promotes successful communication and enhances satisfaction. What follows is the authors’ recommended algorithm for treatment. Figure 1 shows an algorithm when the patient has no other significant compelling factors. In this case, either TCAs or gabapentinoids may be used as first line. If insufficient effect is reached, then consider adding or switching to the other agent. If the patient's goals are still not met, consider adjuncts such as lidocaine, capsaicin, B vitamins and/or tramadol. Figures 2–4 show algorithms based on specific conditions commonly associated with PDPN.
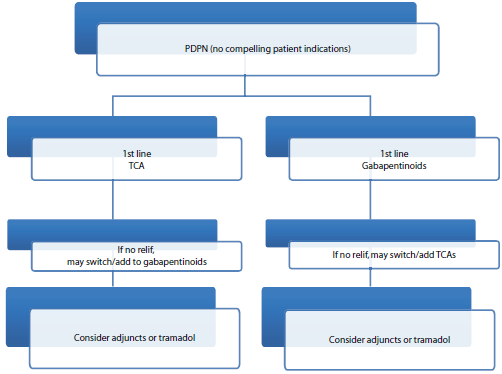
Algorithm for PDPN if no compelling patient indications.
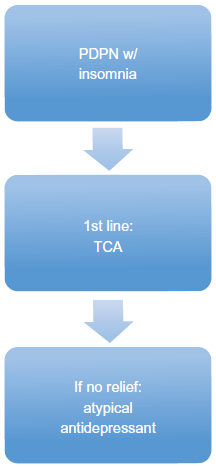
Algorithm for PDPN with insomnia.
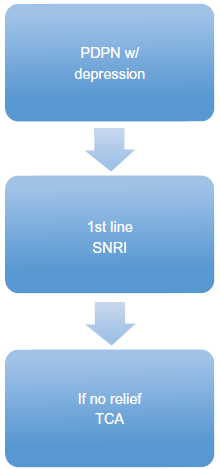
Algorithm with PDPN with depression.
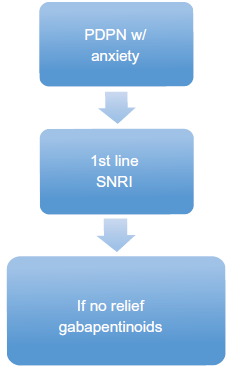
Algorithm for PDPN with anxiety.
Author Contributions
TJL, KV, MT, CMH. Contributed to the writing of the manuscript: TJL, KV, MT, CMH. Jointly developed the structure and arguments for the paper: TJL, KV, MT, CMH. Made critical revisions and approved final version: TJL, KV, MT, CMH. All authors reviewed and approved of the final manuscript.
Funding
Authors disclose no external funding sources.
Competing Interest
Authors disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
The opinions expressed in this article are the opinions of the authors and not that of the United States Air Force.
